Abstract
The ability of current renal replacement therapy modalities to achieve rapid solute removal is limited by membrane surface area and blood flow rate. Extracorporeal membrane oxygenation offers high blood flow and hemodynamic support that may be harnessed to overcome limitations in traditional renal replacement therapy. Using an extracorporeal membrane oxygenation circuit, we describe a high blood flow, high-efficiency hemofiltration technique using in-line hemofilters (hemoconcentrators) and standard replacement fluid to enhance solute clearance. Using this approach and a total of 5 L of replacement volume per treatment, creatinine (Cr) clearances of 8.3 L/hour and 11.2 L/hour using one and two hemoconcentrators, respectively, were achieved. With use of a high blood flow rate of up to 5 L/min, this hemofiltration technique can potentially offer clearance of 30 times that of continuous renal replacement therapy and of 6 times that of hemodialysis which may expand the ability to remove substances traditionally not considered removable via existing extracorporeal therapies.
Keywords
Case
A 62-year-old 62 kg man with a history of hypertension, diabetes, coronary artery disease, and chronic kidney disease presented to an outside hospital with dyspnea. Left heart catheterization (LHC) was performed showing 99% occlusion of right coronary artery. The patient was later transferred to our institution for additional percutaneous coronary intervention. Renal replacement therapy (RRT) was initiated at the outside hospital for non-oliguric acute kidney injury (AKI). During LHC on day of arrival, the patient suffered cardiac arrest and was placed on veno-arterial extracorporeal membrane oxygenation (ECMO) peri-arrest. The patient’s AKI worsened and he developed oliguria within 24 hours.
After emergent initiation of ECMO, consent was obtained for continuation and for initiation of RRT (convective clearance for solute removal). Initially, fluid overload was managed using an in-line hemofilter as is already the standard of care in most ECMO units in the United States. 1 The hemofilter, an in-line Sorin-14 (polyethersulfone membrane) hemoconcentrator (LivaNova USA, Inc., Arvada, CO, USA), had a blood flow throughput of ~1 L/minute per flow meter measurement and was paced before the oxygenator, with proper technique and with close monitoring for safety purpose. The Sorin-14 hemofilter has characteristics that, at minimum, match high-flux hemodialyzers with a sieving cut-off of 65,000 Da and is well suited for high-pressure convective solute clearance.1,2 To establish the validity of baseline convective qualities, effluent electrolyte concentrations from both the Sorin-14 and the PRISMAFLEX M-150 set AN 69 (Baxter International, Deerfeild, IL, USA) conventional continuous renal replacement therapy (CRRT) membrane were concurrently compared in another patient and were nearly identical (data not shown). During patient’s first solute clearance treatment period (treatment one), 5 L of PrismaSATE replacement fluid (Baxter International) was administered and 5 L of hemofiltration was performed using the hemofilter. This was achieved by first delivering 500 cc increments of replacement fluid via the ECMO circuit followed by the exact physician-matched removal of the same effluent volume (500 cc) via the hemofilter using gravity drainage. This process was repeated over the span of 50 minutes until the total effluent removed and replacement fluid volume administered each totaled 5 L. Risk of volume depletion was minimized by exactly matching the amount of replacement fluid given to the amount of effluent removed. Hemodynamics remained stable with mean arterial pressure (MAP) range of 80-90.
For the subsequent treatment (Treatment 2), a second Sorin-14 hemofilter was placed in-line (Figure 1(a)). Both hemofilters, now in parallel with one another, maintained blood flow throughput of ~1 L/minute each as measured by flow meters. A similar procedure was followed to administer 5 L of replacement fluid along with matched effluent volume removal of 5 L via gravity drainage. However, during Treatment 2, effluent was removed simultaneously from both hemofilters via gravity drainage, which doubled the effluent removal rate. This process of replacing and removing 5 L of fluid was completed in 25 minutes, around 10% of time required to achieve standard CRRT clearance goals of 20 cc/kg/hour. 3 The patient remained hemodynamically stable (MAP 70-90) without any adverse effects and there was no net fluid removal. Traditional CRRT was subsequently initiated for further convective clearance and volume management. Basic metabolic panels were obtained pre and post both treatment sessions (Supplemental Table 1a). Effluent fluid was collected and sodium, potassium, and creatinine values were measured from both sessions (Supplemental Table 1b).
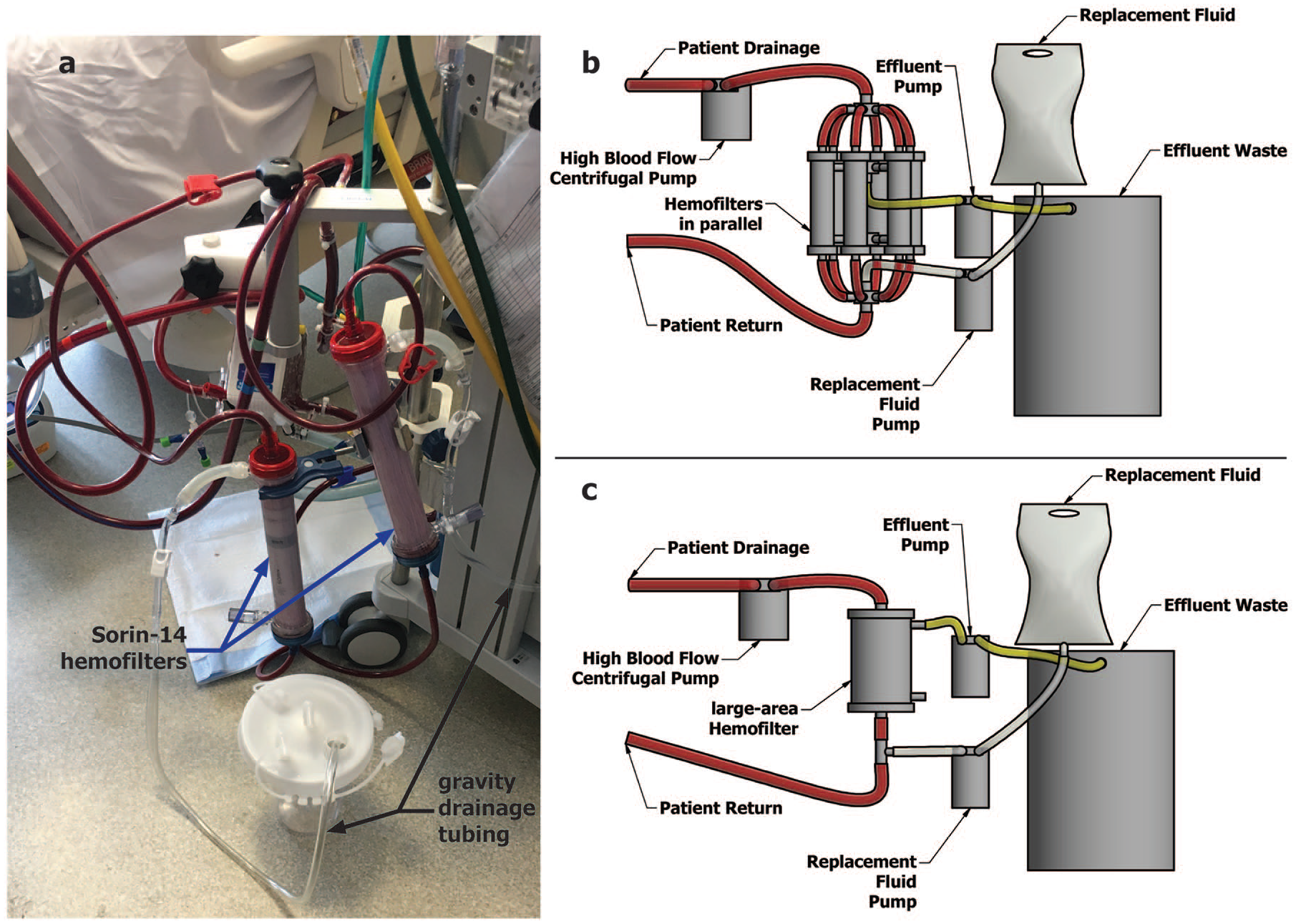
(a) Two Sorin-14 hemofilters (blue arrows) placed in parallel in-line with ECMO circuit with effluent tubing to allow for gravity drainage. Black arrows indicate gravity drainage tubing. (b) Theoretical use of high-flux hemofilters arranged in parallel within a high blood flow circuit (for example, an ECMO circuit). (c) A theoretical large area hemofilter able to accommodate high blood flow throughput (~5-7 L/minute) in series within a high blood flow circuit. Effluent removal is supported by an effluent pump and collects in a waste container. Replacement fluid is delivered via use of replacement fluid pump and is delivered to circuit post-filter (though pre-filter delivery is also possible).
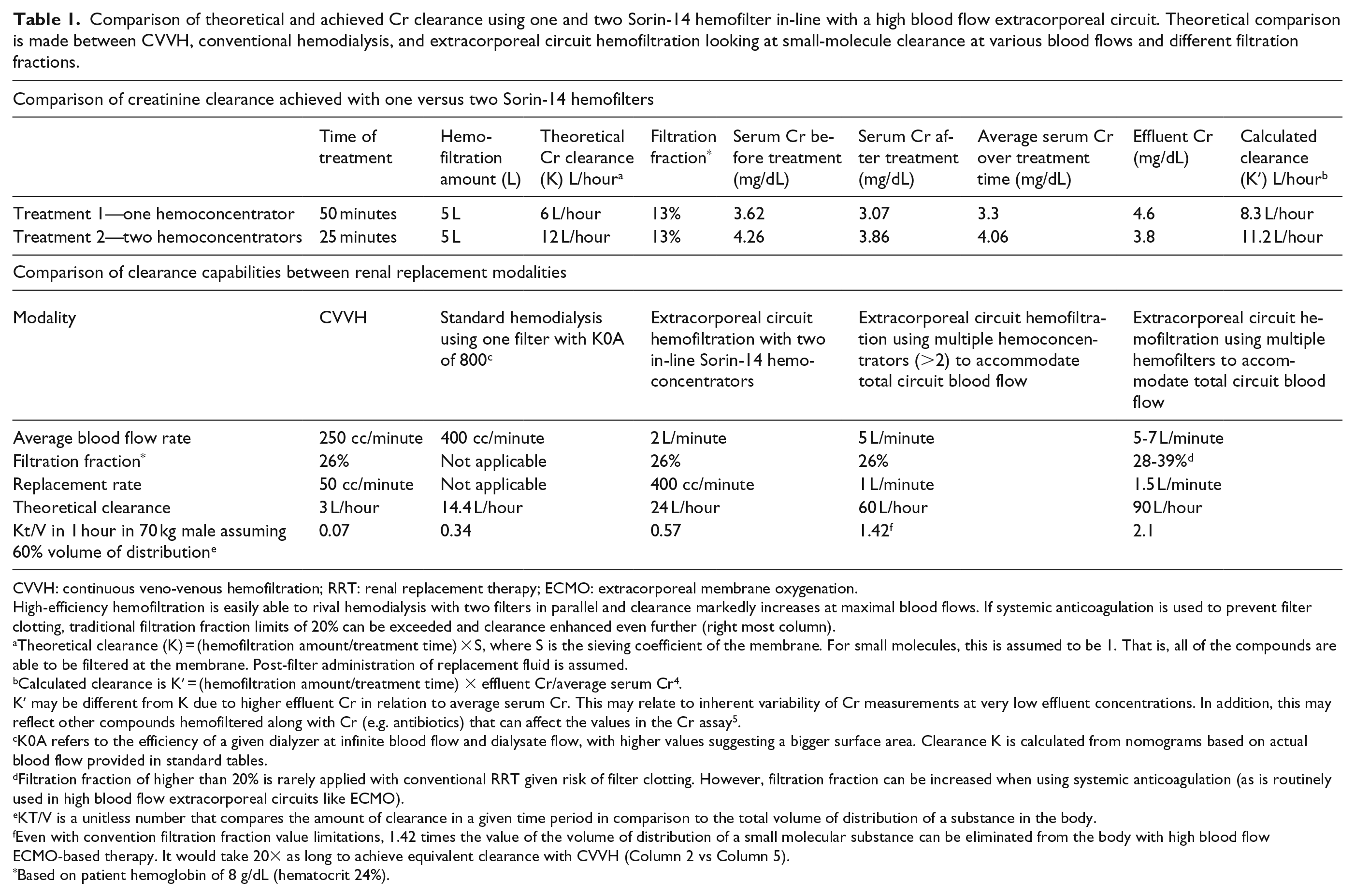
Traditional calculations for clearance in CRRT using post-filter replacement fluid utilize the concept that K (clearance) = hemofiltration rate × S (the ratio of effluent solute concentration/blood solute concentration). 4 Theoretically, S = 1 for most small solutes like Cr given that they freely pass through membrane pores during convection. In our patient, as anticipated, the S value for Cr was nearly one. The above replacement rates (5 L/time of treatment) normalized to 1 hour yielded a theoretical K (clearance) value of 6 L/hour in Treatment 1. In Treatment 2, the K value was twice the value of Treatment 1 (12 L/hour) as expected given that filter area doubled during the second treatment. Clearance can also be calculated as measured mass flux across the membrane (J)/average blood solute concentration (Table 1). With this approach, the Cr clearance for Treatment one was 8.3 L/hour, whereas in Treatment 2, it was 11.2 L/hour. Differences between the theoretical and calculated values may represent inherent lab variability when measuring small concentrations of Cr in effluent samples or drug interference with the Cr assay. 5 The overall trend of both calculations, however, shows near doubling of clearance when two filters were used in parallel approximating the upper limit of what conventional CRRT is currently able to achieve. This was using only 1 L/minute of blood flow per hemofilter, whereas the ECMO circuit can actually flow ~5-7 L/minute. This high blood flow could theoretically allow for even greater convective clearance (Table 1).
Comparison of theoretical and achieved Cr clearance using one and two Sorin-14 hemofilter in-line with a high blood flow extracorporeal circuit. Theoretical comparison is made between CVVH, conventional hemodialysis, and extracorporeal circuit hemofiltration looking at small-molecule clearance at various blood flows and different filtration fractions.
CVVH: continuous veno-venous hemofiltration; RRT: renal replacement therapy; ECMO: extracorporeal membrane oxygenation.
High-efficiency hemofiltration is easily able to rival hemodialysis with two filters in parallel and clearance markedly increases at maximal blood flows. If systemic anticoagulation is used to prevent filter clotting, traditional filtration fraction limits of 20% can be exceeded and clearance enhanced even further (right most column).
Theoretical clearance (K) = (hemofiltration amount/treatment time) × S, where S is the sieving coefficient of the membrane. For small molecules, this is assumed to be 1. That is, all of the compounds are able to be filtered at the membrane. Post-filter administration of replacement fluid is assumed.
Calculated clearance is K′ = (hemofiltration amount/treatment time) × effluent Cr/average serum Cr4.
K′ may be different from K due to higher effluent Cr in relation to average serum Cr. This may relate to inherent variability of Cr measurements at very low effluent concentrations. In addition, this may reflect other compounds hemofiltered along with Cr (e.g. antibiotics) that can affect the values in the Cr assay5.
K0A refers to the efficiency of a given dialyzer at infinite blood flow and dialysate flow, with higher values suggesting a bigger surface area. Clearance K is calculated from nomograms based on actual blood flow provided in standard tables.
Filtration fraction of higher than 20% is rarely applied with conventional RRT given risk of filter clotting. However, filtration fraction can be increased when using systemic anticoagulation (as is routinely used in high blood flow extracorporeal circuits like ECMO).
KT/V is a unitless number that compares the amount of clearance in a given time period in comparison to the total volume of distribution of a substance in the body.
Even with convention filtration fraction value limitations, 1.42 times the value of the volume of distribution of a small molecular substance can be eliminated from the body with high blood flow ECMO-based therapy. It would take 20× as long to achieve equivalent clearance with CVVH (Column 2 vs Column 5).
Based on patient hemoglobin of 8 g/dL (hematocrit 24%).
Discussion
Volume management for patients supported with extracorporeal circulation is often through hemoconcentration via a hemofilter placed in-line with the circuit. 1 However, the use of an in-line hemofilter for solute management has not been studied to our knowledge. Here, we report the novel utilization of in-line hemofiltration with commercial hemofilters and replacement solutions to achieve high-efficiency, high-flux convective solute clearance.
Currently, solute clearance is most often achieved through use of a CRRT circuit placed at a dedicated access site or in-line with an ECMO apparatus. Using CRRT to achieve convective clearance has two significant limitations. Convective clearance is determined by the volume of hemofiltration/time which is limited by the system’s blood flow. 6 Blood flow rates in CRRT circuits do not typically exceed 300 mL/minute due to smaller diameter access catheters and tubing. 4 Currently, achievable hemofiltration rates are also restricted by filtration fraction (hemofiltration rate/blood flow in the circuit) with filtration fractions >20% leading to excessive hemoconcentration and clotting. Both the impediments of filtration fraction ceiling and lower blood flow can be addressed with the use of systemic anticoagulation and large-diameter catheters, respectively.
The second limitation of conventional RRT (continuous and intermittent modalities) is the use of a single standard-size hemofilter or hemodialyzer. These membranes are designed to accommodate blood flow ranges attainable through dialysis catheters (up to 400 mL/minute) or via cannulation of peripheral arteriovenous access sites (up to ~ 600 mL/minute). Since the efficiency of convective solute removal is dependent on both the hemofilter surface area and rate of hemofiltration, proportionally increasing the hemofilter capacity to accommodate higher blood flows of 5-7 L/minute achieved by an extracorporeal circuit, like ECMO, could dramatically enhance the efficiency of solute clearance to 30 times that of CRRT and 6 times that of hemodialysis (Table 1). This could be theoretically accomplished by placing standard high-flux hemofilters in parallel with one another as reported in this case (Table 1, Figure 1(b)). Alternatively, this could be achieved via development of a single larger hemofilter placed in series within a high-flow extracorporeal circuit engineered to maintain the ratios of blood flow to surface area achieved with traditional RRT (Figure 1(c)). Maintenance of these ratios would be essential to preserve the transmembrane pressures and solute sieving properties that standard hemofilter membranes are designed to sustain. In addition, this would prevent complications of hemolysis secondary to increased shear forces created by blood membrane interactions at higher pressures. 7 Finally, to ensure ease and accuracy of replacement fluid delivery, an additional pump could theoretically be incorporated to deliver replacement fluid to the extracorporeal circuit (pre- or post-filter) at a desired rate (Figure 1(b) and (c)). This rate would match the hemofiltration rate unless additional fluid removal was desired.
Here, we demonstrate that use of hemofilters incorporated into a high-flow circuit and in parallel with each other can achieve solute clearance rates that exceed current RRT capabilities. Using this novel method, Cr clearance between 11 and 12 L/hour using two in-line hemofilters was achieved. While traditional RRT provides adequate support for renal failure, its limited efficiency can prevent its application to other critical clinical scenarios. Patients suffering from toxin ingestion, such as drug overdose, endogenous toxic molecule production such as myoglobin in rhabdomyolysis, or cytokine release as occurs in sepsis, may require higher levels of solute clearance for potential therapeutic benefit. 8 Thus, the high-efficiency, high-flux clearance that can be achieved by this approach may broaden the utilization of convective solute removal as a possible therapeutic tool for a variety of disease states mediated by endogenous or exogenous toxins.
Potential limitations of highly efficient solute clearance include two major considerations: first, potential removal of beneficial drugs, electrolytes (e.g. phosphate), and important molecules such as micronutrients (e.g. B-6, B-12, ascorbic acid, etc.)9,10 This would need to be further studied along with consideration of drug dosing modification and development of replacement fluid to include physiologic concentrations of important molecules at risk for depletion. Abrupt change in serum osmolality due to highly efficient solute clearance is another potential risk. This could likely be managed with the addition of urea or an alternative osmole to the replacement fluid to avoid development of hemodynamic instability, cerebral edema, and dialysis disequilibrium. Second, there is risk of poor volumetric control that could lead to hemodynamic instability. 1 This was addressed by first giving volume and then removing it as opposed to the converse, obviating the potential for becoming volume contracted from the beginning of therapy. Strict perfusionist monitoring of hemofiltration via gravity-based drainage (i.e. exactly matching input replacement volume with effluent output volume) further prevented risk of inaccuracy in volume removed. Obviously, for routine practical patient care, this would require an alternative, automated, and accurate means of effluent measurement beyond gravity drainage that is not currently available. Finally, cost and invasiveness of this robust hemofiltration system would need to be weighed against benefit.
Conclusion
Hemofiltration at high blood flow rates with high-flux membranes offers highly efficient solute clearance well beyond current RRT capabilities. Traditional RRT limitations of blood flow dependency and filtration fraction can be overcome to offer rapid clearance of toxins, including those not currently dialyzable (e.g. those with high volumes of distribution). 8 Further study of this high-efficiency, high-flux hemofiltration circuit may maximize the potential utility of this novel form of convective clearance.
Supplemental Material
Supplemental_Table – Supplemental material for High-efficiency, high-flux in-line hemofiltration using a high blood flow extracorporeal circuit
Supplemental material, Supplemental_Table for High-efficiency, high-flux in-line hemofiltration using a high blood flow extracorporeal circuit by Alison Grazioli, Sanjeev R Shah, Joseph Rabin, Rashmikant Shah, Ronson J Madathil, Joshua D King, Laura DiChiacchio, Raymond P Rector, Kristopher B Deatrick, Zhongjun J Wu and Daniel L Herr in Perfusion
Footnotes
Acknowledgements
We would like to thank Nicholas Formica for technical guidance with figure design.
Author’s Note
Part of this work has been presented at the EuroElso Conference in Barcelona, Spain (April 2019).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained for publication of patient’s clinical details and clinical images from the patient. A copy of the consent form is available for review by the Editor of this journal. IRB oversight was also obtained (IRB number HP-00086910).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
