Abstract
Marine engineering components, such as ship stern shafts, are often subject to local overload, wear and seawater erosion. In order to improve the performance and service life of marine engineering components, the CrFeCoNiNb
Introduction
The traditional alloying strategy is to add small amounts of minor elements to one or two major elements. However, this method of major elements severely limits the range of possible element combinations. The high-entropy alloy (HEA) is a type of multi-principal element alloy. HEAs have many advantages based on the high-entropy effect, the lattice distortion effect, the slow diffusion effect and the cocktail effect. The HEA has excellent properties such as high strength, high hardness, good ductility and wear resistance, excellent corrosion resistance and oxidation resistance, etc., and the HEA is expected to be widely used in the aerospace and marine engineering.1–3
As the key properties for practical applications, the mechanical properties and corrosion resistance of HEAs have always been the research focus. In order to enhance the FeCoNiCr HEA coating properties, it is effective to incorporate elements with special properties, such as Nb, Ti, Al, Cu. Several minor elements have been added to improve the mechanical properties and corrosion resistance of FeCoNiCr HEA.
In general, the addition of specific elements will inevitably lead to lattice distortion and phase composition transformation, which directly affects the mechanical properties of the alloy through the combination effect of the solid-solution strengthening, the secondary-phase hardening and the grain refinement.1–5 To improve the mechanical properties, the element Nb was added to the FeCoNiCr HEA coating by Chung et al.
1
The CoCrFeNiNb0.5 HEA was proved to have superior fracture toughness and strength at room temperature due to the hierarchical eutectic structure.
1
The AlCoCrFeNiTi
On the other hand, these minor elements play an important role in resisting corrosion by forming protective oxide films.6–9 The most common types of corrosion are uniform corrosion and local pitting. Pitting preferentially begins at surface inhomogeneities, and oxides formed at these sites or along their periphery generally have no protective effect. In addition, impurity phases, second phase precipitation and segregation are detrimental to the continuity of the passivation film. Therefore, one of the key factors in determining the pitting resistance is the ability to form a continuous passivation film without component defects over the entire surface. The corrosion properties of AlCoCrFeNi HEA coatings were investigated by Li et al. 6 It was found that the absence of Cr-rich intercellular segregation and nanoscale precipitates resulted in a relatively uniform corrosion attack. This differed from the severe galvanic corrosion attack. It was reported that the Al0.1CoCrFeNi HEA showed greatly longer incubation period and lower erosion corrosion rate compared with SS316L steel, which was attributed to the high work-hardening behaviour and stable passive film on the surface. 7 Shi et al. investigated the corrosion behaviour of AlxCoCrFeNi HEAs. They found that the increase in Al caused the passivation film to thicken/disperse, thereby reducing the local corrosion resistance. 8
Moreover, laser cladding, as a surface modification technology, has the characteristics of rapidly cooling and heating, which can promote the formation of simple solid solution. In addition, compared with other surface modification technologies, the thickness of the laser cladding layer is higher (≥0.8 mm). A good metallurgical bonding is formed between the cladding layer and the substrate, which has better performance than the alloy with mechanical bonding. With the further study of FeCoNiCr HEA, Nb is considered as an element to improve hardness and corrosion resistance. It is also an attractive research point to explore the synergistic mechanism of mechanical properties and corrosion resistance of CrFeCoNiNb
Materials and methods
Sample preparation
The 42CrMo with dimensions of 70 mm × 15 mm × 10 mm was employed as substrate, and the surface was polished, then cleaned with alcohol and air-dried. The powders were weighed according to the atomic ratio of CrFeCoNiNb

Coating morphology.
Alloy composition parameters of cladding layer.
Microstructure characterisation
The surface of the prepared sample was cut, ground and polished, and then the sample was corroded by aqua regia for 30s. Subsequently, the grain topography of the sample was examined by scanning electron microscopy (SEM) and energy dispersive spectroscopy (EDS). In addition, X-ray diffraction (XRD) was also performed for structural characterisation.
Performance test
The structure was characterised by Rigaku Ultima IV, and the copper (Cu) was selected as the target material for the experiment, and a voltage of 40 kV and current of 40 mA were applied for the study. The size of the sample was 15 mm wide and about 8 mm long. The Vickers hardness was measured using the HV-1000 Vickers microhardness tester with a load of 10 N for 10 s. Each reported microhardness value was measured from the top of the coating to the substrate with an interval of 0.2 mm. Each reported microhardness value was an average of three measurements. The wear test was evaluated by the UMT-3 TriboLab. The specific parameters were set as follows: the unidirectional displacement was 5 mm, the frequency is 2 Hz, the load was 10 N, and the test duration was 30 min. To ensure that all tests were performed under the same conditions, a 9.525 mm diameter Si3N4 ball was used in dry sliding conditions and a fresh Si3N4 ball was used. Each sample was measured three times as an average value to ensure data accuracy. After the wear test, the abrasion loss of the sample was carried out by laser scanning confocal microscope (LSCM), and the material loss of the wear area was calculated. The samples were sealed in epoxy resin with only one exposed surface area of 1 cm2 prior to electrochemical corrosion test. The potentiodynamic polarisation test was performed in a typical three-electrode cell. The cell consisted of the test sample as the working electrode, a saturated calomel reference electrode (SCE) and Pt as the counter-electrode. The measurements were performed in 3.5 wt% NaCl solution at 25°C under atmospheric pressure, using the CHI660E electrochemical workstation manufactured by Shanghai Huachen. The open circuit potential (OCP) was recorded for 30 min to obtain a stable potential before these tests. Then electrochemical impedance spectroscopy (EIS) data were acquired from 100 kHz to 10 mHz with potential amplification of 5 mV. Each sample was scanned from an initial potential of −1 V to a final potential of 1 V at a rate of 1 mV/s for potentiodynamic polarisation measurements. Electrochemical parameters were obtained using CH Instr. analyst software.
Results and discussion
Phase analysis
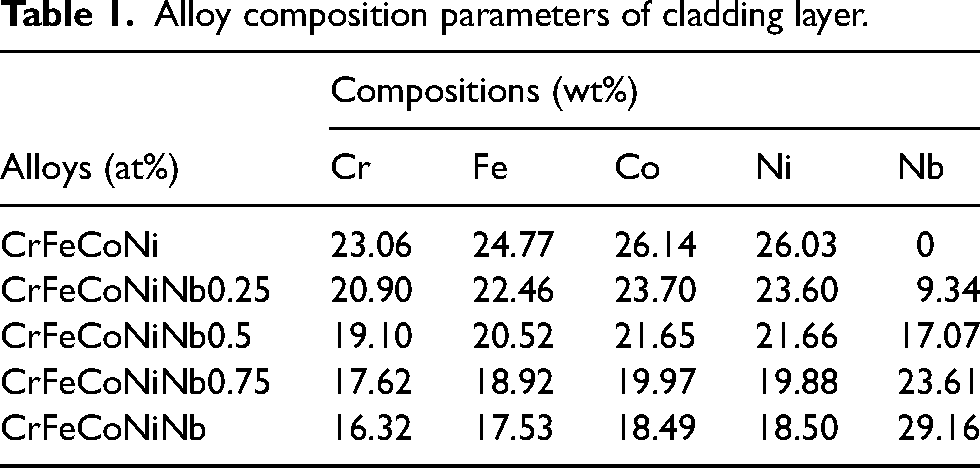
The XRD pattern of the CrFeCoNiNb
XRD patterns and lattice parameter of the CrFeCoNiNb
Microstructure analysis
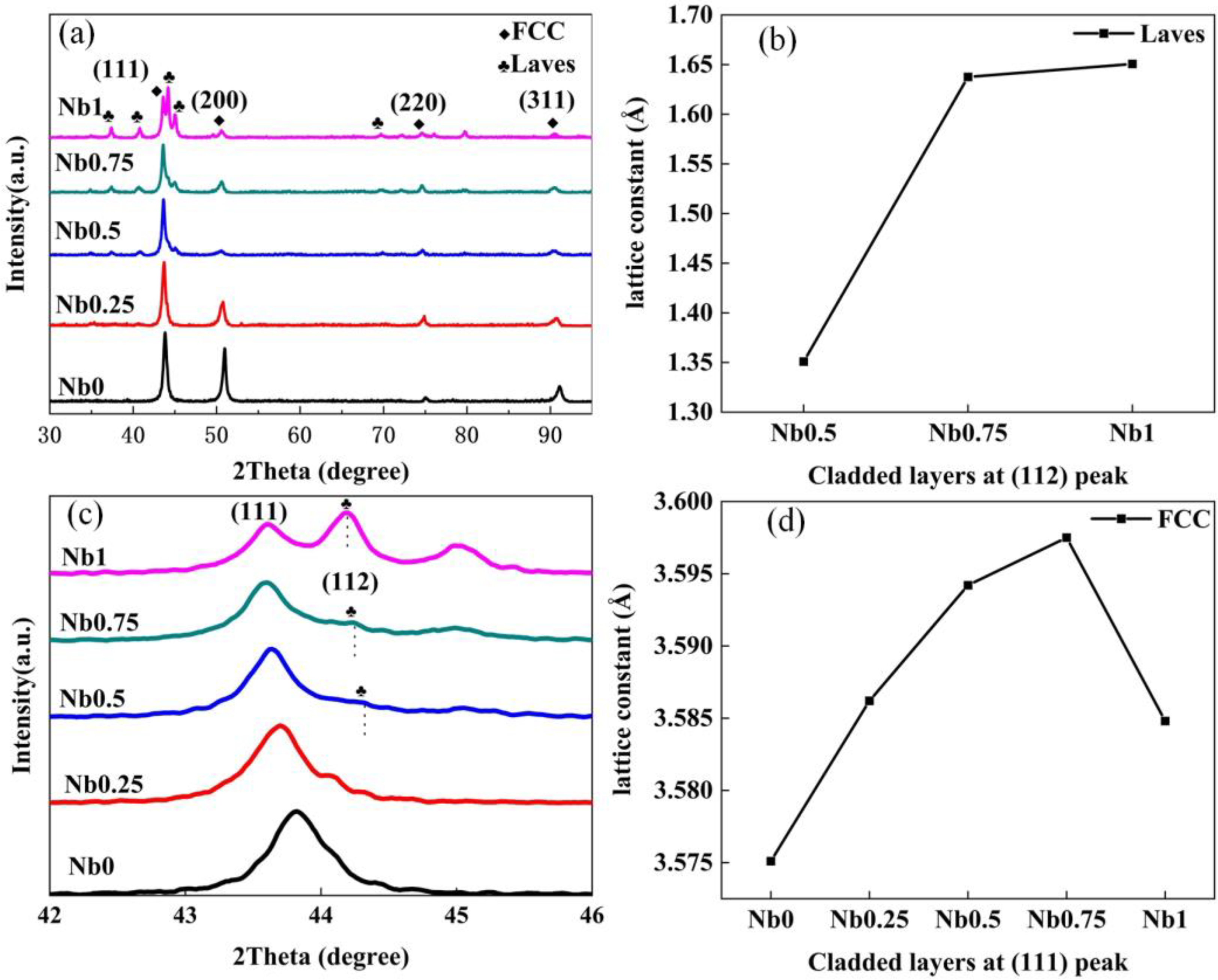
Significant microstructural evolution can be clearly observed in Figure 3 with the increasing of Nb content. The growth of the microstructure follows the principle of dendrite growth, and the cladding layer is generally composed of cellular grains. But there is a clear trend of grain refinement, which is a common feature of
SEM and EDS images of CrFeCoNiNb
Vickers hardness
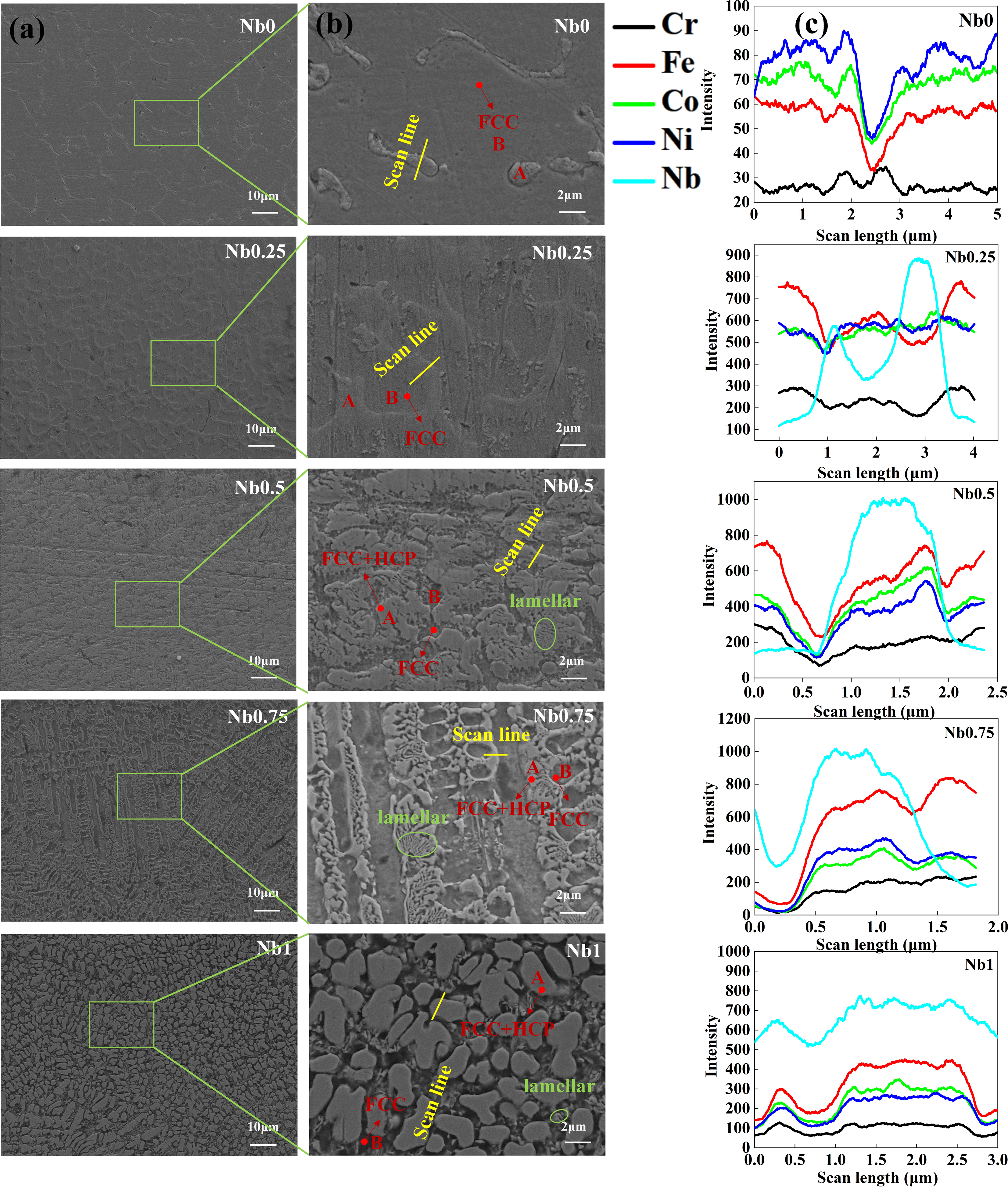
The Vickers hardness values for CrFeCoNiNb
Vickers hardness of cladding layers.
A decrease in the microhardness of the coating (Nb
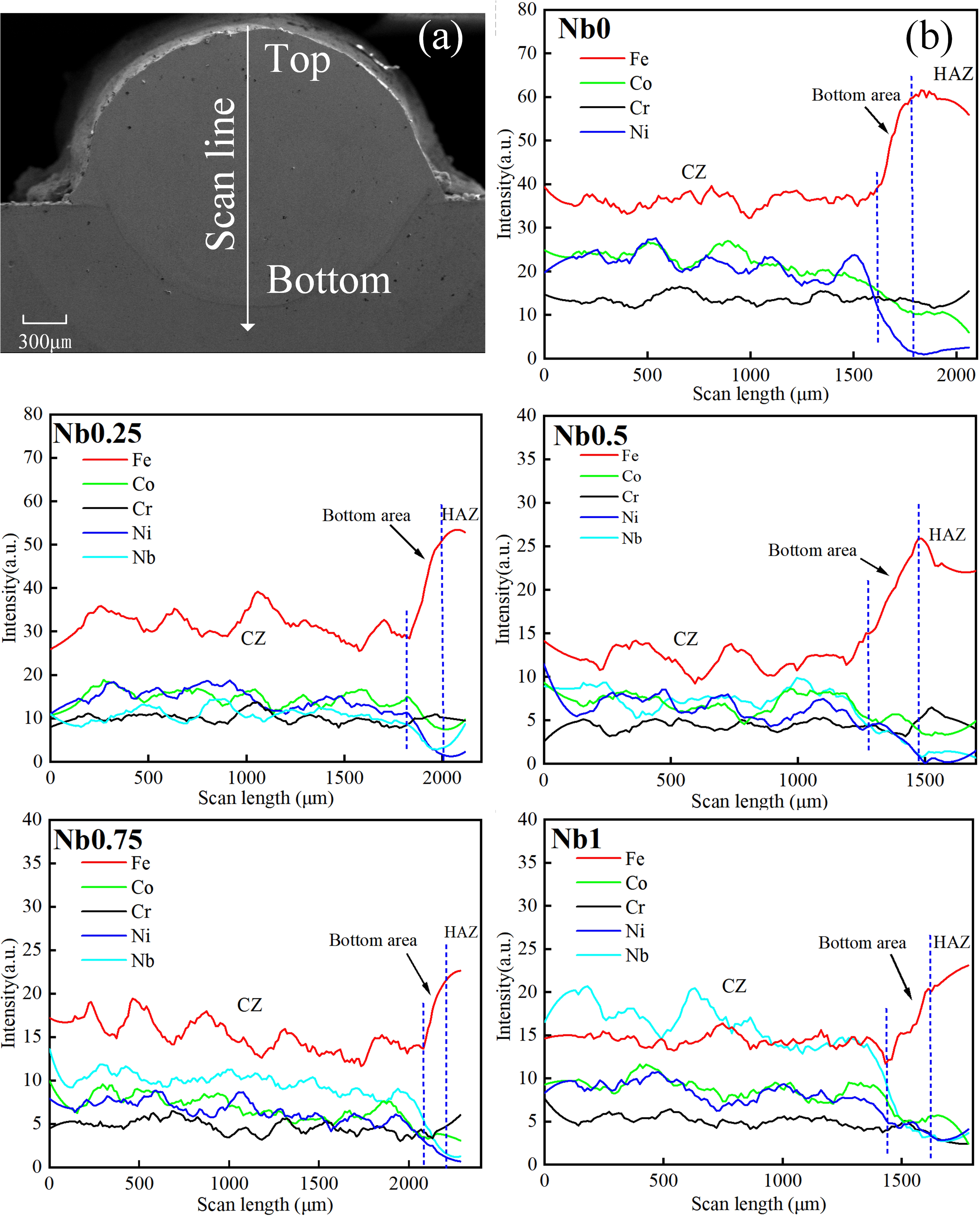
Element distribution of cladding layer: (a) scanning path diagram; (b) EDS scan curve.
Wear resistance
It can be seen from Figure 6(a) that there are grooves, flakes, spalling craters and wear debris on the surface of the friction layer. This indicates that the wear mechanism is adhesive wear and abrasive wear. The factors that affect the wear of the coating are mainly related to the hardness of the coating, and the appearance of the oxide layer will also affect the wear. During the friction process, the wear debris material is compressed and oxidised to form a hard oxide layer, as shown in Figure 6(b), which reduces the wear of the coating, acts as a lubricant
11
and reduces the plastic deformation caused by contact stress during the grinding process. Obviously, the microhardness of the substrate and the cladding layer with

Wear morphology of the substrate and cladding layers: (a) SEM of wear morphology; (b) EDS of friction layer.
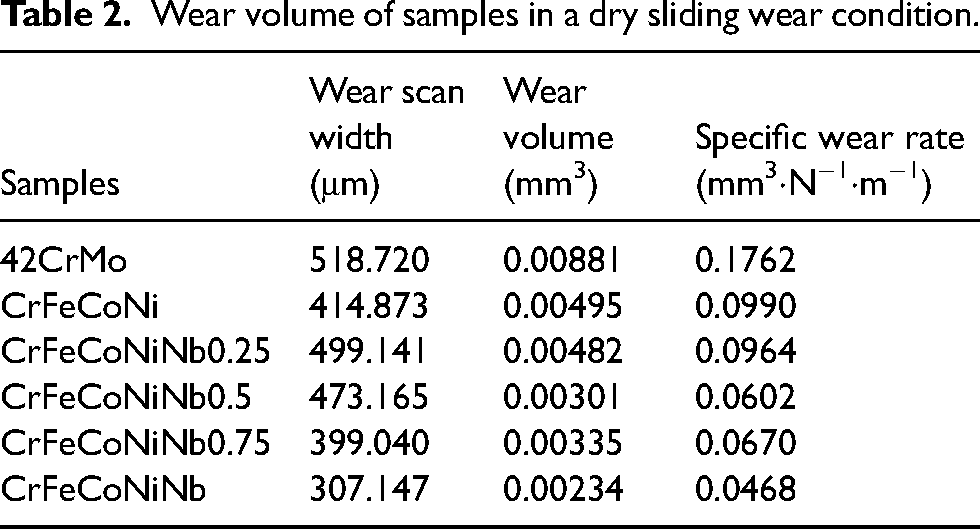
The wear scar width and the volume of the wear scar of the CrFeCoNiNb
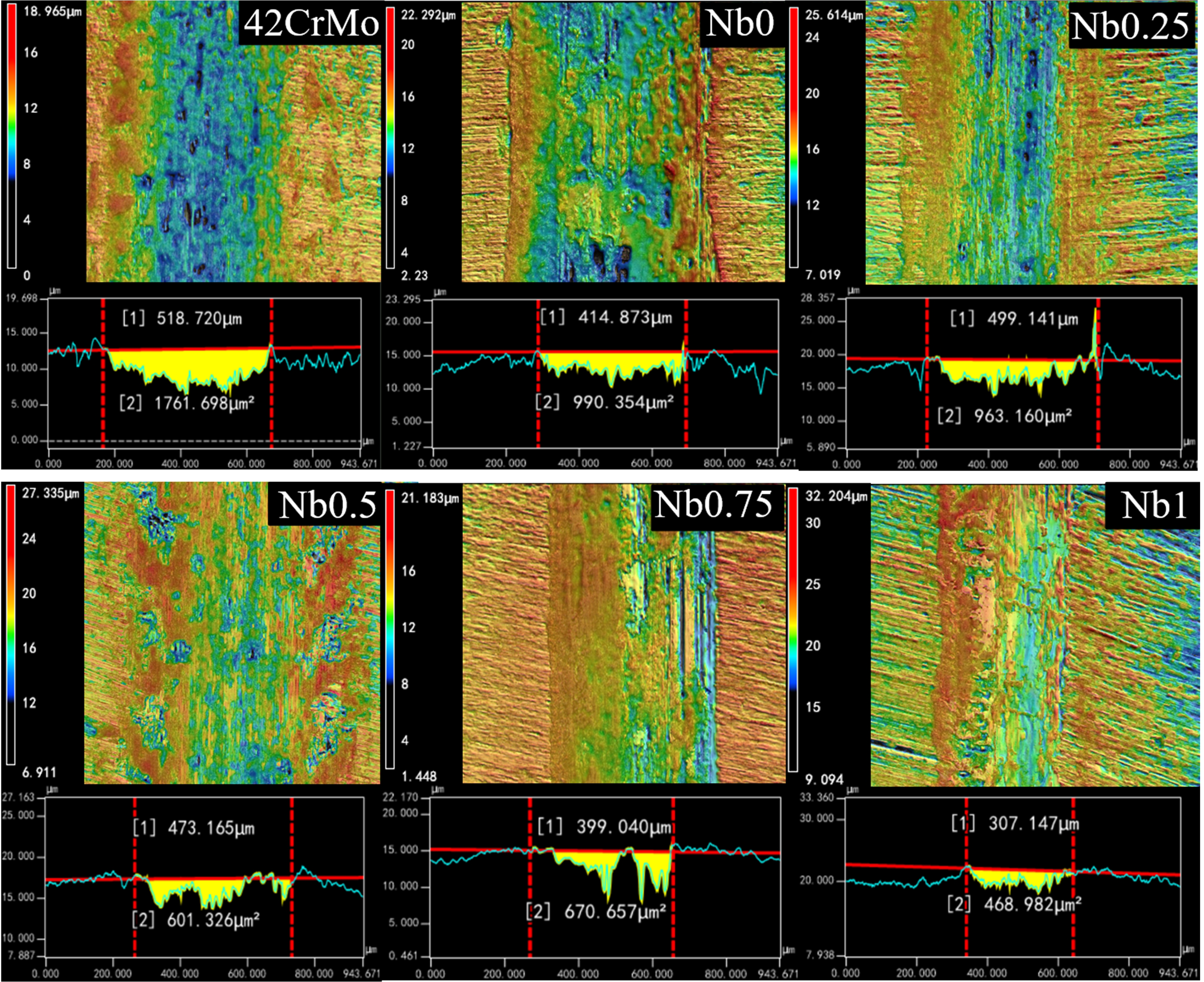
Observation of wear characteristics of CrFeCoNiNb
The relationship between microhardness and wear resistance is illustrated in Figure 8 According to the formula of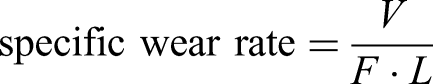
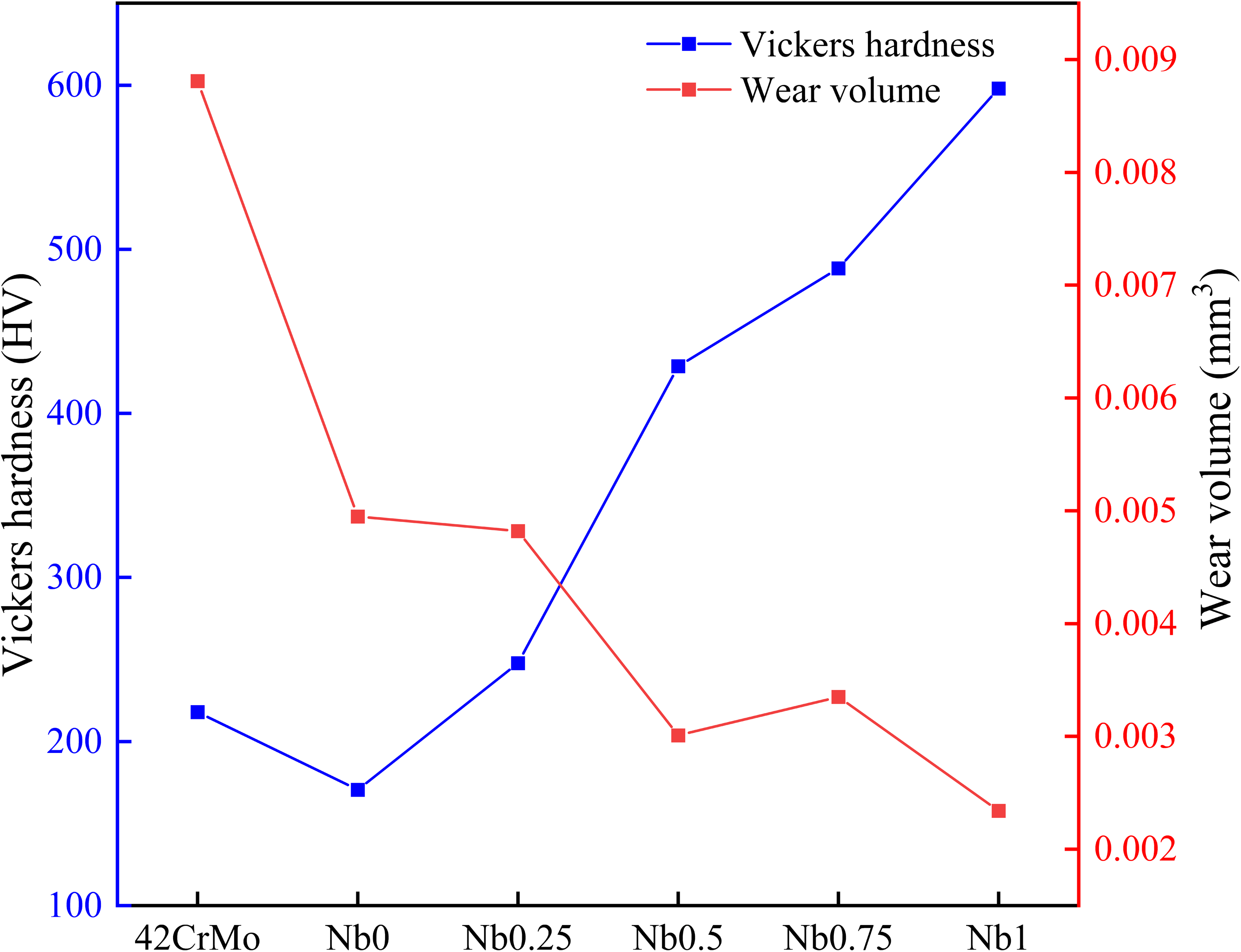
The relationship between microhardness and wear volume.
Wear volume of samples in a dry sliding wear condition.
Corrosion resistance
Using Zsimpwin software to simulate the equivalent circuit can help to understand the actual reaction of metal interface. Figure 9 provides the EIS results in 3.5 wt% NaCl solution as Nyquist plots soaked two hours in advance, the corresponding electrical equivalent circuit (EEC) used to fit the data is also provided. In the circuit simulation process, the capacitive reactance is represented by the constant phase elements (CPE). Due to the dispersion effect, the characteristic value of the CPE element will have a phase angle shift in a wide frequency range, so its characteristics are different from ordinary capacitive elements. The impedance value of CPE is defined by the following formula13,14: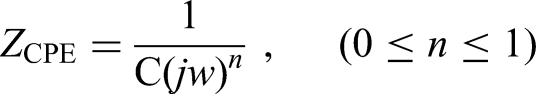
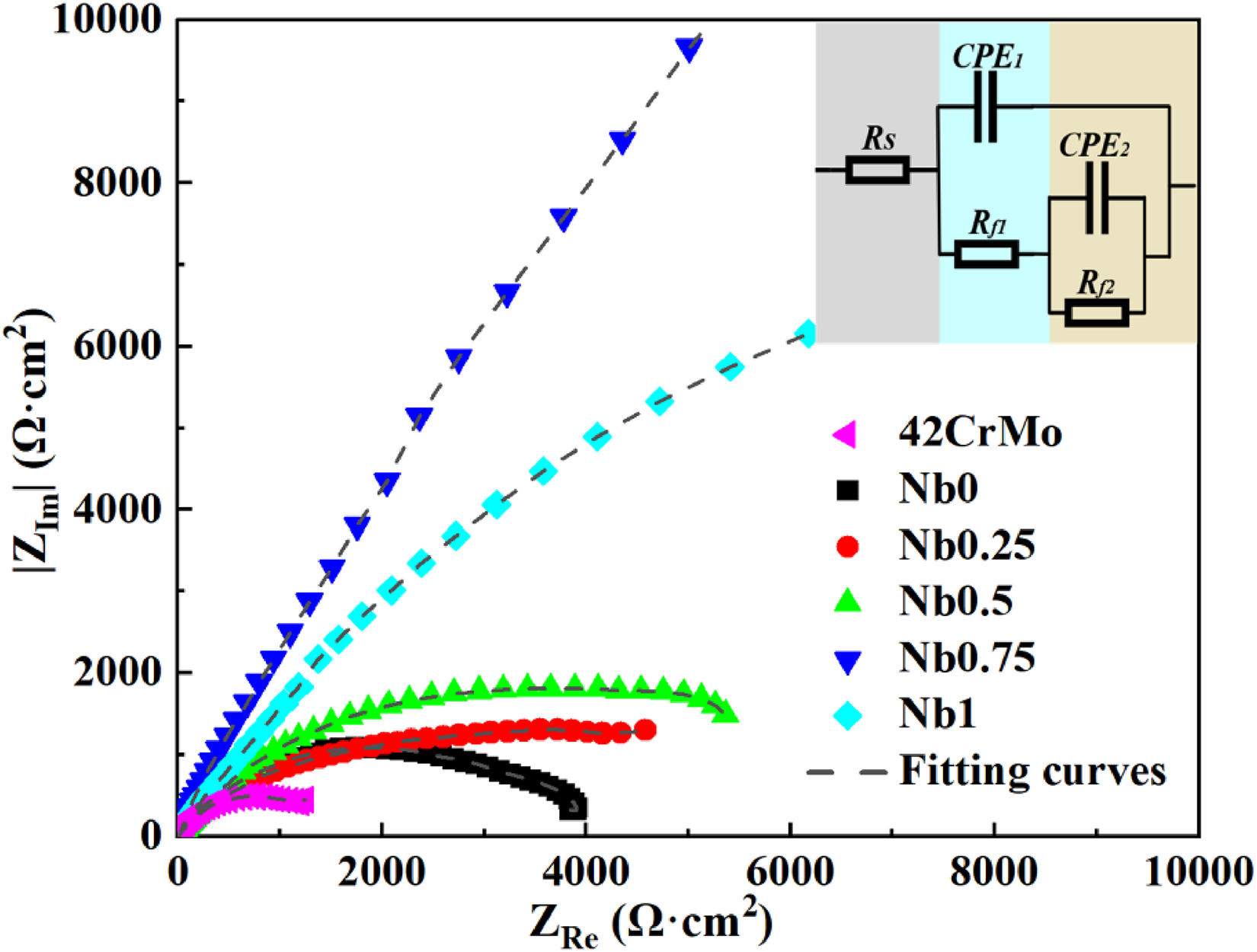
EIS tests as Nyquist plots and equivalent circuit of samples.
If the adsorption of solution molecules or the adsorption of some special atoms has a certain impact on the arrangement of atoms at the reaction interface, there will be a deviation between the CPE element and the ideal capacitance, and the value of
The polarisation curves of CrFeCoNiNb
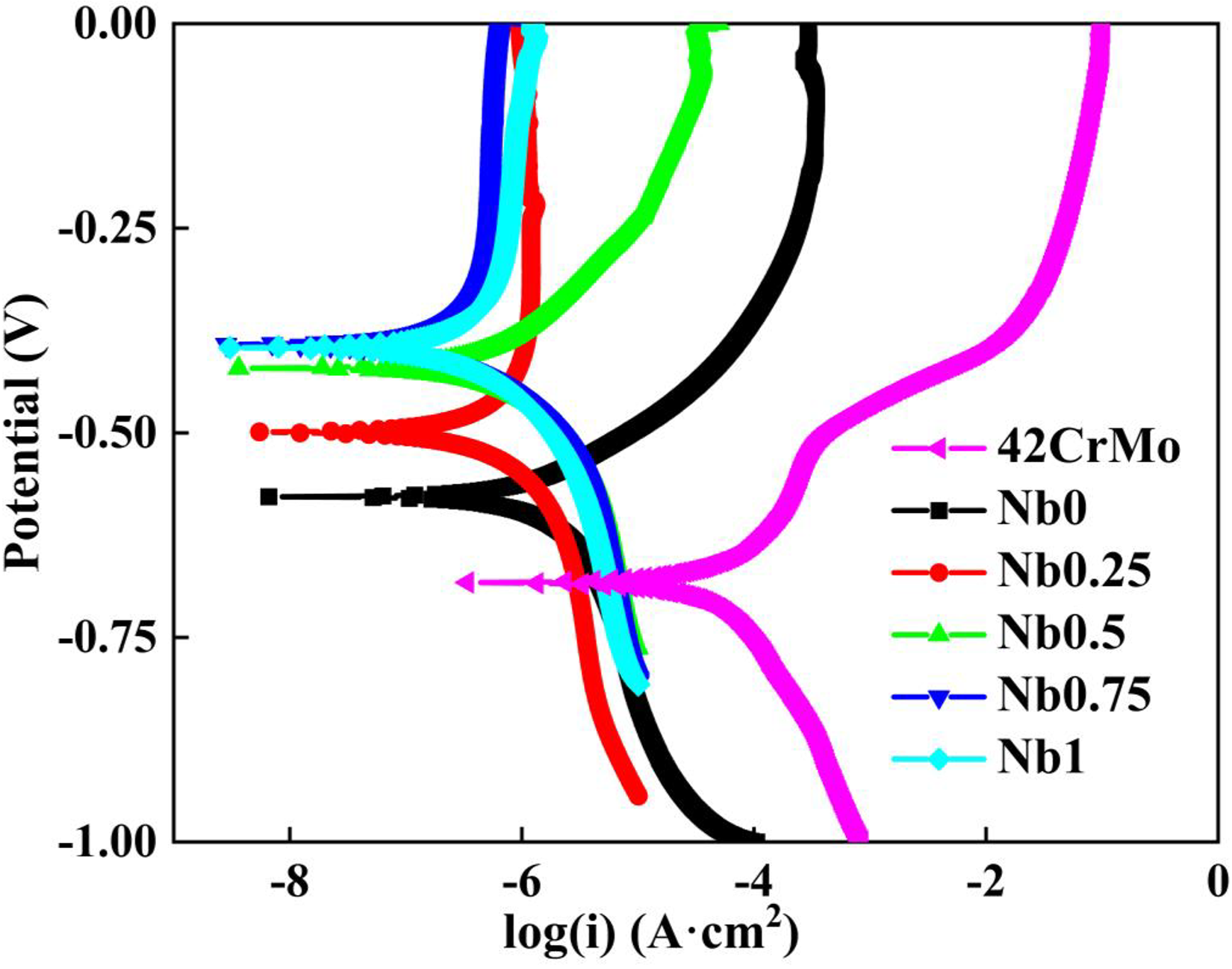
Polarisation curves of samples in 3.5 wt% NaCl solution.
Polarisation data of the alloys in 3.5 wt% NaCl solution.
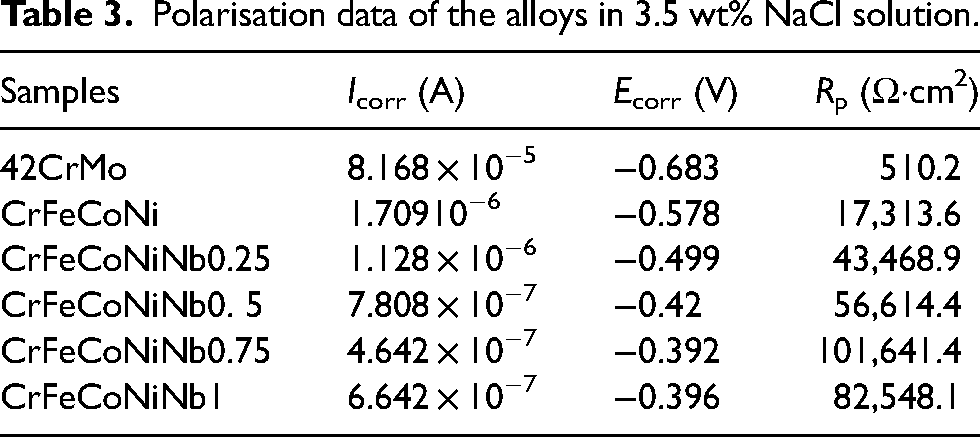
Figure 11 gives the morphologies of the samples after polarisation tests in 3.5 wt% solution. According to the EDS analysis, it is found that the substrate is extremely corroded and a large amount of agglomerated rust is formed. Local corrosion pits, corrosion holes and granular corrosion products appear on the surface of the cladding layer, which also shows that the cladding layer is relatively more resistant to corrosion. Generally speaking, the initiation process of pitting corrosion comes from intergranular corrosion and completes with pits formed by exfoliation of grains. When
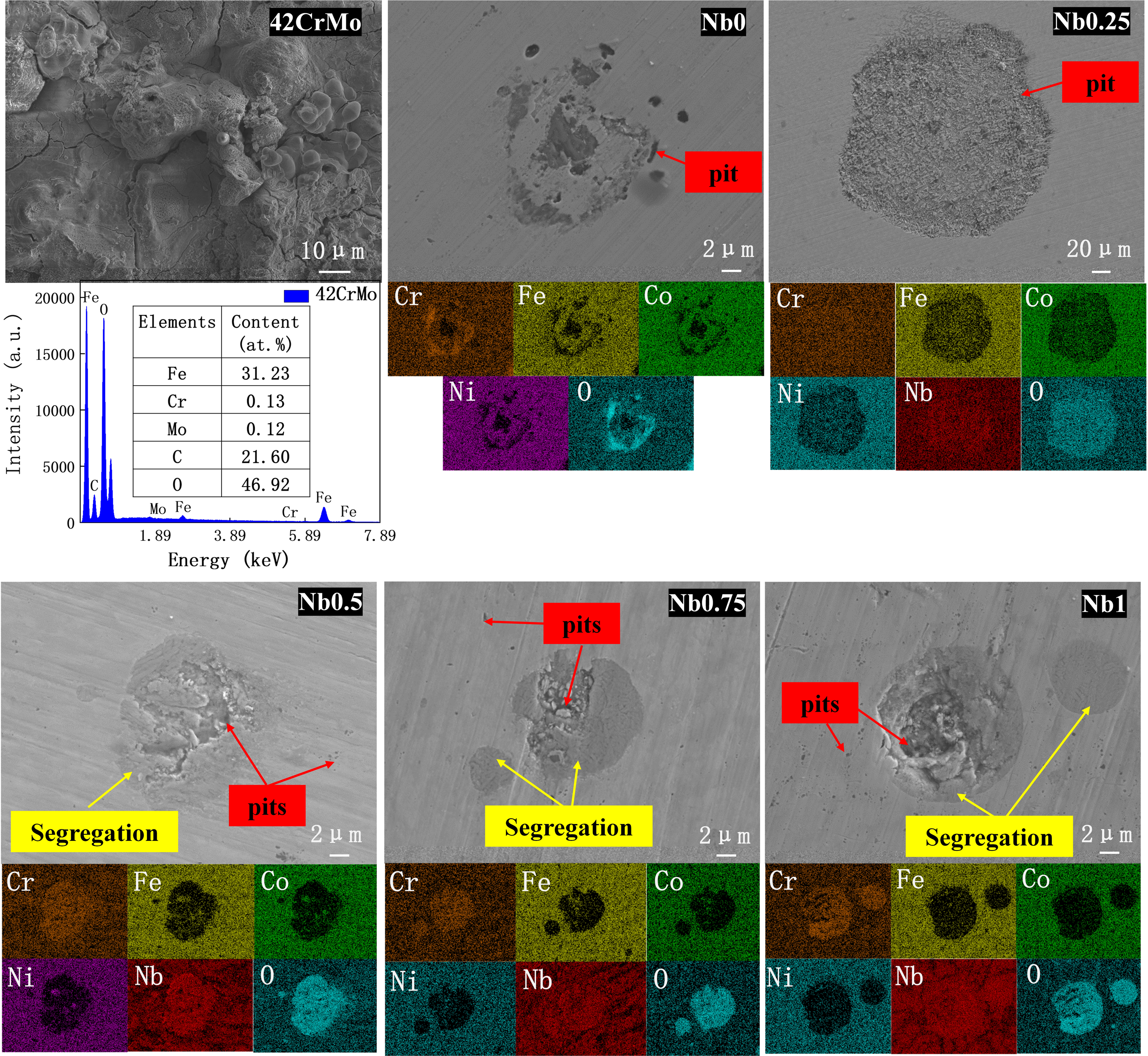
Corrosion morphology after polarisation of samples in 3.5 wt% NaCl solution.
Conclusions
Laser cladding technology and HEAs are combined to extend the service of stern shaft at lower cost. The following are the results of research on the properties of the CrFeCoNiNb The addition of Nb promoted the transformation of the grain structure of the CrFeCoNiNb The microhardness of the CrFeCoNiNb Wear resistance is generally proportional to microhardness and increases with the addition of Nb. It is noteworthy that when Nb content is low, the coating has a hardness similar to that of the substrate. However, due to the presence of an oxide film, wear resistance is improved. CrFeCoNiNb The CrFeCoNiNb
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation (ZR2022ME081, ZR2021ME198), Collaborative Innovation Center for Shandong's Main crop Production Equipment and Mechanization (SDXTZX-21), and also it is Supported by the 111 project (No. D21017).
