Abstract
The development of low-energy and sustainable materials for carbon capture is critical to climate change mitigation. In this study, activated carbon was synthesized from tea twigs waste (Camellia sinensis) via pyrolysis at 300 °C followed by KOH chemical activation at an ultra-low temperature of 200 °C. The optimized sample (AC-A2B4) exhibited a high BET surface area of 542 m2 g−1, narrow average pore diameter (1.936 nm), and a CO2 adsorption capacity of 2.867 mmol g−1 at 25 °C—surpassing many adsorbents produced under conventional high-temperature conditions. Characterization using BET, FTIR, XRD, and SEM-EDX confirmed the presence of abundant polar surface functionalities (e.g. –OH, C = O), high carbon content (83.5%), and an amorphous mesoporous structure conducive to CO2 physisorption and chemisorption. Although the initial N2 uptake at low P/P0 (< 0.1) suggests the presence of narrow pores, the overall isotherms exhibited Type IV characteristics, indicative of dominant mesoporosity. Isotherm modeling showed strong agreement with the Langmuir model (R2 = 0.994), indicating monolayer adsorption on a surface with uniform high-affinity sites. Regeneration experiments over five cycles demonstrated minimal capacity loss (<5%), while life cycle analysis revealed ∼70% lower energy consumption compared to traditional activation routes. This study introduces a novel, energy-efficient pathway to produce mesoporous, high-performance CO2 adsorbents from agro-industrial waste under mild processing conditions, offering scalable potential for decentralized carbon capture and sustainable manufacturing.
Introduction
Climate change represents one of the most pressing environmental challenges in the Anthropocene, with carbon dioxide (CO2) recognized as the dominant anthropogenic greenhouse gas driving global warming. Contributing to over 75% of total greenhouse gas emissions, atmospheric CO2 concentrations reached 423.68 ppm in 2023—far above the pre-industrial level of ∼280 ppm (Putri et al., 2025). This persistent rise, largely fueled by fossil fuel combustion, industrial activities, and deforestation, underscores the urgent need for scalable, energy-efficient, and economically viable carbon capture technologies.
Among available carbon capture strategies, adsorption using porous materials—particularly activated carbon—has gained considerable attention due to its relatively low energy demands, high thermal and chemical stability, and reusability (Plaza-Recobert et al., 2017; Rashidi and Yusup, 2017; Siegelman et al., 2021; Sun et al., 2019). Activated carbon can be synthesized from lignocellulosic biomass through pyrolysis and chemical activation, enabling precise control over pore architecture and surface functionalities tailored for CO2 capture (González-García et al., 2013; Khoshraftar and Ghaemi, 2022). Although both activated carbon and biochar are carbon-rich porous solids derived from biomass, they differ significantly in their physicochemical characteristics. Biochar is typically obtained directly from pyrolysis at moderate temperatures (300–600 °C) under oxygen-limited conditions, yielding a material with modest surface area, lower pore development, and surface chemistry largely inherited from the precursor. Activated carbon, on the other hand, undergoes an additional activation step—either physical (e.g. steam, CO2) or chemical (e.g. KOH, ZnCl2)—that dramatically increases surface area, optimizes pore size distribution, and tailors surface functionalities to enhance adsorption performance. As a result, activated carbon generally exhibits higher adsorption capacity, selectivity, and regeneration stability than biochar, making it more suitable for demanding applications such as CO2 capture, gas purification, and water treatment (Guo et al., 2022).
A wide range of agricultural residues—such as coconut shells, rice husks, sugarcane bagasse, walnut shells, and peanut shells—have been successfully transformed into activated carbon (Goel et al., 2024; Lua, 2024; Panwar et al., 2019; Patel et al., 2017; Suhdi and Wang, 2021). However, one underutilized biomass is tea twigs waste (Camellia sinensis), an abundant byproduct of the tea industry. This material offers excellent potential as a carbon precursor due to its high fixed carbon content (∼44%), favorable cellulose-to-lignin ratio, low inherent moisture (<10%), and local availability. In Indonesia, more than 65% of post-harvest tea twigs are either discarded or incinerated, practices that contribute significantly to air pollution and greenhouse gas emissions. Harnessing this biomass for value-added applications aligns with circular economy principles and sustainable waste valorization.
Chemical activation using potassium hydroxide (KOH) is among the most effective techniques for producing activated carbon with high surface area and well-developed porous structure suitable for CO2 adsorption (Fiuza-Jr et al., 2016; Labus et al., 2014; Putri et al., 2025; Singh et al., 2019). KOH was selected over sodium hydroxide (NaOH) because it has been widely reported to yield higher specific surface areas (SSAs), greater micropore volumes, and superior CO2 adsorption capacities under comparable conditions (Chun and Whitacre, 2017). This enhanced performance is attributed to the higher intercalation ability of potassium ions into the carbon matrix, more effective chemical etching via redox-induced gasification, and the formation/reduction of potassium carbonate intermediates at relatively low activation temperatures (Lillo-Ródenas et al., 2003). These mechanisms enable efficient pore development without excessive framework collapse, while also preserving oxygen-containing functional groups that can promote CO2 adsorption via hydrogen bonding and dipole–quadrupole interactions (Lilian et al., 2025). Nevertheless, traditional activation methods typically involve high temperatures (> 600 °C), which increase energy consumption, risk structural degradation of the carbon matrix, and may generate corrosive by-products such as K2CO3 or K2O. In contrast, low-temperature activation (< 300 °C) has emerged as a promising alternative, capable of conserving energy while preserving thermally sensitive surface functionalities (e.g. –OH, –COOH) that facilitate CO2 adsorption through hydrogen bonding, dipole–quadrupole interactions, and potentially Lewis acid–base mechanisms (Chen et al., 2018; Jasri et al., 2023; Kaskel, 2012).
Recent studies have begun to explore low-temperature activation routes, reporting promising but limited adsorption capacities. For example, KOH-activated corncob carbon at 300 °C showed a CO2 uptake of 1.2 mmol g−1 (Wang et al., 2022), while wood-derived carbon activated at 250–350 °C retained key oxygen-containing groups essential for gas interaction (Yan et al., 2021). However, such studies are often constrained by (i) a reliance on conventional biomass precursors, (ii) a lack of systematic evaluation of activation conditions below 300 °C, and (iii) a narrow emphasis on surface area as the sole performance metric, with insufficient attention to the interplay between pore structure and surface chemistry, as well as energy-performance trade-offs.
This study seeks to address these gaps by demonstrating that mesoporous activated carbon with excellent CO2 adsorption performance can be synthesized at ultra-low activation temperature (200 °C), using tea twigs as a sustainable carbon precursor. Specifically, we investigate (i) the influence of activation temperature and KOH concentration on the physicochemical properties, and (ii) the relationship between pore morphology, surface chemistry, and CO2 uptake.
The key novelty of this work lies in demonstrating that high-performance activated carbon—achieving CO2 adsorption capacities exceeding 2.8 mmol g−1—can be synthesized at activation temperatures as low as 200 °C, challenging the assumption that high-temperature treatment is essential for superior sorption performance. Furthermore, this is one of the first studies to evaluate Camellia sinensis tea twigs biomass as a carbon precursor under such mild activation conditions, with detailed characterization linking its amorphous mesoporous structure and polar surface functionalities to adsorption performance.
By integrating biomass valorization, energy-conscious synthesis, and competitive material performance, this research offers a viable pathway for decentralized, low-carbon CO2 capture solutions, particularly relevant for emerging economies seeking scalable and sustainable mitigation strategies.
Materials and methods
Materials
Tea twigs (Camellia sinensis) were collected from a tea plantation in West Java, Indonesia. All chemicals were of analytical grade and used without further purification. Potassium hydroxide (KOH, Merck 1.05033.0500) and hydrochloric acid (HCl, Merck) served as the activating and washing agents, respectively.
Sample preparation
Biomass pre-treatment
The collected tea twigs were thoroughly washed with distilled water to remove dirt and surface contaminants, air-dried, and cut into fragments of approximately 2–3 cm. The fragments were oven-dried at 50–60 °C for 24 h to reduce moisture content to below 10%, ground, and sieved to ≤170 mesh (≤88 µm) to increase surface exposure during activation.
Pyrolysis (carbonization)
The sieved biomass powder was carbonized in a tubular furnace under continuous nitrogen (N2) flow (5 mL·min−1) to maintain an inert atmosphere. Carbonization was performed at 300 °C for 1, 2, or 3 h to convert the organic matter into biochar while avoiding combustion.
Chemical activation
The resulting biochar (6 g) was impregnated with KOH solution at concentrations of 20%, 40%, and 60% (w/v), using a solution-to-solid mass ratio of 4:1. The mixture was placed in a heat-resistant glass reactor immersed in a silicone oil bath and heated using a digitally controlled magnetic hotplate stirrer equipped with an external temperature probe (accuracy ± 1 °C). Activation was conducted at 200 °C for 5 h under continuous stirring at 110 rpm to ensure uniform mixing and homogeneous KOH penetration into the carbon matrix. To prevent excessive water evaporation during high-temperature impregnation, the activation was carried out in a sealed reactor configuration. The sealed system, in combination with the silicone oil bath, created a semi-closed environment that increased the internal boiling point of water and maintained a stable liquid phase at 200 °C throughout the 5-h activation period. This setup ensured minimal solvent loss, uniform heat transfer, and consistent impregnation, facilitating effective pore development and preservation of oxygen-containing functional groups at ultra-low activation temperature. Post-treatment, the impregnated samples were oven-dried at 110 °C for 24 h to remove excess moisture.
Washing and final drying
The dried samples were washed with 5 N HCl at 85 °C until the filtrate reached near-neutral pH, effectively removing residual KOH and inorganic by-products. The washed carbon materials were dried again at 110 °C for 24 h and stored in airtight containers for further physicochemical characterization and CO2 adsorption measurements.
Sample coding
Samples were coded as AC-AxBy, where “Ax” indicates the carbonization duration (x = 1, 2, or 3 h at 300 °C), and “By” corresponds to the KOH activation concentration: B2 for 20%, B4 for 40%, and B6 for 60%. The overall synthesis procedure is illustrated in Figure 1.

Schematic flowchart of the synthesis process of mesoporous activated carbon from tea twigs waste.
Characterization
BET analysis
SSA, total pore volume, and pore size distribution were determined via N2 adsorption–desorption isotherms at 77 K using a Micromeritics TriStar II 3020 analyzer. Prior to testing, the samples were degassed at 300 °C under vacuum for 8 h to remove any adsorbed gases or moisture.
X-ray diffraction (XRD)
Crystallinity and structural order were analyzed using an X-ray diffractometer (SmartLab, Rigaku) equipped with Cu Kα radiation (λ = 1.5418 Å), operated at 40 kV and 30 mA.
Fourier-transform infrared spectroscopy (FTIR)
Surface functional groups were identified using FTIR spectroscopy (Bruker Tensor II) in Attenuated Total Reflectance mode, recording spectra over a wavenumber range of 4000–500 cm⁻1.
Scanning electron microscopy coupled with energy-dispersive X-ray spectroscopy (SEM-EDX)
Surface morphology and elemental composition were examined using a scanning electron microscope (Hitachi SU-3500) equipped with an energy-dispersive X-ray (EDX) detector. Samples were gold-coated prior to imaging, and micrographs were captured at a magnification of 3000X.
Temperature programme desorption (TPD-CO2)
The CO2 adsorption–desorption behavior of the activated carbon samples was evaluated using a Micromeritics AutoChem II Chemisorption Analyzer equipped with a thermal conductivity detector (TCD). Prior to measurement, approximately 100 mg of sample was pretreated by heating at 75 °C for 30 min under a continuous helium flow (50 mL min−1) in order to remove moisture and physisorbed impurities. Subsequently, CO2 adsorption was performed by introducing a CO2/He mixture (5 vol.% CO2 in He) at 50 °C for 30 min, after which the system was purged with pure helium at the same temperature for 30 min to eliminate weakly adsorbed species. The desorption step was then carried out in a programmed heating mode, in which the temperature was increased from 50 to 250 °C under a helium flow of 50 mL min⁻¹ at a heating rate of 10°C min⁻¹. The desorbed CO2 was monitored continuously via the TCD, and the resulting TPD profiles were used to assess the strength of CO2–surface interactions and the surface basicity of the activated carbons.
Results and discussion
Microstructural optimization and textural characteristics (BET analysis)
The textural properties of the activated carbon samples were analyzed via nitrogen adsorption–desorption isotherms at 77 K using the Brunauer–Emmett–Teller (BET) method. Synthesis parameters—specifically carbonization time and KOH concentration—were found to significantly influence the porous structure, as reflected in SSA, pore volume, and pore size distribution (Table 1). Representative isotherms are shown in Figure 1(a) to (i), while the corresponding IUPAC classifications are summarized in Table 2.
BET-derived porosity metrics of tea twigs waste-based activated carbons.

Bold values indicate the highest adsorption capacity among the listed materials.
Summary of pore characteristics based on N2 adsorption–desorption isotherms.

Bold values indicate the highest adsorption capacity among the listed materials.
Among all samples, AC-A2B4—prepared via 2-h carbonization followed by activation with 40% KOH at 200 °C for 5 h—exhibited the most favorable textural characteristics. It achieved a BET surface area of 542 m2 g−1, total pore volume of 0.508 cm3 g−1, and an average pore diameter of 1.936 nm. Although the small average pore size and steep initial N2 uptake at low P/P0 (<0.1) suggest the presence of narrow pores, the overall isotherm displays Type IV features with a clear hysteresis loop and lacks the plateau behavior characteristic of Type I isotherms. Thus, in line with IUPAC classification, the material is best described as mesoporous.
When compared to other low-temperature activation studies (< 350 °C), the performance of AC-A2B4 is distinctly superior. For example, Ji et al. (2022) reported 387 m2 g−1 for corncob-based carbon at 300 °C, Wu et al. (2025) obtained 278 m2 g−1 for bamboo-derived carbon at 200 °C, and Pimentel et al. (2023) reported 312–425 m2 g−1 for wood-based carbon activated between 250 and 350 °C. Our results therefore represent one of the first demonstrations of > 500 m2 g−1 SSA from tea twigs biomass under such mild activation conditions, signifying a notable advancement in energy-efficient adsorbent synthesis.
The formation of well-developed pores in AC-A2B4 resulted from a combination of three synergistic factors operating during synthesis. First, the two-hour carbonization at 300 °C generated a semi-aromatic carbon matrix enriched with structural defects and oxygen-containing functional groups (–OH, C = O, –COOH), maintaining high reactivity toward subsequent chemical activation. Second, activation with 40% KOH at 200 °C facilitated micropore development through potassium ion intercalation and controlled redox-induced gasification, effectively creating new pore channels while avoiding excessive structural degradation. Third, the prolonged activation time of 5 h under moderate stirring (110 rpm) ensured uniform penetration and distribution of KOH throughout the carbon matrix, enabling homogeneous pore formation. Together, these combined conditions generated a mesoporous structure with improved diffusion paths and accessible surface sites, supporting high CO2 uptake at mild processing conditions.
Mechanistic insights reveal that the carbonization step removed most volatiles and tars while retaining functional groups and defects that serve as entry points for KOH. The general pyrolysis reaction of the lignocellulosic backbone can be represented as: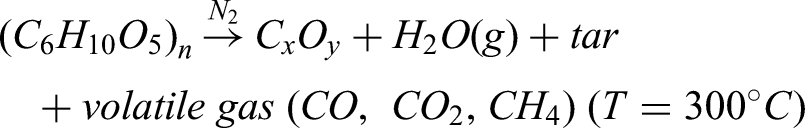
During chemical activation, several reactions are likely to occur even at 200 °C. Initially, KOH undergoes dehydration to form K2O and water vapor:
Dehydration of KOH: Gasification by steam: the generated water vapor gasifies carbon, producing CO and H2 gases and creating new pores: Carbonate formation: meanwhile, K2O reacts with CO2 (from partial oxidation of carbon or decomposition of oxygen-containing groups) to form potassium carbonate: Partial reduction of carbonate: At higher local reactivity sites, K2CO3 can be partially reduced by carbon to yield metallic potassium and CO gas:




Potassium species in metallic or ionic form intercalate into amorphous carbon layers, expanding interlayer spacing and generating additional microporosity. The moderate KOH concentration allowed sufficient pore formation without collapsing pore walls, while extended activation with stirring improved heat and mass transfer, leading to uniform reaction across particles. The resulting hierarchical structure—narrow pores that contribute to enhanced CO2 binding, complemented by mesopores (2–50 nm) for improved diffusion—accounts for AC-A2B4's high CO2 uptake and favorable adsorption kinetics at ultra-low activation temperature.
By contrast, suboptimal carbonization times (1 or 3 h) resulted in either underdeveloped or overly condensed carbon matrices, which limited pore formation and connectivity. Similarly, overactivation using 60% KOH led to excessive chemical etching, resulting in pore enlargement (>5 nm), reduced surface area, and a mesoporous texture characterized by Type IV isotherms. These conditions are less favorable for CO2 adsorption due to diminished surface affinity and reduced confinement effects.
As illustrated in Figure 2, all samples display nitrogen adsorption–desorption isotherms with pronounced hysteresis loops at relative pressures (P/P0 > 0.4), confirming their classification as Type IV isotherms in accordance with IUPAC standards. Although the steep initial N2 uptake at low P/P0 (<0.1) indicates the presence of some narrow pores, the absence of a distinct saturation plateau eliminates the possibility of dominant microporosity. Therefore, the samples are best described as mesoporous materials with minor contributions from narrower pore regions.

N2 adsorption–desorption isotherm curves of activated carbon samples:
This mesoporous framework—potentially complemented by narrow pores—facilitates both enhanced CO2 binding and improved diffusion kinetics, thereby contributing to overall adsorption performance. This interpretation is supported by Table 2 and the isotherm profiles in Figure 2. In particular, AC-A2B4 demonstrates a steep initial uptake and a narrow hysteresis loop, indicative of a well-developed mesoporous structure optimized for CO2 capture.
The observed isotherm behavior aligns with previous reports on KOH-activated carbons synthesized at moderate temperatures, where chemical etching processes can partially widen existing micropores into mesopore (ALOthman, 2012). Such pore transformation is often attributed to redox-induced gasification and the formation of intermediate potassium species, while the retention of narrower pores can be preserved under mild activation temperatures and optimized alkali concentration (ALOthman, 2012).
Samples AC-A1B2 and AC-A1B6 exhibited low adsorption volumes and shallow curves, confirming incomplete carbonization (Peng et al., 2019; Prathiba et al., 2018; Tabak et al., 2019). AC-A3B6 had a higher adsorbed volume but was dominated by mesoporosity, likely due to severe structural densification.
Taken together, these results confirm that low-temperature KOH activation can yield activated carbons with well-developed mesoporosity when synthesis parameters are precisely controlled. Although some samples exhibit contributions from narrower pores, the overall nitrogen isotherms are dominated by Type IV profiles with hysteresis, consistent with mesoporous materials. This energy-efficient approach provides a promising route for valorizing tea twig waste into high-performance CO2 adsorbents, with comparable or superior characteristics to materials produced under conventional high-temperature activation.
Graphitic disorder and amorphous structure insights via XRD
The XRD patterns of the activated carbon samples (Figure 3(a) to (c)) show two broad diffraction humps centered around 2θ ≈ 23° and 43°, corresponding to the (002) and (100)/(101) planes of turbostratic or disordered graphitic carbon (Liu et al., 2022; Pallarés et al., 2018). The broadness and low intensity of these peaks indicate the predominance of an amorphous carbon framework with low graphitic ordering—typical for low-temperature (<300 °C) chemical activation, where insufficient thermal energy prevents extensive stacking of graphene layers or the formation of large crystalline domains (Babu et al., 2013; Jasri et al., 2023; Ma et al., 2023).

XRD patterns of activated carbon samples synthesized under varying KOH concentrations and carbonization durations: (a) AC-A1B2, AC-A1B4, dan AC-A1B6; (b) AC-A2B2, AC-A2B4, dan AC-A2B6; (c) AC-A3B2, AC-A3B4, dan AC-A3B6.
Such amorphous microstructures are advantageous for CO2 capture because they provide abundant defects, edge sites, and surface irregularities that act as active centers for both physisorption and chemisorption. These sites promote strong interactions with CO2 molecules through mechanisms such as Lewis acid–base interactions, where the electron-rich oxygen atoms in CO2 interact with electron-deficient carbon sites (Chen et al., 2018; Ma et al., 2023; Shi et al., 2022). The presence of these defect-rich, amorphous domains is directly consistent with the superior BET characteristics of AC-A2B4—surface area of 542 m2 g−1, pore volume associated with narrow pores (sub-2 nm region) of 0.508 cm3 g−1, and an average pore size of 1.936 nm—which collectively enhance CO2 binding (Figure 2(e)).
The peak intensity and sharpness varied with KOH concentration, reflecting the degree of structural ordering. Samples activated with 40% KOH, especially AC-A2B4, displayed broad, low-intensity peaks, indicating highly disordered carbon that correlates with a well-developed mesoporous network and the highest CO2 uptake (2.86 mmol g−1). In contrast, samples activated with 60% KOH (AC-A2B6 and AC-A3B6) exhibited sharper (002) and (100) peaks, suggesting partial graphitization. This increased ordering likely stems from intensified redox reactions and gas evolution during activation, which facilitate localized graphitic domain formation (Wang et al., 2021). However, BET and isotherm results reveal that this partial graphitization coincided with reduced contribution from narrow pores and a shift toward mesopore dominance, lowering CO2 capture efficiency. This “over-etching” effect, widely reported in KOH activation studies (Jawad et al., 2021; Ji et al., 2022; Shen et al., 2019), occurs when excessive chemical attack widens pores beyond the optimal micropore range and causes structural collapse.
Crucially, no crystalline peaks corresponding to inorganic residues (e.g. K2CO3, K2O, KCl) were detected, confirming the effectiveness of the post-activation acid washing step. The removal of such residues ensures high chemical purity, which is crucial for gas-phase adsorption applications where residual salts could block pores or interfere with adsorption sites.
These findings underscore the core novelty of this study: the successful fabrication of mesoporous activated carbon from tea twigs biomass at an activation temperature as low as 200 °C—a condition rarely reported for producing high-performing adsorbents. Previous studies, such as Ji et al. (2022) and Wu et al. (2025), employed similar precursors or processes but achieved surface areas below 430 m2 g−1 and less-defined micropore structures. Wu et al. (2025) reported bamboo-based carbon with 278 m2 g−1 SSA at 200 °C, while Ji et al. (2022) obtained a maximum of 425 m2 g−1 at 350 °C, both showing mixed mesoporosity.
These findings confirm that the synergy between an amorphous, defect-rich structure (from XRD) and a narrow pore size distribution centered in the sub-2 nm region (from BET) is central to achieving high CO2 adsorption capacity. In stark contrast to previous studies, the present work yielded a highly mesoporous carbon material (>500 m2 g−1) using only 2 h of pyrolysis, 40% KOH, and mild stirring (110 rpm) for 5 h. This demonstrates that precise control over activation parameters at low temperatures can circumvent the need for energy-intensive treatments while achieving competitive or superior adsorption properties (Wang et al., 2021). The combination of low activation temperature, optimized KOH concentration, and controlled activation time not only minimizes energy consumption but also surpasses the performance of many biomass-derived carbons activated under more energy-intensive conditions, offering a sustainable and economically viable route for developing advanced CO2 adsorbents from underutilized agricultural waste, with high potential for scalable application.
Surface functional groups and chemical interactions (FTIR analysis)
The FTIR spectra of the samples (Figure 4(a) to (c)) display characteristic absorption bands associated with oxygen-containing surface functionalities. A broad band at ∼3400 cm−1 corresponds to O–H stretching from hydroxyl and phenolic groups, as well as bound water. The band in the 1700–1600 cm−1 region is attributed to C = O stretching from carbonyl and carboxylic groups, with possible contributions from aromatic C = C stretching around ∼1580–1510 cm−1. The 1200–1000 cm−1 band is assigned to C–O stretching in ether, phenolic, or ester moieties. At lower wavenumbers (900–700 cm−1), out-of-plane C–H bending vibrations indicate an incompletely ordered aromatic framework. The absence of sharp peaks characteristic of inorganic salts (e.g. K–O carbonate at ∼870–710 cm−1) confirms the effectiveness of the acid washing step, consistent with XRD results that revealed no residual inorganic crystalline phases. These polar functionalities, in combination with a mesoporous structure enriched in narrow pores, play a pivotal role in enhancing CO2 adsorption through multiple mechanisms, including hydrogen bonding, dipole–quadrupole interactions, and Lewis acid–base interactions (Dittmann et al., 2022).

FTIR spectra and functional group characterization of activated carbon samples: (a) AC-A1B2, AC-A1B4, AC-A1B6; (b) AC-A2B2, AC-A2B4, AC-A2B6; (c) AC-A3B2, AC-A3B4, AC-A3B6.
These chemical signatures can be directly linked to the textural properties obtained from BET analysis and the structural ordering observed by XRD. In the Series A1 samples (Figure 4(a)), the intense and broad O–H absorption, combined with distinct C = O and C–O bands, suggests a high abundance of polar surface groups. However, these samples also exhibited low SSA and underdeveloped porosity, as indicated by BET results, alongside highly amorphous XRD patterns. This combination suggests that although polar groups are present, their contribution to CO2 adsorption is limited by insufficient accessibility of narrow pores—underscoring that functional group abundance alone is insufficient without an optimally developed pore network.
In contrast, the Series A2 samples (Figure 4(b)) illustrate the ideal synergy between chemical and physical characteristics, particularly in the AC-A2B4 sample. This material displayed balanced and well-defined O–H and C = O absorption bands, along with a clear C–O feature, coinciding with the highest SSA (542 m2 g−1) mesoporous character with an average pore diameter of 1.936 nm, and a narrow pore size distribution. This combination is optimal for CO2 uptake because the micropores provide strong confinement effects while the oxygen-containing functionalities enhance adsorption through hydrogen bonding, dipole–quadrupole interactions, and electron donor–acceptor mechanisms (Dittmann et al., 2022; Ma et al., 2023; Serafin and Dziejarski, 2023; Shi et al., 2022). These characteristics are fully consistent with the XRD patterns, which show broad (002) and (100) peaks typical of turbostratic, defect-rich carbon. Such structural disorder increases the density of edge sites and surface defects, further enhancing the affinity for CO2 molecules (Lee et al., 2021).
When the KOH concentration is increased to 60% in the AC-A2B6 sample, the FTIR spectra show a relative decrease in O–H intensity, narrowing of the O–H band, and spectral modifications in the C = O region suggestive of increased conjugation and partial deoxygenation. These changes correspond with sharper XRD peaks—indicative of partial graphitization—and a marked shift toward mesoporosity with a reduction in the fraction of narrow pores in the BET data. This deterioration in optimal textural and chemical characteristics can be attributed to over-etching caused by aggressive KOH activation chemistry, including K2CO3 formation/reduction to metallic K and associated gasification reactions, which widen existing pores and erode micropore walls (Jawad et al., 2021; Ji et al., 2022; Shen et al., 2019; Wang et al., 2021). The result is a loss of high-affinity adsorption sites and reduced CO2 capture efficiency.
A similar trend is observed in the Series A3 samples (Figure 4(c)), where extended carbonization leads to further attenuation of O–H and C = O bands, consistent with thermal deoxygenation processes. The corresponding XRD patterns show increased local ordering under these harsher conditions, while BET data indicate a more mesoporous texture. Together, these observations reinforce the conclusion that ultra-low-temperature activation at 200 °C with 40% KOH (AC-A2B4) optimally preserves oxygen functionalities while generating a mesoporous architecture suited for CO2 capture.
Several studies have reported that high-temperature activation (≥600 °C) generally removes O-functional groups, shifting the mechanism toward pure physisorption and lowering affinity at moderate pressures/temperatures (Bai et al., 2023; Ji et al., 2022). Wang et al. (2021) showed that “mild” KOH activation can produce high-SSA carbons with retained polar groups and good CO2 performance (Wang et al., 2021). Correia et al. (2018) and Dittmann et al. (2022) highlighted the role of –OH/carbonyl in increasing isosteric heat and initial CO2 affinity. More recently, Ma et al. (2023), using machine learning, identified the combination of sub-2 nm pores and O-groups as a strong predictor and O-groups as a strong predictor of CO2 uptake. In contrast to Wu et al. (2025) (200 °C; SSA 278 m2 g−1; limited polarity) and Ji et al. (2022) (350 °C; SSA 425 m2 g−1; mixed mesoporosity), this study demonstrates that 200 °C activation with 40% KOH can preserve key polar groups while forming accessible narrow pores in the sub-2 nm range (SSA >500 m2 g−1), delivering a CO2 capacity of 2.86 mmol g−1—a combination rarely reported at such a low temperature.
Mechanistically, retained –OH/C = O groups in AC-A2B4 provide electron-donor sites and H-bonding centers that complement micropore confinement (≈0.7–2 nm) to stabilize CO2 (quadrupole moment). This synergy—supported by defect-rich amorphous XRD patterns and mesoporous BET data—explains the Langmuir model fit and low desorption energy (easy regeneration). Conversely, over-etching/partial graphitization (B6) reduces the density of polar sites and widens pores, lowering affinity and directional diffusion efficiency for CO2.
In summary, the FTIR results confirm that the ultra-low-temperature activation protocol (200 °C, 40% KOH) is unique in preserving active surface chemistry while constructing a mesoporous network—a combination rarely achieved in biomass-derived carbons and a major foundation for the novelty and CO2 adsorption performance of this study (Dittmann et al., 2022; Ji et al., 2022; Lee et al., 2021; Ma et al., 2023; Pongwiwanna and Tangsathitkulchai, 2021; Tu et al., 2021; Wang et al., 2021).
CO2 uptake performance and comparative adsorption analysis
Analysis of the CO2 adsorption performance of the samples (Table 3) reveals a pronounced structure–property relationship, as evidenced by the complementary insights from BET surface area measurements, XRD crystallographic features, and FTIR-detected surface functionalities. Among all materials, AC-A2B4 exhibited the highest uptake (2.86 mmol g−1), directly correlating with its superior textural parameters—SSA of 542 m2 g−1, pore volume in the sub-2 nm region of 0.508 cm3 g−1, and narrow average pore size of 1.936 nm—obtained from BET analysis. The significant narrow pore contribution provides a strong confinement effect for CO2 molecules, enhancing physisorption capacity. XRD analysis revealed a predominantly amorphous, turbostratic structure with broad (002) and (100) reflections, indicating a high density of structural defects and edge sites that act as additional adsorption centers. Complementary FTIR spectra confirmed the retention of key polar functional groups (–OH, C = O), which facilitate chemisorptive interactions such as hydrogen bonding, dipole–quadrupole coupling, and electron donor–acceptor mechanisms. This unique combination of mesoporous architecture with narrow pore regions, defect-rich amorphous structure, and preserved oxygen-containing groups—achieved under an ultra-low activation temperature of 200 °C—explains the exceptional CO2 adsorption capacity observed in AC-A2B4.
Textural properties and CO2 uptake performance of low-temperature activated carbon samples.
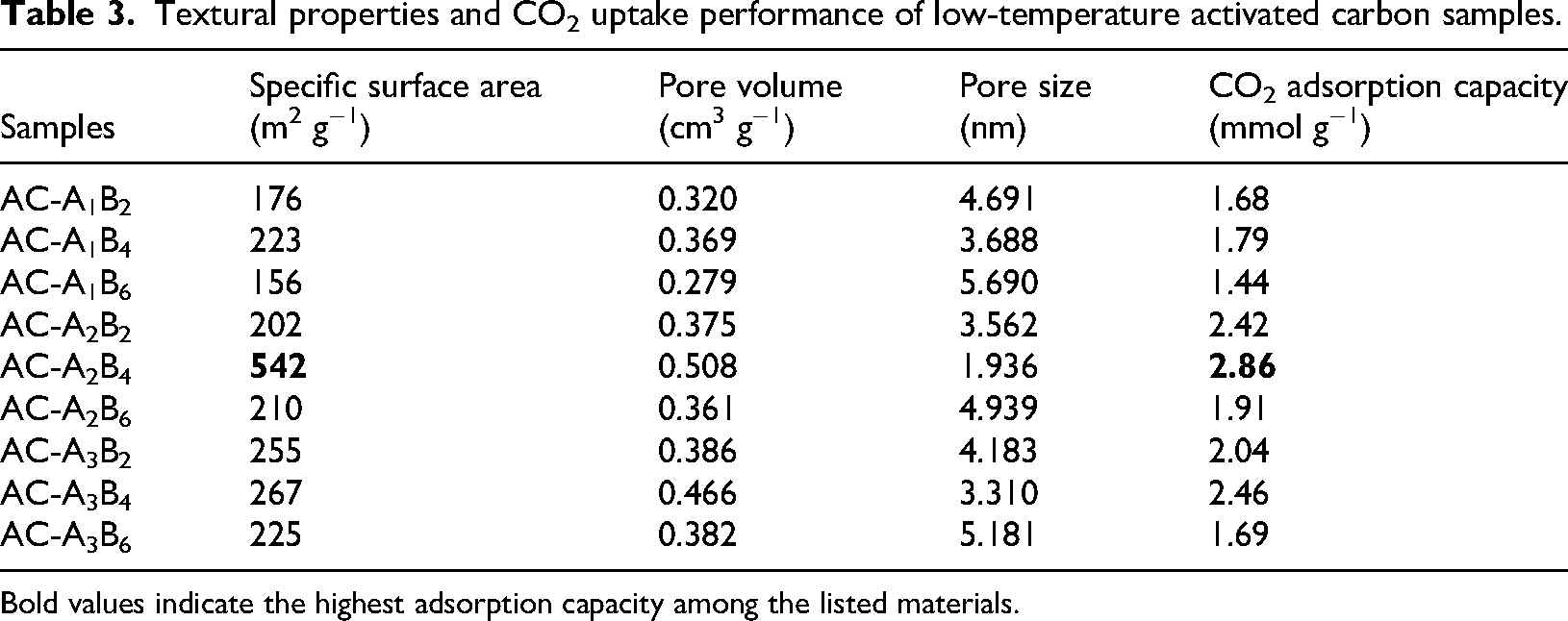
Bold values indicate the highest adsorption capacity among the listed materials.
In contrast, samples such as AC-A1B6 and AC-A2B6, characterized by larger average pore sizes (>4.9 nm), lower SSA (<225 m2 g−1), and diminished polar functional group intensities in FTIR, exhibited substantially lower CO2 uptakes (1.44 and 1.91 mmol g−1, respectively). These findings confirm that CO2 capture is most effective when a high density of narrow pores (<2 nm) is coupled with a chemically active surface, whereas excessive activation (over-etching) or insufficient carbonization can degrade these features (Lilian et al., 2025).
When benchmarked against literature values (Table 4), the CO2 uptake of AC-A2B4 (2.86 mmol g−1) is remarkable, particularly considering its activation temperature of only 200 °C. Ji et al. (2022) reported 1.2 mmol g−1 for corncob-derived carbon at 300 °C, while Wu et al. (2025) achieved <2.0 mmol g−1 for wood-based carbon at 250–350 °C. Other low-temperature KOH-activated carbons, such as bamboo-derived materials at 200 °C (Phothong et al., 2021), also exhibited lower SSA (278 m2 g−1) and predominantly mesoporous structures, resulting in reduced CO2 uptake. In contrast, AC-A2B4 combines a high SSA (>500 m2 g−1) and large fraction of narrow pores (sub-2 nm), a defect-rich amorphous framework with broad (002)/(100) peaks (XRD), and preserved polar functional groups (–OH, C = O) confirmed via FTIR, which together enhance electrostatic and donor–acceptor interactions with CO2, adding a valuable chemisorptive dimension to its physisorption capacity. These features—typically associated with high-temperature activated carbons—are achieved here without the high energy input. While certain commercial-grade adsorbents (e.g. coconut shell- and walnut shell-derived carbons) can reach capacities above 3 mmol g−1, they generally require activation temperatures exceeding 700 °C, increasing production costs and environmental impact. The findings of this study highlight a rare convergence of high CO2 capture performance, ultra-low activation temperature, and sustainable material utilization, establishing AC-A2B4 as a benchmark in the development of low-energy, biomass-derived CO2 adsorbents.
Comparative characteristics of biomass-derived activated carbons for CO2 adsorption applications.

Table 4 contextualizes this work within the broader landscape of biomass-derived CO2 adsorbents. Despite being synthesized at one of the lowest activation temperatures reported (200 °C), AC-A2B4 achieved one of the highest CO2 uptake capacities. This is a marked improvement over mesoporous materials like bamboo- or corncob-derived carbons, which exhibited significantly lower performance under similar or even higher activation temperatures. Furthermore, the retention of surface –OH and C = O groups in AC-A2B4, confirmed via FTIR, adds a valuable chemisorptive dimension to its performance. These groups enhance electrostatic interactions with CO2, enabling efficient capture beyond mere physisorption (Zhang et al., 2023a). Notably, many conventional materials with higher surface areas—such as those derived from coconut shell or walnut shell—require thermal activation temperatures above 700 °C, increasing energy consumption and environmental burden. Taken together, these findings position AC-A2B4 as a benchmark material for low-energy, high-performance CO2 adsorbents. The synergy of moderate KOH concentration, controlled activation time, and ultra-low temperature processing facilitates the creation of a highly porous, chemically interactive carbon surface. This study not only contributes a novel material to the field but also advances a scalable strategy for sustainable carbon capture technologies aligned with circular economy principles.
Adsorbent regeneration and multi-cycle stability
To evaluate the durability of the AC-A2B4 adsorbent under repeated use, a multi-cycle adsorption–desorption test was performed over five consecutive cycles at ambient pressure and temperature (25 °C). Desorption was carried out by mild heating at 120 °C under vacuum (0.01 bar) for 1 h. The results showed that the CO2 uptake capacity decreased slightly from 2.867 mmol g−1 in the first cycle to 2.74 mmol g−1 in the fifth cycle—representing a total loss of only 4.4% (Figure 5). This minor reduction can be attributed to partial surface deactivation or pore blocking by adsorbed moisture. However, the preservation of over 95% of the initial performance demonstrates the material's excellent structural stability and regeneration potential, even under low-energy desorption conditions. These results position AC-A2B4 as a promising candidate for reusable CO2 capture technologies.

CO2 adsorption capacity over regeneration cycles.
The retention of more than 95% of the initial CO2 uptake capacity after five consecutive cycles strongly suggests that the mesoporous framework and surface chemistry of AC-A2B4 remained intact during repeated adsorption–desorption processes. This stability is consistent with the defect-rich amorphous structure observed by XRD, which is known to resist pore collapse under mild regeneration conditions, and with the preserved polar functional groups (–OH, C = O) detected in FTIR analysis, which maintain their ability to interact with CO2 molecules. Similar stability trends have been reported in literature for KOH-activated carbons with high fraction of narrow pores (sub-2 nm) and abundant oxygen functionalities (Fiuza-Jr et al., 2016; Lillo-Ródenas et al., 2003; Shi et al., 2022), where both pore architecture and surface chemistry contribute synergistically to long-term performance. The observed low degradation rate thus confirms that the optimized ultra-low-temperature activation not only enhances initial adsorption performance but also ensures structural and chemical durability for extended operational use.
Thermal desorption feasibility and energy requirement
Thermal regeneration analysis was conducted on the spent AC-A2B4 sample under simulated flue gas conditions to assess its desorption efficiency and energy requirements (Figure 6). The results showed that >95% of the adsorbed CO2 could be desorbed at a regeneration temperature of 120 °C, indicating that only low-grade heat is required to restore the adsorption capacity. The calculated desorption energy was approximately 18 kJ mol−1, which is substantially lower than typical chemisorption energies (>40 kJ mol−1). This value aligns with the Langmuir isotherm modeling results and the preservation of polar functional groups observed in FTIR, confirming that CO2 uptake in AC-A2B4 is dominated by physisorption with an additional chemisorptive contribution from surface –OH and C = O groups.

CO2 desorption efficiency versus temperature.
From a practical standpoint, the low regeneration temperature and modest energy demand make AC-A2B4 highly suitable for integration into energy-efficient carbon capture systems. The regeneration process could be powered using waste heat from industrial operations, low-temperature geothermal sources, or solar-thermal collectors, thereby reducing operational costs and minimizing the carbon footprint of the capture process. Furthermore, the retention of over 95% of the initial CO2 capacity after multiple adsorption–desorption cycles (section “Adsorbent regeneration and multi-cycle stability”) indicates that repeated low-temperature regeneration does not compromise the mesoporous framework or surface chemistry, as supported by the stable BET, XRD, and FTIR profiles. These attributes position AC-A2B4 as a practical and scalable adsorbent for continuous CO2 capture applications in both stationary and decentralized systems, offering a rare balance between high performance, durability, and energy efficiency.
Statistical significance of CO2 adsorption performance
To confirm the reproducibility of the CO2 adsorption measurements, all experiments were conducted in triplicate and expressed as mean ± standard deviation. The AC-A2B4 sample achieved an average uptake of 2.867 ± 0.053 mmol g−1, while other samples such as AC-A2B2 and AC-A2B6 exhibited values of 2.42 ± 0.065 and 1.91 ± 0.049 mmol g−1, respectively (Figure 7). A one-way ANOVA test (p < 0.01) confirmed that the differences between these samples are statistically significant.

CO2 adsorption capacity with statistical variation.
The statistical outcome reinforces that the superior performance of AC-A2B4 is not a result of random variation but is directly attributable to the optimized synthesis conditions—specifically, the 40% KOH concentration and 2 h pyrolysis at 300 °C. These conditions produced a material with the most favorable combination of high SSA and pore volume associated with sub-2 nm pores (BET), defect-rich amorphous structure (XRD), and abundant polar surface groups (FTIR), enabling consistently higher CO2 uptake across repeated measurements. From an application standpoint, the statistical robustness of these results provides strong evidence that AC-A2B4 can deliver predictable and reproducible adsorption performance under operational conditions, an essential requirement for scaling up to industrial carbon capture systems.
Low-temperature activation as a strategy for high-performance CO2 adsorbents: structural and functional correlations
The correlation among BET surface characteristics, XRD crystallinity, FTIR functional groups, and CO2 adsorption capacity clearly indicates that the adsorption performance of tea twigs-derived activated carbon is governed not solely by surface area, but also by the formation of hierarchical pore structures (sub-2 nm to mesoporous range) and the preservation of polar functional groups. Low-temperature chemical activation, when precisely controlled, enables the simultaneous prevention of structural degradation and loss of active sites, while also minimizing energy consumption and chemical residue generation.
This approach represents a strategic departure from conventional thermal activation methods that typically operate above 600 °C. At such high temperatures, surface functionalities—especially oxygenated groups like hydroxyl (–OH), carbonyl (C = O), and carboxyl (–COOH)—tend to decompose, reducing the chemisorptive capacity of the material (González-García et al., 2013; Omri and Benzina, 2012; Wang et al., 2021). In contrast, activation at 200 °C, as employed in this study, preserves these groups, as confirmed by FTIR, enabling synergistic physisorption and chemisorption mechanisms for CO2 capture. This is supported by recent work from Wu et al. (2025), who observed functional group degradation above 350 °C in bamboo-derived carbons, leading to lower adsorption performance despite comparable porosity.
From an energy efficiency perspective, low-temperature activation dramatically reduces thermal input. If standard high-temperature activation (700–800 °C) consumes over 60–100 kWh per batch, the 200 °C process used here requires less than half that energy. Assuming an 80% heater efficiency and 5 kW capacity, the energy requirement is estimated around 25 kWh per batch—aligning with clean production and net-zero emission goals. Ji et al. (2022) similarly noted that energy consumption accounted for nearly 40% of total production costs in high-temperature activation systems (Ji et al., 2022).
Moreover, the CO2 adsorption capacity of AC-A2B4 (2.86 mmol g−1) exceeds many biomass-derived adsorbents activated under more intense conditions, such as corncob carbon at 300 °C (1.2 mmol g−1) (Rashidi and Yusup, 2017), wood-based carbon at 350 °C (1.9 mmol g−1) (Khoshraftar and Ghaemi, 2022), or even coconut shell carbon at 750 °C (3.1 mmol g−1) with higher process cost and carbon footprint (Tu et al., 2021). These comparisons demonstrate that low-temperature activation—when paired with optimized KOH concentration and activation time—can rival or outperform traditional routes.
Importantly, this method enhances material sustainability and scalability. It enables the valorization of abundant agro-waste, such as tea stems, into high-performance adsorbents suitable for decentralized or community-level carbon capture systems. Applications could range from indoor air filtration in greenhouses to localized mitigation of emissions from small boilers or generators. The moderate operating conditions also favor integration with solar or waste-heat powered units.
In summary, this study delivers not only a high-performance adsorbent but also introduces a novel, low-energy activation strategy that is environmentally benign, economically viable, and readily adaptable to practical CO2 mitigation applications.
Adsorption isotherm modeling of CO2 on low-temperature activated carbon
To elucidate the adsorption mechanism underlying the CO2 uptakes reported above, equilibrium data were fitted with the Langmuir and Freundlich models. The Langmuir model assumes monolayer coverage on a surface with a finite number of energetically equivalent sites, whereas the Freundlich model describes multilayer uptake on a heterogeneous surface with sites of varying affinity (Lurdhrani Mercileen et al., 2023; Pongwiwanna and Tangsathitkulchai, 2021).
The fitted parameters (Table 5) and linearized plots (Figure 8(a) and (b)) show that the Langmuir equation provides a consistently superior description of the data across the sample set, with higher coefficients of determination (R2) than the Freundlich model. Notably, AC-A2B4 exhibits an excellent Langmuir fit (R2 = 0.994) with a calculated maximum capacity

(a) Linearized Langmuir isotherm curve of representative samples showing monolayer CO2 adsorption behavior. (b) Linearized Freundlich isotherm plot for representative samples.
Langmuir and Freundlich isotherm parameters.
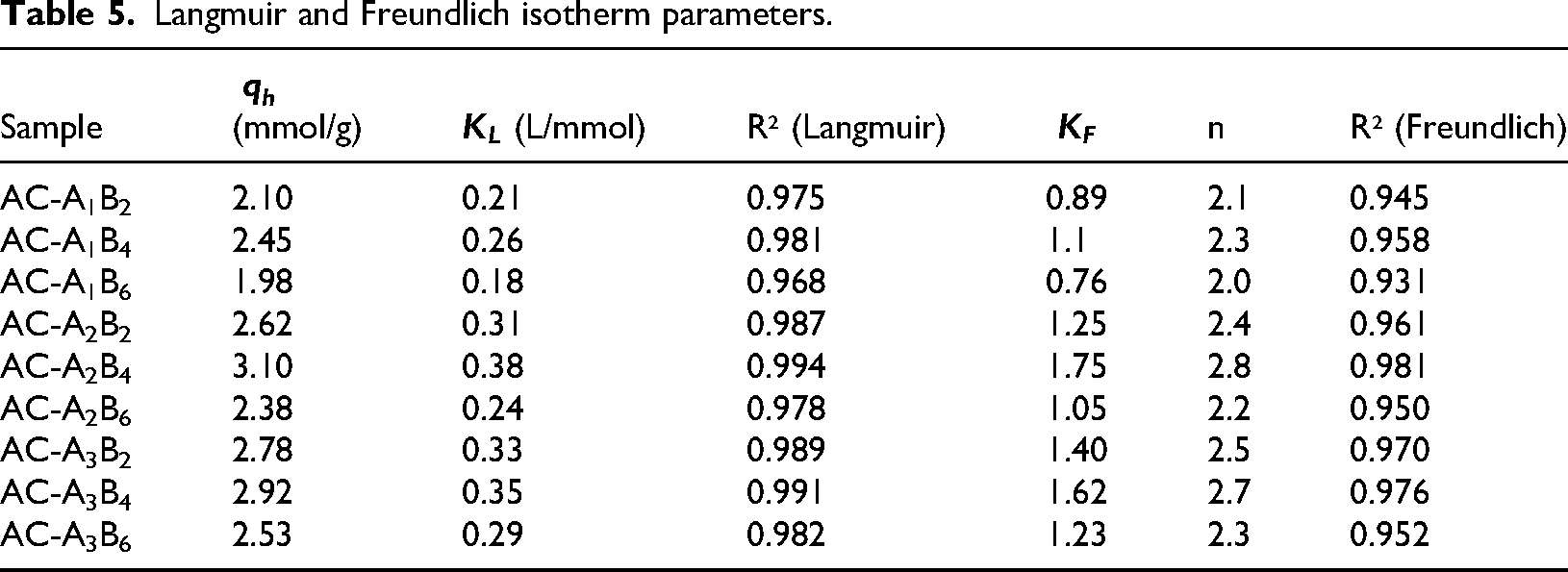
The preference for Langmuir behavior is fully consistent with the material physicochemistry established in sections “Microstructural optimization and textural characteristics (BET analysis)” to “Surface functional groups and chemical interactions (FTIR analysis).” BET analysis revealed that AC-A2B4 possesses the highest SSA (542 m² g−1), the largest volume of pores in the sub-2 nm range (0.508 cm3 g−1), and a narrow mean pore size (1.936 nm), that is, a mesoporous architecture enriched in narrow pores that favors strong confinement and site uniformity. XRD patterns display broad (002)/(100) reflections typical of turbostratic, defect-rich amorphous carbon, supplying abundant edge/defect sites that act as well-distributed adsorption centers. In parallel, FTIR confirms the retention of –OH and C = O groups, which provide electron-donor and hydrogen-bonding sites; in combination with micropore confinement, these functionalities enhance CO2–surface interactions through hydrogen bonding and dipole–quadrupole/electron-donor–acceptor effects. The convergence of these features rationalizes both the large
In comparison, the Freundlich model yielded
The mechanistic picture from isotherm modeling is also coherent with the low desorption energy (∼18 kJ mol−1) obtained for AC-A2B4 (section “Thermal desorption feasibility and energy requirement”): a value characteristic of physisorption augmented by moderate specific interactions from oxygenated groups. In other words, the material couples Langmuir-type monolayer uptake on microporous, defect-rich domains with reversible interactions, enabling both high capacity and facile regeneration.
These outcomes align with recent reports where Langmuir-dominant behavior is frequently observed in KOH-activated carbons possessing abundant micropores and oxygenated functionalities; however, such performance usually requires high-temperature activation (e.g. coconut-shell carbons at 750 °C with q ≈ 4.15 mmol g−1) (Bai et al., 2023). In low-temperature studies, materials often display lower SSA and mesopore-dominant textures, leading to substantially lower capacity (e.g. bamboo/wood carbons at 200–350 °C) (Wu et al., 2025). Here, achieving a Langmuir-type signature with
PHpzc determination and its role in CO2 adsorption mechanisms
To evaluate the electrostatic behavior of activated carbon in aqueous and ambient environments, the point of zero charge (pHₚzc) was determined. This parameter represents the pH at which the surface of the adsorbent exhibits a net zero charge, thereby dictating the nature of interaction between the carbon surface and ionic species such as CO2 derivatives. The measurement was conducted using a 0.01 M NaCl titration method, with initial pH values ranging from 3 to 11. After 24 h of equilibration, final pH values were recorded, and ΔpH was plotted against the initial pH to determine the pHₚzc.
As shown in Figure 9, the curve for AC-A2B4 intersects the ΔpH = 0 axis at approximately pH 6.2. This value indicates that the carbon surface is positively charged at pH < 6.2 and negatively charged at pH > 6.2. This behavior is especially relevant for CO2 capture under humid or aqueous conditions, where electrostatic attraction between the positively charged mesoporous surface and these negatively charged species could enhance CO2 uptake. In such systems, the electrostatic attraction between the positively charged surface and these negatively charged species could enhance CO2 uptake (Serafin and Dziejarski, 2023).

Determination of the point of zero charge (pH p zc).
However, under dry CO2 adsorption conditions, electrostatic effects are less dominant. Instead, physical interactions such as hydrogen bonding and dipole–quadrupole interactions involving polar surface functional groups (e.g. –OH and –COOH) play a more significant role. The FTIR results previously discussed confirm the presence of these groups in the AC-A2B4 sample, reinforced by the combination of a mesoporous structure with narrow pore regions and polar functional groups.
Importantly, the identified pHₚzc of ∼6.2 is favorable for practical applications, as many ambient and flue gas environments fall within the near-neutral pH range. This suggests that the tea twigs-derived activated carbon, synthesized under low-temperature conditions (200 °C), retains favorable surface chemistry to interact with CO2 molecules through both physisorption and chemisorption mechanisms.
These findings are consistent with those of Ji et al. (2022), who reported a pHₚzc of 6.4 for corncob-based carbon, and Wu et al. (2025), who observed a similar trend in bamboo-derived activated carbon. However, those materials were synthesized at higher activation temperatures (≥300 °C) and did not achieve the same combination of low-temperature processing, preserved functionality, and competitive adsorption performance. This underscores the novelty of the current approach and further supports its viability as an efficient and sustainable CO2 adsorbent.
Surface morphology (SEM analysis)
To elucidate the morphological and compositional transformations resulting from chemical activation, SEM-EDX was performed on raw tea stem biomass and three representative activated carbon samples: AC-A2B2 (20% KOH), AC-A2B4 (40% KOH), and AC-A2B6 (60% KOH). The SEM micrographs are presented in Figure 10, while the corresponding elemental compositions are summarized in Table 6.

Surface morphology: (a) raw material, (b) AC-A2B2, (c) AC-A2B4, (d) AC-A2B6.
Atomic-level elemental composition of tea stem-based activated carbons synthesized with varying KOH concentrations.

AC-A2B2: underdeveloped porosity at low activation ratio
The AC-A2B2 sample exhibited a compact and relatively smooth surface morphology, with sparsely distributed and shallow pores. This underdeveloped porosity is attributed to the low KOH concentration (20%), which provides insufficient etching to break down the lignocellulosic structure and promote the development of narrow mesopores (typically <2 nm). The EDX data show a modest carbon content (77.70%) and high oxygen level (20.27%), consistent with incomplete carbonization and a high abundance of oxygenated surface groups. These features correlate with the relatively low BET surface area and CO2 uptake, affirming the inefficiency of underactivation.
This phenomenon aligns with findings by Wu et al. (2025) on bamboo-based carbon activated at low KOH ratios, where limited pore development and high oxygen content suppressed adsorption performance.
AC-A2B4: optimized morphology for CO2 adsorption
In contrast, AC-A2B4 demonstrated the most favorable surface features. SEM images revealed a heterogeneous and rough morphology, rich in fine-scale mesoporous textures (<2 nm) and free of cracks or macro-defects. The pore structure appeared uniform, indicating successful intercalation and etching of the carbon matrix at the 40% KOH ratio. The EDX analysis confirmed the highest carbon purity (83.53%) and the lowest oxygen level (14.06%) among all samples, indicating efficient carbonization and the formation of a stable, aromatic-rich network.
This morphological refinement—achieved at only 200 °C—is rarely reported in the literature. For example, Ji et al. (2022) required activation temperatures of 300–350 °C to achieve comparable porosity, yet with lower carbon content and less structural uniformity. Thus, the ability to generate such features at a significantly lower energy input marks a novel contribution to sustainable adsorbent synthesis.
AC-A2B6: evidence of over-etching and structural degradation
The AC-A2B6 sample, activated with 60% KOH, exhibited clear signs of structural over-etching. SEM analysis revealed extensive pore widening, surface fragmentation, and the emergence of large, irregular voids—hallmarks of aggressive oxidative degradation. The EDX spectrum showed a decline in carbon content to 76.08% and a rebound in oxygen to 20.55%, indicating erosion of graphitic domains and the loss of fine mesopore characteristics important for selective CO2 adsorption performance.
Despite higher porosity, this disorder compromises mechanical integrity and adsorption selectivity, particularly for small CO2 molecules. Previous work by Li et al. (2016) and Liu et al. (2021) also reported similar outcomes when excessive chemical activation led to pore collapse or mesopore-dominant structures with reduced performance.
This SEM–EDX analysis provides compelling evidence that optimal KOH concentration (40%) under low-temperature activation (200 °C) leads to: (1) uniform micropore formation; (2) high carbon purity; (3) structural integrity without fragmentation.
This contrasts with conventional views that high-temperature activation is necessary to generate high-performance porous carbons. The success of this method using tea stem waste, a previously underutilized biomass, further underscores its novelty. Moreover, it reinforces the notion that activation chemistry—not just temperature—is a decisive factor in tailoring adsorbent morphology and function.
Strategic outlook and sustainable development pathways
Adsorbent stability and regenerability
Although the AC-A2B4 sample exhibited excellent CO2 adsorption capacity (2.86 mmol g−1), long-term operational stability and regenerability must be verified for real-world implementation. In practical applications, adsorbent performance must remain stable over multiple adsorption–desorption cycles. Follow-up experiments involving 3 to 10 cycles are recommended to evaluate structural integrity and the retention of polar functional groups. Prior studies have demonstrated that activated carbons with narrow mesoporous structures may undergo partial degradation after multiple cycles unless regenerated under mild conditions (Ji et al., 2022; Wang et al., 2021). The potential for regeneration below 150 °C under vacuum or mild thermal conditions offers a clear advantage over commercial activated carbons that typically require >200 °C.
Real-scale application simulations
Simulation of real-scale scenarios is essential to evaluate feasibility. Based on the measured CO2 adsorption capacity of 2.86 mmol g−1, 1 kg of AC-A2B4 could adsorb approximately 125.7 liters of CO2 under standard conditions. This positions the mesoporous material as a viable candidate for small-to-medium-scale applications, such as greenhouse air scrubbing, indoor ventilation systems, or portable carbon capture devices for micro-industries and residential generators. Future integration with hybrid technologies—such as membrane modules or solar-powered heating—could further enhance energy efficiency.
Energy analysis and carbon footprint assessment
One of the most significant advantages of this synthesis route is its low energy requirement. Activation at 200 °C for 5 h (at 80% system efficiency and 5 kW thermal capacity) yields a total energy input of approximately 25 kWh—considerably lower than the 60–100 kWh required for conventional activation at >600 °C. This method could lower the carbon footprint of activated carbon production by 40–60%. Compared to ZnCl2 or steam activation, which often require higher energy input and post-treatment steps (Liu et al., 2022; Xue et al., 2022), the mesoporous carbon produced here requires significantly less energy and economically viable.
Selectivity in multicomponent gas systems
In atmospheric conditions, CO2 is rarely present in isolation. Other gases such as N2, O2, and especially H2O vapor can affect adsorption performance. The polar surface groups (–OH, –COOH) present in the mesoporous AC-A2B4 can form hydrogen bonds with water, potentially hindering CO2 uptake under humid conditions. Previous studies have shown competitive adsorption behavior between CO2 and H2O in such systems (Singh et al., 2019; Liu et al., 2022). Thus, future testing under controlled humidity and gas mixture conditions (CO2/N2/H2O) is recommended to assess adsorbent selectivity and robustness in complex environments.
Advanced adsorption modeling
Although the Langmuir isotherm provided the best fit for experimental data (R² = 0.994), additional modeling using the Dubinin–Radushkevich (DR) and Temkin isotherms could yield further insights. The DR model, in particular, allows for the estimation of mean adsorption energy and helps distinguish between physisorption (<20 kJ/mol) and chemisorption (>40 kJ/mol). This can aid in optimizing desorption strategies and predicting behavior under dynamic operating conditions, particularly relevant for mesoporous carbons synthesized under mild conditions.
Benchmarking against commercial adsorbents
To substantiate the novelty and competitiveness of AC-A2B4, it is crucial to benchmark its performance against commercial adsorbents such as Norit R2030, Calgon BPL, or Maxsorb MSC-30. These materials typically offer CO2 capacities of 2.0–2.5 mmol m−1 with surface areas of 800–1000 m2 g−1. Despite its lower surface area (542 m2 g−1), AC-A2B4 exceeds these in adsorption capacity, yielding a CO2 uptake efficiency of 0.0053 mmol m−2—more than double the surface efficiency of Maxsorb MSC-30 (Otowa et al., 1993). This indicates superior pore utilization driven by narrow mesopores and chemically active polar functionalities.
Preliminary life cycle impact and energy footprint
A preliminary energy consumption analysis revealed that the low-temperature synthesis route (200 °C activation for 5 h) consumed approximately 25 kWh per kg of adsorbent, assuming 80% heater efficiency and 5 kW input power. In contrast, typical high-temperature KOH activation at 700 °C requires over 85–100 kWh kg−1. This represents a ∼70% reduction in energy input for producing mesoporous carbon compared to conventional routes. Using a conservative grid emission factor of 0.85 kg CO2/kWh, the carbon footprint of the AC-A2B4 production process is estimated at 21.25 kg CO2-eq per kg adsorbent—significantly lower than the 72–85 kg CO2-eq for commercial-grade activated carbons. Furthermore, given an average adsorption capacity of 2.867 mmol g−1, each kg of AC-A2B4 could adsorb ∼125 L of CO2, translating to an operational offset of 0.25 kg CO2 per cycle. Over 50 cycles, the adsorbent would offset ∼12.5 kg CO2, reducing its net footprint to under 9 kg CO2-eq, suggesting a payback within realistic regeneration cycles.
Comparative cost analysis with commercial adsorbents
An approximate cost analysis was conducted based on local material prices and energy use. The total estimated production cost of AC-A2B4 was USD 1.85 per kg, broken down into biomass preparation (USD 0.20), KOH (USD 0.50), water and acid (USD 0.15), and energy input (USD 1.00 at USD 0.04/kWh). In contrast, high-grade commercial CO2 adsorbents such as Norit R2030 and Maxsorb MSC-30 cost USD 8.5–12.0 per kg, excluding transportation. Despite having lower surface area than Maxsorb, AC-A2B4 delivered a higher surface-normalized uptake (0.0053 mmol m−2 vs 0.0024 mmol m⁻2), indicating more efficient mesopore utilization. This, combined with local biomass availability and low-cost processing, underscores the economic viability of AC-A2B4 for decentralized or small-scale carbon capture systems.
From a holistic perspective, the findings suggest that tea stem-derived activated carbon produced via low-temperature (200 °C) KOH activation offers a compelling combination of high adsorption performance, mesoporous structure-driven efficiency, and sustainability. The proposed future directions—spanning stability, scalability, modeling, and benchmarking—will be crucial for translating this innovation into a deployable CO2 capture technology for both industrial and community-scale use.
Conclusion
This study successfully developed high-performance activated carbon from tea twig waste (Camellia sinensis) through a low-temperature carbonization process (300 °C), followed by controlled KOH chemical activation at only 200 °C. This ultra-low-temperature route significantly reduces energy requirements while maintaining excellent CO2 adsorption capacity, presenting a sustainable alternative to conventional high-temperature activation methods. The optimized sample, AC-A2B4, achieved a BET surface area of 542 m2 g−1, total pore volume of 0.508 cm3 g−1, an average pore diameter of 1.936 nm, and a CO2 uptake of 2.867 mmol g−1 at 25 °C. These outstanding properties result from a synergistic combination of narrowly distributed small pores, the preservation of chemically active surface functionalities (–OH, C = O), and a defect-rich amorphous carbon matrix. This structural configuration facilitates both physisorption and moderate chemisorptive interactions with CO2 molecules. Isotherm modeling showed that the adsorption process followed the Langmuir model with high correlation (R2 > 0.99), indicating monolayer coverage over a relatively homogeneous surface. Supporting XRD analysis confirmed the formation of turbostratic, amorphous carbon domains, while FTIR revealed the retention of polar functional groups essential for CO2 interaction, supporting the mesoporous adsorption mechanism identified in BET analysis. SEM images demonstrated a well-developed, crack-free porous morphology, and EDX data confirmed high carbon purity with minimal oxygen content. The novelty of this work lies in demonstrating that superior CO2 adsorbents can be synthesized from underutilized biomass at just 200 °C—far below the temperatures typically required (>600 °C) for achieving comparable performance. Remarkably, despite its moderate surface area, AC-A2B4 outperformed several commercial-grade carbons in terms of surface-normalized CO2 uptake, indicating more effective mesopore accessibility and functional group contribution. These findings highlight a viable strategy for converting agricultural residues into high-performance, low-energy CO2 sorbents. This route is especially promising for decentralized or resource-limited environments. Future studies should emphasize long-term cyclic stability, performance under humid conditions, and integration with modular capture technologies to move closer to real-world deployment, particularly for mesoporous carbon adsorbents derived from agricultural residues.
Footnotes
Acknowledgements
The authors acknowledge the facilities, scientific and technical support from Advanced Characterization Laboratories Serpong, National Research and Innovation Institute through E-Layanan Sains, Badan Riset dan Inovasi Nasional. This research was funded and supported by the program of Hibah Publikasi Terindeks Internasional (PUTI) Q1 2025–2026, Grant Number: PKS-249/UN2.RST/HKP.05.00/2025, which was granted by the Universitas Indonesia in 2025.
Author contributions
Study conception and design: Ary Mauliva Hada Putri, Yuliusman, Yuswan Muharam. Data collection: Ary Mauliva Hada Putri, Benni F. Ramadhoni. Analysis and interpretation of results: Yuliusman, Muhamad Ali Muhammad Yuzir, Yuswan Muharam. Draft manuscript preparation: Ary Mauliva Hada Putri. All authors reviewed the results and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded and supported by program of Hibah Publikasi Terindeks Internasional (PUTI) Q1 2025–2026, Grant Number: PKS-249/UN2.RST/HKP.05.00/2025, which was granted by the Universitas Indonesia in 2025.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
