Abstract
Nitrate pollution originates from wastewater discharge and agricultural and industrial activities and is becoming a serious environmental problem. Current treatment methods often need help achieving high efficiency while maintaining reasonable costs. While nano zero-valent iron (nZVI) is receiving attention as a potential chemical reduction method, challenges such as particle agglomeration, corrosion, and unwanted ammonia formation have limited its widespread application. Support materials that provide a large surface area for mass transfer, such as biochar, zeolite, layered double hydroxides, and mesoporous silica, can be used to enhance the stability, reactivity, and dispersion of nZVI. Thus, such support materials can increase the nitrate removal capacity of nZVI. The effectiveness of carrier-supported bimetallic nanoscale zero-valent iron in nitrate reduction still needs to be demonstrated more clearly to provide valuable insights to enhance nitrate treatment technology and effectively solve water quality problems. Efficient nitrate removal while ensuring high selectivity for nitrogen gas will be achieved through efforts to develop zero-valent iron-based materials to support the development of more sustainable solutions for water treatment.
Introduction
Nitrates discharged into the environment during agricultural practices and industrial processes substantially contaminate surface water and make water systems (Huang et al., 2020; Lee et al., 2007). Therefore, nitrate contamination resulting from sources such as fertilizers, livestock manure, and industrial and domestic wastewater has attracted increasing concern (Liu and Wang, 2019; Tugaoen et al., 2017). Because of its high solubility in water, nitrate nitrogen can contribute to eutrophication and have other detrimental effects on water quality (Shen and Wang, 2011).
Furthermore, nitrate has the potential to cause serious health risks, affecting human physiological function and increasing the likelihood of diseases such as methemoglobinemia, commonly known as “blue baby syndrome,” and carcinogenic, has been linked to colorectal cancer when nitrate levels in drinking water are elevated (Martínez et al., 2017). Therefore, advanced, cost-effective, and environmentally friendly treatment technologies should be developed to remove nitrates from water and wastewater (Humans, 1997).
Conventional methods for removing nitrates from water have low effectiveness levels because nitrates have high solubility and resist precipitation in water. Moreover, materials in water have low adsorption capacity for nitrates because many substances, such as chloride, carbonate, sulfate, and phosphate, compete with nitrates for active adsorption sites (Islam et al., 2010). Scholars have proposed several methods, including biological nitrate reduction, chemical reduction, ion exchange, adsorption, and membrane separation, to overcome these problems; however, these methods also have drawbacks, such as the need for frequent regeneration and the generation of secondary pollutants, which lead to the depletion of resins and adsorbents and the failure of nitrate removal (Vilardi and Di Palma, 2017; Peng et al., 2015; Liu and Wang, 2019; Bijekar et al., 2022). Biological denitrification is often favored because of its cost-effectiveness in wastewater treatment. However, this method requires external organic sources to facilitate the growth of specific microbial communities and provide [H] or exogenous electron donors to reduce nitrates when available organic sources are insufficient. In addition, this method is time-consuming, results in sludge generation, and has high maintenance costs (Cecconet et al., 2018; Tugaoen et al., 2017; Curcio et al., 2022). Chemical denitrification can overcome some of the aforementioned problems because of its simplicity and high reaction rate (Song et al., 2020).
The denitrification process requires various inorganic electron donors for nitrate reduction; this process involves converting nitrates from a high-valence state to ammonia or N2 by using diverse reductants such as H2, formic acid, and (nZVI) (Hamid et al., 2017; Zhu et al., 2024; Zhang et al., 2023a). Of these reductants, nZVI is an environmentally friendly and cost-effective material that has attracted increasing interest in wastewater treatment owing to its core–shell structure (Rodríguez-Rasero et al., 2024; Nyabadza et al., 2024; Fang et al., 2023). Because of its unique and strong reduction ability, nZVI can be used to remove numerous water-soluble pollutants (Maamoun et al., 2021; Zhao et al., 2016; Li et al., 2021; Liu et al., 2024a). Furthermore, nZVI is widely applied in water treatment, including in NO3− removal, owing to its exceptional electron-donating capacity, abundant sources, environmentally benign nature, low cost, and high in situ reactivity (Song et al., 2017; Namakka et al., 2024; Liu et al., 2024b). The reaction of zero-valent iron (ZVI; Fe0) with NO3− results in the oxidation of Fe0 to Fe2+ and the reduction of NO3− to NH3 or N2 gas (Li et al., 2010). The relevant reaction pathways are expressed as follows:
After iron is oxidized, the reduction of nitrate ions by Fe0 is further facilitated by iron oxide (including magnetite) through the following pathway:
To guarantee an in-depth assessment of the literature on enhanced nitrate removal and selective N2 removal by carrier-material-supported bimetallic nanoscale zero-valent iron, the authors conducted a systematic search on science direct.com by keeping the year limit “2013 to 2024,” with keywords of zero-valent iron, nitrate. The results showed that there were about 6807 articles up to October 14, of which 1206 were published in 2024, 1034 were published in 2023, 1020 in 2022, 881 in 2021, and 2666 from 2020 to 2013. Furthermore, out of these 6807 articles, 4304 were research articles, 1558 were review articles, 637 were book chapters, 68 were encyclopedias, 53 were short communications, and the remaining 187 were others.
Through the above investigation, as evidenced by the continuous and strong increase in the number of articles in the last 10 years, it is evident that nitrate treatment based on zero-valent iron is still one of the topics of great interest in the scientific community globally. Some studies on nitrate removal using zero-valent iron are shown in Table 1. Most studies focus on laboratory-scale experiments, with only a few studies simulating real conditions (groundwater), indicating a gap in large-scale or real-world applications. The materials used involve bimetallic systems (e.g., Cu, Pd, Ni) and support materials (e.g., biochar, zeolite, polymers, clay minerals), which enhance nitrate removal efficiency. The reported removal rates are generally high, often exceeding 90%, with variations depending on pH, nitrate concentration, and ZVI dosage. Acidic to neutral pH conditions (3.0–7.16) are commonly investigated, suggesting that ZVI performs well under these conditions. The highest removal rates (close to 100%) are associated with catalytic and composite-supported ZVI materials.
Some studies on nitrate removal using ZVI.

Although ZVI has several advantages, its practical application is nonexistent because of its intrinsic property, which is the susceptibility to agglomeration (Zhou et al., 2022; Di et al., 2023). ZVI particles are attracted to each other, and this results in them agglomerating to form microscopic particles, which reduces their mobility, effective surface area, corrosion resistance, electron selectivity, and reactivity (Guan et al., 2015; Ryu et al., 2011; Yang et al., 2024; Guo et al., 2023; Shi et al., 2015). The identification and control of the end products of NO3– reduction (i.e., NH3–N or N2–N) have attracted substantial research attention (Song et al., 2017; Wang et al., 2021a; Grau-Martínez et al., 2019; Pei et al., 2024). Extensive research has attempted to improve sustainability and stability and enhance nitrogen conversion to overcome the problems associated with ZVI-based denitrification.
A standard method involves adding a second metal (Pd, Ni, Ag, Cu) with an approximately spherical shape that forms chains like Fe nanoparticles, which arise from magnetic and electronic interactions between nanoparticles to nZVI to create additional electron transfer channels with synergistic effects, thereby facilitating the creation of micro–macro electrode systems; such systems can accelerate nZVI corrosion, thus increasing NO3− conversion efficiency (Huang and Zhang, 2005; Liou et al., 2005; Khalil et al., 2016; Dlamini et al., 2023; Venkateshaiah et al., 2022). In addition, support materials that provide a large surface area for mass transfer, such as biochar (BC), zeolite, layered double hydroxides, and mesoporous silica, have been used to enhance the stability, reactivity, and dispersion of nZVI (Kim et al., 2013; Petala et al., 2013; Su et al., 2016; Shi et al., 2013; Zeng et al., 2017; Zhang et al., 2011). In particular, BC is an environmentally friendly material with a high ion-exchange capacity, abundant oxygen-containing functional groups on its micropore surface, a large surface area, and high dispersion ability. BC is often used as a matrix material for nZVI to increase the stability, reduce the agglomeration, and facilitate the practical application of nZVI as well as to reduce metal leachability and bioavailability (Wu et al., 2013; Choppala et al., 2013; Zhao et al., 2024; Wang et al., 2024).
Considering the prevalence of research articles in this field, further investigations are needed to address the limitations of ZVI as well as to increase the orientation of the final product to N2 gas of the denitrification process. Therefore, the authors have attempted to narrow the scope of the research by providing state-of-the-art information on using carrier-material-supported bimetallic nanoscale zero-valent iron to enhance nitrate removal and N2 selectivity. Specifically, the authors focused on selecting and evaluating articles that simultaneously discussed information such as the application of ZVI combined with a second metal at nanoscale and supported on the carrier material for nitrate removal and to enhance the final product orientation to N2. Accordingly, the specific objectives of this review include the following: (1) summarize and classify the synthesis types of BnZVI through liquid reduction; (2) assess the effect of carrier materials; (3) discuss the influencing factors on the selective reduction of nitrate, including pH, reaction time, and the second metal and their ratio when combined with nZVI; and (4) illustrate the potential reduction in performance of materials over many successive reaction cycles without regeneration.
Synthesis of BnZVI through liquid reduction
The widely applied BnZVI production process is shown in Fig. 1, including liquid phase reduction and green synthesis (Zhu et al., 2018; Abdelfatah et al., 2021; Farooqi et al., 2022; Xu et al., 2024). The present review mainly focused on the liquid-phase reduction method, which results in the generation of BnZVI with high chemical stability, uniform particle distribution, high chemical activity, and strong adsorption ability for various pollutants (Deng et al., 2022; Xing et al., 2022; Ding et al., 2024). The addition of a second metal to BnZVI through doping enables the creation of galvanic cells in which iron acts as the anode and the second metal acts as the cathode. The second metal can donate electrons to pollutants, thereby enhancing the reduction potential and overall efficiency of the system in wastewater treatment (Liu et al., 2022b; White et al., 2024; Schrick et al., 2002; Gao et al., 2016; Abolhassani et al., 2021; Gong et al., 2022; Lei et al., 2022; Fu et al., 2014). Metal cation–ZVI nanoparticles can be synthesized using two widely employed approaches: (i) coreduction of catalytic metals and Fe2+/Fe3+ in sodium bromide (NaBH4)/potassium borohydride (KBH4) and (ii) deposition of catalytic metals on preformed Fe0 nanoparticles (Han and Yan, 2014; Yan et al., 2024). Among all metals, Pd, Ni, and Cu-doped nZVI exhibit the highest removal efficiency (Liu et al., 2022b).

Universal synthesis method for BnZVI (Ding et al., 2024).
In the coreduction approach, iron and additive metal precursors undergo reduction simultaneously in a single reaction pot. This process results in the generation of coreduced particles with potentially different intraparticle distributions and catalyst dispersions to those of particles prepared through the solution deposition approach.
Several studies have highlighted the advantages of various support materials in enhancing the performance of bimetallic systems. For example, Cai et al. (2014) examined the effect of kaolin as a support material on the final properties of nickel-containing Fe-based nanoparticles. Moreover, Li et al. (2017) employed sugarcane bagasse and steel cleaning waste as support materials to improve the properties of nanoparticles through a sustainable approach. The findings of these studies indicate that support materials enhance the stability and reactivity of nanoparticles.
Among various surface-modified metals, Cu is a mild yet effective catalyst; therefore, studies have employed various support materials and synthesis methods to create Fe–Cu bimetallic systems with improved properties. Zeng et al. (2017) used NaY zeolite as support and emphasized the importance of controlling the Cu/Fe ratio appropriately for achieving optimal catalytic performance. Shubair et al. (2018) introduced CuCl₂ into the synthesis process to adjust the Cu/Fe ratio. Tang et al. (2019) precisely adjusted the Cu/Fe ratio and used D407 as the substrate to fine-tune the properties of Fe–Cu nanoparticles. To develop tunable synthesis processes and expand the exploration of support materials, Han et al. (2021) used BC as a support material for the Fe–Cu bimetallic system; they varied the pyrolysis temperature from 500 to 900 °C (500, 700, and 900 °C). The aforementioned versatility in synthesis techniques indicates that the Fe–Cu system has broad potential applications in nitrate removal. Fe2+ and Cu2+ are reduced to nanoscale zero-valent metal through the following reaction (Hwang et al., 2011; Belay et al., 2015):
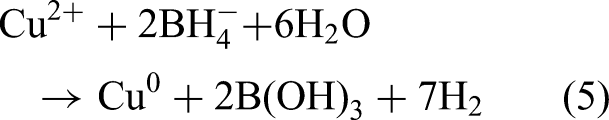
In the deposition approach, NaBH₄/KBH₄ is used to reduce Fe²⁺/Fe³⁺ solution to synthesize monometallic Fe nanoparticles. This process is conducted under the same settings as those used in the coreduction approach. After Fe nanoparticles form, they are added to a solution containing metal cations (such as Cu²⁺ and Ni²⁺), which results in the formation of bimetallic nanoparticles through the deposition of cationic metals on the Fe nanoparticle surface (Ding et al., 2024). The reactions involved in the deposition method are expressed as follows:

The deposition of the secondary metal onto the Fe core is precisely controlled, which improves the stability and reactivity of the resulting bimetallic nanoparticles. Moreover, the nanoparticles generated through the deposition approach have a more uniform coating and superior properties compared with those generated through the coreduction approach. Various studies have provided valuable insights regarding the synthesis of bimetallic nanoparticles through the deposition approach. For example, Choi et al. (2009) used a ZVI-containing acidic palladium solution as the base material to prepare Pd–Fe bimetallic nanoparticles. They observed that the mass fraction and specific surface area of palladium were critical factors determining the efficiency of the produced bimetallic nanoparticles. Moreover, they affirmed the importance of characterization in optimizing the synthesis process. Singh et al. (2012) extended the study of Choi et al. by synthesizing bimetallic ZVI nanoparticles. They reduced Fe2+ solution by using NaBH₄ and then added Ag₂SO₄ solution to the reduced Fe2+ solution to produce Ag–Fe nanoparticles. Additionally, they employed ethylenediaminetetraacetic acid as a catalyst to enhance nanoparticle stability, prevent agglomeration, and protect the specific surface area required for effective pollutant removal.
Su et al. (2014) doped nZVI with Cu, Ag, and Au to enhance the reactivity of nZVI for nitrate removal. They reduced FeCl₃ by using NaBH₄ to obtain nZVI and then added metal precursors to produce doped nanoparticles. These nanoparticles were sonicated, washed, and used in batch tests to evaluate their reactivity with nitrates. Similarly, Lubphoo et al. (2015) successfully synthesized nZVI through the aqueous-phase reduction of ferric chloride by using sodium borohydride. Bimetallic nanoparticles (Cu–nFe0 and Pd–nFe0) were prepared by mixing catalyst precursors with the synthesized nFe0 particles. Finally, trimetallic nanoparticles ((Cu–Pd)–nFe0 and (Pd–Cu)–nFe0)) were produced through the sequential addition of precursors. The redox reactions occurring between Cu²⁺, Pd²⁺, and nFe0 are expressed as follows (Suzuki et al., 2012):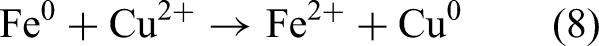

Bae et al. (2016) prepared monometallic and bimetallic nZVI catalysts through sodium borohydride reduction. nZVI was synthesized, and precursor Cu and Ni solutions were then added to create monometallic nZVI catalysts with a 0.5 wt.% loading of each metal.
Guo et al. (2018) employed a liquid-phase chemical reduction method to fabricate two types of bimetallic Fe–Cu particles: ordinary bimetallic Fe–Cu particles and nanoscale bimetallic Fe–Cu particles. To prepare the ordinary particles, iron powder was added to a copper sulfate solution, which was stirred under a nitrogen atmosphere and then filtered. The filtered solution was stored in deoxygenated deionized water. To prepare the nanoparticles, iron sulfate was mixed with an ethanol–water solution containing polyethylene glycol (PEG-4000) and then reduced using a KBH4 solution. Subsequently, a 100-mL solution of CuSO4·5H2O was gradually added to the nanoscale iron suspension and stirred under a nitrogen atmosphere.
In a recent study by Zhang et al. (2024), after the pristine nZVI particles were successfully prepared by solution NaBH4, different amounts of CuSO4·5H2O or PdCl2 were added into the nZVI suspension. Then, the mixtures were continuously stirred for 2 h to ensure that Cu2+ or Pd2+ could be completely reduced and doped on the nZVI surface. Finally, the resulting products were filtered, washed, and freeze-dried for 24 h to obtain Cu–nZVI and Pd–nZVI nanoparticles.
The coreduction and deposition approaches have unique advantages and disadvantages. Specifically, the coreduction approach enables the simultaneous reduction of multiple metals, thereby facilitating homogeneous dispersion and the control of synthesis parameters; however, it exhibits drawbacks related to scalability and nonuniform particle distribution. By contrast, the deposition approach enables precise control over the metal loading process and the sequential addition of metals, which enables the creation of catalysts with tailored compositions; however, this approach involves more steps than the coreduction approach, which may increase material costs.
Novel BnZVI nanoparticles supported by support matrices
Although depositing noble metals onto nZVI surfaces can enhance nitrate reduction, N₂ yields often remain below 10% (Young and Lee, 2005; Zeng et al., 2017). Various support matrices, including rectorite, bentonite, palygorskite, silica materials, zeolite, porous carbon materials, resin, and exfoliated graphite, have been proposed to mitigate the aggregation of nZVI during its synthesis, to increase its dispersity (thereby enhancing nitrate removal), and to ensure that it has high selectivity toward N2 (Hou et al., 2021; Wang et al., 2021b). A support matrix decreases the aggregation of iron particles, which is crucial because aggregation can reduce their reactivity. Such a matrix also facilitates the concentration of reactants at one location, thus aiding electron transfer reactions and promoting product formation. The aforementioned phenomena lead to improved mass transfer.
The chelating resin
Shi et al. (2016) used the chelating resin DOWEX M4195 (DOW 3N) to synthesize bimetallic Fe–Pd nanoparticles by loading Fe and Pd through two methods. To prepare DOW 3N-Fe/Pd, the aforementioned resin was impregnated with Fe3+ solution, then immersed in Pd2+ solution, and subsequently reduced using NaBH4. In addition, DOW 3N-Fe/Pd-R was prepared by reversing the loading sequence of Pd and Fe. Transmission electron microscopy (TEM), energy-dispersive X-ray spectroscopy (EDS), and X-ray diffraction (XRD) analyses were performed to evaluate the morphological differences, size differences, and nitrate removal performance of the supported bimetallic nanoparticles. The TEM results indicated that for both DOW 3N-Fe/Pd and DOW 3N-Fe/Pd-R, Fe/Pd nanoparticles with sizes predominantly between 3 and 5 nm were well dispersed on the surface of DOW 3N (Fig. 2). Moreover, EDS mapping (Fig. 3) indicated that Fe and Pd had a uniform distribution on the DOW 3N surface, which signified that the support matrix provided an optimal environment for the dense and uniform deposition of these metals. For both DOW 3N-Fe/Pd and DOW 3N-Fe/Pd-R, the supported Fe–Pd bimetallic nanoparticles exhibited high nitrate removal efficiency (>90%), demonstrating the crucial role of DOW 3N in minimizing nanoparticle aggregation and promoting uniform particle distribution. The proximity of Pd and Fe on the DOW 3N-Fe/Pd surface potentially reduced the density of low-coordination sites (e.g., edges and corners), which are favorable for NH3 formation, thereby leading to an increase in N2 selectivity owing to the presence of more terrace Pd sites with high coordination.

Transmission electron microscopy (TEM) images and size distribution of the supported nanoparticles prepared by Shi et al. (2016): (a) DOW 3N–Fe/Pd–R and (b) DOW 3N–Fe/Pd.

Energy-dispersive X-ray spectroscopy (EDS) spectra of DOW 3N–Fe/Pd (a,b) and DOW 3N–Fe/Pd–R (c,d) (Shi et al., 2016).
Tang et al. (2019) synthesized a novel composite material, namely Fe–Cu/D407, by depositing nZVI and copper onto the D407 chelating resin to remove nitrate from water. First, CuSO4·5H2O was dissolved in deionized water, and D407 resin was then added to the solution. Subsequently, the solution was mixed with FeSO4·7H2O, with the Cu/Fe ratio being adjusted according to the initial concentration. Finally, the resulting product was reduced using KBH4 under a nitrogen atmosphere. The relevant reaction equations are given as follows:


In this study, Fe–Cu/D407 maintained a high nitrate removal efficiency of at least 90.0%, even in alkaline conditions. By contrast, nZVI requires a low solution pH to remove nitrate effectively. The results obtained for Fe–Cu–D407 are attributable to D407, which provides many acidic functional groups that alter the buffer capacity of the solution and mitigate the negative effect of increasing pH on nitrate removal (Mak et al., 2009; Nie et al., 2018). The Fourier transform infrared spectrum of D407 contained bands at 3400, 1726, 1630, and 1394 cm−1, all of which were noted to correspond to iminodiacetic acid groups (Fig. 4). Moreover, D407 minimized nanoparticle aggregation, promoting a uniform distribution of Fe and Cu particles, which led to closer contact between Fe and Cu particles on the surface of Fe–Cu/D407. Consequently, Cu active sites, which contributed to N2 formation, exhibited enhanced exposure. In general, D407 created an optimal environment for the selective reduction of nitrates to nitrogen, thus ensuring high nitrogen removal performance even at high pH values.
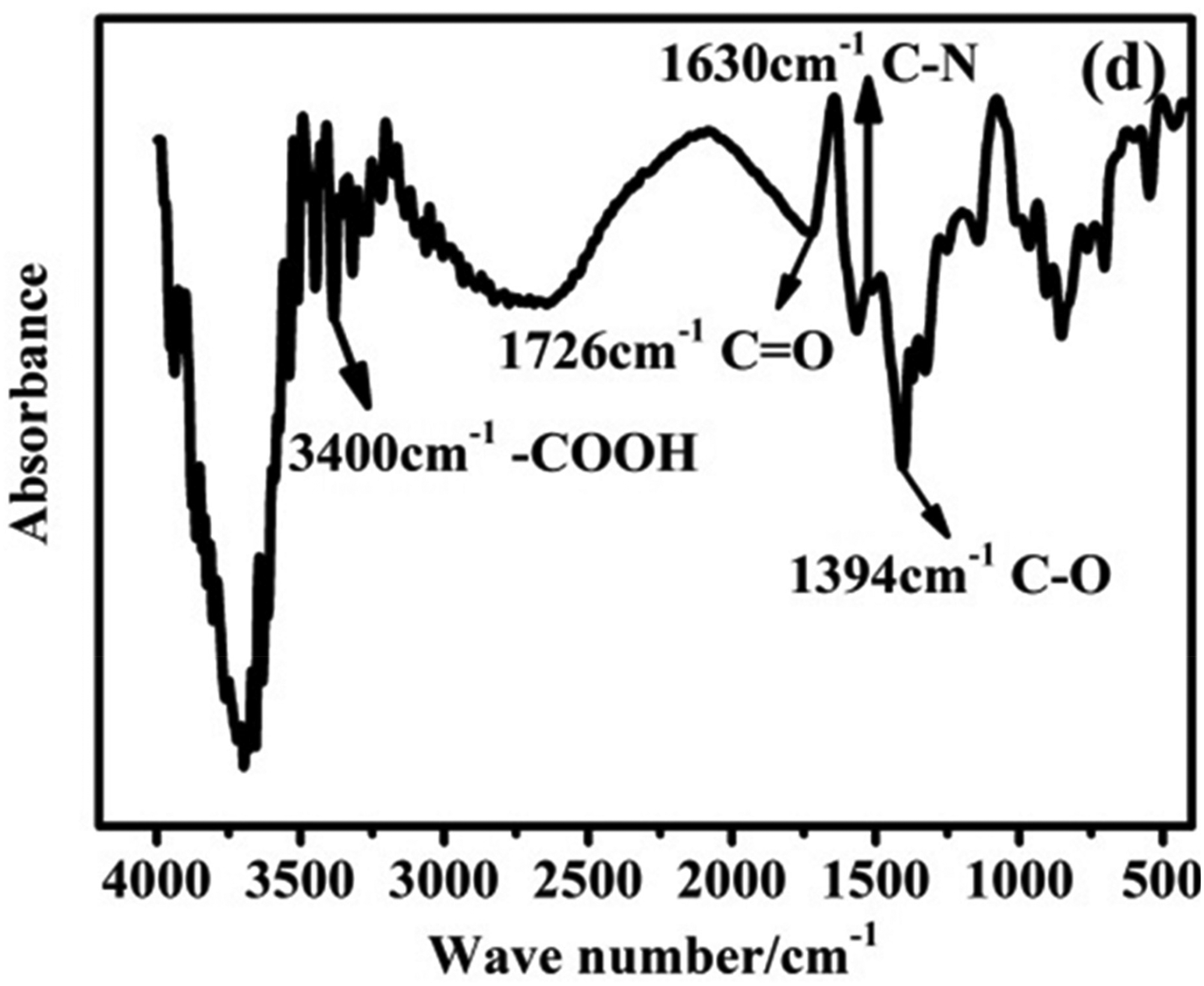
Fourier transform infrared spectrum of D407 in the study of Tang et al. (2019).
The zeolite
Through a process involving two-step ion exchange and liquid-phase reduction, Zeng et al. (2017) successfully synthesized Fe, Cu–Fe, and Mn–Fe nanoparticles supported on NaY zeolite (denoted as F@Y, CF@Y, and MF@Y, respectively). This zeolite had a SiO₂/Al₂O₃ molar ratio of 5.1, a surface area of 900 m2/g (measured in the laboratory as 625 m2/g), and a pHpzc value of 7.1. The aforementioned synthesis involved mixing metal salts with NaY in deionized water in a 1:1 mass ratio, followed by NaBH4 reduction. The ion-exchange and reduction reactions involved in the synthesis are expressed as follows (Liou et al., 2009):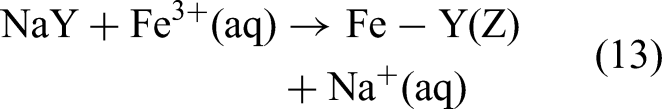

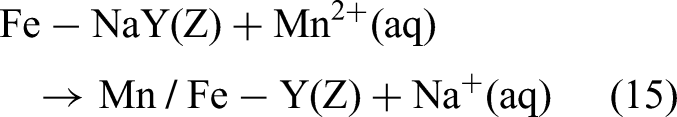
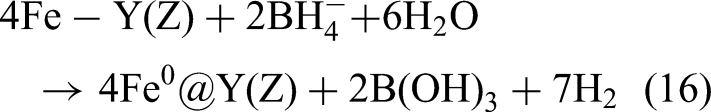
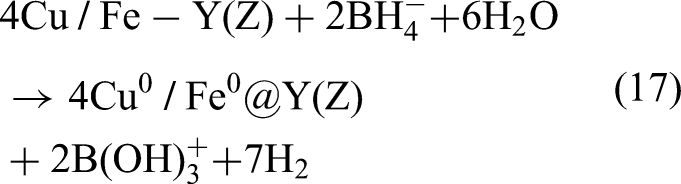

Figure 5 illustrates the nitrate removal performance and selectivity toward NH4+ and N2 for different materials. This figure indicates that CF@Y and MF@Y had considerably higher nitrate removal percentages than did the other materials, achieving 100% nitrate removal within 6 h. Moreover, CF@Y had high N2 selectivity. The negatively charged framework of the NaY zeolite, which was composed of faujasite ((Na2, Ca, Mg)3.5Al7Si17O48·32H2O), enabled metal ions to be deposited through electrostatic attraction on the surface and within the internal structure of the zeolite (Motamedi et al., 2014). Consequently, synergistic coupling was established between the metal nanoparticles and the NaY zeolite, which further promoted the in situ reduction of metal ions by NaBH4. Therefore, surface and structural modification of NaY zeolite with Fe, Cu–Fe, or Mn–Fe nanoparticles can enable the production of nanocomposites with high nitrate removal efficiency.

Nitrate removal percentages of different materials (initial conditions: uncontrolled pH = 5.7, material dosage = 6 g/L, reaction time = 6 h, and initial nitrate concentration = 100 mg/L) in the study of (Zeng et al., 2017).
Fateminia and Falamaki (2013) prepared ZVI-coated zeolite (FeCLP) from 3 g of commercial clinoptilolite obtained from Semnan, Iran, and then subjected the prepared zeolite to ion-exchange reactions by using 50 mL of a 3000-ppm FeCl2 solution. The wet solids produced after these reactions were transferred to a beaker containing 50 mL of a 24 mM NaBH4 solution, and the mixture was subjected to continuous stirring for 1 h at 20 °C and 200 rpm. To produce Cu–Fe-coated zeolite (CuFe-CLP), the prepared FeCLP was exposed to CuCl2 solutions of varying concentrations for 20 min. According to Choe et al. (2004), the precipitation of iron hydroxide on the surface of ZVI particles results in the formation of a passive layer that substantially reduces the nitrate removal efficiency, and an increase in solution pH intensifies this process. However, the initial solution pH did not have a notable effect on the nitrate removal efficiency in the study of Fateminia and Falamaki (2013). In their study, the nitrate removal efficiency primarily depended on the available surface area of Fe0 or Fe0/Cu0 particles. When clinoptilolite was used as a support, a shell developed around the zeolite agglomerate, and iron or copper was deposited on zeolite crystals (Fig. 6). The shell contained interconnected mesopores with an average diameter of 20 nm. The mesoporous structure of the shell allowed rust to form primarily on the outer surface while ensuring that the inner active sites continued to facilitate interactions between metal particles and nitrate ions (Azar and Falamaki, 2012). Furthermore, zeolite was observed to exhibit high selectivity for NH4+ ions, easily exchanging ions with covalently bonded Fe2+ or other cations such as Na+, K+, Ca2+, or Mg2+. Therefore, no NH4+ ions were detected in the solution after the reaction when zeolite was used as a substrate during nitrate removal (Lee et al., 2007; Liou et al., 2005).

Creation of a shell comprising Fe0 nanoparticles around a zeolite agglomerate. Hydrogen bubbles prevent the further reaction of NaBH4 with the inner agglomerate core (Fateminia and Falamaki, 2013).
The biochar
Li et al. (2017) compared the nitrate removal performance of two materials prepared using different loading methods: BC@Fe/Ni and BC@Nano Fe/Ni. BC@Fe/Ni was synthesized through coreduction; the process involved mixing BC, steel pickling waste liquor, and nickel chloride in ethanol and then reducing the mixture by using sodium borohydride. By contrast, BC@Nano Fe/Ni was prepared using a fractional precipitation method; in this method, BC and steel pickling waste liquor were dissolved in ethanol, the mixture was subjected to reduction reactions by using sodium borohydride, and nickel chloride solution was added dropwise to the reduced mixture to obtain Ni0. The final nitrate removal efficiency of BC@Fe/Ni reached approximately 99.55%, and its reaction rate (0.0648 ± 0.002 min−1) was 11.7 times higher than that of BC@Nano Fe/Ni. This result can be attributed to the replacement of Ni0 by Fe0 requiring a longer time when BC@Nano Fe/Ni was used. This phenomenon led to the oxidation of BC@Nano Fe/Ni, which negatively affected its reaction rate.
Hou et al. (2021) prepared Fe–Cu/polyvinylpyrrolidone (PVP)/BC to enhance NO3⁻ removal and N2 selectivity. First, BC was mixed with a CuSO4·5H2O solution, and the resultant solution was then washed and combined with FeSO4·7H2O. Subsequently, Fe0 and Cu0 were effectively immobilized on the surface of BC by adding KBH4 and PVP to the solution to obtain Fe–Cu/PVP/BC. BC promoted the uniform distribution of Fe and Cu particles on its surface and within its pores, as indicated by TEM and scanning electron microscopy (SEM) images (Figs. 7 and 8, respectively). Moreover, it reduced the distance between these particles, which was crucial for the formation of N2 (Ye et al., 2017). The optimal ratio of NO3− to N2 (31%) was achieved when the Fe–Cu/PVP/BC dosage was 1 mg/L and the Fe ratio was 1:2:3. The study of Hou et al. (2021) not only advanced knowledge in the field of water pollution treatment but also opened new directions for the application of BC as an effective catalyst in nitrate treatment technology.

TEM images and particle size distributions of Fe–Cu/ polyvinylpyrrolidone (PVP)/biochar (BC) in the study of (Hou et al., 2021): (a) Fe–Cu/PVP/BC500, (b) Fe–Cu/PVP/BC700, and (c) Fe–Cu/PVP/BC900.

Scanning electron microscopy images and EDS data of Fe–Cu/PVP/BC in the study of Hou et al. (2021): (a) Fe–Cu/PVP/BC500, (b) Fe–Cu/PVP/BC700, and (c) Fe–Cu/PVP/BC900.
The carbon nanotubes
Multiwalled carbon nanotubes (MWCNTs) have attracted considerable research attention because of their excellent adsorption properties. Wang et al. (2021c) used Fe–Pd–Fe3O4/MWCNTs for nitrate removal, which involved the rapid adsorption of nitrate, the gradual reduction of intermediates, and the readsorption of final products. Initially, Fe(II) and Fe(III) adhered to the surface of the MWCNTs when FeSO4⋅7H2O, FeCl3⋅6H2O, and the MWCNTs were sonicated in water, which led to surface precipitation. After the mixture was washed to remove excess ions, NaBH4 solution was added to the mixture, following which K2PdCl6 was loaded onto the iron surface. The relevant reactions are given as follows:

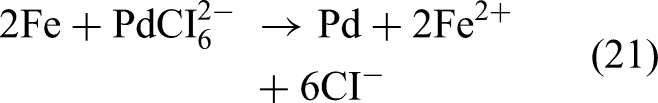
The use of the MWCNTs considerably enhanced the NO3−–N reduction activity and N₂ selectivity of the metal nanoparticles. This improvement occurred because the MWCNTs prevented the agglomeration of metal particles during the reaction, which increased the efficiency of mass transfer. Consequently, the Fe–Pd–Fe3O4/MWCNTs completely removed NO3−–N within 2 h, with 39% and 25% of NO3−–N being converted into NO2−–N and NH4+–N, respectively.
The attapulgite
Attapulgite (ATP), a crystalline hydrated magnesium silicate, has been combined with nZVI for the removal of NO3−–N from aqueous solutions, and promising results have been obtained. Specifically, Zhang et al. (2023b) used a liquid deposition–reduction method to prepare an ATP–nZVI composite. They characterized this composite through SEM, XRD, and X-ray photoelectron spectroscopy and observed that the aluminum silicate particles within ATP function as efficient carriers for nZVI. Thus, ATP can minimize the self-oxidation and aggregation often exhibited by nZVI, ultimately enhancing its nitrate removal performance. The optimal mass ratio of ATP to nZVI was found to be 1:1, which resulted in the highest nitrate removal efficiency. In particular, the NO3−–N removal efficiency achieved by ATP–nZVI under the aforementioned mass ratio was 78.61%, which was higher than that achieved by ATP, iron powder, and nZVI alone.
In summary, the incorporation of support matrices such as chelating resin, zeolite, and carbon materials considerably enhances the nitrate removal efficiency of nZVI. Furthermore, the use of bimetallic nanoparticles on these support matrices results in a substantial improvement in the nitrate removal efficiency and an increase in the production of the final product, namely nitrogen gas. Finally, innovative methodologies, including those involving the use of Fe–Pd–Fe3O4 nanoparticles on MWCNTs and nZVI-coated ATP composites, exhibit promise for application in nitrate remediation.
Influence factors of nitrate removal and N2 selectivity by carrier-material-supported BnZVI
The metal loading
Metal loading can improve the nitrate removal efficiency and N2 selectivity of catalysts in water by preventing the oxidation of iron particles. Bimetallic systems exhibit synergistic effects that improve electron transfer and catalytic performance, which enhances the conversion of nitrates into nitrogen gas (Shi et al., 2014; Li et al., 2023). This suggests that optimizing metal loading and selecting appropriate support materials can enhance the performance of catalysts in nitrate remediation applications.
Fateminia and Falamaki (2013) observed that the nitrate removal efficiency of FeCLP varied with its copper (Cu) loading, with the highest efficiency of 78% being achieved when the Cu/(Cu + Fe) ratio was 50.0% (Fig. 9). Thus, metal ratio optimization can enhance the effectiveness of nitrate remediation systems utilizing FeCLP-supported catalysts.

Effect of Cu loading on nitrate removal kinetics (Fateminia and Falamaki, 2013).
Lubphoo et al. (2015) experimentally investigated the influence of Pd–Cu loading on NO3− reduction reactions. Their results indicated that the nitrate removal efficiency increased with the Pd–Cu loading. To verify this, the specific surface area of the material at different metal loadings was measured and presented in Table 2.
Specific surface areas of nFe0 and (Pd–Cu)–nFe0 in the study of (Lubphoo et al., 2015).

Note: The analyzed Pd–Cu–nFe0 had a Pd:Cu ratio of 2.
Particle agglomeration can reduce the contact between the catalyst surface and nitrate ions, thus reducing the efficiency of nitrate reduction. Lubphoo et al. (2015) reported that when Pd/Cu nanoparticles with a Cu loading of 5% were used, the nitrate removal efficiency reached 100% within 20 min (Fig. 10); they also observed that Cu loading rates of 1% and 20% led to nitrate removal efficiencies of only 78% and 60%, respectively.

Effect of Cu loading in the (Pd–Cu)–nFe0 system in the study of Lubphoo et al. (2015).
Similarly, Shi et al. (2016) noted that the Pd/Fe ratio on the surface of DOW 3N had a crucial effect on the reduction properties of DOW 3N-Fe/Pd. They investigated this effect under an initial solution pH of 6.75 by using four Pd loading values: 2, 4, 6, and 8 wt.%. They observed that the N2 selectivity increased with the Pd loading, reaching a maximum value of 69.2% at a Pd loading of 8 wt.%.
Guo et al. (2018) examined the effect of copper (Cu) loading on the nitrate removal efficiency of Fe/Cu nanoparticles. They observed that the nitrate removal efficiency increased with the Cu loading up to a certain threshold, with the highest nitrate removal efficiency of 100% being achieved (within 20 min) when the Cu loading was 5% (Fig. 11). When the Cu loading was increased beyond this value, the nitrate removal efficiency decreased. Thus, the optimal Cu loading must be identified to maximize the nitrate removal efficiency.

Effect of Cu loading on the nitrate removal efficiency of different materials (nitrate–nitrogen concentration = 80 mg/L) in the study of Guo et al. (2018).
Tang et al. (2019) explored the effect of the Cu/Fe ratio on the nitrate reduction efficiency of Fe–Cu/D407. They determined that the highest nitrate removal efficiency and N2 selectivity were achieved when the Cu/Fe ratio was 1:2. An increase or decrease in this ratio resulted in a decrease in the nitrate removal efficiency; thus, optimizing the metal ratio is crucial for maximizing the nitrate removal efficiency (Xiong et al., 2017; He et al., 2018).
The optimal Cu loading rate for nitrate removal varies with the adsorbent composition and experimental conditions. Tang et al. (2019) and Fateminia and Falamaki (2013) have identified the optimal Cu/Fe ratio, whereas Guo et al. (2018) determined the optimal Cu loading.
The concentration ratio of Fe, metal cations, and the support material must also be optimized to maximize the nitrate removal efficiency and minimize the formation of undesirable by-products such as NH4+ and NO2−. Hou et al. (2021) explored the effects of the Fe:Cu:BC ratio on the nitrate removal efficiency and N2 selectivity, and they noted that a Fe:Cu:BC ratio of 1:2:3 resulted in the highest N₂ selectivity and a high nitrate removal efficiency (Fig. 12). They also determined that Fe, Cu, and BC in Fe–Cu/PVP/BC caused considerable enhancements in the NO3− removal efficiency and N2 selectivity; these results are consistent with those of other studies (Krasae and Wantala, 2016; Wei et al., 2018).

Effects of the (a) Fe:Cu:BC ratio and (b) Fe–Cu/PVP/BC700 dosage on the NO3—removal efficiency and the selectivity for N, NO2−, and NH4+ in the study of Hou et al. (2021). The influent NO3−–N concentration was 15 mg L− 1.
The Cu–nZVI material in the study of Zhang et al. (2024) completely removed nitrate within 2 h at a minimum Cu loading of 0.05 wt% as shown in Fig. 13(a) while the time was shortened to 1 h when the Cu content was increased to 2.0 wt% and further increase did not enhance the reactivity of nZVI. For the Pd–nZVI system, a similar strengthening effect of Pd was observed in Fig. 13(c). However, the accelerating effect of Pd on nZVI reactivity is much lower than that of Cu, although Pd is always considered a better catalyst than Cu. While as shown in Fig. 13(b) and (d), ammonium was consistently the dominant final product in both Cu–nZVI and Pd–nZVI systems, suggesting that despite variations in reactivity, the overall product distribution remained similar.

Effect of different modification methods on the performance of nZVI toward NO3− reduction (a; c) and the corresponding NO2− and NH4+ evolution (b; d) in the study of Zhang et al. (2024). Reaction conditions: [Fe0] = 1 g/L, [NaCl]0 = 1 mM, [KNO3]0 = 2 mM, pH0 = 4.0.
Studies have shown that bimetallic catalysts, especially the Fe–Cu and Fe–Pd systems, can produce a synergistic effect that improves electron transfer and increases catalytic activity. However, if the metal loading is too high, the particles may aggregate, reducing the catalytic efficiency. Therefore, further research is needed to optimize both the metal type and ratio to improve nitrate removal efficiency.
The initial pH
pH is a crucial factor regulating the corrosion behavior of nZVI; pH not only determines the differentiation of iron species in the Fe–H₂O system but also substantially affects the reactivity of nZVI (Tang et al., 2024). A low-pH environment increases the reactivity of nZVI by promoting iron corrosion and the dissolution of passivated surface layers, whereas a high-pH environment reduces the reactivity of nZVI because such an environment contains abundant OH− ions, which encourage the precipitation of ferrous hydroxide on nZVI, thus hindering electron transfer processes (Tang et al., 2019). Chen et al. (2004) examined the role of pH in the reduction of nitrates by ZVI; specifically, they examined its influence on the ZVI corrosion rate and the distribution of reaction products. Wang et al. (2006) noted that low pH promotes the reduction of nitrates by nZVI by facilitating the dissolution of the ferrous hydroxide coatings on the surface of nZVI, thereby exposing more reaction sites for nitrate reduction. Moreover, an alkaline environment promotes the formation of ferrous hydroxide precipitates, which inhibit electron transfer from nZVI to nitrates, thereby hindering nitrate reduction.
Shi et al. (2016) investigated the effects of pH on the nitrate removal efficiency and product formation observed when DOW 3N-Fe/Pd nanoparticles were used for nitrate removal. They observed that although a decrease in pH accelerated nitrate removal, the nitrate removal efficiency remained stable at various pH values. However, when the solution pH reached 8.67, the nitrate conversion efficiency decreased considerably because of H⁺ consumption and ferrous hydroxide formation during the reduction reaction (Fig. 14).

Nitrate removal efficiencies of DOW 3N-Fe/Pd (8 wt.% Fe and 8 wt.% Pd) under different initial pH values in the study of Shi et al. (2016): (a) NO3−–N, (b) NH4+–N, and (c) pH variation.
Shi et al. (2016) reported that an increase in pH resulted in a decrease in nitrate conversion efficiency but an increase in N2 selectivity, with the maximum N2 selectivity (71.0%) occurring at a pH of 8.67 (Table 3). This trend in N2 selectivity is consistent with those observed in other studies, which have indicated that the ratio of nitrite to hydrogen affects the selectivity of nitrate reduction products, with higher pH promoting enhanced N2 formation (Prüsse and Vorlop, 2001; Mikami et al., 2003; Guy et al., 2009; Yoshinaga et al., 2002; Shuai et al., 2012).
Nitrate removal efficiencies and N2 selectivity values achieved with supported nanoparticles under different experimental conditions in the study of Shi et al. (2016).

Rnitrate: nitrate removal efficiency; Cnitrate: nitrate conversion rate; SN2: N2 selectivity; Anitrate and Anitrite: ratios of the quantities of NO3−–N and NO2−–N (mg), respectively, adsorbed on the composites to the corresponding initial quantities (mg).
Zeng et al. (2017) observed that the nitrate reduction efficiency remained high within a broad pH range when zeolite was used as a support material; this can be attributed to the unique ion-exchange properties and mesoporosity of zeolite (Fig. 15). Zeolite-supported Fe, Cu–Fe, and Mn–Fe particles exhibited high nitrate conversion rates, even under alkaline conditions, because of the buffering effect of zeolite against pH changes.

Effects of initial solution pH on (a) nitrate removal efficiency, (b) NH4+ and NO2− concentrations, and (c) NH4+ and N2 selectivity in the study of Zeng et al. (2017); initial conditions: ph = 3.0–9.0, dosage of composite materials = 4 g/L, dosage of nanoscale zero-valent iron = 8 g/L, reaction time = 6 h, and initial nitrate concentration = 100 mg/L).
Li et al. (2017) examined the effect of initial solution pH on the nitrate degradation process by reacting aqueous nitrate solutions having different initial pH values with a 4-g/L BC@Fe/Ni solution. They achieved a maximum nitrate removal efficiency of 99.2% at a pH of 3, and this efficiency value was more than 17% higher than that achieved at a pH of 9 (82%). Iron oxide forms when nFe0 comes into contact with dissolved oxygen in water. At a low pH value, H+ dissolves ferrous hydroxide and other protective layers on the surface of nFe0 (a process called acid washing), yielding more fresh active sites for NO3− reduction. In addition to Li et al. (2017), several other researchers have suggested that an acidic environment enhances the exposure of active sites on the surface of nZVI, thereby intensifying the degradation of nitrates by nZVI (Lubphoo et al., 2015; Wang et al., 2021c; Krasae and Wantala, 2016; Han et al., 2016; Fateminia and Falamaki, 2013; Bae et al., 2016).
The reaction time
The impact of reaction time on the change in solution pH as well as the conversion of nitrogen species during denitrification has always been carried out in studies on the efficiency and sustainability of bimetallic systems. During nitrate reduction, reaction time plays a crucial role in pH evolution. Fateminia and Falamaki (2013) observed that with CuFeCLP, the pH increased sharply within the first hour, reaching approximately 9.2 regardless of the initial pH, then gradually declined and stabilized at around 8.5 after 30 h. Similarly, Zeng et al. (2017) reported that for NaY zeolite-supported Cu/Fe and Mn/Fe nanoparticles, the pH rose rapidly within the first 30 min, reaching about 8.0, and then stabilized between 8.0 and 8.5 after 2 h. Guo et al. (2018) pointed out that nanoscale bimetallic Fe/Cu particles caused the pH to increase from 7.24 to nearly 10 within 10 min and remained constant thereafter. These results indicate that the most significant pH changes occur in the early stages of the reaction, driven by proton consumption and hydroxyl ion generation. Over time, the system reaches a steady-state alkaline condition, which transforms nitrogen species by promoting ammonium formation and limiting nitrite accumulation.
The conversion of NO3− to various nitrogen species, including NO2− and NH4+, follows a distinct trend over time, as observed in a study of Zeng et al. (2017) (see Fig. 16). Initially, nitrate concentration decreases sharply within the first 150 min, indicating rapid reduction. However, beyond this point, the reduction rate decreases and gradually stabilizes at a constant concentration. The initial fast reduction phase is likely due to high reactive surface sites on the catalysts, which become gradually passivated over time. NO2− concentration, a key intermediate, follows a transient pattern. It is either undetectable or appears at very low concentrations before disappearing entirely within 30 min. This suggests that nitrite is an intermediate product, rapidly undergoing further reduction to ammonium or N2. Notably, at 20 min, nitrite was only detected at trace levels (0.31 mg/L at pH 5.7 and 0.17 mg/L at pH 7.0 for CF@Y), indicating an efficient and nearly complete conversion mechanism within a short timeframe. NH4+ concentration follows an increasing trend over time. It rises steadily in the first 20 min, stabilizes after 150 min, and remains constant after 300 min, suggesting that all nitrogen transformations have reached completion. Similarly, the reaction proceeds according to the stepwise reduction pathway presented by Guo et al. (2018): NO3−–N → NO2−–N → NH4+ –N. Nitrate concentration continuously decreased, achieving nearly 100% removal within 40 min. Throughout the reaction, 22.4% of the initial nitrate was converted to nitrite as an intermediate, which was subsequently reduced to ammonium. By the end of the reaction, 7.7% of nitrite remained in solution, while 86.1% of the nitrogen was converted to ammonium. The total nitrogen content initially decreased and then stabilized, with an overall reduction of 5%, suggesting partial nitrogen loss as gaseous N₂. Although nitrate is rapidly reduced, the residual nitrite indicates incomplete conversion, highlighting that the reaction proceeds through a sequential reduction mechanism. In agreement with this observation, total nitrogen in study of He et al. (2020) decreased from 20 mg/L to 9.79 mg/L after 10 min, indicating that most nitrate was reduced to nitrogen gas by Z-Fe/Pd and escaped from the solution early in the reaction. As shown in Fig. 17, ammonium increased to 5.22 mg/L after 20 min and then gradually decreased to 2.08 mg/L by the end of the reaction. Nitrite appeared only in the first 2 min at 0.35 mg/L, suggesting it was an intermediate rapidly reduced further. Pd0 played a crucial role in enhancing nitrate reduction and N₂ selectivity through a galvanic effect between Fe0 and Pd0.

Nitrogen conversion and formation kinetics at initial nitrate concentration 150, 200 mg/L in study of Zeng et al. (2017) : (a) F@Y, (b) CF@Y, (c) MF@Y. (initial conditions: dosage 4 g/L and pH uncontrolled 5.7).

Simultaneous removal kinetics of nitrate (dosage, 0.5 g/L; initial solution pH, 3.0; concentrations of nitrate 20 mg/L; reaction time, 6.0 h) in study of He et al. (2020).
The results emphasize the essential influence of reaction time on pH changes and the transformation of nitrogen species during nitrate reduction. The most significant pH changes occur in the early stages, driven by proton consumption and hydroxyl ion generation, before stabilizing in an alkaline state. This pH shift directly influences the stepwise reduction of nitrate to nitrite and ammonium.
Reusability of BnZVI materials for nitrate reduction
The ability of BnZVI materials to be reused is essential for evaluating their suitability for large-scale applications. Studies have shown a gradual decline in nitrate removal efficiency over reuse cycles due to the depletion of active sites, the formation of less reactive oxide layers, material loss, and the reduction in adsorption capacity. Zeng et al. (2017) investigated the performance decline of F@Y, CF@Y, and MF@Y over three successive reaction cycles without regeneration under conditions of 4 g/L metals@Y dosage, 100 mg/L NO3−, and an initial uncontrolled pH of 5.7. Results showed that nitrate removal efficiency decreased from 100% to 75.0%, 84.2%, and 76.3% for F@Y, CF@Y, and MF@Y, respectively, after three cycles (18 h). The decline was attributed to four factors: (1) partial exhaustion of active sites on the nanocomposite surfaces; (2) formation of a less reactive rust layer due to dissolved oxygen; (3) material loss during recycling; and (4) leaching of nano Fe particles from the zeolite support.
Wang et al. (2021c) investigated the reusability of Fe–Pd–Fe₃O₄/MWCNTs nanocomposites through NO₃⁻–N removal efficiency. As shown in Fig. 18, a decline from 63% to 43% with increasing cycles, indicating insufficient reducing power from Fe0. NH₄+–N adsorption efficiency also decreased from 96% to 53%, suggesting a gradual depletion of adsorption sites on MWCNTs. Although Fe0 was consumed, NO3⁻–N reduction was slightly enhanced by FeOOH/Fe3O4 formation via Fe0 oxidation, as previously reported by Xu et al. (2012). These findings highlight the gradual efficiency loss of the nanocomposite due to material consumption and adsorption site depletion. Additionally, the presence of strongly magnetic Fe₃O₄ enhances the ease and efficiency of nanocomposite recovery.

The reusability of nanocomposite for NO3−–N removal in the study of (Wang et al., 2021c).
Conclusions
This review provides crucial insights regarding the optimization of nitrate removal processes through the use of carrier-material-supported nZVI systems; it highlights advancements in improving the effectiveness and stability of these systems. The findings of this review are as follows. First, lower pH values increase nitrate reduction rates by facilitating the dissolution of ferrous hydroxide coatings on nZVI, whereas alkaline conditions inhibit nitrate reduction because they promote the formation of iron hydroxide precipitates. Second, support materials such as rectorite, bentonite, and silica prevent the aggregation, improve the dispersity, and facilitate the mass transfer of nZVI, thereby enhancing the nitrate removal efficiency. Third, metal loading in nZVI considerably influences its reactivity and selectivity, and bimetallic nanocomposites exhibit enhanced nitrate removal performance because of the presence of metal cations. Additionally, not only the crucial role of reaction time in pH variations and the conversion of nitrogen species during nitrate reduction but also the impressive reusability of nZVI is highlighted. In summary, the findings of this review indicate that nZVI has potential for application in advanced strategies for removing nitrates from water, thus contributing to environmental protection and human health. These findings can be used as a reference to develop more efficient and sustainable approaches for mitigating the nitrate pollution of water sources.
Footnotes
Abbreviations
Acknowledgments
The authors acknowledge financial support from the Taiwanese National Science and Technology Council.
Author contributions
Huu-Khiem Nguyen was responsible for searching and storing references, selecting references relevant to the research content, receiving comments, and revising the final version of the manuscript. Chih-Chi Yang was responsible for editing the manuscript and controlling the progress of the research. Yung-Pin Tsai was accountable for planning the work, reviewing the entire manuscript, and coordinating personnel.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Ethical issues including plagiarism, informed consent, misconduct, data fabrication and/or falsification, double publication and/or submission, and duplication have been fully addressed by the authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taiwanese National Science and Technology Council [NSTC 112-2221-E-260 −002 -MY3].



