Abstract
Dry reforming of methane (DRM) offers a promising pathway towards carbon neutrality by converting the greenhouse gases methane (CH4) and carbon dioxide (CO2) into valuable syngas (CO + H2). This sustainable process not only mitigates climate change but also contributes to a circular carbon economy by utilizing waste gases as valuable feedstocks. However, the successful industrial implementation of DRM hinges on the development of stable and efficient catalysts. This study investigated the influence of the ceria support source on the catalytic performance of Ni/CeO2 catalysts. Three commercially available ceria supports from Germany, Canada, and the USA were employed, denoted as Ni-P, Ni-M, and Ni-C, respectively. These supports were impregnated with nickel and characterized using a suite of techniques, including XRD, FTIR, SEM, N2 adsorption-desorption, and TGA. Catalytic activity and stability were evaluated within a temperature range of 550 to 750 °C. Our findings revealed that the catalytic performance is significantly influenced by the physicochemical properties of the catalyst. The Ni/CeO2 (Ni-C) catalyst demonstrated superior activity and stability, exhibiting minimal carbon deposition as evidenced by TGA analysis and a low deactivation factor. This research provides valuable insights into the critical role of support materials in optimizing Ni/CeO2 catalyst performance for DRM. The development of highly stable and active catalysts, such as the Ni/CeO2 (Ni-C) catalyst, is crucial for the successful industrial implementation of DRM, contributing to a more sustainable and environmentally friendly energy future.
Introduction
The escalating global energy demand, coupled with the pressing need to mitigate climate change, has intensified the search for sustainable and environmentally friendly energy solutions (Chava et al., 2021; Gattie, 2020). The over-reliance on fossil fuels has resulted in a significant rise in atmospheric greenhouse gas (GHG) concentrations, primarily driven by the release of carbon dioxide (CO2) and methane (CH4). The depletion of fossil fuel reserves and their detrimental environmental impact, including global warming and climate change, necessitate the exploration of alternative, cleaner energy sources (Leba and Yıldırım, 2020; Mageed et al., 2025).
These alarming trends underscore the urgent need for a transition towards cleaner and more sustainable energy sources (da Silva Pinto et al., 2022). Biogas, produced from the anaerobic digestion of biomass such as agricultural residues, forest waste, and food waste, offers a promising renewable energy option (Chava et al., 2023; da Silva Pinto et al., 2022). However, biogas typically comprises a mixture of 35–75% CH4, 25–55% CO2, and minor amounts of other impurities such as H2S, N2, O2, CO, and trace amounts of water vapor, NH3, siloxanes, aromatics, and dust particles (Chava et al., 2022; Yao et al., 2023). The presence of CO2 and other impurities can significantly reduce the energy output efficiency of biogas when used in applications like combined heat and power (CHP) engines (Qi et al., 2022).
To address this challenge and enhance the value of biogas, various reforming technologies can be employed to convert its constituents (CH4 and CO2) into synthesis gas (CO + H2). These technologies include dry reforming, steam reforming, partial oxidation, and autothermal reforming (Ahmad et al., 2021; Bian et al., 2021; Gao et al., 2021; He et al., 2022). Among these, dry reforming of methane (DRM) offers several advantages:
Effectively utilizes two major greenhouse gases, CH4 and CO2, transforming them into valuable syngas Compared to steam reforming and partial oxidation, DRM requires less methane consumption The syngas produced from DRM can be utilized as a versatile feedstock for various industrial processes, including the production of fuels, chemicals, and electricity.
Catalyst deactivation is a major concern in DRM, leading to a decline in catalytic activity and stability over time. Several factors contribute to deactivation, including:
Dry reforming of methane (DRM) presents a viable solution by utilizing both CH4 and CO2, two major greenhouse gases, to produce valuable syngas (CO + H2) according to the following reaction Equation (1):
This endothermic reaction not only mitigates the environmental impact of these greenhouse gases but also provides a sustainable route to produce syngas, a versatile feedstock for various industrial processes, including the production of fuels, chemicals, and electricity.
Despite its promise, DRM faces several challenges, including the endothermic nature of the reaction necessitates high operating temperatures, which can increase energy consumption and operational costs. Catalyst deactivation remains a major concern in DRM, significantly impacting process efficiency and long-term operability.
Nickel (Ni)-based catalysts have emerged as frontrunners in DRM due to their high activity and relatively low cost. However, these catalysts suffer from inherent limitations, including deactivation at high temperatures caused by catalyst sintering and carbon deposition (Abdulrasheed et al., 2019; Bai et al., 2023; He et al., 2024). Optimizing DRM performance hinges on two crucial factors: (1) the nature of the interaction between nickel and the support material and (2) the degree of nickel particle dispersion within the support. Unfortunately, many commercially available Ni-based DRM catalysts still struggle with issues such as low dispersion, limited surface area, restricted accessibility to active sites, and uneven nickel particle distribution accessibility of active sites (Niu et al., 2021). These limitations significantly impede their widespread industrial application.
Fortunately, various strategies can be employed to address these limitations. One promising approach involves the incorporation of promoters at the molecular level to enhance the functionality and structure of the catalyst interface (Cao et al., 2023; Deng et al., 2022; Zhang et al., 2023). Smaller nickel particles, achieved through optimized preparation methods, can create a larger metal-support interface, facilitating CO2 dissociation and carbon oxidation, thereby maintaining catalyst functionality. Therefore, controlling nickel particle size is crucial for developing more efficient and thermally stable catalysts for high-temperature DRM reactions.
Furthermore, establishing a strong bond between the metal (Ni) and the oxide component (ceria) during catalyst preparation has been demonstrated to effectively inhibit carbon deposition and enhance the functionality of Ni-based catalysts in DRM (Gong and Bao, 2017; Suchorski et al., 2018). Ceria (CeO2) has attracted significant attention due to its exceptional oxygen storage and release capabilities, along with its redox properties. The reduction of Ce+4 to Ce+3 in CeO2 creates oxygen vacancies, which can significantly enhance CO2 adsorption and dissociation. Additionally, density functional theory (DFT) data suggests that the strong metal-support interaction (SMSI) between Ni and CeO2 can promote C-H bond cleavage at lower temperatures. This Ni-CeO2 interaction further improves the DRM process by accelerating reaction kinetics and minimizing carbon buildup.
This study delves into the impact of the ceria source on the catalytic performance of Ni/CeO2 catalysts for CH4 reforming with CO2. The catalysts were synthesized using the wet impregnation method, as detailed in the following section. We employed various characterization techniques, including X-ray diffraction (XRD), Brunauer-Emmett-Teller (BET) surface area analysis, Fourier-transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), thermogravimetric analysis (TGA), and deactivation factor (DAF) measurements, to comprehensively evaluate the influence of the ceria source.
Materials and methods
Catalyst preparation
Ni/CeO2 catalysts were prepared by the wet impregnation method. Three different commercial CeO2 supports were obtained from Sigma Aldrich (Germany), M K Impex Corp. (NPD, Canada), and Merck KGaA (USA). Briefly, the desired amount of nickel nitrate hexahydrate was dissolved in deionized water and impregnated onto the CeO2 support. The impregnated samples were dried overnight at 120 °C and subsequently calcined at 750 °C for 5 h. Detailed catalyst synthesis, detailed characterization techniques, and experimental procedures are provided in the Supplementary Information.
Results and discussions
Characterization of calcined catalysts
N2 adsorption-desorption isotherms
The specific surface area, pore size distribution, and pore volume of the catalysts were derived from N2 adsorption-desorption isotherms at liquid nitrogen temperature. The particular surface area was determined using the BET surface area analysis method. The quantity of N2 molecules adsorbed and desorbed on the catalytic surface is estimated using these techniques.
. These methods estimate the amount of N2 molecules adsorbed and desorbed on the catalytic surface. There is usually internal diffusion of reactant's mass on the catalyst surface which means molecules have to pass through the pores to reach activity sites. Figure 1 displays the N2 adsorption-desorption isotherms for the catalysts. The isotherms were classified according to the guidelines set out by IUPAC when addressing the characteristics of pores (Singha et al., 2017).

N2 adsorption-desorption isotherm of all Synthesized Catalysts
A type IV isotherm for mesoporous materials is suggested by the properties of the Ni/CeO2 catalysts shown in the combined isotherm. This sort of isotherm characterizes monolayer-multilayer adsorption on the mesoporous walls, with pore sizes that are in the 2–50 nm range. The reported results are almost similar to those reported in the literature (Deng et al., 2014; Singha et al., 2017).
In the region of low relative pressure (Figure 1) showed that the catalysts show very low adsorption levels, thus suggesting that the monolayer region is not filled. Relative pressure (P/P0) can vary from 0 < 0.1 as the above expression indicates and corresponding values of the above two expressions can be calculated 1, there is the monolayer formation with gas adsorption, but the amount of gas that can be adsorbed is still minimal. At relatively high-pressure regions, multilayer adsorption takes preemi nence (Ibrahim et al., 2022).
Since the relative pressure P/P0 is proportional to the fractional coverage of the adsorbent surface with adsorbate molecules, it can be concluded that an increase in the coverage by N2 molecules in the CO2/N2 system takes place at P/P0 around 0. 75 in the latter half of the isotherm (Figure 2) can be ascribed to capillary condensation inside the pores or within the gaps between the catalyst particles belonging to the mesopores and macropores in nature (Teh et al., 2021). This typically occurs when the adsorbate's critical temperature is higher than the adsorption temperature. Furthermore, the support materials themselves are usually mesoporous materials that can quickly accelerate the gasification of these active carbon molecules.
The quantity of N2 adsorbed differed markedly from the one another catalyst systems, whereas NI-M and NI-P possessed a low N2 adsorption capacity of 262cm3/g, 220 cm3/g and NI-C possessed a high N2 adsorption capacity of 315 cm3/g respectively. This equates to specific surface areas of 37.98 m²/g, 30.10 m²/g, and 28.10 m²/g for catalysts NI-C, NI-M and NI-P respectively. Better catalytic activity is often ensured by the growth of large surface area and pore volume, particularly through substrate adsorption and improved metal dispersion. Therefore, in terms of catalytic behavior, Ni-C should counterpoise over Ni-M and Ni-P. Based on the adsorption data, the pore size distribution was calculated using the Barrett-Joyner-Halenda (BJH) adsorption cumulative pore volume model to explain the isotherms. The hysteresis loop that is now being seen is of the H2 type, which is indicative of the capillary condensation in the mesopores based on the IUPAC classification (Teh et al., 2021).

Pore volume distribution.
XRD analysis
X-ray diffraction (XRD) analysis was employed to characterize the crystalline phases present in the Ni/CeO2 catalysts (Figure 3). The XRD patterns revealed the presence of crystalline peaks corresponding to both NiO and CeO2. All NiO peaks were indexed to the cubic system (JCPDS 01-081-0792), with characteristic reflections observed at 2θ values of 37.29° and 43.45°.
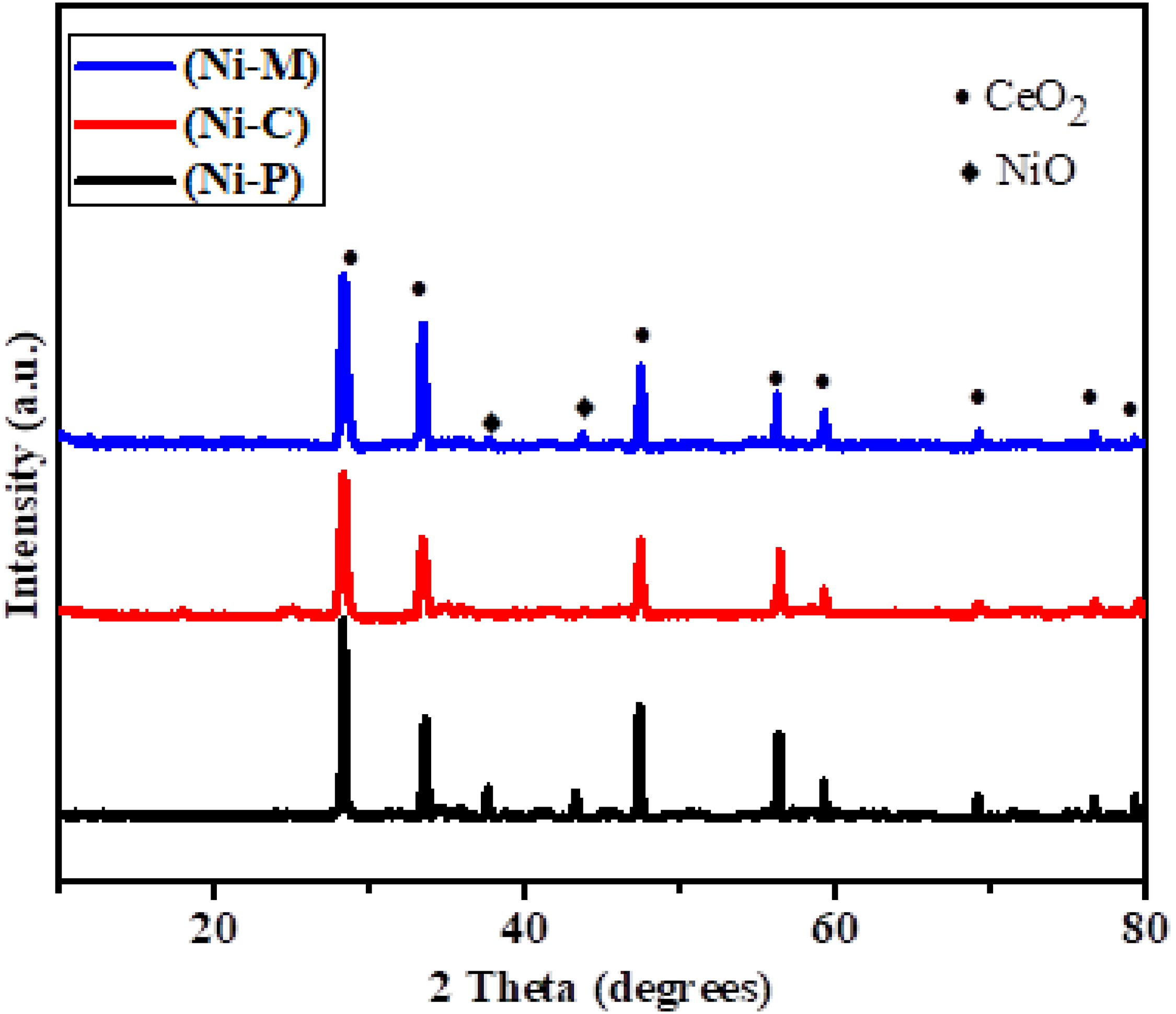
XRD Spectra of all synthesized catalysts.
The characteristic peaks of CeO2 (JCPDS 34-0394) were detected at 2θ values of 28.4°, 33.18°, 47.42°, 56.45°, 59.18°, 69.45°, 76°, and 79°. These observations are consistent with previous studies (Mierczynski et al., 2023). The broader CeO2 (111) peak observed in the NI-C catalyst compared to NI-M and NI-P suggests the presence of smaller ceria crystallites or a distorted lattice structure (Mosinska et al., 2020). This, coupled with the relatively broad and low-intensity CeO2 and NiO diffraction peaks, indicates the presence of smaller crystallites of both phases, consistent with the high BET surface area observed for this catalyst. Smaller crystallite sizes generally correlate with higher surface areas, providing more active sites for the DRM reaction.
The NI-M catalyst exhibited the highest intensity NiO peaks, suggesting the presence of larger, potentially aggregated NiO particles on the CeO2 support surface. Larger NiO particles may have limited accessibility and reduced active surface area, potentially impacting catalytic performance.
The XRD pattern of the NI-P catalyst suggests the formation of larger NiO particles during the calcination step. These larger particles could potentially lead to lower catalytic activity and stability due to reduced metal-support interaction and increased susceptibility to sintering.
These XRD findings provide valuable insights into the structural characteristics of the synthesized catalysts, correlating crystallite size with surface area and potentially influencing catalytic activity.
FTIR analysis
On the catalyst surface, functional species can be found by employing FTIR spectroscopy. Figure 4 displays the FT-IR spectra of the calcined Ni/CeO2 catalysts.

FTIR analysis of all synthesized catalysts.
Comparing the two spectra, the band at 3420–3431 cm⁻¹ can be identified as the O-H stretching in hydroxyl (OH) species, which, in this case, might originate from weakly adsorbed water or H-chemisorbed species on the surface metals (Mierczynski et al., 2023). At 1630 cm⁻¹ and 1018 cm⁻¹, new bands were seen. These groups are known as the asymmetric stretching (νas(CO3)), symmetric stretching (νs(CO3)), and the OCO-bending (δ(OCO)) of carbonate ion respectively (Mosinska et al., 2020; Souza et al., 2011). These bands can indicate that some carbonate species might result from the calcination process, while others may be formed via CO2 adsorption during the exposure.
Bands of the post-exposed catalyst are seen at 1376 cm⁻¹ and 1470 cm⁻¹. These bands are assigned to the CH-bending (δ(CH)) and asymmetric CO2-stretching (νas(COO)) modes of formate species; corresponding to bidentate chelating formates. Furthermore, a band located in the regions of 495–540 cm⁻¹ is indicative of the NiO bending vibrations (Mierczynski et al., 2020). The dispersed carbonates and formates comprise the whole reaction intermediates in the CO2 hydrogenation process, which forms methane, according to the characterization of the calcined catalysts. The formation of these larger NiO particles might have been able to reduce the specific surface area, which in turn might be able to affect the catalytic properties negatively (Mosinska et al., 2021).
SEM analysis
SEM images (Figure 5) provide valuable insights into the surface morphology of the catalysts. The analysis revealed that the highly ordered and cubic structure of the ceria support was well-maintained across all samples, even after the deposition of nickel nanoparticles.

SEM analysis all synthesized catalysts.
The images indicated the presence of well-dispersed, aggregated metal nanoparticles on the surface of the ceria support. Furthermore, cross-sectional images suggested a consistent distribution of nickel nanoparticles throughout the support volume. This uniform distribution of metal particles is crucial for maximizing metal-support interactions, which are essential for optimal catalytic performance in the dry reforming of methane. Effective metal-support interactions can enhance CO2 activation, facilitate electron transfer, and minimize carbon deposition.
The observed morphology, characterized by well-dispersed nanoparticles and a consistent distribution throughout the support, is likely to contribute to the observed catalytic activity and stability. These morphological features may facilitate efficient mass transport within the catalyst bed, enhancing reactant accessibility to the active sites.
Performance study of catalyst
Catalytic activity
This Figure examines the effects of the reaction temperature on the conversion of methane (XCH4) and carbon dioxide (XCO2), as well as the molar ratio of H2/CO. With an increase in temperature, the Ni/Ce catalysts system's methane reaction conversion rises from 10% at 500°C to 35% at 750°C (Figure 7a). Likewise, the transformation of the CO2 increases from 5% for the reaction at 500 °C to 50% for the reaction at 750 °C (Figure 7(b)). Specifically, Ni catalyst shown on Ni-C come closer to the higher phenomenal conversion of carbon dioxide and methane over the range of temperatures examined and, therefore, they can be referring as more active compared to Ni-M and Ni-P.
These observations are particularly in synergy with the prior findings that were made. The Ni under-layer is not the only reason why the Ni-C catalyst is more active than the other two ceria-supported catalysts in this reaction; the catalyst's increased surface area and active site density both contribute to its action (Kambolis et al., 2010). In addition, ceria is widely known for its remarkable efficiency in terms of activating CO2 (Araiza et al., 2021). According to others, the high concentration of oxygen vacancies on the surface of the ceria system, for example, contributes to CO2 adsorption and activation sites (Jin et al., 2024; Yang et al., 2008). Electrons from the support move to the adsorbed molecule when the carbon dioxide adsorbs through the oxygen atom, causing the C-O bond to relax and dissociate (Florez et al., 2008) as shown in Figure 6. Therefore, it can be concluded that the number of oxygen vacancies and reactivity have a significant impact on these catalysts’ catalytic performance with a direct result on the activation of CO2 and the corresponding higher DRM rate in Figure 7.
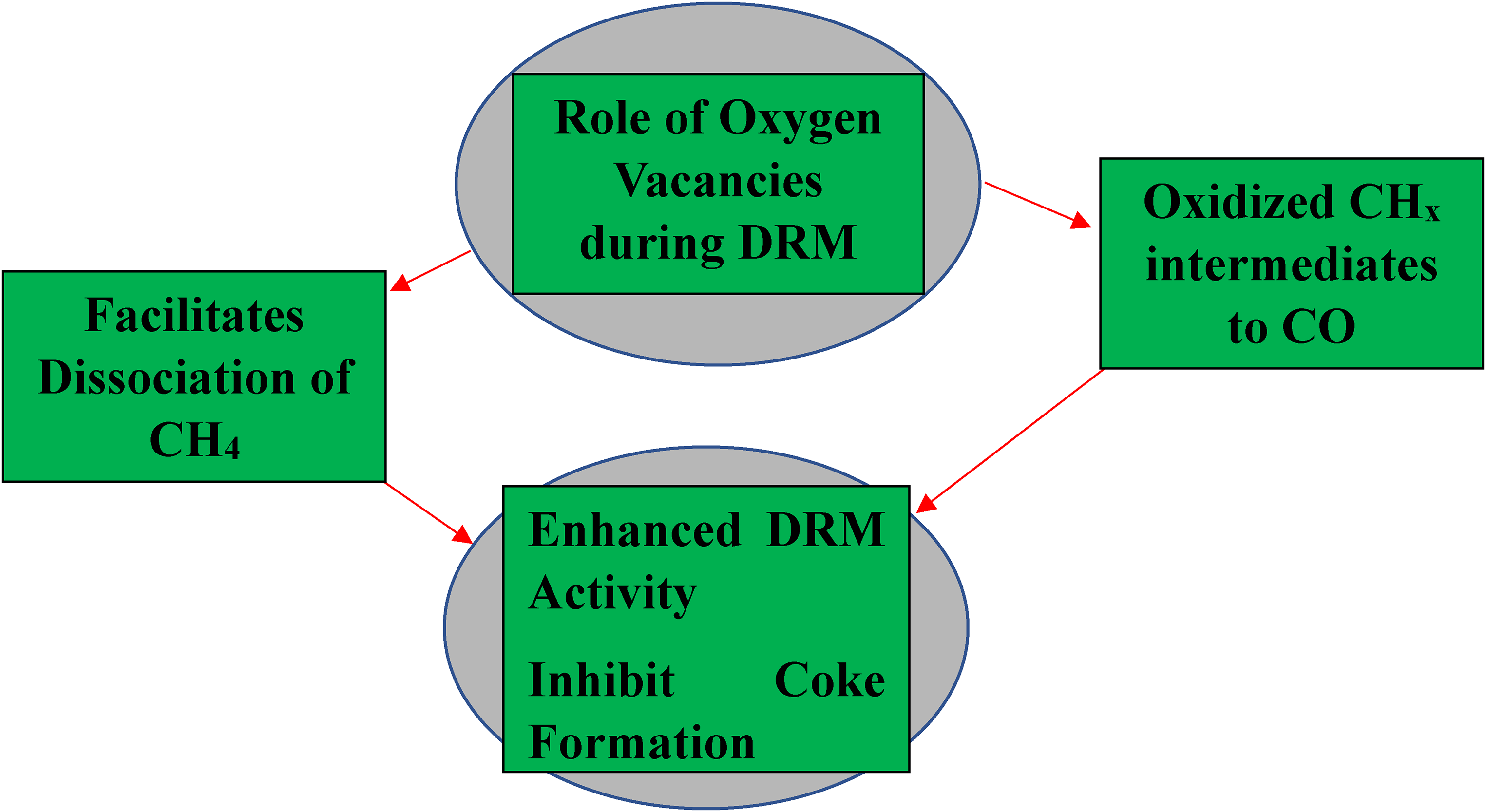
Schematic diagram showing the role of oxygen vacancies in DRM reactions.
Importantly, it has also been demonstrated that ceria is an effective catalyst for the water-gas shift reaction (WGS) at both high and low temperatures (Smal et al., 2022). In particular, this reaction is endothermic and falls within the category of dry reforming of methane (DRM) reactions. Raising the reaction temperature increases the yield of H2 and CO as well as the conversion of CH4 and CO2 (Vera-Hincapie et al., 2023). Notably, the reaction of the CO2 to CH4 conversion is higher throughout all the catalysts and temperature ranges in the analysis.
According to this, it can therefore be ascribed to the reverse water-gas shift process (RWGS). Nonetheless, the greatest conversions of CO2 and methane were attained at 84% conversion. About 56% and 89. 34% and 34%, respectively, were obtained at 750°C for the catalyst containing Ni, which was supported on CeO2 with 10% Ni loading. Other research has also shown the same findings of increased CO2 conversion compared to CH4 at lower temperatures as this work (Gandhi and Patel, 2015). Coke may occur when the conversion of methane increases since the dissociation rates for CH4 are aligned to increase as the reaction temperature is raised and exceeded.

Performance of catalysts at different temperature in terms of (a) methane conversion (%) (b) carbon dioxide conversion (%). (c) Methane conversion (d) Carbon dioxide conversion (e) H2/CO ratio Versus Time on stream at given reaction Temperature.
As the temperature increases CH4 dissociation emerges as a more favorable process than CH4 abstraction by O2.
H2/CO molar ratio
The H2/CO molar ratio for the Ni-C, Ni-M, and Ni-P catalysts as determined by the DRM technique is thus displayed in Figure 7(e). The H2/CO ratio is kept below 1 in all three catalytic systems; therefore, the RWGS reaction somewhat interferes with the product yield. This finding is consistent with other researchers’ investigations into the characteristics of ceria-based and ceria-zirconia-supported Ni catalysts, using feed gas compositions similar to the current study (Araque et al., 2015; Ebiad et al., 2012; Kumar et al., 2007).
However, the rates of the DRM and Boudouard reactions are the other factors that help determine the H2/CO ratio. This is because the efficiency of the said approach depends on the occurrence of several concurrent reactions among which are the RWGS, methane decomposition, and the Boudouard reactions. Carbon formation on the catalyst surface may either be through the Boudouard reaction (equation (3)), which relates the conversion of CO to carbon and CO2, or through methane cracking (equation (2)) in which the transforms CH4 to carbon and 2H2. The reverse Boudouard reaction (C + CO2 → 2CO) or surface oxygen on the support (C + O2 → CO2) can reoxidize these deposited carbon molecules to CO by gaseous CO2 (Damyanova et al., 2012; Sokolov et al., 2012; Taufiq-Yap et al., 2013). Similarly, as pointed out by (Radlik et al., 2015), the ratio comprised of H2/CO smaller than 1 for Ni/Ce catalysts most probably results from the RWGS reaction and carbon oxidation by CO2.
Catalyst stability
About 420 min on stream (TOS) were used to analyze the catalytic performances of the Ni/CeO2 catalysts for CH4 reforming in this experiment. Their reaction temperature was set to 750°C, and the feed rate of the reactants while the pressure was 1 atm. The outcome is depicted in Figures 7 (c, d), it is clear that the presence of ceria derived from the support source has a significant impact on both activity and stability. The Ni-C catalyst proved to achieve the highest first conversion of CH4 of 63%. With an average conversion of 53%, which proved effective throughout the TOS. Ni-M and Ni-P catalysts were found to have a lower initial conversion of roughly 42.9%. The Ni-C catalyst's excellent performance can be attributed to a number of factors: One can infer from the results in Table 1 that the catalyst favors its high BET surface area, which may be the result of the catalyst and support material interacting properly as it was observed from XRD, SEM, FTIR, and DFA analysis. This conforms with the fundamental rule that as the surface area that exists between a substrate and a catalyst increases catalytic activity will improve (Gomes et al., 2019).
Texture properties of catalysts.

Comparison of catalysts performance during DRM
As stated earlier, the efficiencies of different catalysts can also be evaluated through the CO2 conversion in the CH4 reforming process as presented in Figure 7 above. Keep in mind that the rates of CH4 conversion are lower than those of CO2, with an average difference of 5–10% for all catalysts. Equation 4 demonstrates that this is caused by the Reverse Water-Gas Shift Reaction (Liu et al., 2019). Among all these catalysts, the Ni-C catalyst showed the highest first value average of CO2 conversion (around 66%) which is slightly greater than Ni-M and Ni-P catalysts with average CO2 conversion value of approximately 52%, and 32. 8%, respectively. Table 2 shows the performance of catalysts with the literature.
Comparison of catalyst performance with other studies.

The deactivation factor, the extent of weight loss (from TGA analysis), and other items of interest for each catalyst are given in Table 3. Catalyst deactivation is a significant challenge in industrial DRM processes, leading to a gradual decline in catalytic activity over time and ultimately necessitating catalyst replacement. Key deactivation mechanisms include (Martín et al., 2022; Otor et al., 2020): The formation of carbonaceous species (coke) on the catalyst surface is a primary deactivation mechanism. Coke can block active sites, hinder reactant diffusion, and even cause catalyst fragmentation. High operating temperatures can lead to the sintering of metal nanoparticles, resulting in a decrease in active surface area and a subsequent loss of catalytic activity. Impurities in the feedstock, such as sulfur compounds, can poison the active sites of the catalyst, leading to a significant decrease in catalytic activity.
Conversion of CH4 and CO2, deactivation factor and weight loss (%).

The deactivation factor quantifies the extent of catalyst deactivation by comparing the initial catalytic activity with the activity after a certain period of operation. A lower deactivation factor indicates higher stability and, consequently, a longer catalyst lifetime (Lin et al., 2022).
Weight loss during the reaction can be attributed to several factors, including carbon deposition, volatilization of catalyst components, and sintering (Gromotka et al., 2021). Thermogravimetric analysis (TGA) can be employed to quantify weight loss and provide valuable insights into the extent of carbon deposition.
Catalysts with lower deactivation factors generally exhibit higher stability and longer lifetimes. This implies that they maintain their activity for extended periods, reducing the frequency of catalyst replacement.
Lower weight loss during the reaction, as observed in the Ni/CeO2 (Ni-C) catalyst in this study, indicates minimal carbon deposition. This is crucial for maintaining catalyst activity and prolonging catalyst lifetime. Reduced weight loss also suggests less sintering of the metal particles, which further contributes to enhanced stability
Characterization of spent catalyst
TGA analysis
Thermogravimetric analysis (TGA) provides valuable insights into catalyst deactivation by quantifying mass loss during a controlled temperature program (Ferrandon et al., 2022; Palma et al., 2020). In the context of DRM, weight loss primarily arises from: (a) The formation of carbonaceous species (coke) on the catalyst surface is the most significant contributor to weight loss. This can occur through various mechanisms, including methane cracking, the Boudouard reaction, and polymerization reactions. (b) Minor weight loss can also occur due to the volatilization of certain catalyst components at elevated temperatures.
Catalysts exhibiting lower weight loss during TGA analysis generally demonstrate higher resistance to coking (Borgschulte et al., 2024; Rahmati et al., 2020). This translates to: (I) Reduced coke formation leads to prolonged periods of stable catalytic activity, minimizing the frequency of catalyst replacement (II) Longer catalyst lifetimes result in higher reactor throughput and reduced downtime for catalyst regeneration or replacement (III) Lower catalyst replacement frequency and improved reactor productivity contribute to significant cost savings in industrial DRM processes.
Minimizing carbon deposition aligns with environmental sustainability goals by reducing waste generation and minimizing the environmental impact of catalyst disposal.
In this study, the Ni/CeO2 (Ni-C) catalyst exhibited the lowest weight loss during TGA analysis, indicating minimal carbon deposition as shown in Figure 8. This observation strongly correlates with its superior stability and prolonged activity during the DRM reaction. The reduced carbon deposition on the Ni/CeO2 (Ni-C) catalyst suggests that it is more resistant to deactivation, making it a more promising candidate for industrial applications.

TGA analysis of all spent catalysts.
The type of catalyst and its chemical formula profile such as ceria-zirconia-based components can affect the C formation mechanism priorities concerning CH4 activation or CO2 activation as well as the reactivity of the formed C with oxygen-containing species (Makri et al., 2016). The available oxygen in the DRM catalyst surface can provide a favorable environment where CO2 activation, as well as cleavage of the C-O bond, can occur without much hindrance from carbon layer formation, leading to deactivation. These open brasses, often described as lattice oxygen, might spark the oxidation of coke to CO (Marinho et al., 2020).
Conclusion
This study investigated the impact of ceria support origin on the catalytic performance of Ni/CeO2 catalysts for dry reforming of methane (DRM). Three commercially available ceria supports were utilized to synthesize Ni/CeO2 catalysts with a 10 wt% Ni loading. The catalytic performance of these catalysts was evaluated in a fixed-bed reactor at 750 °C.
The results unequivocally demonstrated that the source of the ceria support significantly influenced both the activity and stability of the Ni/CeO2 catalysts. Among the three catalysts, Ni/CeO2 (NI-C) exhibited superior performance, characterized by higher CH4 and CO2 conversions and enhanced stability compared to Ni/CeO2 (NI-M) and Ni/CeO2 (NI-P).
A comprehensive characterization of the catalysts, employing techniques such as BET surface area analysis, XRD, FTIR, SEM, and TGA, revealed that the superior performance of Ni/CeO2 (NI-C) could be attributed to several factors. These include a higher surface area, smaller particle size, and potentially stronger metal-support interactions, leading to enhanced catalytic activity and stability.
Furthermore, TGA analysis provided crucial insights into catalyst deactivation. The Ni/CeO2 (NI-C) catalyst exhibited the lowest weight loss during TGA analysis, indicating minimal carbon deposition. This observation is of paramount importance for industrial applications, as coke formation significantly reduces catalyst lifetime and increases operational costs.
To further enhance the performance of Ni/CeO2 catalysts for DRM, several avenues for future research can be explored to investigate alternative ceria synthesis methods, such as hydrothermal or sol-gel methods, which could enable the tailoring of support properties, including surface area, porosity, and oxygen vacancy concentration, to further enhance catalytic performance. Exploring mixed oxide supports, such as CeZrO2 and CeLaO3, or incorporation of promoters, such as alkali or alkaline earth metals, could further enhance catalyst performance by promoting CO2 adsorption, facilitating oxygen vacancy formation, and mitigating carbon deposition could potentially offer synergistic effects, leading to improved stability and activity.
Supplemental Material
sj-docx-1-adt-10.1177_02636174251330376 - Supplemental material for Role of stable Ni nano catalysts for dry reforming of methane
Supplemental material, sj-docx-1-adt-10.1177_02636174251330376 for Role of stable Ni nano catalysts for dry reforming of methane by Subhan Azeem, Muhammad Ashraf and Sadiq Hussain in Adsorption Science & Technology
Footnotes
Acknowledgment
Author Acknowledge the technical and analytical support from the NFC Institute of Engineering and Technology and Institute of Chemical Engineering and Technology, University of the Punjab.
Authors contributions
Dr Subhan Azeem did all the experimental work, collected the data, and drafted the initial paper.
Dr Muhammad Ashraf did data analysis and its interpretation, followed by several critical revisions of the main draft.
Dr Sadiq Hussainmade revisions in the introduction and experimental sections.
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

