Abstract
The development of adsorbents for removing contaminants from wastewater, which have not only high efficiency, but also the capacity for regeneration and reuse is extremely necessary. In this study, sustainable and efficient adsorbents with perovskite (LaFeO3) and pyrochlore (La2Zr2O7 and La2Fe0.3Zr1.7O7) structures were developed for removing organic pollutants (such as the turquoise blue (TB)) from the aqueous media. They were synthesized by a previously reported synthesis method, and have a specific surface area between 12 and 29 m2 g−1, pH at a point of zero charge from 3.5 to 6.9, and a surface charge density negative value. The adsorbents presented high efficiency for the removal of TB contaminant, with values of up to 98% (q ≅ 294), and they maintained the adsorption capacities and efficiency high (E% > 95%; q > 115) in all three studied regeneration-reuse of adsorbents cycles using the methodology 2 and in five cycles (E% = 70 to 98, q = 27 to 165) using methodology 1. They also proved to be efficient in removing a pollutant compound from real effluent collected from a small local industry, as well as showing capacity for regeneration and reuse in three cycles studied. The perovskite and pyrochlore structures are maintained after adsorption test and regeneration and the TB compound adsorbed is degraded during thermal treatment. The removal mechanism may involve acid–base interactions, hydrogen bonds, electrostatic interactions, and cation–π interactions.
Introduction
In the industrial environment, optimizing time and costs is of paramount importance for processes, but the way companies deal with waste and potential pollutants in effluents is a growing concern, which has led to a growing demand for studies of methods and materials for effective removal of pollutants from aquatic environments (Dehghani et al., 2024; Garba et al., 2020). Among several processes for removing pollutants from aqueous media is the adsorption process, which can be highly efficient for treating different types of contaminants from aqueous media (Dehghani et al., 2024; Garba et al., 2020; Lei et al., 2023; Xiao et al., 2021), but may have the disadvantage of generating secondary pollution due to transfer of the pollutant to the solid with generation of new solid waste with adsorbed pollutant. To avoid this problem it is necessary to eliminate the adsorbed substance and regenerate the adsorbent, however, this is not a simple task and in many cases the regeneration and/or reuse of the adsorbent is unfeasible. Therefore, the need to use sustainable adsorbents to remove pollutants from aquatic environments is clear, that is, those that present not only high efficiency, but a long useful life with a high capacity for regeneration and reuse.
One of the contributors to the contamination of aquatic environments is compounds used in fiber pigmentation. However, during the processes, waste is produced and needs to be treated, because these compounds can present high toxicity to health and the environment, in addition to potentially showing mutagenic behavior for human health. In this context, several forms of textile effluent treatment are studied and applied, in order to reconcile low cost and high efficiency (Dehghani et al., 2024; Gade et al., 2018; Garba et al., 2020; Sousa et al., 2018; Xiao et al., 2020, 2021).
The main characteristics of these effluents are color, high pH, and large amounts of salts. In addition to the toxicity of the compound used in the process and the lack of natural degradation, the color of contaminated water makes it difficult for sunlight to pass through and limits the photosynthesis process of aquatic flora, even when present in small quantities (1 ppm) (Gade et al., 2018; Garba et al., 2020; Sousa et al., 2018; Xiao et al., 2020, 2021). For the removal of the compounds used in fiber pigmentation, conventional methods are as follows: adsorption, coagulation, electrochemical destruction, electrocoagulation, irradiation, membrane filtration, photochemical oxidation, ozonitation, and Fenton process (Dehghani et al., 2024; Gade et al., 2018; Garba et al., 2020; Lei et al., 2023; Lemos et al., 2023; Sousa et al., 2018; Xiao et al., 2020, 2021).
Usually, the materials used in the adsorption processes, for the proposed report, are activated carbon, clays, zeolites, alumina, biomass, agricultural residues, and industrial by-products among others (Dehghani et al., 2024; Gade et al., 2018; Garba et al., 2020; Lei et al., 2023; Lemos et al., 2023; Sousa et al., 2018; Sudiana et al., 2022; Xiao et al., 2020, 2021). Activated carbon has been widely used in wastewater treatment for a long time due to its advantage of higher adsorption capacity for the removal of different pollutants from aquatic environments. However, its use can be limited by the difficulty of the regeneration process after use. Other adsorbents such as zeolites, chitosan, montmorillonite, and cellulose are also designed as pollutant adsorbents from the water, however, the adsorption capacity can be reduced due to the limitation of surface charge of zeolites, the solubility of chitosan in acidic solution, the affinity of montmorillonite to many pollutants are particularly weak. The difficulty of regeneration of all of them by thermal treatment is also a reported aspect, and this can be overcome with the addition of composites containing metallic ions, generally oxides (Dehghani et al., 2024; Liu et al., 2021; Xiao et al., 2020).
Recent works have presented mixed oxides as excellent adsorbents to remove pollutants from aquatic environments (Dehghani et al., 2024; Deng et al., 2019; Gade et al., 2018; Garba et al., 2020; Lei et al., 2023; Lemos et al., 2023; Luu et al., 2016; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Sousa et al., 2018; Souza et al., 2021; Xiao et al., 2020, 2021). Among several mixed oxides studied are those with perovskite and spinnel structures, but studies with pyrochlore structures for use as adsorbents are scarce.
Mixed oxides of the pyrochlore type are ceramic materials with the formula A2B2O7, in which A can be occupied by trivalent cations such as rare earth elements and B is usually tetravalent transition metals from the d-block (Matovic et al., 2023; Subramanian et al., 1983; Thomson et al., 1999). The sum of the charges of the metallic cations must total +14 for the total charge of the structure to be neutral, normally the structures correspond to A3+ and B4+ (Figure 1) or A2+ and B5+. Another factor for the formation of the oxide, the Shannon stability criterion must be considered, with atomic radius values (RA/RB) between 1.46 and 1.80 for A3+ and B4+ and 1.40 to 2.20 for A2+ and B5+ (Matovic et al., 2023; Subramanian et al., 1983; Thomson et al., 1999), below these established values, the structure is classified as fluorite. In addition to the cubic shape, materials with a pyrochlore structure may present symmetry distortions.

Representation of ions coordination in the perovskite (ABO3) and pyrochlore (A2B2O7) structures.
The perovskite structure is another class of materials that can also be called mixed oxide, which structure combines metallic and non-metallic elements. The form that gives rise to other oxides with a perovskite structure has the general form ABX3, where A is occupied by large cations, usually alkali metals, alkaline earth metals, or lanthanides. In B, smaller metallic cations are occupied as transition elements of the d block and X is occupied by oxygen (Garba et al., 2020; Lemos et al., 2023; Moure and Peña, 2015). The sum of the charges must total 6+, considering that sites A and B commonly have 3+ oxidation states (Figure 1). The Goldschmidt tolerance factor is a measure used to evaluate the compatibility of ion sizes in a crystalline structure and stipulates that the value must be in the range of 0.75–1. Within this range, the structure may suffer distortions, meaning that the maximum value indicates an ideal crystalline configuration, which is cubic (Moure and Peña, 2015).
The efficiency of a material of oxide mixed type for a specific application may be related to the synthesis method and composition. Furthermore, the stage of finalizing the synthesis with heat treatment (calcination) can determine the purity and is an important step in obtaining the oxide. Most of the synthesis routes go through heat treatment and calcination to obtain the crystalline network, in which, through high temperatures, the diffusion of ions and decomposition of the precursor material occurs (Garba et al., 2020; Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021). Depending on the method of preparing the mixed oxides, there may be a greater number of secondary phases that also influence the final application. Recent work has demonstrated the use of mixed oxide prepared by modified proteic method and with perovskite structure as adsorbents and/or catalysts for the removal of contaminants from the aqueous medium, including possible reuse of the material in a new process (Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021).
Therefore, in this article, the synthesis of materials with pyrochlore and perovskite structures-based lanthanum, zirconium, and iron by an optimized synthesis method is presented, and the promising use of the prepared materials as adsorbent for the removal of environmental contaminants and with the possibility of reuse in several cycles. Among several environmental contaminants, the turquoise blue (TB) compound was chosen as the model compound and a study comparing the efficiency of the mixed oxides synthesized with the commercial adsorbents was also conducted. With molecular formula C32H16CuN8Na2O6S2, the TB compound is often used as a dye in the textile industry, and it is classified as an anion and its chromophore group is phthalocyanine with a maximum wavelength of 624 nm (Lemos et al., 2023; Sudiana et al., 2022) and has solubility <100 g L−1 at 25 °C. It is a non-biodegradable compound and is considered one of the most toxic compounds that can cause environmental problems and carcinogenic effects in humans.
Several materials were studied for the removal of the TB compound from aqueous media among them, the oxides have gained significance in the removal of this compound with satisfactory results of efficiency and equilibrium time (Lemos et al., 2023; Noreen et al., 2022; Ramesh et al., 2017; Silva et al., 2015). However, regeneration and reutilization studies still are scarce. Lemos et al. (2023) showed that LaNiO3 adsorbent had a high efficiency for removing the TB compound from the aqueous medium (removal efficiency, E% > 90%), and the adsorbent was regenerated and maintained high efficiency (E% > 90%) in six reuse cycles studied. Furthermore, the equilibrium time was relatively short. Therefore, the mixed oxides are sustainable adsorbents, because they combine high efficiency, regeneration, and reuse capacity with highly efficient maintenance.
Materials and methods
Development of materials
To prepare the La2Zr2O7 material, 3.589 g of ZrO(NO3)2·H2O (Scientific ACS-97.0%, Brazil; referring to site B of the oxide structure) was dissolved in 150 mL of distilled water at room temperature and kept under magnetic stirring for 30 min. Subsequently, 6.054 g of La(NO3)3·6H2O (Dinâmica-99.9%, Brazil; referring to site A of the oxide) was added and stirred for another 30 min at room temperature. After this, the system temperature was changed to stabilize at ∼70 °C to add the complexing agent, the collagen (9.643 g). This mass was defined in a 1:1 ratio in relation to metallic cations. After addition, the system remained under stirring for 1 h and 30 min with a constant temperature of around 70 °C. Then, the system was sent for pre-calcination treatment in a muffle furnace for 2 h at 350 °C using a heating rate of 10 °C min−1. The result is a precursor powder which is macerated and calcined at 900 °C for 2 h with a heating rate of 10 °C min−1 in order to obtain the final product. The same procedure was repeated to prepare the material LaFeO3 but using the 7.135 g of La(NO3)3·6H2O and the 6.787 g of Fe(NO3)3·9H2O (Synth-98.0%, Brazil; associated with site B in the oxide structure) and for the La2Fe0.3Zr1.7O7 in which the procedure differs only in the addition of nitrates of metals B at the same time due to two metals present in site B in the structure. The mass used were 6.168 g of La(NO3)3·6H2O, 3.108 g of ZrO(NO3)2·H2O, and 9.716 g of Fe(NO3)3·9H2O. The synthesis procedure was adapted from previous work (Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021). The powders calcined at 900 °C were named LZ-9C for La2Zr2O7, LFZ-9C for La2Fe0.3Zr1.7O7, and LF-9C for LaFeO3 material.
Characterizations of materials
To identify the crystalline phases of the materials, a PANalytical EMPYREAN diffractometer was used with CuKα radiation (λ = 1.540562 Å), 2θ in the range of 10° to 60° and scanning 5° min−1. The phases were identified using one database contained in the Inorganic Crystal Structure Database (ICSD). To determine the average crystallite size, the Scherrer equation was used, through the average of the three main peaks of the pyrochlore or perovskite phase.
The scanning electron microscopy (SEM) images of the calcined samples were observed using a Hitachi High Technologies scanning electron microscope, model TM-3000. The samples were deposited on a copper tape and the images were captured at magnifications in the range of 1000 and 2000 times with a power of 5 kV, using carbon coating.
The specific surface area (SSA) was obtained using Quantachrome equipment (NOVA 1200e model) and the Brunauer–Emmett–Teller mathematical method. Before the analysis, the samples were subjected to degas at 300 °C for 1 h.
The determination of the pH at the point of zero charge (pcz) for the samples obtained after calcination was carried out using the equilibrium method in a batch system. For this, 20 mg of adsorbent (previously dried for 30 min at 60 °C) was added to a 50 mL beaker, and 20 mL of 0.1 mol L−1 NaCl solution was added. The system was subjected to pH adjustments (3, 5, 7, 9, and 11) using 0.1 mol L−1 NaOH or 0.1 mol L−1 HCl solutions. The system was subjected to magnetic stirring for 1 h at room temperature and subjected to the common filtration process on qualitative filter paper. The final pH of the solutions was measured with a bench pH meter (Micronal). To determine the value of the pH at the point of zero charge, a graph of the pH variation versus initial pH was plotted, where the point that intercepted the zero value is the interested value.
Tests for removing pollutants from water
The LZ-9C, LFZ-9C, and LF-9C synthesized materials were tested as adsorbents, seeking better pH and mass, where the studies were carried out with 20 mL of the aqueous solution of the TB (at 3 × 104 μg L−1) and 2 cg of the adsorbent material. The adsorbent used was previously dried at 60 °C for 30 min. First, the pH of the TB solution was adjusted (with NaOH or HCl 0.1 mol L−1) and then the dry adsorbent mass was added. The system was subjected to agitation for 1 h and 30 min. At the end of the time, the solution was subjected to the centrifugation process at 4000 r/min for 10 min and the supernatant was analyzed in the ultraviolet–visible (UV–Vis) spectrophotometer in the range of 400–700 nm. The study was carried out in triplicate at pH values equivalent to 1, 3, 5, 7, 9, and 11. The adsorbent mass study was carried out in Erlenmeyer flasks containing 20 mL of the TB solution (at 3 × 104 μg L−1) with the pH of the natural environment and where 1 cg of adsorbent material was added. The system was kept for stirring for 1 h and 30 min. At the end of the time, the solution was centrifuged at 4000 r/min for 10 min and the supernatant was read in the UV–Vis spectrophotometer in the range of 400–700 nm. The test was carried out in triplicate and the same process was repeated using masses of 2 cg and 3 cg. The TB removal efficiency from water and the amount of adsorbed TB were calculated using the equations mentioned in the literature (Lemos et al., 2023; Santos et al., 2018; Sousa et al., 2018). In this work, the values of efficiency of removal (E%) of pollutants from water are given in percentage, and it is calculated from the initial and in a given time concentration of the model compound. The adsorption capacity (q) of the adsorbents was calculated from the initial and equilibrium concentration of the adsorbate, the volume of the solution, and the mass of the adsorbent, and the q values are given in μg/cg. In order to reduce the amount of residue after the adsorption tests, a small volume of the solution of model pollutant compounds was used, which influences the q values calculated.
The adsorption tests with the effect of contact time were carried out in a Dubnoff bath with Tecnal stirring and heating control, with a temperature of 30 °C using Erlenmeyer flasks containing 20 mL of the TB solution at natural pH (at 3 × 104 μg L−1) and 2 cg of the adsorbent mass. The adsorbent used was previously dried at 60 °C for 30 min. The systems were subjected to magnetic stirring during contact times of 10, 20, 30, 50, 70, and 90 min. In the end, the solution was subjected to the centrifugation process for 8 min and 4000 r/min. The supernatant solution was analyzed by Shimadzu UV–Vis absorption spectroscopy, model UV-1800 in the wavelength range of 700–400 nm. Each analyzed solution was transferred to a 3.5 mL quartz cuvette with an optical path of 1 cm. The entire study was carried out in triplicate. And the procedure was repeated changing the temperatures to 40 °C and 50 °C. After the tests, the adsorbent used in each Erlenmeyer flask was recovered by simple filtration, the content was dried in an oven and the material was stored.
TB solution stability tests against light and solution stirring time (compound photostability) were developed under the same conditions, but without adsorbent. This study was carried out to evaluate the behavior of the compound in solution when exposed to light and agitation.
Real effluent was collected from the textile industry and sent to the laboratory for analysis and adsorption tests. The tests were carried out under the same conditions as the kinetic study presented previously, but at a temperature of 30 °C and using also commercial adsorbents, such as active carbon (CA; Synth-Brazil) and silica gel (SG; Carvalhaes-Brazil).
Other compounds potentially used in fabric dyeing, which may also be potentially contaminating, were also studied, such as methyl violet (MV) 10B(C25H30ClN3)—(Wang et al., 2021), Congo Red (CR) (C32H22N6Na2O6S2)—(Santos et al., 2018; Souza et al., 2021; Wang et al., 2011), TB (C32H16CuN8Na2O6S2)—(Lemos et al., 2023), methylene blue (MB) (C16H18N3SCl)—(Aghashiri et al., 2022; Xiao et al., 2021) and methyl orange (MO) (C14H14N3O3SNa)—(Wang et al., 2021; Xiao et al., 2021). The structures of all studied are presented in Figure 2. The pH measured from each solution was MV (pH = 6), CR (pH = 5), TB (pH = 5), MB (pH = 4), and MO (pH = 4). The studies were conducted at 30 °C, a test time of 90 min, the solution of compounds at 3 × 104 μg L−1, using a mass of 3 cg of LF-9C adsorbent and using also commercial adsorbents, such as CA and SG.
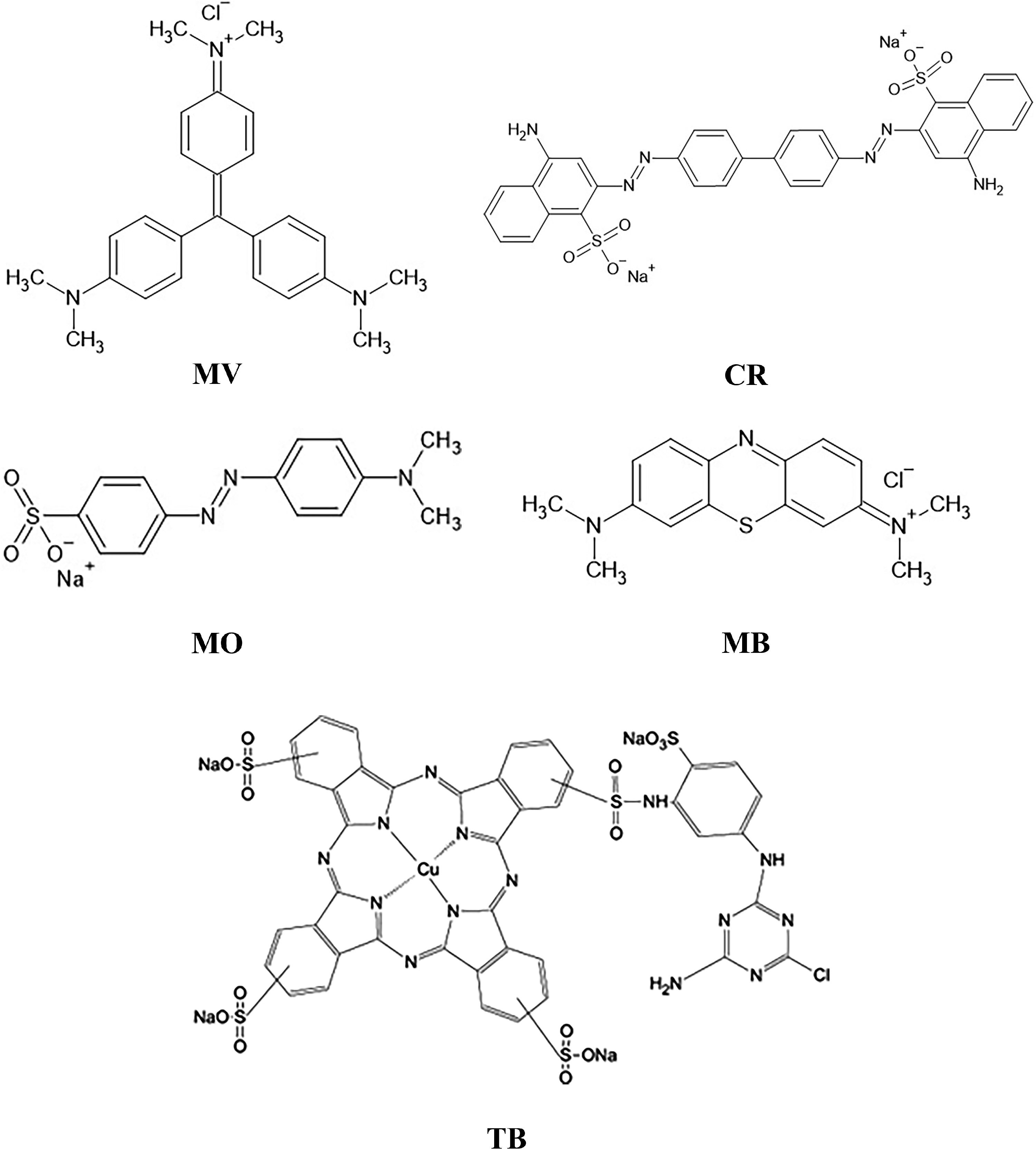
Structures of some potential pollutants for water environments: MV 10 B, CR, TB, MB, and MO.
Adsorbent regeneration and reuse tests
The adsorbent reuse study was developed using the recovered masses of the adsorbent used in the adsorption test performed previously. For this reuse study, Erlenmeyer flasks containing 20 mL of the TB solution (at 3 × 104 μg L−1) and 20 cg of adsorbent material recovered were used. This adsorbent was previously dried at 80 °C for 10 min. The system was subjected to agitation for 90 min and transferred right away to the centrifugation process, the supernatant formed was analyzed by UV–Vis absorption spectroscopy; the result is presented as removal efficiency of the organic compound for cycle 1 of adsorbent reuse. The resultant suspension was subjected to filtration to recover the adsorbent and the resulting filtrate was dried in an oven (80 °C for 10 min) immediately afterward a part of the mass obtained (15 cg) was applied again to a new solution of TB with the same characteristics as the previous solution. This result is presented as the removal efficiency of the organic compound for cycle 2 of adsorbent reuse. This same procedure was repeated four times with each material recovered in the cycle, although using masses of 10, 7, 5, and 2 cg of adsorbent. The results are presented as removal efficiency of the organic compound for cycles 3, 4, 5, and 6 of adsorbent reuse, respectively. This methodology of recovery and reuse of the adsorbent was called methodology 1.
For methodology 2 a similar study was carried out but using 20 cg of adsorbent material recovered which was subjected to calcination in a muffle furnace at 900 °C for 1 h. For cycle 1 of the adsorbent reuse 15 cg of this adsorbent calcined was used, 20 mL of the TB solution at 3 × 104 μg L−1, and the system was subjected to agitation for 90 min, followed by centrifugation and analysis. The difference is that after filtration and drying to recover the adsorbent mass, the solid material obtained is subjected to calcination in a muffle furnace at 900 °C for 1 h. A part of the mass obtained (10 cg) is treated with a new solution of TB with the same characteristics as the previous solution. This same procedure was repeated three times using the mass of 15, 10, and 5 cg in each procedure. The results are presented as removal efficiency of the organic compound for cycles 1, 2, and 3 of adsorbent reuse, respectively.
For the study, a real effluent from the local textile industry was used in methodology 2 for adsorbent recovery, with calcination before each cycle of reuse adsorbent and the masses used were 8, 5, and 3 cg; and the adsorption test was made as mentioned previously.
The materials recovered in methodologies 1 and 2 were characterized by the X-ray diffraction powder method.
Results and discussion
Development of materials
Among various material synthesis methods, the modified protein method is an alternative and low-cost synthesis route that has been used to produce mixed oxides (Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021). The formation of the mixed oxide occurs due to the complexation of the ligand groups with metallic cations, similar to what is reported in conventional methods, however, in the modified protein method, the chelating agent is a substance with a high protein content, so that the reaction takes place with fewer steps, as the interaction occurs directly with the amino acids of the protein composition. Considering the excellent results obtained with the formation of mixed oxides with a perovskite-type structure (Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021), this method becomes relevant for the production of materials with a pyrochlore-type structure. The results presented in Figure 3 shows that it is possible to synthesize a crystalline material from this synthesis method and calcination conditions used.

X-ray diffraction pattern of the LZ-9C, LFZ-9C, and LF-9C materials.
Figure 3 reveals the X-ray diffractogram (XRD) patterns of LZ-9C, LFZ-9C, and LF-9C materials. For the LZ-9C material, the XRD indicates the presence of peaks with values close to standard chart number ICSD-26,851, with a maximum peak at 2θ = 28.46°, attributed to the La2Zr2O7 phase with pyrochlore structure. The other peaks also attributed to phase presented 2θ values at 32.89°, 47.28°, 56.11°, and 58.84°, with pyrochlore structure. The XRD of the LFZ-9C material resembles the diffractogram of the undoped material (LZ-9C), except for the presence of other weak peaks attributed to the presence of impurities at 2θ = 31.83° and 45.79° such as ZrO2 and/or La2O3. In the XRD patterns of this sample, peaks are not identified due to the presence of iron oxides. The presence of weak peaks related to secondary phases may be an indication of the excess or deficiency of reagents. On the other hand, as there are no peaks due to secondary phases of iron oxides, the isomorphic substitution of some Zr (IV) ions by iron (III) ions may have occurred or not in the structure.
For the LF-9C material, the XRD indicates the presence of peaks with values close to the standard chart number ICSD-7794, with a maximum peak at 2θ = 32.21°, attributed to the LaFeO3 phase. The other peaks also attributed to the LaFeO3 phase presented 2θ values at 22.66°, 39.76°, 46.23°, and 57.45°, with perovskite structure. The values are consistent with the literature reported (Aghashiri et al., 2022).
As seen in Table 1, the pyrochlore phase of the LZ-9C and LFZ-9C materials presented small and similar crystallite size values of 14 and 16 nm, respectively. However, the perovskite phase of the LF-9C material has higher average crystallite size values, being 50 nm. These values are consistent with the literature when it comes to mixed oxides (Cheng et al., 2009; Josserand et al., 2022; Lemos et al., 2023), which indicates that several factors can influence the crystallinity and size of the crystal, such as the form of preparation, the metals present in the structure, dopants, among others.
Crystallite size, specific surface area, the pH at point of zero charge, and surface potential (ES) of materials.

ES = surface potential.
Among several factors that influence the adsorption process in the liquid phase, are the pH of the medium and the surface charge of the solid in an aqueous medium at different pH values. Determining the pH at a point of zero charge (pcz) can be useful in adsorption studies in the liquid phase, as it provides indications of conditions that could facilitate the interaction between adsorbate and adsorbent. The pH at point of zero charge values were 6.9, 3.9, and 3.5 for the materials LZ-9C, LFZ-9C, and LF-9C, respectively, also shown in Table 1. Relating these values with the behavior of the adsorbent materials in the presence of different compounds in aqueous solution, it is expected that the adsorption will preferably be of anionic species from the solution when the solution studied is below this pH (6.9, 3.9, and 3.5). This predilection occurs because the surface of the adsorbent must be loaded with cationic species, which facilitates interactions with negatively charged species, as reported in other works (Badawy et al., 2019; Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Souza et al., 2021; Tahir et al., 2020). Furthermore, it is notable that the samples present varying values and that doping and the type of metal ion influence the pH found, since the presence of iron reduces the pHpzc value (pH of the medium in which the charge on the surface of the adsorbent is neutral). This behavior is common in mixed oxides containing iron (Liang et al., 2022) and may be also associated with possible vacancies caused by dopants or even due to the type of surface species that Fe(III) ions induce.
The surface charge density or surface potential (E or ES) in volts was calculated based on the equation proposed by Cao (2004) which is related to pH, the Nernst equation (Equation 1), and pzc and considering room temperature in simplified form:
The micrographs obtained by SEM of all materials calcined at 900 °C can be seen in Figure 4 and showed relative irregularity in powder size, that is, the samples are not in uniformity similar to the report in literary works (Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021). It is also notable that the materials have defined pores on their surface, which can influence the adsorptive behavior when applied as an adsorbent or catalyst.

Micrographs obtained by SEM of LZ-9C (a, b), LFZ-9C (c, d), and LF-9C (e, f) materials.
The SSA values of the LZ-9C, LFZ-9C, and LF-9C materials are 23, 29, and 12 m2 g−1, respectively, as shown in Table 1. The SSA values are consistent with literature data, since, in general, the area values of these oxides are low due to the high temperatures involved in synthesis and calcination. It is also notable that all materials have a slightly higher area value than typically reported in the literature for materials with these structures (Gaur et al., 2011; Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021). It can be seen that the SSA values of the materials are in accordance with the crystallite sizes, as larger crystallites result in smaller areas. This aspect may be related to the synthesis method used, as during the calcination step, the decomposition of collagen produces a significant number of volatiles, which contributes to the generation of porosity in the material (as seen in the SEM images) and subsequently this contributes to an increase in the SSA, as observed in other studies that used collagen during synthesis (Lemos et al., 2023; Souza et al., 2021).
Removing pollutants from water
To evaluate the potential of the materials synthesized in this study as adsorbents, the TB was used as a model compound to simulate a pollutant from water environments. This compound, in addition to being non-biodegradable, is one of the most toxic compounds that can cause environmental problems and carcinogenic effects in humans, besides this, it also contains copper in the structure, which can further aggravate the environmental problems.
The study begins by optimizing the conditions for adsorption tests, such as the pH of the medium, adsorbent mass, and evaluating the photostability of the compound. The TB solution stability study in relation to light (photostability) was carried out in a similar way to the tests, however, without the presence of the adsorbent. Figure 5(a) shows the spectra of the TB solution at different test times. It can be seen that the absorbance values at 626 nm are not reduced or increased considerably over time, which means that the compound in solution is resistant to light and exposure to the environment during the test time.

Spectra UV–Vis of the TB in an aqueous medium submitted at different times of agitation (a); TB solid, in aqueous solution, and in dyed cotton fiber (b).
The adsorption tests for the TB in an aqueous solution began with the study of the ideal pH of the solution for removing the compound of the mean. From the results obtained, it can be stated that the adsorption of the TB is favored in the following order of acidity of the medium for all adsorbents studied: pH 1.00 > pH 3.00 > pH 5.00 > pH 7.00 > pH 9.00 > pH 11.00. In other words, the higher the acidity of the solution, the better the adsorption of the TB in the materials. This behavior can be seen in Figure 6(a), which shows the adsorption capacity of adsorbents for the removal of the TB compound as a function of pH of the middle.

Influence of pH of the medium and mass of the adsorbent (a) and of time of contact (b) in the turquoise blue (TB) removal from aqueous media using the LZ-9C, LFZ-9C, and LF-9C adsorbents.
The TB compound is an anionic species in aqueous solution and when the pH of the medium is below the pHPZC of the adsorbents, the affinity is greater because in an acidic medium, positive charges are created on the surface of the adsorbent and anionic species are attracted more easily, therefore, the adsorption is favored in acidic media, a similar result was reported previously (Lemos et al., 2023; Noreen et al., 2022). However, the adjustment to pH = 1 consumed a considerable amount of hydrochloric acid, and the same occurs for pH = 3, therefore, pH = 5 was ideal considering the moderate adsorption capacities presented and no need for consumables.
The pH is one of the factors that influence the adsorption phenomenon in solution. Different materials have already been used to adsorb TB compound and the pH of the solution has been modified in several studies. For example, biochar created from Prosopis (Ramesh et al., 2017), cashew bagasse (Silva et al., 2015), and mesoporous silica-iron (Tahir et al., 2020) required pH adjustments of the solution, equivalent to pH 6, 2, and 8, respectively.
The mass of adsorbent also proved to be decisive in the adsorption of several compounds in aqueous media. According to Figure 6(a), the adsorption capacities of adsorbents decrease when there is a greater amount of adsorbent mass used in the system. With an increasing dosage of adsorbent, the adsorption capacity of the adsorbent is expected to be lower, considering that the adsorbent mass is used in the denominator of the equation, in addition to the possibility of particle aggregation and a decrease in the number of active sites as previously reported (Noreen et al., 2022). However, an opposite behavior can be observed and has been previously reported (Gaur et al., 2011; Ramesh et al., 2017) and can be attributed to the number of available reactive adsorption sites that cause more adsorbate species to be adsorbed (Xiao et al., 2020).
The results shown in Figure 6(a) and (b) indicate that the adsorbents that contain iron in composition have higher values of adsorption capacities of TB from the aqueous medium. These results indicate a trend contrary to what was expected considering the pzc of the LFZ-9C and LF-9C adsorbents and the pH of the medium. These results suggest that electrostatic interactions are not the predominant factor for interactions between the TB compound and the adsorbents. On the other hand, the results are coherent considering the surface charge density (ES) values, as it follows the trend of the more negative the ES value, presented in Table 1, the higher the adsorption capacity value. Comparing the results presented in Figure 6 with the other data presented in Table 1, such as crystallite size and SSA, we have not found any direct correlation.
Figure 6(b) shows the adsorption capacities of LZ-9C, LFZ-9C, and LF-9C adsorbents to remove TB compound as a function of contact time. The three adsorbent materials were efficient in removing contaminates from the aqueous medium, with adsorption capacities (q) values ranging from 204 to 294 (and efficiency removal values ranging from 68% to 98%) in the equilibrium time. The results indicate that the highest adsorption capacity and removal efficiency values are found in materials containing iron in the composition. For materials with pyrochlore structure, LZ-9C and LFZ-9C, it can be seen that the insertion of Fe(III) ions in the composition causes a significant increase in the efficiency of the material. And for materials with a perovskite structure (LF-9C), a significantly high efficiency is achieved.
Although the time to reach equilibrium was very close when applying the three adsorbent materials, the materials containing iron in the composition, after 20 min of testing already show an adsorption capacity (q) of around 174 to 228 and a TB compound removal efficiency of around 50% to 80%, while when using the LZ-9C adsorbent at this time, the adsorption capacity (q) is 60 and removal efficiency value is 20%. The results indicate that the type of ion present in the composition of the mixed oxide affects the application studied for the materials. When a metal ion is inserted into the composition/structure of a mixed oxide, its properties and characteristics can be inhibited or enhanced. There are reports in the literature of mixed oxides with dopants that enhance existing adsorption properties (Nascimento et al., 2021; Ribeiro et al., 2022; Souza et al., 2021).
The kinetic study was applied using pseudo-first-order (PFO), pseudo-second-order (PSO), and variable constant (VC) models. All applied kinetic models presented determination coefficient values very close to 1, which indicates the effectiveness of applying the models in the study of tests. However, the experimental data presented a better fit for the PSO model. The values of the constants obtained by the PSO model are found in Table 2. PSO equation considers that its mechanism is based mainly on the sharing or exchange of electrons between the adsorbent and the adsorbate, assuming chemical adsorption.
Parameters obtained from the PSO, PFO, and VC models for adsorption of TB using the adsorbent materials.

PFO: pseudo-first-order; PSO: pseudo-second-order; VC: variable constant; TB: turquoise blue.
kn = adsorption velocity constant; qn = maximum adsorption capacities (μg cg−1); R2 = determination coefficient; χ2 = chi-square.
The parameters obtained by the PSO model were q2 which is the maximum adsorption capacity and k2 which is the adsorption rate constant. The determination coefficient (R2) and the non-linear test or chi-square test (χ2) also were obtained. The results confirm that adsorbents containing iron ions in the composition are more effective for the process, as in addition to having greater efficiency for removing the TB compound, they have faster kinetics. The results also indicate that the LZ-9C adsorbent has a significantly higher adsorption capacity than the value found experimentally, although the adsorption rate constant is much lower. This suggests that a long time will be required to reach this maximum adsorption capacity.
In addition to contact time, system temperature also affects the efficiency of adsorbents for removing model compounds from aqueous systems. Aiming to evaluate this parameter, studies were conducted at three different temperatures at equilibrium time (150 min) and Table 3 shows the adsorption capacities of the materials and removal efficiency of TB from the water at different temperatures using LZ-9C, LFZ-9C, and LF-9C materials as adsorbents. When LZ-9C was used the values of E% were 68%, 66%, and 48%, while q values were 224, 200, and 150, for LFZ-9C material the values of E% were 87%, 91%, and 77% for removal efficiency, but q values were 261, 270 and 220 at temperatures of 30 °C, 40 °C, and 50 °C, respectively. It is notable that there is a small variation in efficiency with increasing temperature, but at 50 °C there is a decrease in efficiency for both materials. On the other hand, when using the LF-9C material as an adsorbent, the temperature has practically no effect on the efficiency of the adsorbent to remove the TB compound from the aqueous medium.
Values of adsorption capacity (q) and removal efficiency (E) of turquoise blue (TB) at different temperature tests, using adsorbents synthesized.

E = removal efficiency (%); q = adsorption capacity (μg cg−1).
Again, the results show that the insertion of iron ions in the composition of the mixed oxide results in a more efficient material at any temperature of the system that was studied. This suggests that a greater load of iron ions in the composition of the structure not only results in a more efficient material, but also a more stable one.
LZ-9C adsorbent showed moderate removal efficiency and adsorption capacity, while the LFZ-9C and LF-9C materials proved to be highly efficient in removing the TB, and with greater adsorption capacity, with iron(III) doping being possibly responsible for this behavior.
Several materials have already been used as adsorbents for the TB, such as composites/nanocomposites (Li and Wang, 2017; Noreen et al., 2022; Tahir et al., 2020) alternatives such as active charcoal (Gaur et al., 20011; Silva et al., 2015), biochar (Ramesh et al., 2017), graphene waste (Ambika and Srilekha, 2021), among others (Lemos et al., 2023). The materials generally presented good adsorbents in removing the TB. Although not all samples present enhanced efficiency, mixed oxides with pyrochlore and perovskite structures are promising materials for contaminant removal, as they are highly stable and have the possibility of regeneration after adsorption tests, as previously reported for the other mixed oxides (Garba et al., 2020; Lemos et al., 2023; Nascimento et al., 2021; Ribeiro et al., 2022; Santos et al., 2018; Souza et al., 2021).
The adsorbents LZ-9C, LFZ-9C, and LF-9C were tested in removing a textile dye collected from the effluent from a local textile industry and compared their efficiency for removing the dye from the effluent with other adsorbents such as CA and SG, commercially used materials, and the results are presented in Figure 7. The LZ-9C, LFZ-9C, and LF-9C materials showed 21, 27, and 36 adsorption capacity of textile dye, while the CA and SG adsorbents obtained 75 and 33 adsorption capacity for removal of the environmental contaminant. It is worth noting that the pH of the solution collected in the textile effluent was 7.0, which is not the most favorable pH to obtain better results with adsorbents. Despite having lower or similar values than commercial ones, the results are promising as there is the possibility of reusing the adsorbents.

Adsorption capacity of the LZ-9C, LFZ-9C, and LF-9C and commercial (CA and SG) adsorbents in removing dye from textile effluent.
The relatively low values of adsorption capacities of the adsorbents to remove dye from the textile industry effluent (q < 76) using different adsorbents may be related to several factors, including the presence of compounds that are added during the industrial fabric dyeing process and also during adjustment of the pH of the medium to discard the solution. Therefore, substances other than the dye present are affecting the adsorption efficiency of the dye on the adsorbents, and the pH of the medium is also not very suitable for maximizing the efficiency of the adsorbents.
Adsorbent regeneration and reuse tests
The reuse study of the materials in cycles was carried out, and the adsorption capacity of the LZ-9C, LFZ-9C, and LF-9C adsorbents (Figure 8(a)) and the TB removal efficiency results (E%) reveal excellent behavior in six reuse cycles (Figure 8(b)), using centrifugation, filtration, and oven drying as the adsorbent recovery procedure, recovery methodology 1.

Adsorption capacity of the LZ-9C, LFZ-9C, and LF-9C adsorbents (a, c) and turquoise blue removal efficiency (b, d) during the reuse cycles of adsorbents applying adsorbent recovery methodology 1 (a and b) and recovery methodology 2 (c and d).
The results presented in Figure 8(a) show that there is a gradual increase in adsorption capacity with each subsequent cycle, except for sample LFZ-9C, which did not show trend-following behavior when it went from cycle 5 to cycle 6. Considering that with each cycle there is a gradual decrease in the adsorbent mass, this result was expected. When comparing the adsorption capacity of the materials in reuse cycle 6 (Figure 8(a)) with the adsorption capacity of the virgin adsorbents (Figure 6(b) and Table 3) at equilibrium time, it can be observed that the adsorbents LZ-9C, LFZ-9C, and LF-9C presented a decrease in the adsorption capacity of 50%, 37%, and 67%, respectively. These data indicate that the structure of the material exerts an important influence on the stability in the reuse cycles, where the pyrochlore structure showed greater stability. Furthermore, the presence of iron ions in the composition may be influencing the unexpected behavior of the LFZ-9C sample in cycle 6. The results presented in Figure 8(b) indicate that there is a decline in the removal efficiency of TB as the cycles go by, being more pronounced from cycle 4 onwards.
For the LZ adsorbent, it can be observed that there is a small increase in removal efficiency when going from cycle 1 to cycle 2, and high values are maintained (E > 80%) until cycle 4 (Figure 8(b)). However, from cycle 4 to cycle 6 there is a decrease in removal efficiency, having an equivalent removal efficiency value (E%) in reuse cycle 5 and when using the virgin LZ-9C adsorbent (Table 3), but reaching a total drop in the last cycle of 50% in relation to the virgin adsorbent. On the other hand, doping with iron ions in the material with a pyrochlore structure (LZ) resulted in a more stable material, the LFZ material, as the adsorption capacity and the removal efficiency of TB, in addition to being greater than that of other adsorbents, E (%) is also kept stable in cycles 1 to 4 (Figure 8(b)). After the four cycles, there is a gradual drop in efficiency until the sixth cycle, but the drop is smaller when compared to the undoped material. The LF material showed high-efficiency values in cycles 1, 2, and 3, but it presents a more pronounced drop over the cycles than the other materials, resulting in a drop from the first to the sixth cycle of approximately 67% (Figure 8(b)). This significant decrease in efficiency after six cycles probably occurs due to the difficulty of more species of the TB compound adhering to already occupied sites in the adsorbents. This methodology for recovering and reusing adsorbents results in drops in the material's efficiency throughout the cycles, however, the material with iron ion doping is more stable.
The results of the material reuse study presented in terms of adsorption capacity and TB removal efficiency (E%) using calcination methodology for recovery and regeneration of the adsorbent, methodology 2 (Figures 8(c) and 8(d)) revealed excellent behavior in all cycles studied. The results presented in Figure 8(c) show that there is a gradual increase in the adsorption capacity of all adsorbents with each subsequent cycle, but a more significant increase when it went from cycle 2 to cycle 3. This occurred due to the gradual decrease in the adsorbent mass with each cycle. The adsorption capacities are identical in each cycle, except for the LF-9C material which showed a slightly lower capacity in cycle 3. Figure 8(d) shows that values of removal efficiency of TB are close to 100% for all adsorbents. The results suggest the possibility of the TB used having thermally degraded with each calcination suffered. It can be observed that the LF-9C adsorbent showed a drop of around 12% from the second to the third cycle. In this aspect, it can be suggested that the structure and the ions that compose it play an important role in the stability of the material for regeneration and reuse cycles.
The results indicate that this methodology for recovering and reusing adsorbents in cycles does not result in a decrease in the efficiency of materials for removing TB from the aqueous medium when the structure is of the pyrochlore type in materials with and without doping (LZ and LFZ), however, there is a small decrease when the structure is of perovskite-type (LF material).
Figure 9 shows the XRD patterns of the adsorbents LZ-9C, LFZ-9C, and LF-9C (virgin, with TB adsorbed and subsequent calcination). The peaks that refer to the pyrochlore or perovskite phase are marked with the letter “p.” When comparing the virgin materials with those that underwent calcination after the adsorption tests, it can be noted that the peaks that refer to the main phases are maintained, with values of 2θ = 28.5°, 31.83°, 32°, 47°, and 58° for the pyrochlore phase (Figure 9(a) and (b)) and 2θ = 22.66°, 32.21°, 39.76°, 46.23°, and 57.45° for the perovskite phase (Figure 9(c)). This confirms that the oxide structure is preserved and it is suggested that the TB compound thermally degrades during calcination at 900 °C.

XRD patterns of the LZ-9C, LFZ-9C, and LF-9C materials (virgin, with TBAC); image of the color of the powders as synthesized and with TB adsorbed.
The adhesion of the TB to the powder does not affect the crystallinity and structure of the adsorbent materials, as there were no peaks from other phases with considerable intensity or signs of amorphous material present, except in the XRD of the LFZ-9C and LZ-9C materials, a few low-intensity peaks were identified, which could possibly be due to the crystalline component of the adsorbed dye. In view of the above, it is assertive to state that the prepared materials can be reused in new adsorption tests, given that calcination allows the release of sites occupied by the TB model compound. The color of the powder of the adsorbent materials changed significantly with the adsorbed TB compound in relation to the virgin (V) powder, as can be seen in Figure 9, which can also be a confirmation of the adsorption process.
Through thermogravimetric analysis, it was possible to confirm that the dye is degraded during the calcination step, in previous studies (Lemos et al., 2023). Although all organic material from the dye is not decomposed at 900 °C, a significant part is degraded/decomposed.
The reuse study of synthesized and commercial adsorbents after use in removing dye from industrial effluent and after calcination cycles (Figure 10) revealed that the adsorbent materials synthesized in this work (LZ-9C, LFZ-9C, and LF-9C) have good or excellent behavior in reuse cycles, unlike the commercial SG material, which showed significative decay after the first cycle and the commercial adsorbent CA that did not withstand the high calcination temperature and was decomposed leaving only ash in the crucible, making it impossible to study reuse cycles after calcination methodology for regeneration.

Adsorption capacity of the LZ-9C, LFZ-9C and LF-9C and commercial silica gel (SG) adsorbents (a) and efficiency of dye removal (b) from industrial effluent during reuse cycles of adsorbent materials using adsorbent recovery methodology 2.
Comparing the results presented in Figure 10(a) (use of recovered, calcined, and reused adsorbents) with those presented in Figure 7 (use of virgin adsorbents), for LZ-9C and LFZ-9C adsorbents, it can be seen that during cycles 1 and 2 the adsorption capacities values are greater than when is used the virgin adsorbents. An opposite result was expected considering that a greater mass of adsorbent is used during cycles 1 and 2. It can be attributed to the greater number of available reactive adsorption sites that cause more adsorbate species to be adsorbed. While with the use of LF-9C recovered material (in cycles 1 and 2) the value is lower than when the virgin adsorbent was used. On the other hand, in cycle 3, the amount of adsorbent is the same as that was used for the virgin adsorbents, but in this case when using the materials recovered LZ-9C and LFZ-9C the values are practically the same, while with the use of LF-9C recovered material the value is lower. The results indicate that the SG only maintained the adsorptive capacity at significant values in the first cycle, as in the second and third cycles the drop was drastic and within the experiment's margin of error. In other words, the SG only tolerated one calcination cycle.
Figure 10(b) shows the efficiency of the dye removal from industrial effluent during reuse cycles of adsorbent materials using adsorbent recovery methodology 2. The results indicated that after cycle 1 there is a gradual drop in efficiency until the third cycle, except for SG that have a more significant drop. In cycles 1 and 2, the removal efficiency values are greater than in cycle 3 as the amount of adsorbent is greater in cycles 1 and 2. The results presented from the third reuse cycle indicate that the LZ-9C and LFZ-9C materials have an efficiency slightly higher, while the LFZ-9C material has a slightly lower efficiency compared to the virgin adsorbents.
Comparing the results shown in Figures 8(d) and 10(b) (and Figures 8(c) and 10(a)) (both dealing with adsorbent reuse cycles, but using TB as a model compound and using a real effluent), it can be seen that during the regeneration and reuse cycles, the results did not follow the same trend. The observed drop in E% values (Figure 10(b)) and in adsorption capacities values (Figure 10(a)) can possibly be attributed to other species present in the industrial effluent that affect not only the E% and q values but also the regeneration of the adsorbents.
Studies carried out by Deng et al. (2019) reported the use of LaFeO3 perovskite oxide decorated active carbon fibers (LFO-ACFs) to remove Rhodamine B (RhB) dye from the water with excellent results. The adsorption kinetics obeyed the PSO model and with excellent regeneration of LFO-ACFs realized through an easy pyrolysis method.
Garba et al. (2020) presented a review of the preparation, characterization, and potential application of perovskites as adsorbents for wastewater treatment. The results clearly show a very good performance by the perovskite materials for the adsorption of various adsorbates. Several advantages of perovskite materials as adsorbents were presented as renewable, effective, economical, environment-friendly, and sustainable materials.
Previous studies by Lemos et al. (2023), Souza et al. (2021), Santos et al. (2018), Nascimento et al. (2021), and Ribeiro et al. (2022) showed that adsorbents with a perovskite structure had high efficiency for removing different compounds used in dye textile (TB, CR, blue and red Bezaktiv matrix) from the aqueous medium, and with a capacity of regenerated and maintained high efficiency in several reuse cycles.
Given the results presented, it can be proposed that all the synthesized materials are very promising for this particular application, with emphasis on the materials containing iron in the composition: LF-9C and LFZ-9C. Considering that the LFZ and LF materials were prepared by the same synthesis method and that the characteristics applied for the removal of the model compound from aqueous media and capacity for regeneration and reuse are similar, however, the cost of iron(III) salts for the preparation is much smaller than zirconium(IV) salts, it can be considered that the LF material has the best cost–benefit ratio.
Removing pollutants from water environments using LaFeO3 material
Considering the efficiency results for removing contaminants from the aqueous medium and the reusability capacity obtained with the LF-9C material, and considering the lower cost in the production of this material compared to the LFZ-9C material, studies were carried out on the removal of different compounds from the aqueous medium using the LaFeO3 (LF-9C or LF) material as an adsorbent. The compounds studied are shown in Figure 2 and the results of the adsorption capacities of the adsorbents from the aqueous media using LF-9C (LF) and commercial (CA and SG) adsorbents are shown in Figure 11. It is important to highlight that the experiments were conducted without adjusting the pH of the medium and that they were all carried out under the same conditions (temperature, time, concentration of the model compound solution, and mass of adsorbent).

Adsorption capacity of the LF and commercial (CA and SG) adsorbents in removing different organic compounds from the aqueous media.
Of the materials studied, two adsorbents are widely used and studied, CA and SG. The CA adsorbent showed high efficiency and adsorption capacity for removing all compounds studied with values above 96% (E% > 96 and q > 190), as expected since CA is an efficient adsorbent for several types of different compounds, such as dyes, pesticides, and metal ions, in accordance with already related (Dehghani et al., 2024; Deng et al., 2019; Lei et al., 2023; Xiao et al., 2020, 2021). On the other hand, SG showed a moderate adsorption capacity for removing MB and MV and a very low adsorption capacity for removing MO, CR, and TB. The LF-9C material developed in this work showed a high adsorption capacity for removal of TB and CR from the aqueous medium, a low adsorption capacity for removing MV, and was not efficient for removing MO and MB from the aqueous medium, considering the adsorption capacity was very low.
The LaFeO3 material with a perovskite structure was studied in the literature to remove some compounds as dyes from the water environment (Deng et al., 2019; Garba et al., 2020). Deng et al. (2019) reported the use of LFO-ACFs in the removal of RhB dye from the water. They suggested that several factors influence the adsorption process, such as the presence of La3+ and Fe3+ ions in the perovskite structure as well as the presence of several surface OH− groups, promoting various interactions between RhB and LFO-ACFs, such as electrostatic and hydrogen bonding, π–π stacking, and cation–π interactions.
Liu et al. (2021) developed sugar-based carbon using lanthanum and cobalt bi-metal species (La/Co-SC) for effective adsorption of MO. They proposed that La/Co-SC exhibits an excellent adsorption performance with the adsorption process being endothermic and it obeyed the PSO kinetic model. Moreover, La/Co-SC shows a good regeneration and, in the mechanisms, proposed, the driving forces of MO adsorption by La/Co–SC are not only attributed to electrostatic interactions, there are hydrogen bonding and π–π interaction throughout the adsorption process. They proposed also several possibilities of interaction between the adsorbent and adsorbant, such as hydrogen bonding (O and H atoms of La/Co-SC with N and O atoms of MO). Acid–base interaction between acid groups in the La/Co–SC and base groups in MO species.
The compounds studied (shown in Figure 2) have different structures and are typically used as soluble compounds in aqueous media, which can give them cationic, anionic, or neutral characteristics when in aqueous solution. In this group, there are cationic, anionic, or even without load on the structure. The CA adsorbent used in this study showed high adsorption capacity and efficiency for removing all compounds whether cationic, anionic, or neutral in an aqueous medium. On the other hand, the different values found for the adsorption capacities of different compounds when using the LF-9C mixed oxide suggest that there are different interactions between them and the LF-9C adsorbent synthesized in this work. The adsorption capacities to remove the MB (typically considered cationic) and MO (typically considered anionic) compounds from the medium were extremely low, within the margin of error of the experiment, while that to remove the TB and CR (typically considered anionic) were high. However, this is not the only factor that affects the interaction between the adsorbent and the studied compounds since MV is also considered a cationic compound and there is removal of this compound from the aqueous media. If only the pzc value is considered for the LF-9C adsorbent and the pH of the aqueous medium in which the studied compounds are found, preferential adsorption of cationic species, that is, of the MB and MV compounds, should occur. As expected, and related in the literature (Deng et al., 2019; Garba et al., 2020; Xiao et al., 2021), the interaction between opposite charges is not the only factor governing the interaction between the compounds studied and the adsorbents. Once again, we found that attractive electrostatic interactions are not the predominant factor of interaction between the studied compounds (CR, TB, MV, MO, and MB) and the LaFeO3 adsorbent.
Comparing the results presented in Figure 11 with the structures of the compounds shown in Figure 2, we found that the compounds that have sulfonic groups (CR and TB) in the structure were removed from the aqueous medium by the LF-9C adsorbent very effectively (showed high adsorption capacities values) and those that do not have this group (MB and MV) the values were very low or median. On the other hand, the MO compound has this group in its structure but its removal from the medium was insignificant using LF-9C adsorbent.
In previous studies (Lemos et al., 2023; Santos et al., 2018; Souza et al., 2021), it was highlighted that the interaction between the sulfonic groups of the studied model compound (CR and TB) and the cations of the mixed oxide structure (La3+ and Ni3+) exerts a significant influence on the removal of compounds from the aqueous medium by adsorption. In addition, the pH of the medium and pzc and ES of the adsorbent also favor or not a greater electrostatic interaction between the compound and the adsorbent. Simultaneously, there must also be cation–π interactions, acid–base interactions, and hydrogen bonds as illustrated in Figure 12 of the possible interactions between the ions of the mixed oxide structure and the TB compound.

Possible interactions between the ions of the mixed oxide structure and turquoise blue (TB) compound in aqueous media.
Therefore, the interaction between the TB and CR compounds with the La3+ and Fe3+ ions present in the LaFeO3 adsorbent structure results in very high values of adsorption capacity (and removal efficiency) because several factors that govern the interaction are more suitable. The studies using the MO and MV compounds and LF-9C adsorbent suggest that the pH of the medium significantly disfavored the interaction of the MO compound with the adsorbent and that is why attractive electrostatic interactions are weaker than repulsive ones. The higher adsorption capacity for removing other compounds (MB, MO, and MV) using the LF-9C adsorbent can be obtained according to the variation of experimental conditions such as adsorbent mass, pH of medium, experiment time, and temperature are adjusted.
Conclusions
Mixed oxides of the type La2Zr2O7 and La2Fe0.3Zr1.7O7 with pyrochlore structure and LaFeO3 with perovskite structure can be formed when they are prepared by the modified proteic method followed by calcination at 900 °C for 2 h. The choice of metals and the proportion of doping with Fe(III) proved to be adequate, since the structure of the desired oxides was confirmed. The SSA and zero charge point values of the calcined materials ranged from 3.5 to 6.9 and the SSA ranged from 12 to 29 m2 g−1. From the adsorption tests, it was found that the pH = 5 for the TB solution and that the mass of 2 cg of adsorbent is suitable for removing the TB from the water. LZ-9C, LFZ-9C, and LF-9C materials showed high adsorption capacity (q ≅ 294) and removal efficiency of TB (values of up to 98%) in the maximum test time at different medium temperatures. It was also found that the PSO kinetic model is the one that best suits the values obtained for the adsorption kinetics. The result of reuse studies of the adsorbents, using different recovery and regeneration methodologies, are very promising, as the materials maintained excellent adsorption capacities and efficiency values in removing contaminants from the synthetic solution and industrial effluent, they maintained the adsorption capacities and efficiency high (q > 115; E% > 95) in all three studied regeneration reuse of adsorbents cycles using the methodology 2 and in five cycles (q = 27 to 165; E% between 70 and 98) when applied methodology 1. The perovskite and pyrochlore structures of the adsorbents synthesized are maintained after the adsorption test and regeneration and the compound adsorbed is degraded, which indicates that the materials are very promising as they can be regenerated to be reused several times, maintaining efficiency high level for application on an industrial scale. The LaFeO3 is selectivity to several compounds that are contaminants from the aqueous media and the mechanism of interactions is not only attributed to electrostatic interactions, but may also involve acid–base interactions, hydrogen bonds, electrostatic interactions, and cation–π interactions. In future studies, the authors intend to use the data obtained in this work to study the scale-up of experiments with the design of a fixed-bed continuous flow adsorption column.
Footnotes
Data availability
All raw data used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CNPq for the development of this research (Proc. 313486/2020-4 and 308989/2023-6). This work was carried out with the support of the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES—Financing Code 001 and by CLQM (Multi-user Chemical Laboratory Center) of the Federal University of Sergipe for the support and analytical infrastructure provided.
