Abstract
The increasing environmental concerns associated with textile dye pollution necessitate the development of sustainable and eco-friendly treatment methods. Among various natural biosorbents, Avicennia marina (true mangrove) emerges as a promising candidate due to its robust growth in coastal ecosystems and its potential for dye adsorption. This research delves into the feasibility of employing A. marina as a green biosorbent for the decolorization of methyl red (MR), a prevalent textile dye. Through comprehensive experimentation, we examined the effects of various parameters, including the contact time (20–120 min), pH (1–10), temperature, dose (25–150 g/L), and initial dye concentration (20–600 mg/L), on the adsorption capacity of the biosorbent. Characterization of the prepared biosorbent was done using atomic force microscopy(AFM), scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and X-ray diffraction analysis (XRD). The FTIR results show phenobarbitone containing OH, –CH, –NH2, COOH, –CO, C=C=N, NO, and C=C groups. SEM image indicated long-fiber-bundle and honeycomb-structure parenchyma cell structure found between 1 µm and 2 µm, and XRD peaks exhibited hemicellulose at 2Ɵ=15.01 and cellulose at 2Ɵ=22.03. A. marina exhibits excellent adsorption capacity for MR (73% decolorization at a concentration of 500 ppm occurred in highly acidic conditions (pH = 1) after 75 min of contact time), making it a promising candidate for industrial wastewater treatment. The study emphasizes pseudo-second-order kinetics and the Freundlich adsorption isotherm as the best-fitted model for MR adsorption. Furthermore, the adsorbent demonstrated a commendable removal efficiency for MR dye in actual water samples. The removal efficiency of dye in industrial wastewater, CETP, and Sabarmati river discharge is 67.18%, 82.07%, and 77.55% respectively, with a time duration of 20–120 min. The findings underscore the potential of harnessing natural resources like A. marina for sustainable water management, contributing to the broader goal of achieving zero-waste economies.
Introduction
The widespread industrial applications of dyes have undeniably revolutionized numerous sectors, from textiles to pharmaceuticals, by offering vibrant colors and esthetic appeal (Bharagava et al., 2020). However, this convenience comes at a price, as the disposal of dye-laden wastewater into natural ecosystems has raised significant environmental concerns (Shindhal et al., 2021). The industry that emits dye as effluent is ranked as the tenth most polluting, accounting for 17–20% of total water pollution (Johansen et al., 2022; Mao et al., 2021). Due to their widespread use in the textile, paint, paper, and food sectors, some 5000–10,000 tons of dye waste are dumped into water bodies each year (Saharan et al., 2023). The discharge of dye-contaminated wastewater interferes with photosynthesis in autotrophic organisms, resulting in a decrease in the productivity of aquatic plants and animals (Hashem et al., 2020). Many of these dyes possess complex aromatic structures, rendering them resistant to degradation by heat, light, oxidizing agents, and microbial attack (Selvaraj et al., 2023). Furthermore, some dyes demonstrate high toxicity and are recognized as allergenic, mutagenic, teratogenic, and potentially carcinogenic to humans (Deb et al., 2023; Mariyam et al., 2021).
Among these dyes, methyl red has attracted particular attention due to its ecological and health hazards. It is widely utilized in the textile and printing industries (Okoro et al., 2022). Because of its vivid color in aqueous environments, MR is commonly employed as a model dye for numerous experimental research. The release of MR into receiving bodies influences aquatic and human life, resulting in various harmful consequences (Ahmad et al., 2015; Hanafi and Sapawe., 2020; Islam et al., 2023. The presence of benzene rings limits the dye's capacity to degrade naturally. Methyl red is mutagenic; it experiences biotransformation into 2-aminobenzoic corrosive and N-N'-dimethyl-p-phenylenediamine (Chung and Enzinger., 1981) and carcinogenic, mutagenic, and teratogenic substance known to cause respiratory issues, digestive system infections, skin and eye irritation, and teratogenesis (Khan and Khan., 2018). Conventional wastewater treatment methods often prove inadequate in effectively removing such persistent dyes, necessitating the exploration of innovative, sustainable, and eco-friendly alternatives (Ramaraju et al., 2014; De et al., 2016. Several efficient techniques are available for treating industrial wastewater tainted with dyes. Various methods can be employed to treat wastewater, such as oxidation, electrocoagulation, filtration, ion exchange, activated sludge, and microbial attack.
Recent research has emphasized the potency of these approaches. To remove dye contaminants from wastewater, various physical and chemical methods have been utilized (Doondani et al., 2022; Sharma et al., 2022; Wei et al., 2021), including adsorption, filtration, membrane separation, and phytoremediation. There are several methods for removing harmful dyes, but adsorption is the most efficient, cost-effective, and accessible. Dye decolorization can be accomplished sustainably with natural biosorbents. In recent years, utilizing natural biosorbents has emerged as a promising strategy to mitigate the environmental impact of synthetic dyes. Natural biosorbents from raw renewable materials have demonstrated remarkable potential for adsorbing and eliminating various pollutants, including synthetic dyes, from aqueous solutions. Within this burgeoning field, Avicennia marina, commonly known as the gray mangrove, has emerged as a particularly intriguing candidate for the decolorization of methyl red. Avicennia marina, a true mangrove species renowned for its adaptability and resilience in coastal ecosystems, has garnered attention for its potential in dye adsorption. Among them, the adsorption technique is considered to be one of the superior techniques because of its cost effectiveness, easy design, efficiency, availability of numerous sustainable adsorbents, and ability to treat different types of pharmaceuticals effectively (Deb et al., 2023).
A prior literature study shows that mangrove resources can be used as a green biosorbent to remove various dyes. The results indicate that chemically treated and raw mangrove leaves remove 98% and 75% of 304 methyl violet 2B dye (Oloo et al., 2020). It also emphasizes the critical role that mangrove species play in the phytoremediation of pollutants, such as dyes, that may be discharged into marine ecosystems. The removal of methyl red by Avicennia marina involves various mechanisms, including physical adsorption, electrostatic interactions, ion exchange, and chemisorption. The abundance of functional groups on the surface of Avicennia marina contributes to the strong affinity for Methyl Red molecules (Tan et al., 2010). Many authors have reported in their studies that decolorizing dyes from mangroves is a natural phytoremediator. The study (Bano and Zahir., 2016) looks at the adsorption characteristics of acidic and basic dyes on chemically treated mangrove bark. An acidic formaldehyde solution was treated with the powdered Rhizophora apiculata barks to produce a non-soluble dye adsorbent. The study showed that the modified barks had a monolayer adsorption capacity of 129.87 mg g−1 for the basic dye malachite green and 12.72 mg g−1 for the acid dye sunset yellow FCF. The adsorption kinetics of both dyes were found to fit the pseudo-second-order kinetic model. These findings imply that mangrove bark might effectively remove textile dyes from effluents at a low cost (Bano and Zahir., 2016).
The use of mangrove bark has been bolstered because of its wide availability as charcoal industry waste. This industrial waste is burned for energy generation or discarded on landscapes, and no high-value applications have been discovered yet. First, it transforms undesirable and surplus agricultural waste (billions of kg per year) into value-added adsorbents. Second, the adsorbent may be obtained cheaply from raw materials. Therefore, this studies aimed at increasing the value of such a residue are critical for the economies of nations where this species grows abundantly. Mangrove bark was chosen for this study because it is inexpensive, plentiful, and environmentally acceptable, and it includes phytochemicals such as steroids, tannin, flavonoids, saponins, and phenolic acids, which function as reducing agents. It also includes hydroxyl, carboxylic, and phenolic groups, which may absorb dyes at suitable pH and temperature conditions.
This paper aims to comprehensively investigate A. marina's capabilities as a green biosorbent for MR decolorization, focusing on optimizing the adsorption process and evaluating its economic implications. Several factors were examined, including pH, concentration, contact time, and ionic strength. Despite being a natural adsorbent, the adsorbent proved highly efficient at removing dye. Moreover, it demonstrated strong MR dye adsorption in textile inlets, outlets, seawater, and river water. This study aims to mitigate dye pollution caused by industrial effluents in aquatic bodies by using Avicennia marina as an adsorbent.
Material and method
Material
The study site was a mangrove ecosystem and a coastal wetland near Purna Estuary in Navsari district, a southern part of Gujarat. It ranges from longitude 72°44’ E to 72°55’ E and latitude 20°55’ N to 21°01’ N (Vaish and Pathak., 2020). The mangrove plants and bark were collected from the Navsari region near the Purna River. GEER Foundation, Gujarat, characterized the mangrove species. In the present study, Avicennia marina (mangrove) bark is used as a natural biosorbent whereas an anionic dye methyl red (M/s Sigma> 95%), KBr (M/s Sigma 99.9%), methanol (M/s Himedia; 99.8%), HCl (M/s Himedia;35%), NaOH (M/s Himedia;98%), distilled water, electronic weight balance (M/s BR Biochem Life sciences, Delhi), water bath shaker (M/s York Scientific Industries Pvt Ltd), oven (M/s Sciencetech), pH meter (ROCI, Delhi), UV–VIS spectrophotometer (M/s Shimadzu: UV-1900i), vortex (M/s Tarsons-3020), refrigerator (M/s LG), centrifuge (M/s Remi Electrotecnik LTD), FTIR (M/s Perkin Elmer Model: SP-65), XRD (M/s Bruker; AXS D8), AFM (Model: XE-70 M/s Park System), SEM (M/s Carl Zeiss; EVO 18).
Methods
Preparation of adsorbent and adsorbate
The mangrove plant species were collected from the Purna Estuary, Navsari region. The collected bark sample was washed with distilled water 2–3 times and left to air-dry thoroughly. Before converting into powder form, the bark sample was crushed into small pieces and grinded into a mixture grinder. After grinding, sieve the bark powder using 250 mesh as shown in Figure 1. The biosorbent was then analyzed through various sophisticated instruments for characterization. The prepared biosorbent was stored for further use. Methyl red was purchased from Sigma Aldrich (Figure 2). The stock solution of 500 ppm of dye was prepared with an equal amount of dye in distilled water. The various amounts of methyl red dye prepared the different stock solution concentrations.
Systematic diagram for synthesis of adsorbent from mangrove bark.

Chemical structure of methyl red.
The study investigated the adsorption of methyl red in a batch experiment using mangrove bark species as an adsorbent. During the investigation, 100 mg of the adsorbent was mixed with 10 mL of MR dye and continuously stirred at 298 r/min and room temperature (25 °C). During the decolorization process, factors such as pH, adsorbent size, dosage, adsorbate concentration, and adsorption contact time are also responsible for reaction conditions.
Point zero charge
After undergoing a batch equilibrium procedure with a 0.01 M NaCl solution, they analyzed the point zero charge value of the bark of Avicennia marina. The pH value at point zero charges (ΔpH= pHi– pHf) was plotted against pHi to create a graph. By identifying the intersection of the curve and the x-axis, the adsorbent can calculate the pHZPC value.
Results and discussion
Effect of adsorbent dose on different dye concentration
The number of active sites on the adsorbent determines the performance of the adsorbent for dye removal in removing azo dyes from textile effluents. The amount of adsorbent used in adsorption is crucial because it establishes the system's adsorbate–adsorbent equilibrium and forecasts the adsorbent treatment cost based on the quantity of dye solution (Wekoye et al., 2020). The impact of the adsorbent amount on the bark adsorption was investigated using adsorbent doses ranging from 25 to 150 mg/L and dye concentrations ranging from 20 to 600 mg/L (Figure 3). The findings revealed that the removal efficiency increased significantly as the amount of adsorbent was raised, achieving a peak efficiency of 74% at a dye concentration of 500 ppm with 100 mg of adsorbent. Initially, the removal efficiency rose rapidly from 68% to 74% as the dosage increased to 150 mg/L, after which a decline was observed due to the saturation of active sites. This is due to increasing the amount of adsorbent enhances dye removal efficiency up to a saturation point, this relationship is complex and influenced by both the properties of the adsorbent and the structural characteristics of the azo dyes. Specifically, the observed increase in efficiency between 25 mg and 100 mg of adsorbent can be explained by an increase in available binding sites. However, as saturation occurs, further increases in adsorbent quantity do not yield proportional improvements in dye removal.

Effect of adsorbent doses (25–150 mg) at different dye concentration (20–600 mg/L).
Adsorption of MR influenced by pH
A solution with a concentration of 500 ppm was prepared and then divided into ten beakers, with each beaker containing a total volume of 10 mL. Each solution was maintained in the pH value range between 1 and 10 using 0.5 M HCl and 0.5 M NaOH. Methyl red is acidic, with a pH value of 4.5–6.5. The pH meter altered the dye solution pH with dilute HCl (0.1N) or NaOH (0.1N). Figure 4 shows that the MR decolorization efficiency is vital in an acidic medium. As the pH increases, the adsorption capacity of bark (Avicennia marina) decreases. For MR, the adsorption is high in an acidic medium and decreases as the solution pH increases. Adsorbents contain functional groups that modify dye surface charges. As shown in Figure 4, pH influences the bark's adsorption capacity (%). The 73% adsorption efficiency was highest at pH 1, giving the best results. However, the adsorption capacity decreased significantly as the pH increased from 2 (46%) to 3 (16%). Bark's adsorption capacity decreased gradually until reaching 6% at pH 9 and remained the same at pH 10 (Ahmad et al., 2015). MR is an anionic dye that releases an H+ ion in an acidic medium, so that these cations will occupy the active sites of the adsorbent, and the positive sites of the ions will adsorb the MR molecules. A lower pH value shows the MR dye protonation. This led to electrostatic attraction between the negatively charged dye and the positively charged bark adsorbent. Electrostatic interaction reduces adsorption efficiency by repelling negatively charged dye from positively charged surfaces. At higher pH values, fewer positively interacting sites in the solution lead to less electrostatic attraction between the adsorbate and adsorbent surfaces due to more hydroxyl groups, which will compete with the anionic dye molecules for the adsorption sites (Conde et al., 2020). The study primarily focused on pH 1 to10, and the best optimum result was obtained on pH=1 because it provides valuable insights into adsorption under acidic conditions, the broader pH range effects remaining unexplored. The study's generalizability may be limited as different materials exhibit varying adsorption behaviors. Regarding the experimental part, the study duration was 2 h, potentially overlooking long-term effects and kinetics, especially in real-world scenarios with extended exposure. Hence, it is essential to conduct separate studies to quantify dye adsorption with a wide range of pH of the adsorbates concerning the time duration.
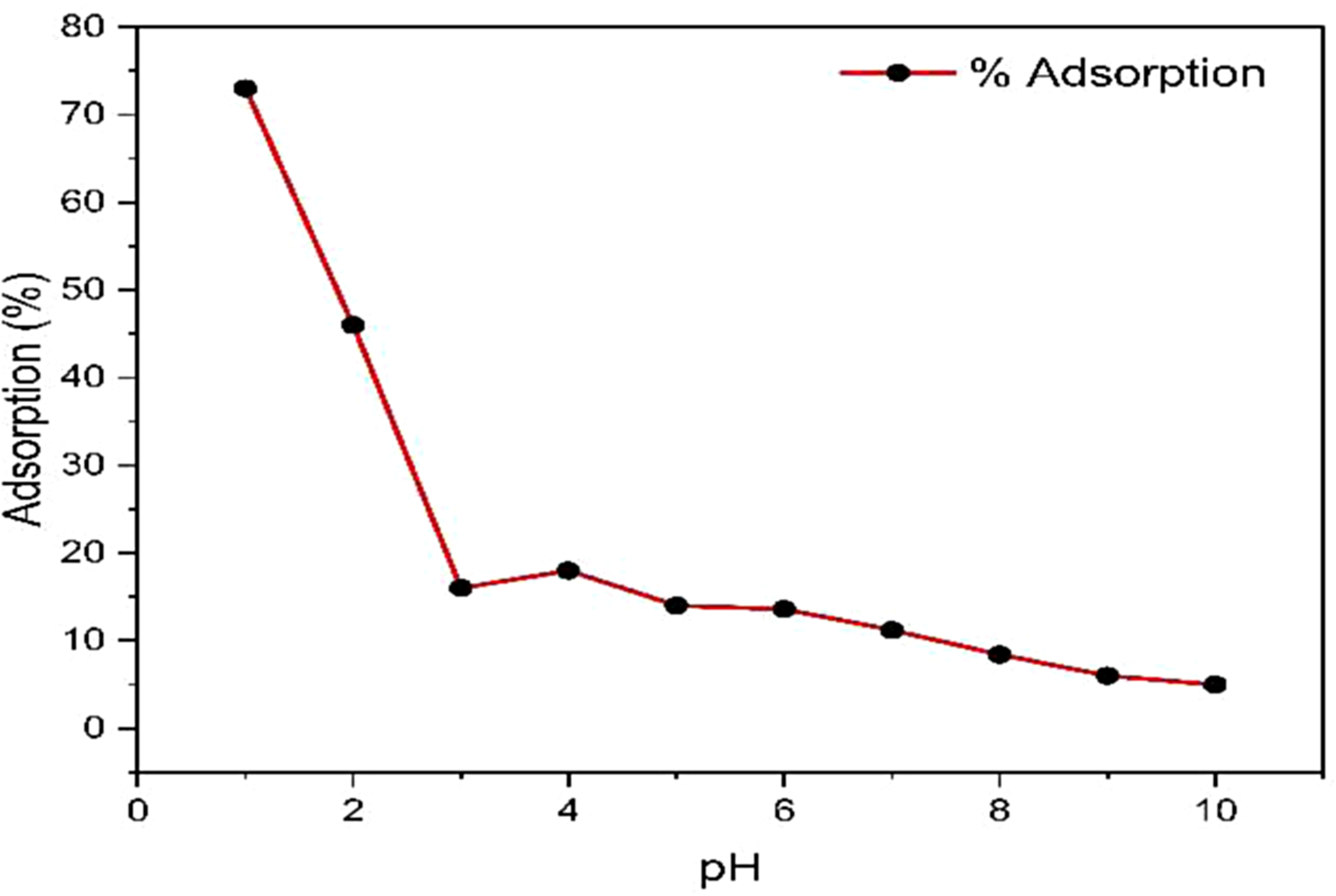
The adsorption efficiency of methyl red at different pH.
Point zero charges of the adsorbent
The concentration of hydrogen ions in a material is closely linked to the surface charge of particles, significantly impacting a range of material properties. This influence extends to the behavior of colloidal materials, affecting their stability, aggregation, and interaction with electrolytes on different substrates. The optimal value of the point of zero charge (pHzpc) for treated bark, an adsorbent, is 4, indicating a globally neutral state with the maximum presence of negatively charged ions. At this point, the adsorbent surface becomes highly conducive to adsorbing cations due to the prevailing negative charge. Significantly, the solution pH surpasses the pHzpc when the surface charge becomes negative, as depicted in Figure 5. This dynamic shift in surface charge has implications for ion exchange capability and the overall adsorption efficiency of the material (Shukla et al., 2002). In this study, the surface charge of the adsorbates are kept similar throughout the study, hence it is important to explore the surface charge dependent dye removal efficiency of the adsorbates, keeping the other parameters such as time duration of the treatment, dose, and pH.

Determination of point of zero charge.
Adsorption of MR influenced by temperature
Temperature has a significant impact on adsorbent adsorption capability. The capacity of the adsorbents to hold onto the dye molecules is decreased by high temperatures, which break the covalent bond. The chemical interaction of the functional groups on the adsorbent surface with the adsorbate largely determines the adsorption capacity (Jayarajan et al., 2011). Temperature influences on dye absorption were investigated using different temperatures, such as 20 °C, 25 °C, and 30 °C, as revealed in Figure 6. The maximum dye removal efficiency of 74.16% was attained at 30 °C within 75 min and then increased somewhat when the temperature was raised further. More studies are needed to study the temperature-based kinetics of dye removal. This study explores only a narrow range of temperatures (20–30 °C).

Effect of temperature (20–30 °C) at different time interval (20–120 min).
Effect of dye concentration on adsorption
Different solutions ranging from 20 to 560 ppm was prepared through 600 ppm stock solution of MR Dye. 0.5 M HCl solution is added to maintain the pH of the reaction. The bark powder of Avicennia marina (100 mg) was added to each solution. After shaking at 350 r/min for 120 min, the suspension adsorbent was centrifuged to separate it from the solution. Figure 7 illustrates that adsorption capacity increases with initial adsorbate concentrations due to effective collisions between adsorbate and adsorbent. Recent findings indicate that the highest adsorption (70%) is achieved when using 100 mg of powdered Avicennia marina bark in a 500-ppm solution. This information can be beneficial in optimizing adsorption processes to remove pollutants from various sources. The dye uptake mechanism mainly shows the equilibrium at the initial dye concentration. The results show that dye concentration increases and that adsorption efficiency by bark decreases due to a decrease in surface area and an increase in dye concentration of the adsorbent active sites; hence, adsorption efficiency was decreased. As this experiment was conducted with a concentration up to 100 mg of adsorbate and 600 mg of dye. Above and below doses of adsorbate and dye need to be explored further.

Influence of Ci on the adsorption of methyl red via adsorbent.
Effect of contact time on adsorption
Different rotation times and initial MR concentrations were used to study the effects of contact time and initial dye concentration on adsorption capacity, as shown in Figure 8. (Dawadi et al., 2020). The findings obtained show that adsorption increases with time in contact (20–120 min) and eventually reaches equilibrium at 75 min (Figure 9). However, the percentage of adsorption increased by 60–73% within 75 min. There may have been unfilled active sites on the surface of the adsorbent, which would account for the initial phase's significant rise in adsorption. The following stage's modest increase is caused by the dye's sluggish diffusion into the adsorbent pores, caused by the outer sites being completely occupied (Hameed and Ahmed., 2009). Additionally, it is hypothesized that when different dye concentrations rise from 20 to 500 mg/L, the adsorption capacity strengthens and then carries on with minor deviations, suggesting absorption saturation.
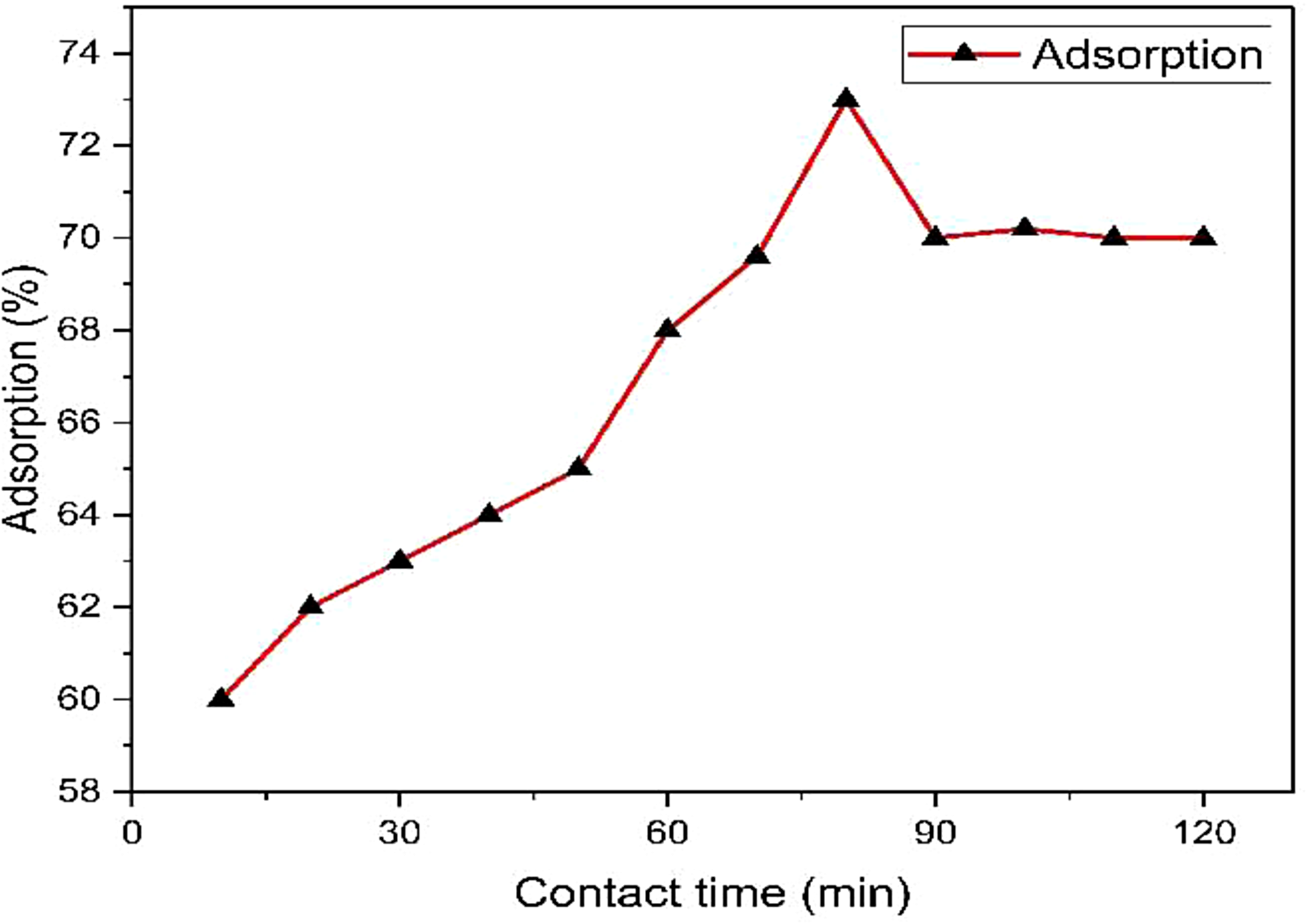
Effect of contact time on MR dye adsorption on A. marina plant bark.

Aspect of ionic strength on adsorption of methyl red dye at bark adsorbent surface.
Effect of ionic strength on adsorption
The adsorption capacity decreased from 79.8% to 76.6%, the sodium chloride concentration increased from 0.1 M to 0.9 M due to the positive charge on the surface of the adsorbent caused by the MR dye. The adsorption capacity is high (Figure 9) in acidic solutions due to the electrostatic interactions between a negatively charged MR dye and a positively charged adsorbent surface. The study focuses on the adsorption characteristics of MR dye, and the findings may not be universally applicable to other dyes or contaminants. This research primarily examines the adsorption on a specific adsorbent material. Different adsorbents may exhibit varied behaviors, and a broader range of materials could enhance the study's applicability.
Avicennia marina is naturally adapted to saline environments dominated by NaCl, making this salt particularly suitable for simulating real-world conditions where the plant thrives (Sivasankaramoorthy, 2012). The biomass production of Avicennia marina (a mangrove species) was higher than that of other salts in seawater (22,000 mg/L) containing NaCl. NaCl is commonly chosen as the salt for studying the effect of ionic strength on the adsorption process of methyl red dye due to its dissociation property and neutral ion behavior. NaCl dissociates completely into Na⁺ and Cl⁻ ions in aqueous solutions, making it a controlled condition without affecting ion size and charge. This ensures that changes in adsorption efficiency are attributed solely to ionic strength rather than to unique chemical interactions that might occur with other salts (Ashihara et al., 1997).
Leveraging mangrove bark adsorbent for effective decolorization of real wastewater
The results presented in Figure 10 demonstrate the superior adsorption capacity of mangrove bark powder in decolorizing water samples. Notably, MB achieved removal efficiencies of up to 50% in industrial wastewater, surpassing current standards for dye removal. When applied to Sabarmati river discharge and CETP effluents, MBP exhibited even greater efficiency, with removal rates of 67.18%, 82.07%, and 77.55% within 20–120 min. Furthermore, physicochemical characterization of wastewater before and after treatment presented in Table 1. These outcomes highlight the potential of MB as a versatile and effective adsorbent for tackling dye pollution and heavy metals in various water sources (Yaseen and Scholz., 2019). The study also acknowledges the variability in treatment duration and efficiency, which may be influenced by the initial dye concentration and its composition.

Mangrove bark adsorbent application to the different wastewater samples.
Physicochemical characterization before and after treatment.

BT, before treatment; AT, after treatment.
Morphological characterization of Avicennia marina bark biosorbent through SEM analysis
Scanning electron microscopy (SEM) was utilized to examine modifications in the surface morphology of the materials. The Figure illustrates the long-fiber-bundle and honeycomb-structure parenchyma cells structure found between 1 µm and 2 µm (Figure 11(a) and (b)). The adsorbent material possesses an irregular structure, which facilitates the adsorption of methyl red dye across various regions of the adsorbent. The visualizations presented in the figures depict the surface texture and porosity of the bark-based adsorbent, featuring the presence of voids and diminutive apertures that enhance the contact area. This characteristic promotes pore diffusion during the adsorption process. (Figures 9(a) and (b), 11(a) and (b)).

SEM images of A. marina bark biosorbent before and after decolorization.
Atomic force microscopy (AFM) morphology analysis of biosorbent
Atomic force microscopy (AFM) provides a powerful tool for investigating the surface topography and roughness of materials at the nanoscale. The AFM images revealed a wide range of particle sizes within the mangrove bark powder sample, indicating a heterogeneous population of particles. This diversity in particle sizes is crucial for adsorption processes, as smaller particles can increase the total surface area available for interaction with target substances. The surface roughness analysis showed that the mangrove bark surfaces exhibited a high degree of irregularities and undulations. Such roughness can enhance the adsorption capacity by increasing the surface area in contact with the adsorbate and potentially trapping particles through mechanical entrapment. However, the overall surface area available for adsorption remained largely unchanged, indicating that the biosorbent retained its capacity for further adsorption cycles. Future work should focus on optimizing the regeneration process to maximize the biosorbents lifecycle and minimize environmental impact (Figure 12(a) and (b)).

AFM images of A. marina bark biosorbent before and after decolorization.
Fourier transform infrared (FTIR) analysis of adsorbent
A Fourier transformation infrared (FTIR) study was carried out to characterize Avicennia marina bark powder before and after treatment with methyl red dye. The FTIR spectrum showed the band peak of functional groups. It was explained that different functional groups are present in the treated and untreated biosorbents. An FTIR analysis was performed to determine the core functional groups on the surface of the adsorbent responsible for dye binding (Muinde et al., 2017). The FTIR spectra of Avicennia marina bark before and after adsorption are shown in Figure 13. The FTIR spectrum showed a wide absorption peak at 3153 cm−1, corresponding to the stretching vibration of hydroxyl groups containing phenol or alcohol. The peak at 2920 cm−1 is caused by stretching vibrations of aliphatic (SP3) hydrocarbon (–CH) groups, whereas the peak at 1643 cm−1 is caused by aromatic C=C bending, and the peak at 1406 cm−1 is caused by –OH bending vibrations. C–O stretching in phenols, ethers, and esters causes a strong peak at 1110 cm−1. The peaks expanded after dye adsorption and a new peak at 2852 cm−1 was seen, indicating the formation of new C-H stretching, which causes the vibrations of the CH, CH2, and CH3 groups (Essaadaoui et al., 2018). The combination of a -OH group and a carbonyl group suggests the existence of a carboxylic group. Peak shifting from 2087 cm−1 to 2073 cm−1 following MR dye adsorption, indicates the formation of a new bond. The adsorbent's surface hydroxyl groups were essential in the adsorption process. These findings are reminiscent of the chemical groups found in lignin, hemicelluloses, and cellulose, which are the primary components of species (Liu et al., 2015).
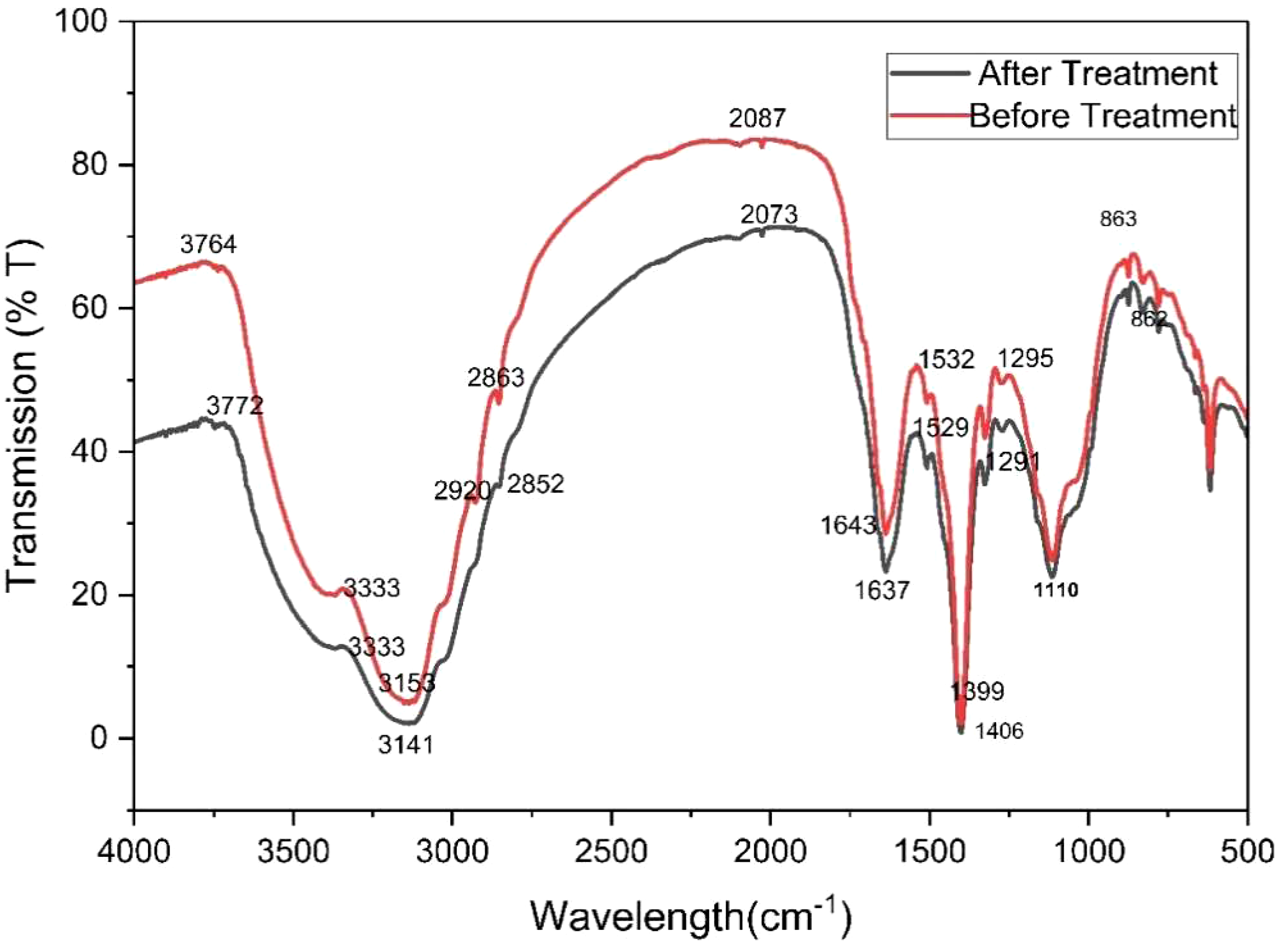
FTIR spectrum of before and after treatment.
The FTIR spectrum confirmed that the natural adsorbent surface has a greater tendency to absorb, which increases the capacity to reduce biosorption from the dye effluents. The graph showed that functional groups changed after treating methyl red dye with Avicennia marina bark powder.
XRD characterization of biosorbent before and after decolorization
The biosorbent prepared for dye removal was analyzed using XRD. Figure 14 illustrates the peaks. The XRD pattern reveals the clear presence of the hemicellulose peak at 2θ =15 and the cellulose peak at 2θ =22, making it evident that both these components are fundamentally important in the material's structure (Isiuku et al., 2014). On the other hand, another peak was also observed before adsorption on 2θ =11.02, 30.65, and 55.36. After loading the biosorbent with MR dye, the XRD pattern changed significantly compared to the unloaded dye with 2θ = 10.97, 15.01, 22.03, 30.60, and 54.96. It indicates that the adsorption process has altered the crystallinity of the biosorbent.
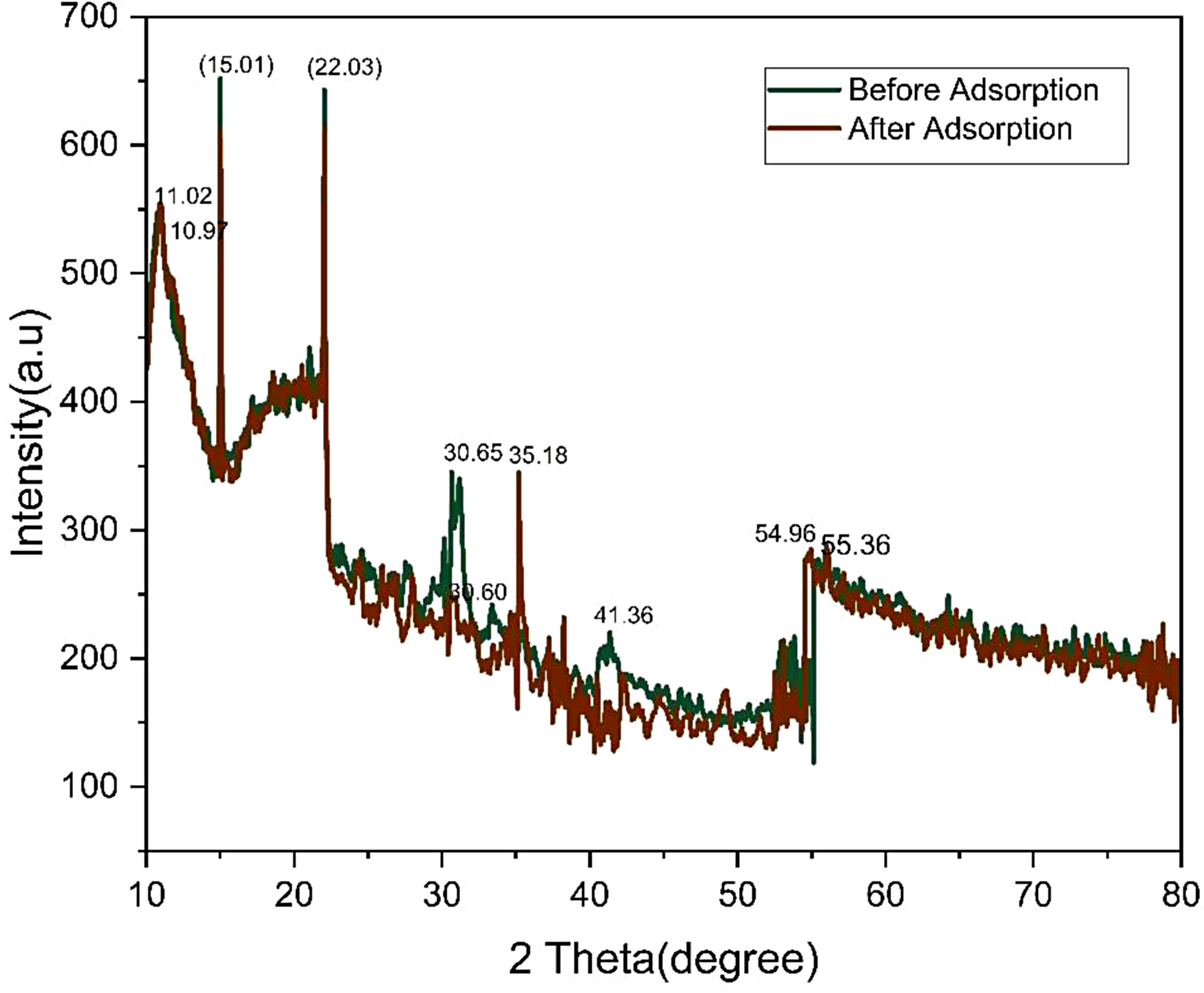
XRD spectrum of adsorbent before and after adsorption.
Adsorption kinetics
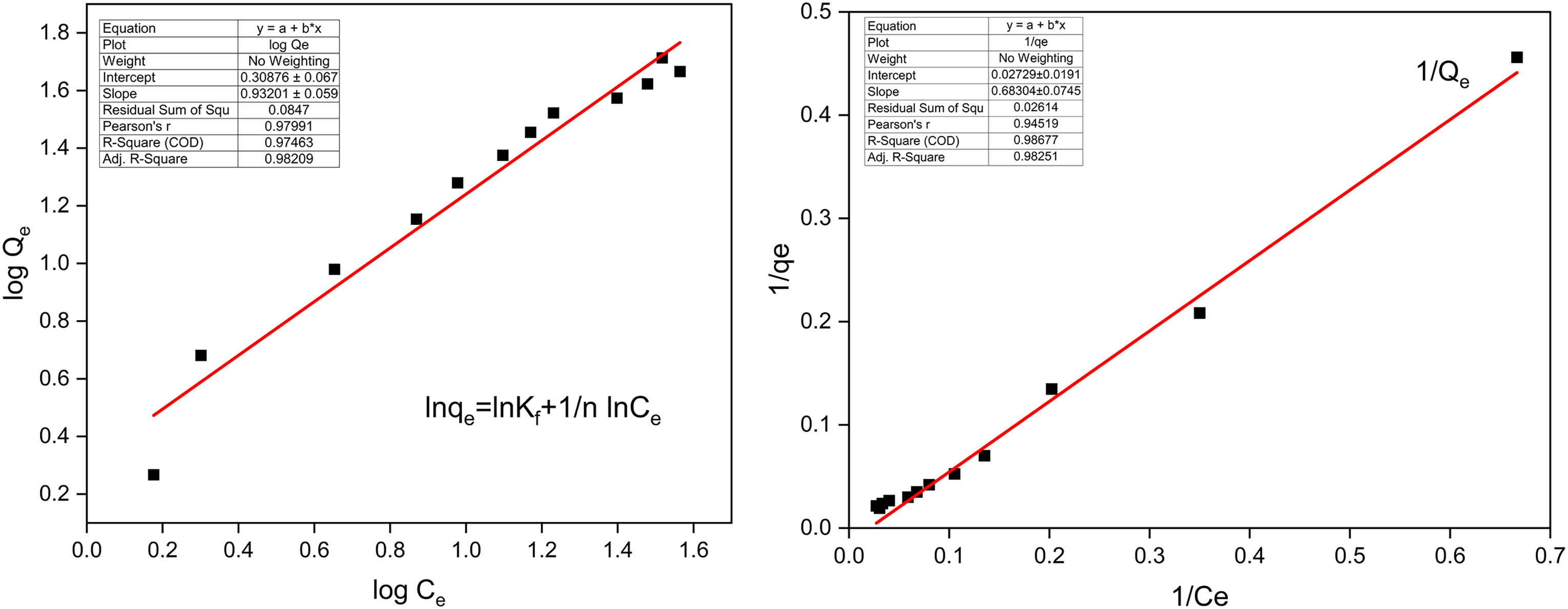
The data in Figure 15(a) and (b) was analyzed using pseudo-first-order and pseudo-second-order models. The findings revealed that Avicennia marina bark powder followed PSO kinetics instead of the pseudo-first-order kinetics. The adsorption process followed pseudo-second-order kinetics, where the values of Qt and Qe influenced the adsorption rate. So, pseudo-second-order adsorption kinetics is the best-fitted model in the adsorption process.
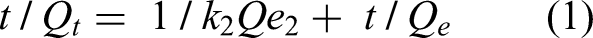

(a) Lagergren pseudo first order and (b) pseudo second order.
The rate constant k2 has dimensions of g mg−1 min−1 in the equation. Table 2 lists the parameters obtained from the slopes and intercepts of the PFO and PSO plots. The pseudo-second-order kinetic equation predicts that Qe is similar to Qexp.
Adsorption kinetics of methyl red.

Adsorption isotherms
The experimental data was analyzed in Table 3 using the Langmuir and Freundlich adsorption isotherms. Langmuir isotherm requires homogeneous adsorbent surfaces and equal adsorbent sites, where a monolayer of adsorbate forms on the surface.
Adsorption isotherm of methyl red.

At equilibrium, the amount of dye concentration (Ce) is proportional to adsorption capacity (Qe) and maximum dye adsorbed (Qm). Freundlich isotherm was utilized to fit the data, considering surface heterogeneity, roughness, and different adsorption sites. The coefficient of determination (R2) for Langmuir isotherm was 0.694, indicating that the system does not meet Langmuir's assumptions.
(a) Langmuir and (b) Freundlich adsorption isotherms.
Thermodynamic studies
Spontaneity of a process can be determined by thermodynamic parameters such as enthalpy change ΔH0, free energy change ΔG0 and entropy change ΔS0. A spontaneous process will show a decrease in ΔG0 value with increasing temperature. The thermodynamic parameters such as change in free energy ΔG0 (J mol−1), enthalpy (ΔH0) (J mol−1) and entropy (ΔS0) (J K−1mol−1) are determined using the following relationships: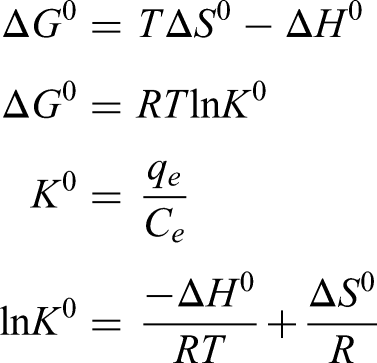
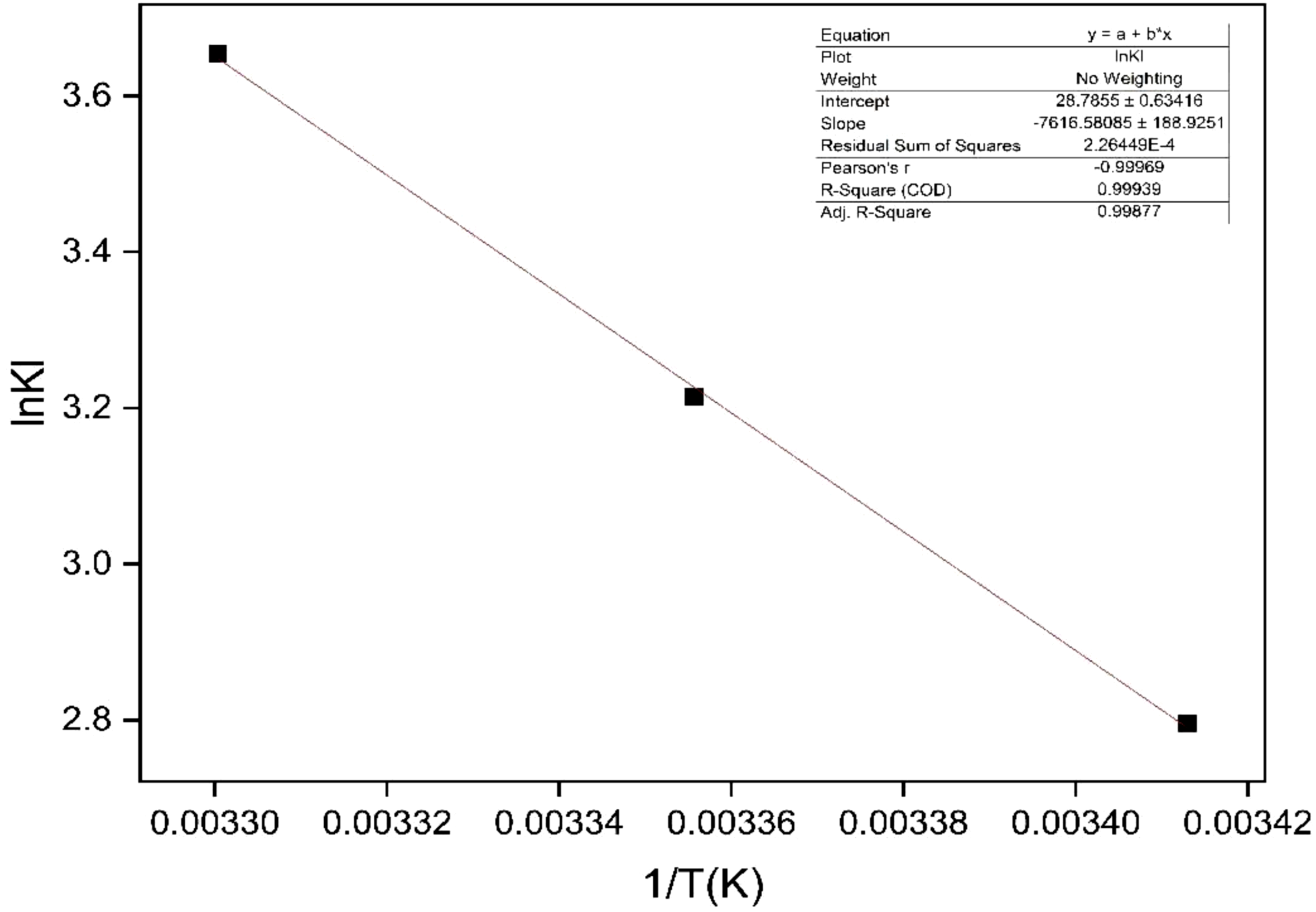
Van't Hoff plot for the adsorption of MR on bark biosorbent.
The pseudo-second order model is identified as the best kinetic model for the adsorption of MR on bioadsorbent surface. Accordingly, the rate constants (K2) of the pseudo-second order model are adopted to calculate the activation energy of the adsorption process using the following Arrhenius equation:
Avicennia marina bark compared to previous low-cost adsorbents
Based on previous studies, Table 4 compares the MR dye adsorption ability of Avicennia marina plant bark powder with other inexpensive adsorbents. The experimental findings indicate that the optimal adsorbent dosage for MR dye solution (500 ppm, pH − 1) is 100 mg per 10 mL, with a contact time of 75 min, resulting in excellent results. Additionally, the adsorbent dose used in this work is lower than those used in earlier studies, and a higher dye concentration can be removed with the lower dose (Table 5).
Thermodynamic parameters for adsorption of mr on bark surface.

Compare the adsorption capacity of Avicennia marina’s bark with other studied adsorbents.

Adsorption mechanism of methyl red dye using synthesize biosorbent
The adsorption mechanism of methyl red (MR) dye on synthesized bark biosorbents relies primarily on the biosorbent's surface properties, including functional groups, porosity, and surface area (Figure 18). The bark material often contains a variety of functional groups, such as hydroxyl, carboxyl, and phenolic groups, which interact with MR through hydrogen bonding, electrostatic interactions, and Van der Waals forces. Methyl red (MR) dye contains two benzene rings with electron-dense regions that facilitate donor–acceptor interactions, promoting π–π stacking onto the biosorbent surface (Wang et al., 2018). Additionally, due to the high porosity of biosorbents, MR molecules may penetrate the biosorbent through pore diffusion, as confirmed by field emission scanning electron microscopy (FESEM) analysis (Gul et al., 2022). These interactions facilitate the binding of MR dye molecules onto the bark surface, particularly in acidic conditions where MR is protonated, enhancing its affinity for the biosorbent. The pKa value of methyl red (MR), a common azo dye pollutant, is 5.1 (Teweldebrihan and Dinka., 2024). This pKa value is significant because it indicates the pH at which MR exists as a mixture of both protonated and deprotonated forms. The dye is red in acidic conditions (pH < pKa) and yellow in basic conditions (pH > pKa). The pKa value of methyl red directly affects its removal efficiency in aqueous solutions. Since methyl red behaves differently under acidic and basic conditions, adsorption and other remediation processes are influenced by pH. For instance, studies show that acidic conditions (below pH 5) generally enhance the removal efficiency, as many adsorbents bind better with the protonated form of MR, increasing the adsorption affinity (Kim et al. 2022). Similarly, a group of investigators led by removal efficiency peaks around pH 4–5 with materials like activated carbon and eggshell powder because these conditions align with the dye's protonated form, enhancing binding and removal efficiency (Amari et al., 2023).
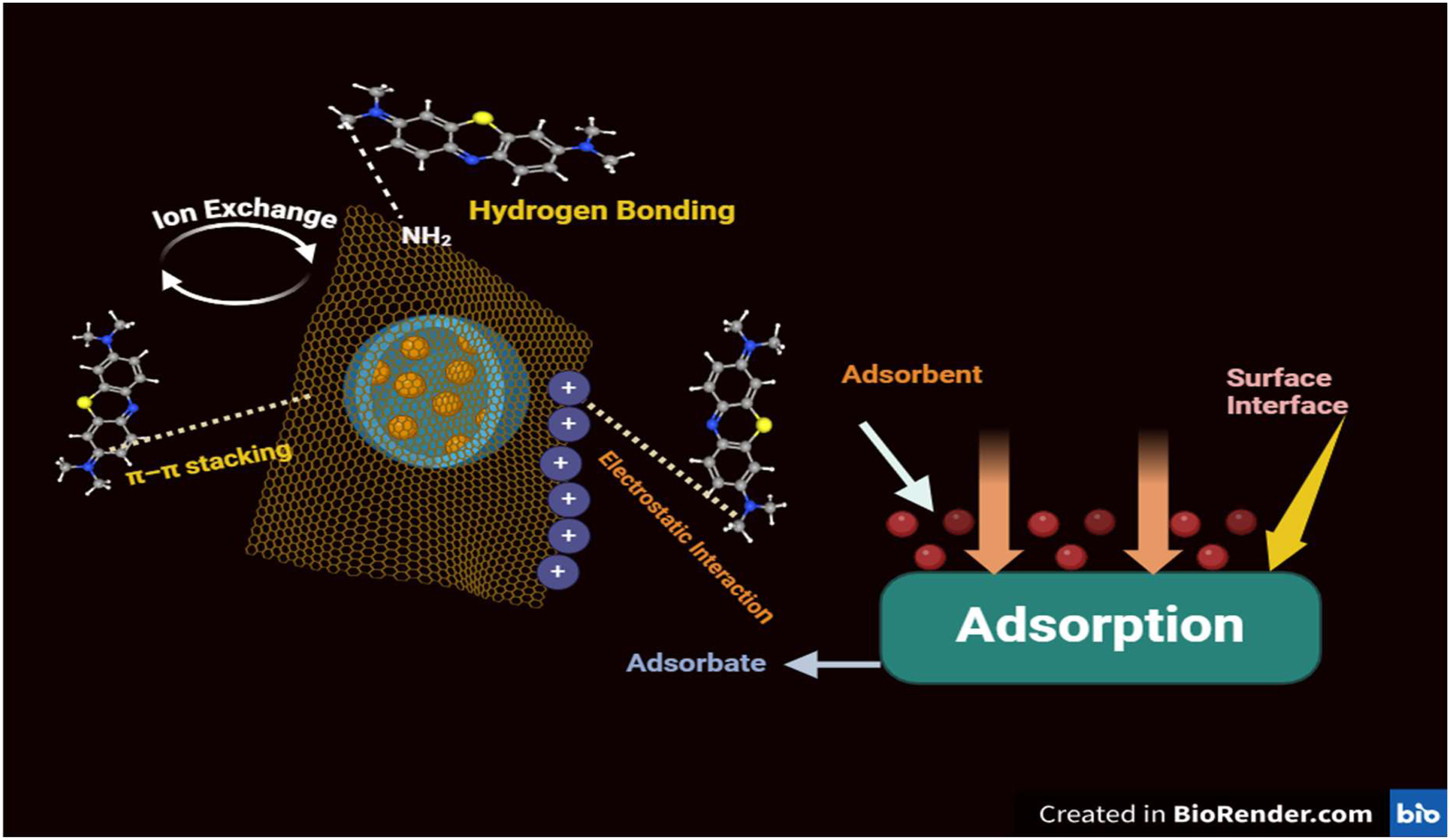
Adsorbate (bark biosorbent)–adsorbent (methyl red) mechanism.
Conclusion
This comprehensive investigation into the use of Avicennia marina as a green biosorbent for methyl red decolorization (74%) underscores the species’ remarkable potential in addressing textile dye pollution. The material possesses a fibrous and porous structure characterized by a large surface area and functional groups that facilitate dye adsorption. Avicennia marina bark powder shows greater adsorption capacity than other biosorbents such as squamosa shells, potato peels, fibers of the banana pseudo stem, custard apple, and lemongrass, which have a value of 61%, 61%, 64%, and 14.46%, respectively. Moreover, equilibrium adsorption follows the pseudo-second-order kinetic model and the Freundlich adsorption isotherm. The findings emphasize the ecological and economic benefits of leveraging natural resources like A. marina in wastewater treatment, offering a sustainable and cost-effective solution to the challenges posed by dye effluents. As the world seeks to balance economic growth with environmental stewardship, the exploitation of A. marina represents a significant stride towards achieving sustainable water management and fostering a greener future.
Footnotes
Acknowledgments
I, Supriya Vaish, highly acknowledge UGC, DST/WOS-B/WWM-7/2021(G), and I-Stem for the financial help. The authors thank the Central Instrumentation Facility, Central University of Gujarat, Gandhinagar, India for all necessary and instrumentation facilities.
Author contributions
Bhawana Pathak: conceptualization, supervision, review, and editing; Supriya Vaish: conceptualization, research experiment, data analysis & compilation and manuscript. Both the authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and analyzed during the current study are available within the article and its supplementary information files or from the corresponding author upon reasonable request.
Funding
The author (Supriya Vaish) received financial support for the research (DST/WOS-B/WWM-7/2021). No funding support for authorship, and publication of this article.
