Abstract
Sugarcane bagasse, a metal-organic framework, and magnetite nanoparticles form a composite that can be used as absorbent materials for the removal of three pesticides (atrazine, carbofuran, and iprodione). Magnetite nanoparticles were synthesized, functionalized, and supported on the SCB while the metal-organic framework grew on the surface. The adsorption process was performed in water; the pH was close to 7, the room temperature was 23 °C, and the separation of the material was promoted by a magnetic field. The maximum adsorption capacities of the composite were 52.38 mg g−1 for iprodione, 55.45 mg g−1 for atrazine, and 48.03 mg g−1 for carbofuran. The adsorption process occurs through hydrogen bridges with certain steric impediments owing to the ramifications and large sizes of the molecules involved.
Introduction
Recently, many natural materials have been used to develop composites, including natural bone (Yunus Basha et al., 2015), kenaf (Kamal, 2014), alginate (Sirviö et al., 2014), and mango kernel (Akram et al., 2017). The use of natural materials to develop composites has advantages, such as low cost, abundance, and environmental friendliness. Natural materials combined with nanoparticles (Toledo-Jaldin et al., 2020), polymers (Mohammed et al., 2015), and metal-organic frameworks (MOFs) (Li et al., 2018) have opened a new horizon for material science.
MOFs are fascinating porous materials. Hundreds, if not thousands, of linkers and metal ions combinations have been reported in recent years (Firooz and Armstrong, 2022; Kitagawa, 2014; Vaitsis et al., 2019; Wang et al., 2022). Research publications describe new MOFs for various applications (Rosales-Vázquez et al., 2022; Wiwasuku et al., 2022). At the same time, other researchers are dedicated to developing composites using MOFs (Chen et al., 2021; Huang et al., 2018; Yang et al., 2023). These new composites exhibit a higher surface area, tailored structure, excellent chemical stability, and other properties that make them outstanding adsorbent materials for removing contaminants (Yang et al., 2023). Magnetic nanoparticles (MNP) are a suitable option for combination with MOF, and their magnetic properties can be used to facilitate separation. To prepare this kind of composite, MNP can be coated using MOF as a shell (Huang et al., 2018; Ma et al., 2018) or MNP can be supported on MOF (Zhou et al., 2017).
Pesticides are organic molecules that are toxic to humans and aquatic living organisms (Dhankhar and Kumar, 2023). MOF composites can be synthesized and applied as efficient porous adsorbents for the adsorption of pesticides (Jalalzaei et al., 2022). For example, natural materials, such as bovine serum albumin, can be combined with an Fe-MOF to remove insecticides (Sheikhi et al., 2021). The results showed that this composite removed parathion-methyl and diazinon from water samples. The maximum adsorption capacities were 370.4 mg g−1 for parathion-methyl and 400 mg g−1 for diazinon at pH 7. Remarkably, it was found that the composite enhanced the adsorption activity compared with the MOF alone.
The choice of MOF to be supported on a natural material depends mainly on the synthesis method. Most reported MOFs are synthesized by a solvothermal process, which requires high pressure and temperature. Therefore, it is necessary that the synthesis conditions do not modify the physical or chemical characteristics of the matrix, particularly if they are natural and sensitive to high temperatures. In this sense, choosing an MOF that can be assembled on a natural material surface by simple contact between the linker and the metal ion is the best choice. One of the MOFs that meet this requirement is MOF-199 or HKUST-1. This MOF, composed of copper metal ions and the linker H3BTC (1,3,5-benzene tricarboxylic acid), easily assembles in an ethanolic solution without altering the composition of the matrix. It does not require high temperature or pressure like in a solvothermal synthesis, which is common for MOF synthesis (Jagódka et al., 2022; Primakov et al., 2022).
Although HKUST-1 is one of the first MOFs reported in the literature, its properties and easy synthesis make it a good candidate for the development of new composites. The formation of the composite can be examined by FTIR, SEM, TEM, and BET. The matrix characteristics, crystallinity of the MOF, nanoparticle size, morphology, and chemical composition of the reinforcement material are essential for the adsorption process (Safa et al., 2021).
In the present study, sugarcane bagasse was selected as a natural matrix for depositing HKUST-1 and MNP. The matrix's characteristics were investigated before and after the deposition. This study demonstrated the potential use of sugarcane bagasse as a natural matrix for a composite that can be used as an absorbent material for pesticides such as atrazine, carbofuran, and iprodione.
Experimental section
Materials and chemicals
All chemicals were of analytical grade and used without further purification. Iron(II) chloride (FeCl2), iron(III) chloride (FeCl3), 1,3,5-benzene tricarboxylic acid (H3BTC), thioglycolic acid (C2H4O2S), and ethanol (C2H5OH) were purchased from Sigma-Aldrich. Cu(II) nitrate (Cu(NO3)2·2.5H2O) was purchased from Fermont. Merck Chemie supplied sodium hydroxide (NaOH). The pesticides atrazine (C8H14ClN5), carbofuran (C12H15NO3), and iprodione (C13H13Cl2N3O3) were provided by SYFATEC S.A. de C.V.
Collection and preparation of natural matrix (NSCB)
Sugarcane bagasse was collected from a local market in Mexico State. The material was washed with water and sun-dried for 5 days. The dried bagasse was ground in a seed mill, sieved (to particle sizes between 0.250 and 0.297 mm in diameter), washed with ethanol solution (10%, v/v) for 24 h, dried for another 24 h in an oven, and finally labeled as NSCB.
Functionalized magnetite nanoparticles (MNPF)
A co-precipitation process was used to synthesize magnetite nanoparticles (MNP). 50 mL of FeCl3 (0.1 M) and 50 mL of FeCl2 (0.05 M) were mixed for 30 min under a nitrogen atmosphere. 50 mL of NaOH (3.5 M) was added dropwise. After the reaction ended, MNPs were separated using a magnet; the remaining solution was decanted, and the solid was washed with deionized water before functionalization.
Oxidation of magnetite nanoparticles in air is a common problem. Therefore, MNP have been functionalized with thioglycolic acid to overcome this concern (Kazemzadeh et al., 2012). MNP were dispersed in ethanol/thioglycolic acid solution (2.9 mM). After stirring for 24 h, functionalized magnetite nanoparticles (named MNPF) were separated by a magnet, washed with ethanol, and dried for 6 h.
Synthesis of the composite MNPF/MOF@NSCB
2.0 g of NSCB and 2.02 g of Cu(NO3)2·2.5H2O were added to 100 mL of ethanol, maintaining stirring for 16 h. It is important to mention that this time was established as the equilibrium time for Cu on bagasse adsorption in a previous work (Toledo-Jaldin et al., 2018). After this time, 0.2 g of MNPF was dispersed by sonication in 2 mL of ethanol and added to the suspension. To form the MOF, 0.875 g of H3BTC in 100 mL of ethanol was added dropwise (stirring for 2 h). Using a small magnetic bar, the composite MNPF/MOF@NSCB was isolated and washed first with ethanol and then with water. The material was dried for 48 h at 100 °C.
Characterization
All equipment used to characterize the material is described in Supplementary Material Section 1.
Adsorption kinetics
Atrazine, carbofuran, and iprodione solutions (10.0 mg L−1) were prepared for the batch adsorption experiments. For this purpose, 10 mg of MNPF/MOF@NSCB was added to 10 mL of the pesticide solutions. After 3, 5, 10, 15, 20, 25, 40, and 45 min the composite was separated and the remaining concentration in the supernatant was determined. The concentration of each pesticide in the liquid phase was determined using a spectrophotometer (UV-VIS, Perkin Elmer Lambda 10). A blank run was conducted to verify that there was no interference during quantification.
The amount of adsorbed pesticide per gram of the material (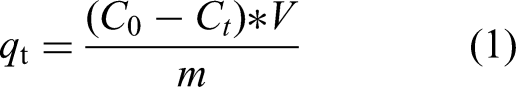
Adsorption data were analyzed using the pseudo-first-order, pseudo-second-order, and Elovich mathematical models (Mohseni-Bandpi et al., 2015).
Adsorption isotherms
Pesticide solutions were prepared at concentrations from 1 to 10 mg L−1. Ten milliliters of each concentration was in contact with 10 mg of the composite MNPF/MOF@NSCB until the equilibrium time (determined before in the kinetic study) was reached.
As described before, equation (1) was used to calculate the amount of pesticide adsorbed per gram of composite. The experimental data were tested by Langmuir, Freundlich, Temkin, Dubini–Radushkevich, Sip, and Redlich–Peterson (R-P) equations (Blanco-Flores et al., 2016).
The intraparticle diffusion model was applied to the experimental adsorption data (equation (2)) (Franco et al., 2020).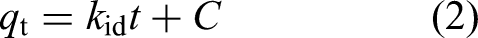
Results and discussion
Characterization
Figure S1a shows the FTIR spectra of the MNPs after (MNP) and before functionalization (MNPF). The spectrum of MNP shows characteristic bands of magnetite (Çıftçı et al., 2017) such as the one observed at 576 cm−1, which corresponds to the Fe–O bond (Bordbar et al., 2014). The signal at 3399 cm−1 corresponds to hydroxyl groups (O–H stretching) on the surface of the nanoparticles. Considering the functionalization model proposed before (Oveisi et al., 2017), two functional groups –OH can be replaced by an –HS from a thioglycolic acid molecule (Figure S1b). Another band that confirms the functionalization process is the S-H signal, which can be observed at 2567 cm−1 (Wu et al., 2012). The peak at 1590 cm−1 suggests the presence of carboxyl groups C=O (Zhan et al., 2018). This model assumed that carboxylic groups could be chemically attached to the magnetite surface.
Figure 1 shows the XPS spectra of the MNPF. As shown in Figure 1a, four characteristic peaks were observed at binding energies of 729.4 eV and 708.2 eV, corresponding to Fe2p1/2, Fe2p3/2, 532.58, and 285.4 eV corresponding to O1s and C1s, respectively, indicating that MNPF is mainly composed of iron and oxygen elements. The intensity of the C1s peak is the sum of the carbons in thioglycolic acid and the possible interference from the carbon tape on which the sample was supported.

XPS functionalized magnetite nanoparticles spectra (a), deconvoluted XPS survey of Fe (b) and S (c).
As seen in Figure 1b, the signal at 711.2 eV is typical of maghemite due to the oxidization of Fe(II) on the magnetite surface to Fe(III) (Cuenca et al., 2016). The peak at 709.4 eV could be attributed to the Fe–O bond. Consequently, the last peak at 708.2 eV could be associated with Fe–S on the surface of the material.
Figure 1c shows that the S2p3/2 peak was deconvoluted into four prominent peaks. The peak at the binding energy of 162.6 eV is related to the S–Fe bond, suggesting that other arrangements of thioglycolic acid could take place aside from the one proposed by Oveisi et al. Another research (Suriamoorthy et al., 2010) put forward that sulfur could be directly bonded to the nanoparticle to develop an interaction between S and O (Figure S2), which can be associated with the signal 164.5 eV (Zheng et al., 2017). However, there is a peak at 164 eV associated with SH– and another peak at 163.7 eV corresponding to the S–C bond (Zhang et al., 2012). These results and the previously described FTIR results indicate that the functionalization process was carried out in two different ways.
In the Supporting Information (Figure S3), six diffraction peaks are observed in the XRD pattern of MNPF. Using the JCPDS file (No. 19-0629) of the magnetite phase, the peaks at 2
The morphology and size of MNPs were characterized by transmission electron microscope (Figure S4). Spheroidal particles with an average size between 2 and 9 nm were identified, while the selected area electron diffraction (SAED) pattern revealed the crystalline form of the sample associated with an inverse spinel structure of Fe3O4, according to the JCPDS file (No. 19-0629), these results agree with the previous XRD analysis. In the high-resolution transmission electron microscopy (HRTEM) image, 4.7, 2.2, and 2.9 Å fringe spacings were associated with (1 1 1), (2 2 0), and (2 2 2) interplanar spacings planes of an FCC structure.
Figure 2a shows the embedding of MNPs in the MOF, forming a composite MNPF/MOF. The SAED pattern (Figure 2b) indicates the crystalline nature of the MOF and MNP. The (1 1 1) magnetite crystallographic plane associated with the most intense peak in the XRD pattern can be observed. The other two crystallographic planes correspond to HKUST-1 (Loera-Serna et al., 2012).

TEM image (a), SAED patterns (b), and HRTEM micrograph of
Figure 2c shows that MNPs and HKUST-1 form a composite. HRTEM lattice fringes of 4.7 and 2.5 Å were associated with (1 1 1) and (3 1 1) interplanar distance
The synthesis of the HKUST-1 results in various sizes of octahedral crystals (Figure S5a). When the synthesis of the HKUST-1 is done in the presence of MNPF, the shape of the MOF becomes rough and irregular (Figure S5b). MNPs may interfere with the MOF crystal growth and the octahedral crystal structure changes. This result was reported before (Tan et al., 2017).
STEM was used to investigate the microstructure and morphology of MNPF/MOF, as described in the Supplementary Material in Section 2 and Figure S6.
EDS analysis (Figure S7) confirmed the presence of Fe from the MNPs and Cu ions associated with the MOF.
Figure 3 shows the FTIR spectra of NSCB, MOF (HKUST-1), MNPF, and the composite MNPF/MOF@NSCB. The spectra of NSCB showed characteristic bands associated with the main components of sugarcane bagasse (cellulose, hemicellulose, and lignin) (Cruz et al., 2013). The stretching vibrations of the O–H group appeared at 3348 cm−1 and could be observed not only in the NSCB but also in the composite.

ATR-FTIR of NSCB, MNPF, MOF, and composite MNPF/MOF@NSCB.
The band at 2898 cm−1 is related to the stretching band of the C–H group of cellulose and the MOF. The peak at 1644 cm−1 in the composite is a combination of the carbonyl band (C=O) at 1629 cm−1 of hemicellulose and the signal at 1645 cm−1 of the deprotonated benzene tricarboxylic acid of the MOF. The band at 1365 cm−1 and close to 1500 cm−1 corresponds to the (C–O2) symmetric stretching modes of the linker in the MOF. The C–O–C of the cellulose and hemicellulose chains is around the strong peak at 1165–1045 cm−1. The vibration band at 727 cm−1 can be attributed to the stretching vibration band of Cu–O (Goyal et al., 2022). Finally, the absorption bands around 518 cm−1 can be assigned to Fe–O stretching modes.
The specific surface area of NSCB was 9.54 m2 g−1, which is higher than that reported for sugarcane bagasse. 1.7 m2 g−1 surface area was reported for bagasse pretreated with hot water (Zanchetta et al., 2018). A higher area could be reached when the material is subjected to a pressure of 2.0 MPa (5.84 m2 g−1) (Liang et al., 2016). However, a lower surface area (0.66 m2 g−1) was determined when sugarcane bagasse was washed only with distilled water (Zhang et al., 2013). Therefore, it is evident that the washed conditions used to clean the sugarcane bagasse are crucial to obtaining a significant surface area. Similar to nanoparticles, MOFs have large surface areas; therefore, it is not surprising that NSCB increases their surface area significantly from 9.54 to 220.4 m2 g−1, which is the surface area of MNPF/MOF@NSCB.
The diffraction peaks in the NSCB are typical XRD patterns for cellulose or lignocellulosic materials (Pereira et al., 2016), which are the main phases in sugarcane bagasse (Figure 4). The crystallographic planes (1 0 1) and (0 0 2) matched diffraction peaks at 15.6° and 22.2°. The phases in the MNPF/MOF@NSCB confirmed the successful synthesis of the composite.

X-ray diffraction of NSCB, MNPF/MOF, and composite MNPF/MOF@NSCB.
The NSCB surface characteristics can be analyzed using SEM. Pores of different sizes were distributed on the surface (Figure 5a). The matrix maintains its characteristics, while MNPF/MOF crystals are observed on the surface of MNPF/MOF@NSCB (Figure 5b).

SEM of NSCB (a) and of composite MNPF/MOF@NSCB (b).
Adsorption kinetics
Various concentrations of pesticides were prepared. A calibration curve of the absorbance against each pesticide concentration is plotted (Figure S8). Using this calibration curve, the concentration of each composite after contact with the MNPF/MOF@NSCB was determined with good precision.
The contact time between each composite and the pesticides was measured in the time range of 5–50 min with the following parameters (adsorbent dose = 0 mg; Co = 15 mg/L;
Equilibrium time in minutes for composite MNPF/MOF@NSCB with atrazine, carbofuran, and iprodione.

The three models described in the Supplementary Material (Section 3) were applied, and each kinetic parameter is in Table 2. The Elovich model for atrazine and carbofuran removal better described the adsorption process of MNPF/MOF@NSCB. The lowest values for RSS and
Parameters of kinetic models with MNPF/MOF@NSCB with atrazine, iprodione, and carbofuran.

The bold type values show the best-fitting mathematical model for each pesticide.
The Elovich model described better the atrazine and carbofuran adsorption, which implies that the adsorption mechanism is controlled by a second-order reaction where an energetically heterogeneous is involved (Pintor et al., 2018). In the case of atrazine and carbofuran, a heterogeneous distribution of activation energy sites is common in composites, where the coverage depends on the morphology of the surface. During the characterization, the heterogenicity of the NSCB surface was described. The cellulose and lignin in the matrix, MNPs, and MOF exhibited different activation energies for possible chemisorption. However, a heterogeneous system can promote chemisorption as well as ion exchange, precipitation, or intraparticle diffusion. Therefore, it is difficult to establish which process is dominant (Riahi et al., 2017). The adsorption rate parameter,
The best-fitted model for iprodione was pseudo-second-order, which describes a chemical adsorption process through the sharing or exchange of electrons.
Adsorption isotherms modeling
To understand the adsorption process, it is essential to establish the most appropriate correlation of the equilibrium curves formed by the isotherms. In the literature, several mathematical models can be found for this purpose; in the present work, isotherms of atrazine, carbofuran, and iprodione with MNPF/MOF@NSCB have been tested by four mathematical models described in section 4 of Supplementary Material. The data with the best-fit model are shown in Figure 6, and all parameters are listed in Table 3 (the statistical parameters are listed in Table S2).

Adsorption isotherms of MNPF/MOF@NSCB for atrazine, carbofuran, and iprodione removal using the best-adjusted model.
Isotherm modeling parameters for MNPF/MOF@NSCB.
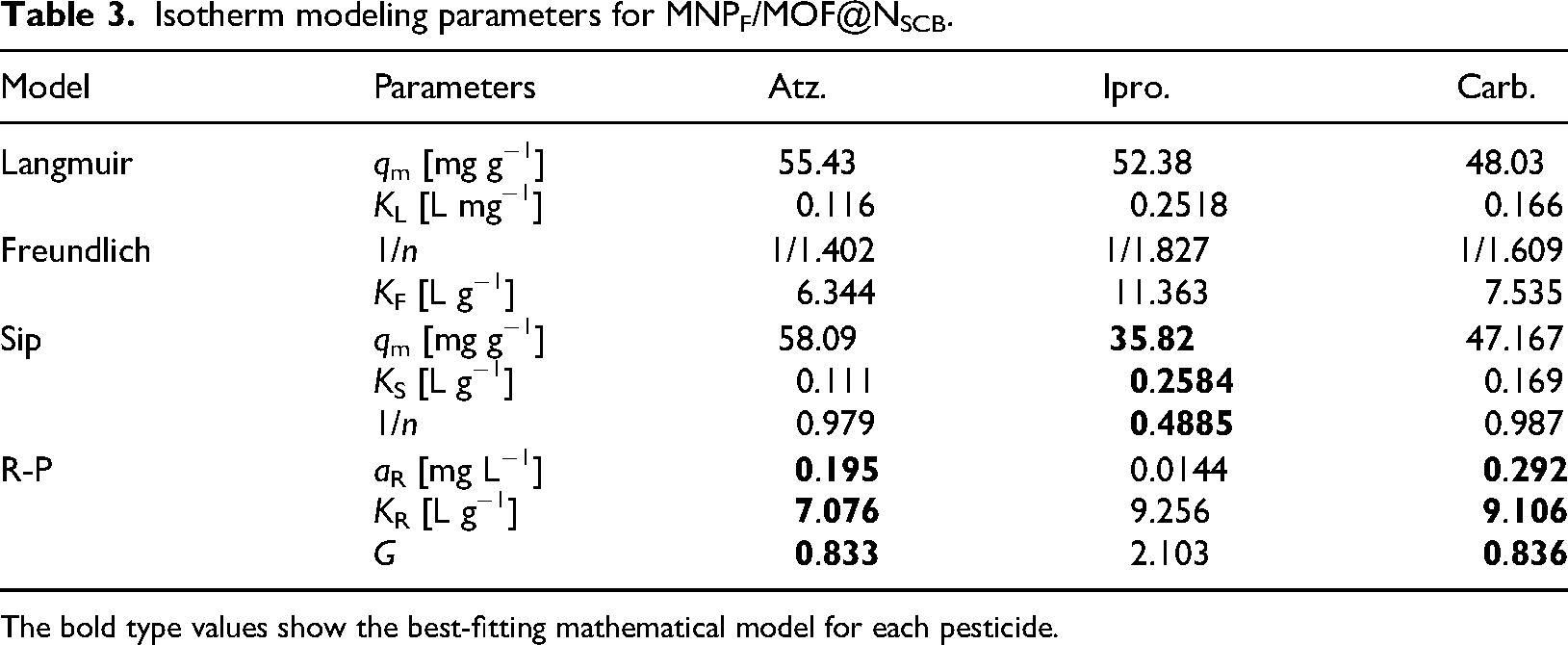
The bold type values show the best-fitting mathematical model for each pesticide.
As shown in Figure 6, the shape of the adsorption isotherms of MNPF/MOF@NSCB for the removal of carbofuran was significantly different from that of the others (red line). The graph shows a curvature or elbow near 30 (mg g−1) with a tendency to form a plateau after this point, while the other two isotherms for atrazine (black line) and iprodione (blue line) seem to have a growing tendency. The carbofuran isotherm fit better with the Sip model, unlike the other two systems, which found the best fit in the Redlich–Peterson model, which it is a modification of the first.
It means that the adsorption of carbofuran occurred through a combination of processes that involve chemisorption, and electrostatic attraction, among others. This confirms the results obtained when applying the kinetic models where the best fit of the model that describes chemisorption processes was involved.
The Sip model 1/n parameter described a higher removal intensity for iprodione. Using the Langmuir Model, the adsorption capacities of the composites MNPF/MOF@NSCB for atrazine, carbofuran, and iprodione were 55.43, 48.03, and 52.38 mg g−1, respectively. Chemical adsorption occurred on heterogeneous surfaces according to the best-fitted mathematical models for MNPF/MOF@NSCB.
The adsorption capacity of the NSCB was evaluated for all three pesticides (Figure S9). Only the Langmuir model was applied to the data to compare the adsorption capacities with those of MNPF/MOF@NSCB. As expected, capacity increased significantly.
Determination of the adsorption process controlling stage with the best material and kinetic adsorption mechanism proposal
Three linear zones were observed in each case using the intraparticle diffusion model for each pesticide. They diffused through the liquid film bordering the adsorbent particle, intraparticle diffusion, and adsorption processes (Figure S10a-c). Because none of the zones had an intercept at zero, intraparticle diffusion was not the controlling step during the process. The
Kinetic model parameters for atrazine, carbofuran, and iprodione adsorption onto MNPF/MOF@NSCB.

The external mass transfer coefficient was obtained by applying the external mass transfer model (EMTM), which assumes that the intraparticle diffusion stage occurs instantaneously (Franco et al., 2020).
The model's principle was verified using kid values. The
The 

Analysis of the first variable in equation (4) indicates less interaction between the adsorbate and adsorbent for carbofuran than for atrazine or iprodione because of steric impediments.
The transport number
As a result, the adsorption process occurs through hydrogen bridges, with certain steric impediments due to the presence of ramifications and the large sizes of the molecules involved. Although the surface area of the material could not be completely occupied, the adsorption capacities were high because multilayer adsorption was favored. This was confirmed by the shape of the adsorption isotherms and adjustments to the mathematical models obtained for each adsorption process with the same material (Figure 7).
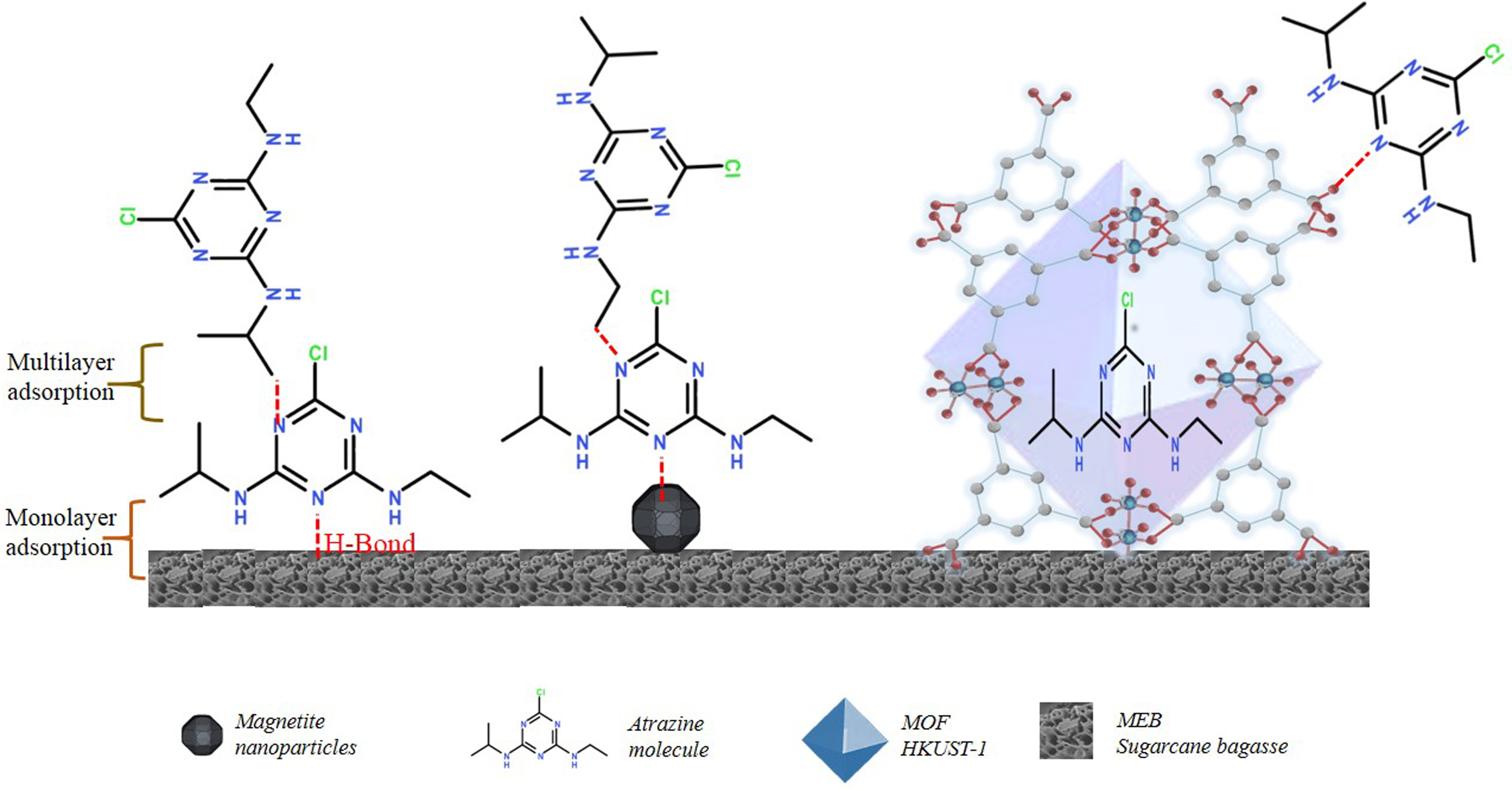
Kinetic adsorption mechanism for pesticides adsorption on MNPF/MOF@NSCB.
In Table 5, some reported material capacities for atrazine, iprodione, and carbofuran are compared with those of the composite MNPF/MOF@NSCB. Atrazine is a widely studied herbicide used in adsorption processes using various materials with unique characteristics. Some of these studies are discussed below: Akpinar and Yazaydin (2018) studied the effects of Zr6-based metal-organic frameworks (MOFs) linkers and topology on atrazine uptake capacity. They found that a mesoporous MOF facilitated atrazine uptake, whereas pyrene-based linkers offered sites for π–π interactions. The maximum adsorption capacity of this MOF was 36.0 mg g−1. In another study by Akpinar et al. (2019), three MOFs and a commercial activated carbon, F400, were compared for atrazine removal. Only one MOF was ineffective for the process, whereas F400 was found to remove 13.73 mg g−1. Another type of composite, Fe/C, derived from iron-crosslinked alginate prepared via pyrolysis, was developed by Lei et al. Batch experiments showed that the removal of atrazine was 64.8 mg g−1 at pH 6 using the Langmuir model.
Comparison of MNPF/MOF@NSCB with previously reported adsorbents.

Few studies have been reported on the adsorption of the fungicide iprodione. One of them is that of Perendija et al. (2021), who achieved a removal of 30.16 mg g−1 with a magnetite (MG)-modified cellulose membrane obtained from nonlinear Langmuir model fitting. However, Huang et al. used four magnetic iron (III)-based framework composites for the sensing of iprodione, with a limit of detection of 0.04–0.4 μg L−1 (Huang et al., 2018). Like the last one, some authors have focused more on iprodione determination than on adsorption.
Table 5 shows that different materials have different adsorption capacities for carbofuran, which, unlike iprodione, have been reported. Materials such as silica-anchored graphene oxide composites have lower adsorption capacities than those reported in this work. In contrast, materials such as steam-activated biochar,
Conclusions
Magnetite nanoparticles were successfully synthesized using a coprecipitation method. Functionalized nanoparticles can be incorporated into MOF, forming a magnetic composite that can be supported on lignocellulosic material. Two different arrangements can be achieved using or not functionalized MNP. At the same time, it can explain the position inside or outside the MOF structure. Pesticide removal occurs via chemical adsorption onto heterogeneous surfaces. Maximum adsorption capacities were 48.03 mg g−1 for iprodione, 55.45 mg g−1 for atrazine, and 52.38 mg g−1 for carbofuran, respectively.
Supplemental Material
sj-docx-1-adt-10.1177_02636174241292559 - Supplemental material for Natural matrix reinforced with a coordination compound and magnetic nanoparticles to remove organic contaminants from water
Supplemental material, sj-docx-1-adt-10.1177_02636174241292559 for Natural matrix reinforced with a coordination compound and magnetic nanoparticles to remove organic contaminants from water by Helen Paola Toledo-Jaldin, Alien Blanco-Flores, Delia Monserrat Ávila-Márquez, Oscar Roberto Montes-Moreno, Alfredo Rafael Vilchis-Nestor, Gustavo López-Téllez and Alejandro Dorazco-González in Adsorption Science & Technology
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Consejo Mexiquense de Ciencia y Tecnología (COMECYT), México (grant number FICDTEM-2023-100).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
