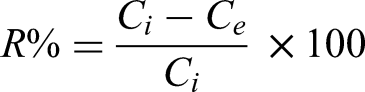Abstract
In response to the rising interest in reducing atmospheric CO2 levels and heavy metals in water, new polyaminal-linked polymer networks were effectively synthesized using a one-pot polycondensation reaction of melamine and 9-anthracenecarboxaldehyde (PAN-AN). The polyaminal porosity parameter was investigated and applied for CO2 adsorption and heavy metal removal. Fourier transform infrared spectroscopy, solid-state ¹³C NMR, and X-ray diffraction were used to confirm the formation of the polymer structure. The porous material structure was determined using a scanning electron microscope and N2 adsorption–desorption methods at 77 K. The polyaminal has a Brunauer–Emmett–Teller surface area of ∼ 230.42 m2/g and a high CO2 uptake of 30.08 cm3/g. PAN-AN was used to investigate the adsorption behavior of several metal cations and showed high sensitivity for Pb(II). The pH, adsorbent dose, initial concentration, and contact time were used to evaluate the synthesized polymer as an adsorbent against the Pb(II) ion. At pH 5.5 and 20 mg of dosage, PAN-AN had a high capability in removing 90.05% of Pb(II). Furthermore, the Freundlich model fits the adsorption isotherm data well, whereas the adsorption kinetics followed the pseudo-second-order kinetic model. The results revealed that the porosity (PAN-AN) influences CO2 adsorption and heavy metal adsorption.
Introduction
Air pollution results from burning coal, oil, and natural gas. CO2 emissions from these sources enhance carbon dioxide levels, producing substantial environmental issues, most notably the greenhouse effect (Rehman and Park, 2018). Considerable research has been conducted to find materials capable of capturing and storing CO2. Zeolitic imidazolate frameworks (Yang et al., 2024), activated carbons (Ligero et al., 2023), and aqueous amines (Liu et al., 2021) are examples of these materials. Furthermore, water pollution impacts the environment and leads to a damaging discharge into ecosystems and hydro systems, including heavy metals, nitrates, phosphates, and pesticides (Sall et al., 2020). Exposure to heavy metals can cause severe diseases such as hematological, gastrointestinal, kidney pathology, hepatocellular, cardiac, reproductive, immunological, and neurological diseases (Munir et al., 2021). Many techniques have been employed for heavy metal removal, including biosorption (Sharifi et al., 2023), hydrogel (Tohamy et al., 2023), membrane separation (Long et al., 2023), and nano adsorbent (Liu et al., 2020). Among the mentioned techniques for CO2 capture and heavy metal removal, porous organic polymers (POPs) have tremendous promise due to their tunable porosity, high catalytic activity, and high chemical-thermal stability (Tang et al., 2018). POPs, which are a class of highly porous materials, have received a lot of interest due to their unique features attained by merging the advanced properties of porous materials and polymers, along with their uses such as the elimination of organic contaminants (Hu et al., 2019), energy and gas storage (Bhanja et al., 2018; Furukawa and Yaghi, 2009), and heavy metal removal (Taskin et al., 2016). Polyaminal-based polymer networks (PANs) are POPs prepared from triazine-based multiamines with different aldehydes (Li et al., 2016; Matias et al., 2023; Zhang et al., 2018; Zhang et al., 2019). Polyaminals are particularly interesting owing to their ease of synthesis via a one-pot catalyst-free polycondensation of aldehyde derivatives with melamine, high nitrogen content, and an abundance of microporous structures (Zhang et al., 2020). The intermediate imine bond (–C = N–) combines with the active amino group to form a stable aminal linkage (–NH–CH–NH–). Wang et al. synthesized PAN-P from the reaction of benzaldehyde and melamine with a Brunauer–Emmett–Teller (BET) surface area of 682 m2/g and a pore size distribution (PSD) of 0.84/2.12, indicating the presence of micro- and mesopores enhancing CO2 uptake up to 11.1 wt% (Li et al., 2016). Manal and co-workers synthesized polyaminal based on melamine and naphthalene (PAN-NA) with SBET 607.46 m2/g, and the pore width peak relied on micropore region at 0.54 nm, which has good adsorption for small CO2 gas molecules up to 67.9 cm3/g and to all metal cations, including Cr(III), Cu(II), Ba(II), Cd(II), and Ni(II), with a superior affinity for the Pb(II) cation with an adsorption capacity of 90.36 mg/g at pH = 6, 20 mg/L concentration, and an adsorbent dose of 20 mg at 20 min. due to the larger surface area, abundant functional groups, and microporosity, resulting in increased capacity in cationic species (Ibrahim et al., 2022). Sandín et al. created new polyaminal networks with phenyl (MA-iPh), pyridine (MA-Py), and thiophene (MA-Th). The adsorption capability of these polyaminals was tested for several hazardous metals such as Cr(VI), Cu(II), Cd(II), and Pb(II). MA-Py, which had the highest amount of nitrogen 37% and SBET 424 m2/ g in this series, also had the most increased adsorption capabilities, with values of 27.22, 26.42, and 53.13 mg/g for Cr(VI), Cd(II), and Pb(II), respectively. The increased adsorption capacity of MA-Py might be due to both its higher specific surface area and its higher N concentration (Sandín et al., 2021). Furthermore, Liu et al. introduced two triphenylamine-based nitrogen-rich polyaminals (TPOAs), indicated as TPOA-F and TPOA-OH, which were readily synthesized using triphenylamine-based triazine-hexamine and fluoro- and hydroxy-monoaldehydes, respectively. The porous polymer materials have substantial particular surface areas of 1179 and 565 m2/g and pore distribution of 0.58 to 5.20 nm. The higher CO2 uptake of TPOA-F compared to TPOA-OH 155.3 mg/g and 72.4 mg/g, respectively, is attributed to its higher BET surface area and narrower PSD. This affords more surface area for adsorption and improves access to adsorption sites, enhancing adsorption performance (Liu et al., 2023). The findings demonstrate the importance of the surface area in improving the adsorption capability of polyaminal networks (PANs) for heavy metal removal. Larger surface areas provide more active sites accessible to adsorption, resulting in higher adsorption capabilities. Furthermore, the existence of micro- and mesopores within PANs contributes to the adsorption of both small CO2 gas molecules and different metal cations. Specific surface area and PSD have a direct impact on adsorption performance, with materials exhibiting larger surface areas and narrower PSDs demonstrating superior adsorption capabilities. These findings highlight the importance of the surface area and pore structure in developing efficient adsorbent materials for environmental remediation applications. Anthracene consists of three linearly fused aromatic rings comprising an extended π- system with 14 delocalized electrons (Van Damme and Prez, 2018). Introducing 9-anthracenecarboxaldehyde, which is rich in electrons and promotes the production of new (PAN-AN), is expected to alter the surface area, porosity parameters, and adsorption capabilities. As a result, the effect of anthracene moieties on the structure of pores and adsorption capacity will be studied. In the present work, anthracene-based polyaminal networks (PAN-AN) are synthesized via the direct one-step polymerization technique using 9-anthracenecarboxaldehyde and melamine without employing a catalyst, as depicted in Scheme 1. Furthermore, CO2 capture and heavy metal removal were utilized to investigate the adsorption capacity of the prepared polymer. The synthesized polymer PAN-AN composition was examined through Fourier transform infrared spectroscopy (FTIR), solid-state 13C NMR, X-ray diffraction (XRD), and thermogravimetric analysis (TGA). Scanning electron microscopes with an energy-dispersive X-ray spectrometer (EDS) were utilized to estimate morphology and elemental analysis. Physical sorption of nitrogen at 77 K was used to evaluate the porosity parameters of PAN-AN.
Synthesis of PAN-AN.
Experimental section
Materials
9-Anthracenecarboxaldehyde (99%) was purchased from Baoji Guokang Bio-Technology Co., Lt, Baoji City, Mainland China; melamine (97.5%), dimethyl sulfoxide (DMSO 99%), NaOH (98%), Cu(CH3 COO)2.H2O (99%), and NiSO4.6H2O (98%) were supplied from BDH Laboratory Reagents, England, UK; tetrahydrofuran (THF ≥ 99.5%) was supplied from Honeywell, Germany; dichloromethane (≥ 99.8%) and Pb(NO3)2 (99%) were purchased from Fisher Chemicals, England, UK; acetone was purchased from Cornaredo (Milano), Italy; HCl (35%) was supplied from LOBA Chemie, Mumbai, India; and ethanol (99.8%) was purchased from Sigma-Aldrich Darmstadt, Germany. All materials were received and utilized without further purification.
Synthesis of anthracene-based polyaminal networks (PAN-AN)
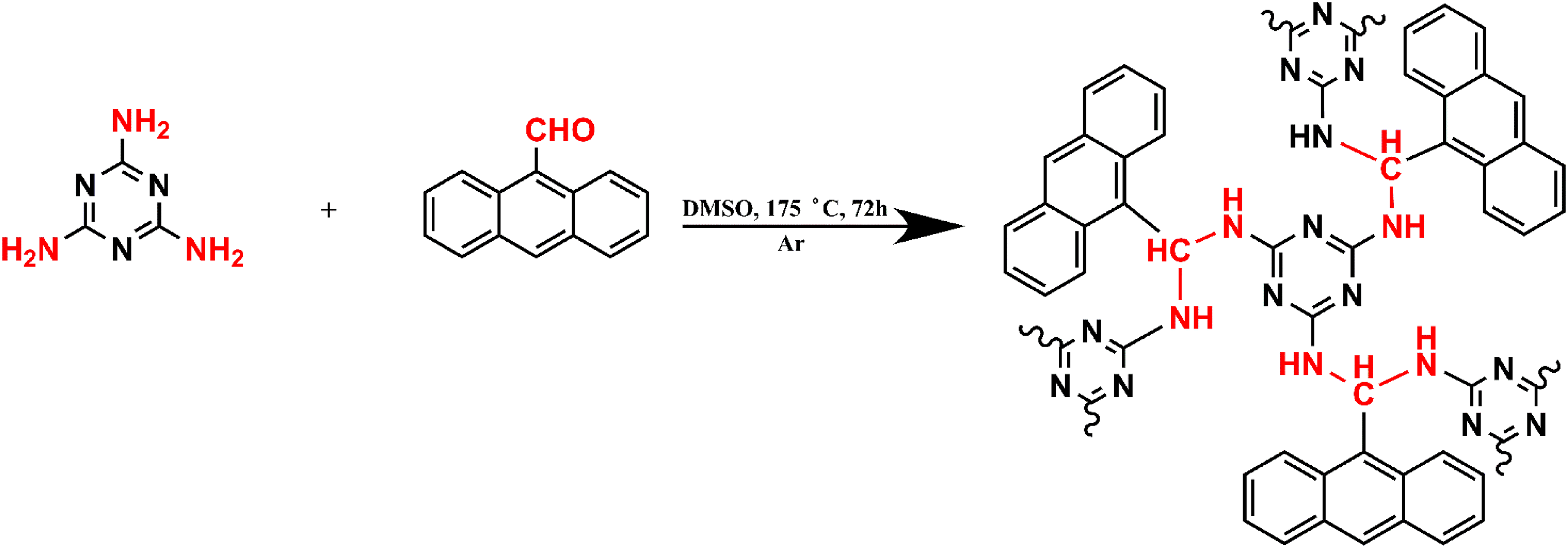
A condenser with a rubber septum installed on a three-necked flask with a magnetic stirrer was degassed using two evacuation–argon-backfill cycles. Under argon flow, melamine (0.5 g, 3.96 mmol), anthracenecarboxaldehyde (1.6 g 7.92 mmol), and 50 mL of DMSO were added and heated at 175 °C for 72 h. Finally, the system was cooled down, and the product was isolated and washed several times with dichloromethane, tetrahydrofuran, and acetone, respectively. The polymer was filtered and dried in a vacuum to yield (1.304 g, 50%).
Adsorption experiments for heavy metal removal
The removal of Pb(II), Cu(II), and Ni(II) was accomplished by preparing 100 mL of a 20-ppm standard solution of each salt, adding 20 mg of PAN-AN, stirring for 60 min, filtering, and collecting the filtrate to measure concentration. The adsorbent removal efficiency R% and adsorption capacity qe are calculated by the following equations:
Instrumentation
FTIR was performed using the KBr disk technique on an FTIR spectrometer (Perkin Elmer). Solid-state 13C NMR spectra were recorded on a Bruker AVANCE I HD 500 MHz spectrophotometer. XRD measurements were detected using a Bruker D8 Advance with Cu Kα at the diffraction angle (2θ) of 10° to 80°. To determine sample morphology, scanning electron microscopy (SEM) studies were performed on an SEM Teneo (Thermo Fisher Scientific) occupied with an EDS to evaluate elemental analysis. TGA was recorded on a TGA4724 with a heating rate of 10 °C min−1 under an N2 atmosphere. On a Micromeritics 3 Flex 3500, N2 adsorption and desorption measurements were performed. The material surface area was calculated using the Langmuir and BET techniques. The micropore surface area was calculated using the t-plot. The material was degassed at 120 °C for 12 h under vacuum before being analyzed. The sample PSD was studied using non-local density functional theory (NLDFT). A Perkin–Elmer Optima 7000 DV Inductively Coupled Plasma Optical Emission Spectrometer was used for the determination of Pb(II), Cu(II), and Ni(II) ion concentrations.
Results and discussion
Synthesis and characterization
Polyaminal PAN-AN polymer brownish powder is insoluble in water, and the stability in various organic solvents, including DMSO, tetrahydrofuran, and dichloromethane, was examined. PAN-AN was prepared using a one-step polycondensation reaction at 175 ˚C and under an Ar atmosphere to obtain a hyper-cross-linked polymer, as shown in Scheme 1.
FTIR, solid-state 13C NMR, SEM, XRD, TGA, and surface area analysis examined the structure of PAN-AN. The FTIR spectra of melamine, 9-anthracenecarboxaldehyde, and PAN-AN are shown in Figure 1. The aminal linkage stretching vibration occurs at 3312 and 1159 cm−1. The stretching vibrations of C–N exist at 1331 cm−1. Triazine rings, characteristic bands of PAN-AN, appeared at 1542 and 1475 cm−1 (Zhang et al., 2020). Melamine exhibits bands at 3470 cm−1 and 3420 cm−1 related to the –NH2 stretching frequency. The spectra revealed the C–O stretching of the carbonyl group of 9-anthracenecarboxaldehyde at 1664 cm−1. Furthermore, the distinctive band of the aldehyde group and melamine cannot be seen in the PAN-AN spectrum. This indicates that the aldehyde group and melamine monomers have been completely transformed into polymer networks (Seong et al., 2019).
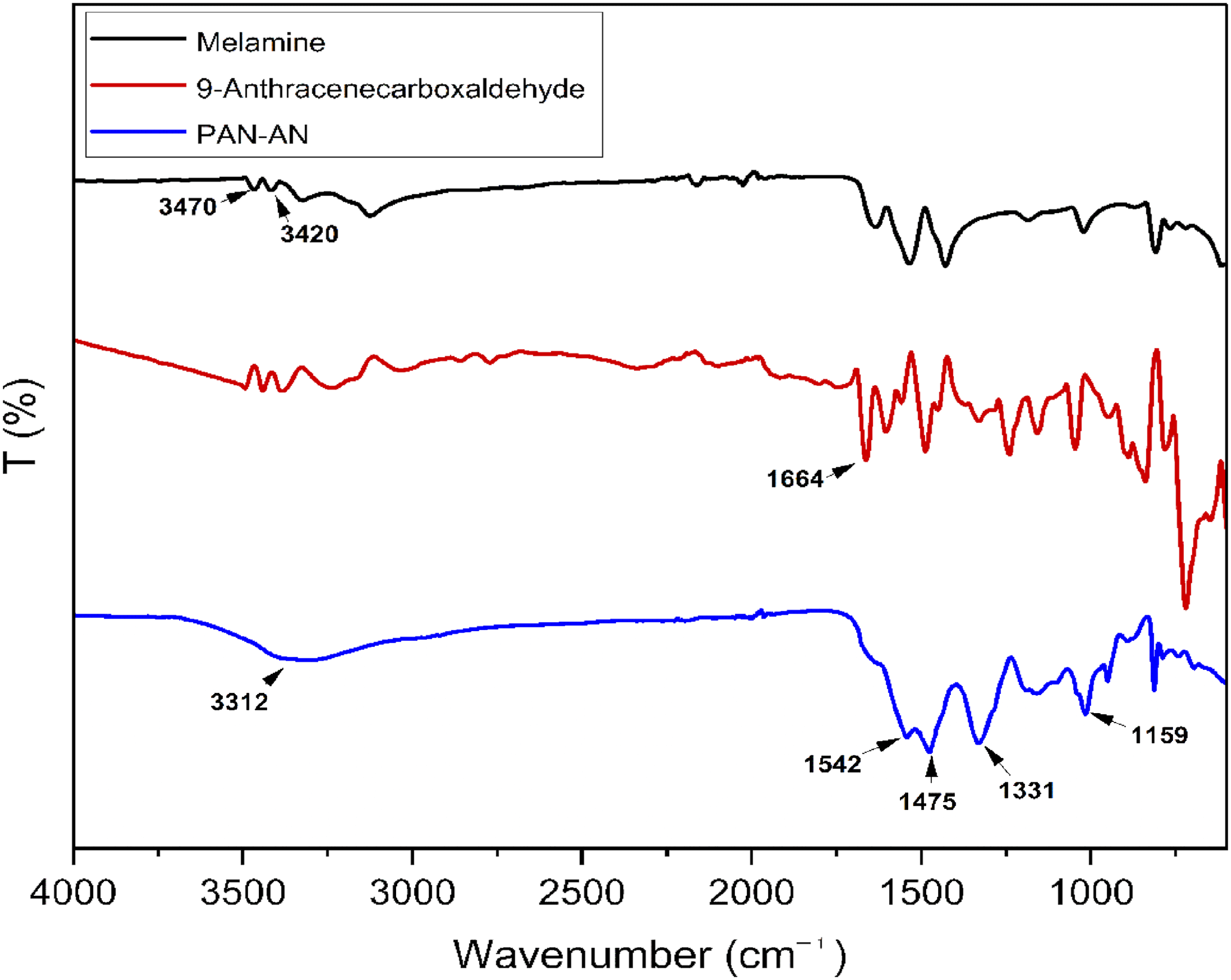
FTIR spectra of melamine, 9-anthracenecarboxaldehyde, and PAN-AN. FTIR: Fourier transform infrared spectroscopy.
The solid-state 13C NMR measurements were performed to further confirm the structures, as shown in Figure 2. The carbon triazine ring signal appeared at 166 ppm. The carbon signal for benzene appeared at 127 ppm. The resonance at 53 ppm is assigned to the tertiary carbon atom produced by aminal linkage (Shunmughanathan et al., 2015).

Solid-state 13C NMR spectra of PAN-AN (*) peak arising from the presence of the solvent DMSO. DMSO: dimethyl sulfoxide.
The XRD pattern confirmed the amorphous nature of PAN-AN by the appearance of distinct broad peaks in the region of 2θ = 20˚, as depicted in Figure 3 (c). Moreover, melamine and 9-anthracenecarboxaldehyde exhibit sharp peaks regarding crystallinity, as shown in Figure 3 (a, b), respectively, indicating that the monomers have reacted to form the polymer networks (Mohan et al., 2022).

XRD pattern of (a) melamine, (b) 9-anthracenecarboxaldehyde, and (c) PAN-AN.
To examine the surface morphology of PAN-AN, a SEM was employed. Figure 4 (a–c) shows the loose cotton-like morphology of the synthesized polymer, which is reported in the previous works (Alkayal et al., 2022; Li et al., 2016). The presence of C, N, and O is demonstrated by the EDX analysis Figure 4 (d), confirming the synthesis of PAN-AN. The elemental analysis revealed that PAN-AN contained (36.97%) carbon, (59.12%) nitrogen, and (3.91%) oxygen, demonstrating the successful synthesis of PAN-AN (Anfar et al., 2020).

SEM images for PAN-AN at (a) = 3 µ. (b) = 2 µ. (c) = 1 µ magnification, and (d) EDX analysis of PAN-AN. SEM: scanning electron microscopy.
A thermal gravimetric analysis study was performed to determine the thermal stability of PAN-AN, as shown in Figure 5. PAN-AN exhibits a three-stage decomposition. The first stage started from 38 to 84 ˚C weight loss of 5% due to solvent loss. In the second stage, at a temperature of 84–266 ˚C, weight loss occurred by 19%, related to the cleavage of the pendant group (Zhang et al., 2018). The last decomposition step at a temperature between 300 and 500 ˚C is attributed to aminal linkage decomposition. Total decomposition appeared at 500 ˚C related to the dissociation of triazine and benzene rings (Liu et al., 2020). The thermogram certifies the thermal stability of PAN-AN up to 300 ˚C.
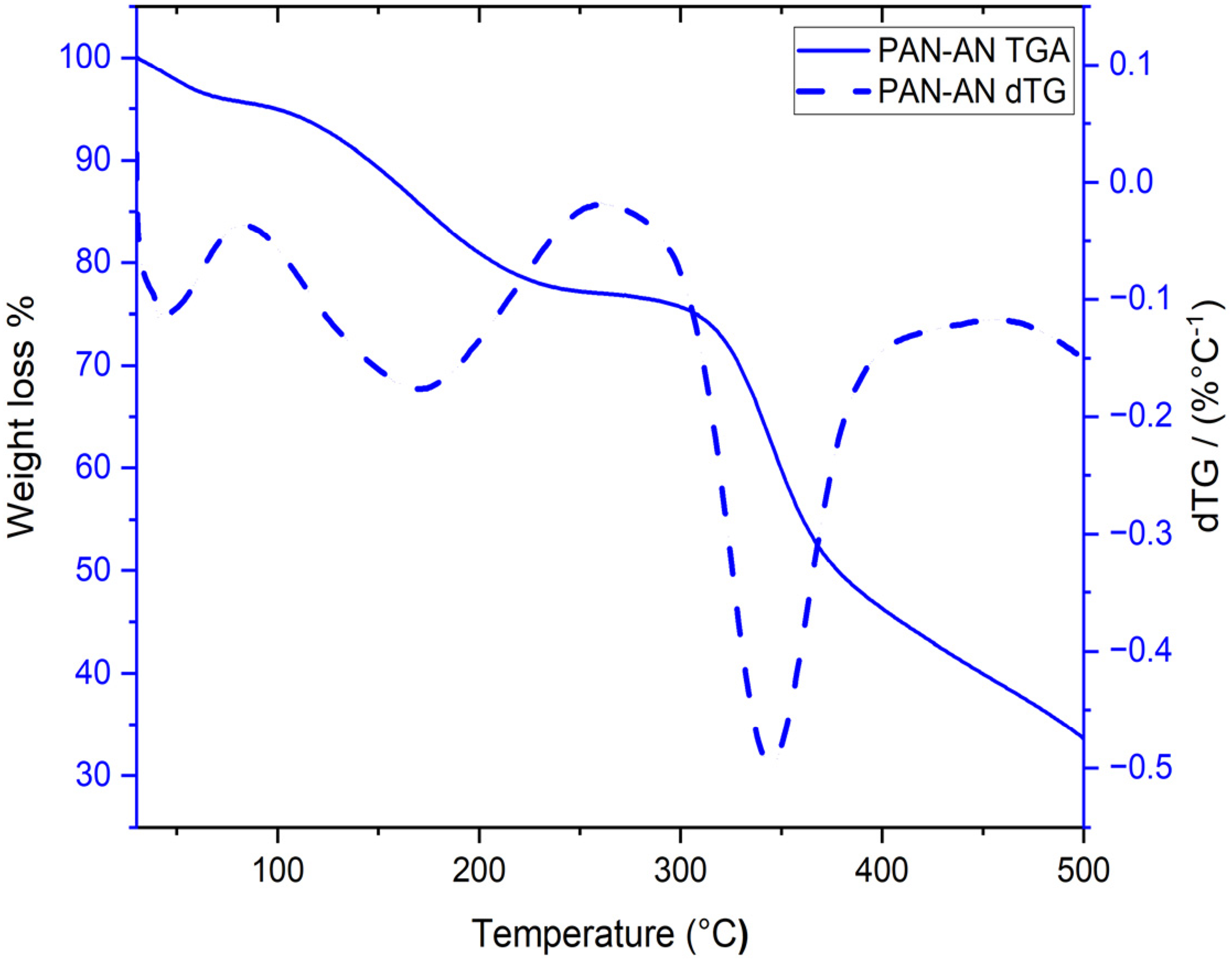
TGA (solid line) and dTG (dashed line) curves of PAN-AN.
The nitrogen adsorption measurements at 77 K were used to analyze the porosity parameters of PAN-AN. PAN-AN appears to contain mesopores, which belong to Type-IV isotherms, as determined by IUPAC classification; in addition, PAN-AN has an H4-type hysteresis loop related to narrow slit pores (Lykiema et al., 1984; Shao et al., 2019). When the relative pressure approaches 0.8, there is a significant increase in nitrogen uptake, which confirms the presence of mesoporous structure, most likely because of inter-particulate voids produced by the loose packing of small particles, as shown by SEM morphology (Li et al., 2016). The isotherm presented in Figure 6 (a) shows that the increase of nitrogen uptake occurs in the (P/P0 < 0.01) region of very low pressure, which indicates the existence of some micropores within the structure. The BET surface area is estimated to be ∼ 230.42 m2/g, while the t-plot external surface area is ∼ 213.74 m2/g, which confirmed the presence of 16.6801 m2/g microporous channels in PAN-AN structure. Figure 6 (b) illustrates the calculated NLDFT PSD, which implies that the pores in PAN-AN are classified as mesopores as the significant peak centered at ∼ 27.43 nm. The total pore volume of pores in PAN-AN is 1.11 cm3/g, and the micropore volume is 0.01 cm3/g.

(a) N2 adsorption–desorption isotherm and (b) pore size distribution of PAN-AN calculated by NLDFT. NLDFT: non-local density functional theory.
CO2 adsorption
The CO2 isotherm at 273 K was measured up to 1 bar, as illustrated in Figure 7, to evaluate the CO2 adsorption–desorption performance of PAN-AN. The CO2 uptake of PAN-AN was found to be between 0.32 and 30.08 cm3/g. The CO2 adsorption rises with pressure and has not reached saturation in the experimental pressure range, showing that a significantly higher adsorption capacity might be achieved at a higher CO2 flow rate (Li et al., 2016). The CO2 uptake of PAN-AN may be attributed to its large surface area and high N content as polar –NH and triazine rings, which results from stronger interactions of polarizable CO2 molecules with the polymer via dipole–quadrupole interactions (Ma et al., 2020). The reactant 9-anthracenecarboxaldehyde had a substantial steric hindrance, making sufficient condensation with melamine difficult; however, the solid intramolecular hydrogen bonding makes the condensation difficult to complete entirely, resulting in decreased cross-linking and the creation of few micro/mesopores channels, which result in a lower CO2 uptake by PAN-AN in comparison with a previously reported PAN-NA that shows a higher uptake at 67.9 cm3/g (Ibrahim et al., 2022; Shao et al., 2019).

CO2 adsorption–desorption isotherm for PAN-AN at 273 K up to 1 bar.
Heavy metal adsorption
The adsorption capacities (mg/g) of PAN-AN toward various metal cations and removal efficiency are shown in Table 1. PAN-AN exhibits low metal ion adsorption toward Cu(II) and Ni(II) but shows high sensitivity for Pb(II), which could be referred to as the ionic radius size, as Sandin et al. reported (Sandín et al., 2021). The removal efficiency increases as the ionic radius size becomes larger Cu(II), 0.69 Å < Ni(II), 0.72 Å < Pb(II),1.20 Å (Bohli, 2013). The Pb(II) cation was used for the following adsorption investigations. Figure 8 (a) shows SEM images of PAN-AN after Pb(II) adsorption; the morphologies demonstrate that PAN-AN maintains its structure stability. The elemental mapping analysis confirmed the presence of C, N, and O components in the PAN-AN adsorbent structure after Pb(II) adsorption. The results of Pb(II) adsorption by PAN-AN adsorbent revealed the presence of Pb(II) (1.70%) on the surface of PAN-AN. The elemental mapping study supports the distribution of Pb(II) ions on the PAN-AN adsorbent surface, as shown in Figure 8 (b) (Melhi, 2022).

(a) SEM images; (b) EDX mapping analysis of PAN-AN after Pb(II) adsorption. SEM: scanning electron microscopy.
Adsorption capacities qe (mg/g) and removal efficiency (R%) of PAN-AN to different metal ions (100 mL of 20 ppm) at an initial pH of 5.5 and an adsorbent dose of 20 mg.

Effect of Ph and adsorbent dosage
The pH of the solution is revealed to be an essential factor in determining the degree of surface charge ionization and adsorbate speciation (Kumar et al., 2014). At pH 3, practically all amine groups are protonated, reducing Pb(II) ion adsorption to a minimum. Lower pH causes protonation of amine groups, resulting in lower adsorption characteristics (Huang et al., 2019). There was a general increase in adsorption with increasing solution pH up to pH 5.5 because the amount of free amine groups in the polymer structure was substantial. The adsorption capabilities of the polymer towards Pb(II) ions are high, with a removal efficiency of 90.05%, as shown in Figure 9 (a) (Cegłowski et al., 2018). At pH 6, the presence of soluble complex Pb(OH)+ or Pb(OH)2 and the ability of these complexes to precipitate at pH > 7 make the study of the adsorption more complicated (Kumar et al., 2014). Therefore, the optimal pH for the entire experiment was conducted at pH 5.5. The removal efficiency of Pb(II) increased with increasing dosage due to more active sites becoming available at higher adsorbent concentrations. As adsorbent dosage increases, particle accumulation occurs, which reduces the overall surface area of the adsorbent, resulting in decreased efficiency of Pb(II) adsorption (Alghamdi et al., 2019). The maximum removal efficiency was found with a dosage of 20 mg adsorbent, as indicated in Figure 9 (b).

The effects of (a) pH and (b) PAN-AN dose on the Pb(II) adsorption.
Adsorption isotherms
In the range of 10–200 ppm of metal concentration, the effect of initial concentration on PAN-AN adsorption efficiency was evaluated. The initial heavy metal concentration highly influences the metal adsorption mechanism. At low concentrations, Pb(II) cations are adsorbed by active sites; however, as metal concentrations increase, the sites become occupied (Mouni et al., 2010). The maximum adsorption capacity qe was found to be 139 mg/g at 100 ppm concentration of Pb(II) cation after that qe started to decrease. The main reason behind this behavior is that when the concentration of Pb(II) was high, adsorption sites became saturated, leaving most metal ions unadsorbed. Thus, at lower concentrations, the adsorption capacity increased due to the availability of a higher adsorbent surface area (Khosravani et al., 2024). The optimal initial concentration of 20 ppm was selected for further experimentation. The influence of initial concentration on the adsorption capacity of PAN-AN is displayed in Figure 10 (a). Langmuir and Freundlich's adsorption isotherm models were used to characterize the adsorption process.

The influence of (a) initial concentrations on Pb(II) adsorption; (b) Freundlich model.
The linearized Langmuir adsorption isotherm is applied in the form:
The Freundlich adsorption isotherm can be stated as follows:
Langmuir and Freundlich adsorption isotherm parameters of Pb(II) by the PAN-AN polymer.

Adsorption kinetics
The change in adsorption capacity with contact time was investigated to evaluate the adsorption kinetics. Over the course of 20–120 min, the kinetics of Pb(II) adsorption on PAN-AN were studied. Pb(II) adsorption occurred rapidly in the first 20 min, as shown in Figure 11 (a), and equilibrium was reached. The experimental data were examined using pseudo-first-order and pseudo-second-order kinetic models. The following equations were used to apply both kinetic models:

The effect of (a) contact time on Pb(II) adsorption; (b) pseudo-second order.
Pseudo-first-order kinetic model.

Kinetic parameters were calculated for pseudo-first-order and pseudo-second-order Pb(II) adsorption on PAN-AN.

The correlation coefficients R2 = 1 and the estimated adsorption capacity (qe = 91.05 mg/g) from the experimental results, which agree with the calculated value (qe = 90.82), indicate that adsorption follows a pseudo-second-order kinetic model, as shown in Figure 11 (b), demonstrating that Pb(II) adsorption on the PAN-AN surface is accomplished through a chemisorption mechanism (Ha et al., 2020).
Comparison of PAN-An network adsorption capacity with various melamine-based POPs studies on Pb(II) removal
The capability of the PAN-AN network tested in this investigation to remove Pb(II) from aqueous solutions was compared to prior studies (Ibrahim et al., 2022; Sandín et al., 2021; Seong et al., 2019), and the results are provided in Table 4. PAN-AN performs well in Pb(II) adsorption, with a duration of 1 h and an adsorption capacity of 91.05 mg/g. The greater capacity found can be attributed to the presence of additional active sites the absorbent material provides to interact with lead ions (Taskin et al., 2016). The following reasons can explain the higher adsorption capacity: (i) the addition of distinct anthracene spacer groups, which could decrease hydrogen bonds and thus promote the numerous amine and triazine groups (Yang et al., 2010); (ii) the amorphous structure and the existence of micropores which is highly advantageous for heavy metal ion adsorption (Yang et al., 2010); and (iii) As coordinating ligands, amine groups can form powerful bonds with the Pb(II) cation (Sandín et al., 2021).
Comparisons of adsorption capacities of PAN-AN polymer with the various melamine-based POPs studies on Pb(II) removal.

BET: Brunauer–Emmett–Teller; POPs: porous organic polymers.
Conclusion
A novel polyaminal-linked porous network was synthesized from melamine and 9-anthracenecarboxaldehyde via one-pot polycondensation. The structure and porosity characteristics of the synthesized polymer PAN-AN were studied. The CO2 adsorption of the adsorbent was evaluated at 273 K and 1 bar. The PAN-AN, with a BET surface area of about ∼ 230.42 m2/g, can capture 30.08 cm3/g CO₂ gas. The polymer's potential to adsorb heavy metals was studied. As a result, the PAN-AN exhibits high adsorption for Pb(II) removal at pH 5.5, 20 mg adsorbent dosage, and rapid removal in the first 20 min, with a removal efficiency of 90.05%. The synthesized polymer is a promising gas adsorbent for environmental applications such as gas storage and separation. It is an excellent adsorbent with good qualities for removing Pb(II) cations, which makes PAN-AN recommended for water treatment applications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.