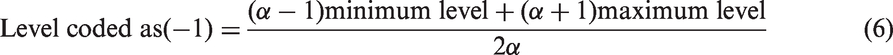Abstract
The present study focuses on the response surface methodology (RSM) for the optimization of lead removal from an aqueous solution by a novel superparamagnetic nanocomposite. A rotatable central composite design and the response surface methodology were used to conduct and to analyze the experiments, respectively. The adsorption process was investigated as a function of the four factors consisting of pH (4.0–6.0), temperature (20℃–60℃), initial lead concentration (10–90 mg/L) and adsorbent dosage (0.2–1.0 g/L). The maximum lead adsorption capacity was obtained to be 124.955 mg/g under the optimal conditions of 5.49, 60℃, 89.08 mg/L, and 0.48 g/L for the solution pH, temperature, initial lead ion concentration, and the adsorbent dosage, respectively. The desirability function was used to find an optimum point where the desired conditions could be obtained. The superparamagnetic nanocomposite could be used as an adsorbent for the removal of toxic heavy metals from water and wastewater.
Introduction
Environmental contamination of heavy metals has become a worldwide problem and a special attention has been given to them because of their high toxicity, nonbiodegradability, and accumulation in the food chain (Javanbakht et al., 2011; Kumar et al., 2014; Kumari et al., 2015; Kumari and Tripathi, 2015; Mendoza-Castillo et al., 2015; Moftakhar et al.; Singh and Prasad, 2015; Song et al., 2011; Xu et al., 2012a). The heavy metal sources from industry include fertilizers, pigments, batteries, and so on (Javanbakht et al., 2014; Mahdavi et al., 2012; Nędzarek et al., 2015; Sarioglu et al., 2009; Şen et al., 2015; Tan et al., 2012). Heavy metals, even at trace level, are very harmful for human beings. Thus, the removal of heavy metals from water systems is a very critical and essential topic and has attracted a considerable attention (Ahmed et al., 2013; Lata et al., 2015).
Different methods have been proposed for the efficient heavy metal removal from water systems, including chemical precipitation, ion exchange, adsorption, membrane filtration, and electrochemical technologies (Salam, 2013; Shen et al., 2012). Among these techniques, adsorption as a cost-effective technology offers flexibility in design and operation, and in many cases it will generate high-quality treated effluent (Gong et al., 2012; Komkiene and Baltrenaite, 2016). In addition, a lot of adsorbents can be regenerated by desorption processes of ease of operation, for multiple use. Thus, the adsorption process has come as one of the major techniques for the removal of heavy metals from water and wastewater effluents (Xu et al., 2012a).
Various natural materials such as clay, seaweed, and adsorbent, as well as synthetic adsorbents such as activated carbon, resin, and mesoporous silica, have been used to remove heavy metals (Pang et al., 2011). Among minerals that possess adsorbent properties, natural zeolite appears as one of the most promising minerals for metal purification (Kurniawan et al., 2006; Monroy-Figueroa et al., 2015). The special structural, physical, and chemical properties of zeolites make them valuable for many industrial, agricultural, and environmental applications (Liu et al., 2013). Natural zeolites are low cost and available materials functioning as excellent cation exchangers (Oliveira et al., 2004). Clinoptilolite (CPL), a hydrated alumina–silicate member of the heulandite group, is one of the most common natural zeolites, being widespread in the world (Dinu and Dragan, 2010).
Recently, magnetic nanocomposites have attracted much attention because of their widespread application in different fields such as adsorption, mineral separation, catalysis, drug delivery, and magnetic resonance imaging (Faraji et al., 2010; Huang and Chen, 2009; Mahdavian and Mirrahimi, 2010; Mahmoud et al., 2013; Panneerselvam et al., 2011; Unsoy et al., 2012). Magnetic adsorbents can be easily separated from the solution by an external magnetic field and therefore, adsorbents combining magnetic separation technique and nanotechnology would remove heavy metals in perfect performance (Chang et al., 2012; Gao et al., 2013; Gong et al., 2009; Xu et al., 2012b). Magnetic composite particles can be designed to have specific physical, chemical, and surface properties that allow the selective attachment of ions (Hakami et al., 2012). Several magnetic nanomaterials, including magnetite and maghemite nanoparticles, functionalized or modified with some compounds, have been explored for the removal of heavy metals ions (Faghihian et al., 2014; Hao et al., 2010).
Chitosan (CS), a nontoxic, biocompatible, and biodegradable material has been investigated as adsorbent for the heavy metal removal (Ngah et al., 2011). It is one of the cheap and abundant biopolymers with the ability to form complexes with metals (Babel and Kurniawan, 2003; Kyzas and Deliyanni, 2013). Numerous studies have been done to the modification of the CS surface by the cross-linking with different functional agents, such as epichlorhydrin, glutaraldehyde sodium, and tripolyphosphate, to prevent CS dissolution in an acidic medium (Dinu and Dragan, 2010; Guibal, 2004).
The objective of this work was to study the effect of pH, temperature, initial lead concentration, and adsorbent dosage on the lead adsorption as a toxic heavy metal using a novel superparamagnetic CS/CPL nanocomposite. We synthesized the nanocomposite (adsorbent) by embedding a magnetite in a matrix of the zeolite microparticles, cross-linking them with glutaraldehyde and coating the product with the chitosan to improve the stability and adsorption capacity of the nanocomposite. Optimization of the lead adsorption by the classical method involves changing one independent variable (i.e., pH, temperature, lead ion concentration, and adsorbent dosages) while maintaining the others at a fixed level. The experimental factorial design and response surface methodology (RSM) can be employed to optimize the lead adsorption onto the magnetic CS/CPL nanocomposite in an aqueous solution. For a better understanding of different stages of the adsorption at a varying pH, temperature, lead concentration, and adsorbent dosages, the RSM (under DESIGN EXPERT software) was used to optimize the lead adsorption.
Materials and methods
Materials
The CPL sample (KNa2Ca2(Al7Si29)O72·24H2O) was collected from Semnan region in the central Alborz Mountains, Iran. The CS as a powder (with a medium molecular weight and a deacetylation degree of 75–85%) was obtained from the Sigma–Aldrich. Pb(NO3)2, NaOH (99%), HCl (99%), HNO3 (99%), and glutaraldehyde solution (25%) were supplied by the Merck company (Germany). FeCl3.6H2O and FeCl2.4H2O were supplied by the UNI-CHEM, Germany. All the solutions were prepared with distillated water. Also, all the chemical reagents were of an analytical reagent grade.
Synthesis of the modified CPL (CPL/Fe3O4)
A known amount of the CPL powder was added to a 30 mL of distilled water. The mixture was homogenized using a vigorously mechanical stirring (350 r/min) for 10 min. The solutions of FeCl2·4H2O and FeCl3·6H2O in water with a molar ratio of 2:1 were prepared and were mixed together and then were deoxygenated by bubbling of Argon (Ar) gas. The initial pH of the solutions was adjusted to approximately 2 with 2 M HCl. About a 9 mL of 1 M NaOH solution was added dropwise to the suspension while the mixture was vigorously mechanical stirred under an ultrasonic irradiation and in Ar atmosphere for 1 h. The prepared composite (zeolite powder, iron oxide with a 1:1 mass ratio) was separated using a magnet. The product was washed with distilled water for four times and then was dried at 80℃ in a vacuum oven for 24 h.
Preparation of the magnetic CS/CPL nanocomposite
In this research, the CPL/Fe3O4 nanocomposite was synthesized by embedding magnetite nanoparticles in a matrix of the CPL microparticles with precipitation method and then, crosslinked the CS around the CPL/Fe3O4 nanocomposite with glutaraldehyde crosslinker to improve the stability and adsorption capacity of the nanocomposite. As the prepared CPL/Fe3O4 nanocomposite (50 mg) was completely washed and dispersed into distillated water, the initial pH of the suspension was adjusted to about 11 with 1 M NaOH. Glutaraldehyde solution (0.200 mL) as a crosslinker was dropped into the suspension with a continual stirring of 200 r/min under the ultrasonic irradiation for 30 min. Next, a solution of chitosan (CS/CPL with a 1:5 mass ratio) in an acetic acid with 1 wt% was added dropwise to the suspension while the mixture was vigorously mechanical stirred under the ultrasonic irradiation for 1 h. As a novel way to prevent deterioration of synthesized magnetite when adding acidic chitosan solution, an alkaline solution was added dropwise to maintain the suspension pH of about 11. After the addition was completed, the stirring was continued for 60 min, then the solid–liquid separation was performed using a magnetic field. The product was washed and then was dried at 70℃ for 44 h. Finally, it was grinded to obtain the nanocomposite (adsorbent).
Analytical methods
The scanning electron microscopy (SEM) (Cam-Scan MV 2300) was used to study the surface of the adsorbent. Prior to the SEM studies, the samples were coated with a thin layer of gold. The crystal structure of nanocomposite was examined by a JEOL JDX-8030 diffractometer using a Cu Kα radiation (λ = 0.1541 nm) in the range of 10–90° (2θ) (X-ray powder diffraction (XRD)). The Fourier transform infrared (FTIR) spectra of the nanocomposite were analyzed using a Bruker Tensor 27 spectrometer. The magnetization was measured at room temperature with a vibrating sample magnetometer (VSM, MPMS-5 SQUID). The concentration of lead solution was determined using a Buck 210VGP atomic absorption spectrophotometer. A stock lead ion solution of 1000 mg/L was prepared by dissolving 1.5980 g of lead nitrate into a 1000 mL of deionized water. The solution was diluted for different considered ion concentrations. The pH of the solutions was adjusted by adding 1 N NaOH and 1 N HNO3 solutions.
Lead adsorption process
By means of the RSM, 30 batch adsorption experiments were designed and the effect of the pH, temperature, initial lead concentration, and the adsorbent dosage on the lead removal was studied. Each experiment was carried out in 50 mL of the lead solution under a mechanical stirring at 100 r/min for 120 min to reach the equilibrium and finally, the adsorbent was separated from the solution using a magnetic field. The separated solutions were analyzed for the residual lead ion concentration. The removal capacity of lead (Y) was given by
Design of experiments
The RSM consists of several experimental techniques dedicated to the evaluation of relationship between a group of controlled experimental factors and measured responses based on one or more criteria (Amini et al., 2008; Ghorbani et al., 2008; Radaei et al., 2014). RSM can be used to evaluate the effects of individual parameters, the interaction of variables, and the optimum conditions for responses (Ghoreishi et al., 2012; Roosta et al., 2014). A five-level, four-factor central composite design (CCD) was used to investigate the effect of the selected parameters on the lead removal capacity by the adsorbent particles. The pH (X1), temperature (X2), initial lead concentration (X3), and the adsorbent dosage (X4), were selected as the independent variables, and the lead removal capacity was considered as the response (dependent variable). The variables were coded according to the following equation
A CCD is made rotatable by the choice of the rotatability factor α which depends on the number of the points in the factorial portion of the design as expresses
The levels and ranges of the independent variables.

Experimental conditions and values obtained through the CCD.

Observed and predicted values.

The independent variables vary between the lowest level of −1 and the highest level of +1. The axial points are located at the distance α from the center and make the design rotatable. The experimental results were analyzed using the Design Expert 7.0.0 (Stat-Ease, Inc., Minneapolis, MN, USA) software and the mathematical relationship between the four independent variables were approximated by the second-order polynomial model (Amini et al., 2008; Ghorbani et al., 2008; Ghoreishi et al., 2012; Radaei et al., 2014; Roosta et al., 2014).

Results and discussion
Characterization of the adsorbent
Figure 1(a) and (b) show the SEM image and the surface morphologies of the modified CPL and the nanocomposite. The white spots in Figure 1(b) show the presence of the chitosan covering the CPL/Fe3O4 nanocomposite surface.
SEM micrograph of (a) the modified CPL and (b) the nanocomposite.
In the FTIR result of the nanocomposite (Figure 2), the band at around 3430 cm−1 can be assigned to the stretching modes of the OH groups on the surface of the magnetite particles or to the N–H extension vibration on the surface of the Cs (Batista et al., 2011). The C–H stretching vibration of the Cs is manifested through a strong peak of 2923 cm−1 while the characteristic band centered at 1634 cm−1 can be attributed to the N–H bending of the NH2 group (Unsoy et al., 2012). The Fe–O–Fe band observed at 575 cm−1 corresponds to the intrinsic stretching vibrations of the metal (Ozkaya et al., 2009; Petcharoen and Sirivat, 2012). The absorption appeared in the regions of around 450 and 800 cm−1 is resulted from the stretching and bending modes of Si–O or Al–O in the CPL framework (Faghihian et al., 2014; Mansouri et al., 2013). The bands at 1417 cm−1 can be attributed to the CH3 bending vibration (Kyzas and Deliyanni, 2013).
FTIR results of the nanocomposite.
The XRD results of the nanocomposite represent the characteristic peaks of the crystalline magnetite at 2θ = 30.4, 35.5, 43.1, 53.4, 57.0, and 62.9° marked by their indices (2 2 0), (3 1 1), (4 0 0), (4 2 2), (5 1 1), and (4 4 0), respectively, of the crystalline CPL at 2θ = 22.5, 26.0, and 42.9°, and of the chitosan at 2θ = 20.2° (Figure 3). There is no significant shift in the characteristic peaks of the magnetite and zeolite in the nanocomposite particles. This reveals that the crystallinity of the magnetite and zeolite is still retained in the nanocomposite. Indeed, for a relative change in the peak intensities ca. the reaction has not altered the crystalline structures of the magnetite and zeolite. It is also suggested that the Cs modification does not change the phase of nanocomposite compositions.
XRD results of (a) magnetite, (b) CPL, (c) Cs, and (d) nanocomposite.
The nanocomposite revealed good magnetic properties in the magnetic field. After the preparation, a test with a magnet showed that the particles were superparamagnetic and were completely attracted to the magnet. The saturation magnetization of the nanocomposite is 9.50 emu g−1 (Figure 4).
The hysteresis loops of the nanocomposite.
Lead adsorption process
In this section, the effects of process parameters on the removal response are presented. The removal of lead as a function of the pH and the adsorbent dosage is given in Figure 5(a). The Figure 5(a) shows that the lead removal decreased with increasing the adsorbent dosage from 0.2 to 1.0 g/L. Although increasing the adsorbent dosage can be attributed to the increased surface area and adsorption sites of adsorbent, the values of lead removal decreased with increasing the adsorbent dosage (Ghorbani et al., 2008). The primary reason is that the adsorption sites remain unsaturated during the adsorption reaction whereas the number of sites available for the adsorption increases by increasing the adsorbent dosage. The agglomeration of the adsorbent can also be a reason for the decrease in the lead removal capacity at the high concentrations of the adsorbent. This means that the higher values of lead adsorption are obtained by simultaneously, the decrease in the adsorbent dosage and the increase in the pH. This may be explained by the increase in availability of the adsorption sites at a higher pH which improved the access of lead to the adsorption sites of the adsorbent. In fact, the protons will combine with the lead ions for the ligands and consequently, will decrease the interaction of lead ions with the adsorbent (Tan et al., 2012).
Response surface plots for the effect of (a) the pH and the adsorbent dosage (g/L); (b) the pH and the temperature (℃); (c) the pH and the initial lead concentration (mg/L); (d) the temperature (℃) and the adsorbent dosage (g/L); (e) the initial lead concentration (mg/L) and temperature (℃); (f) the adsorbent dosage (g/L) and the initial lead concentration (mg/L); on the lead removal.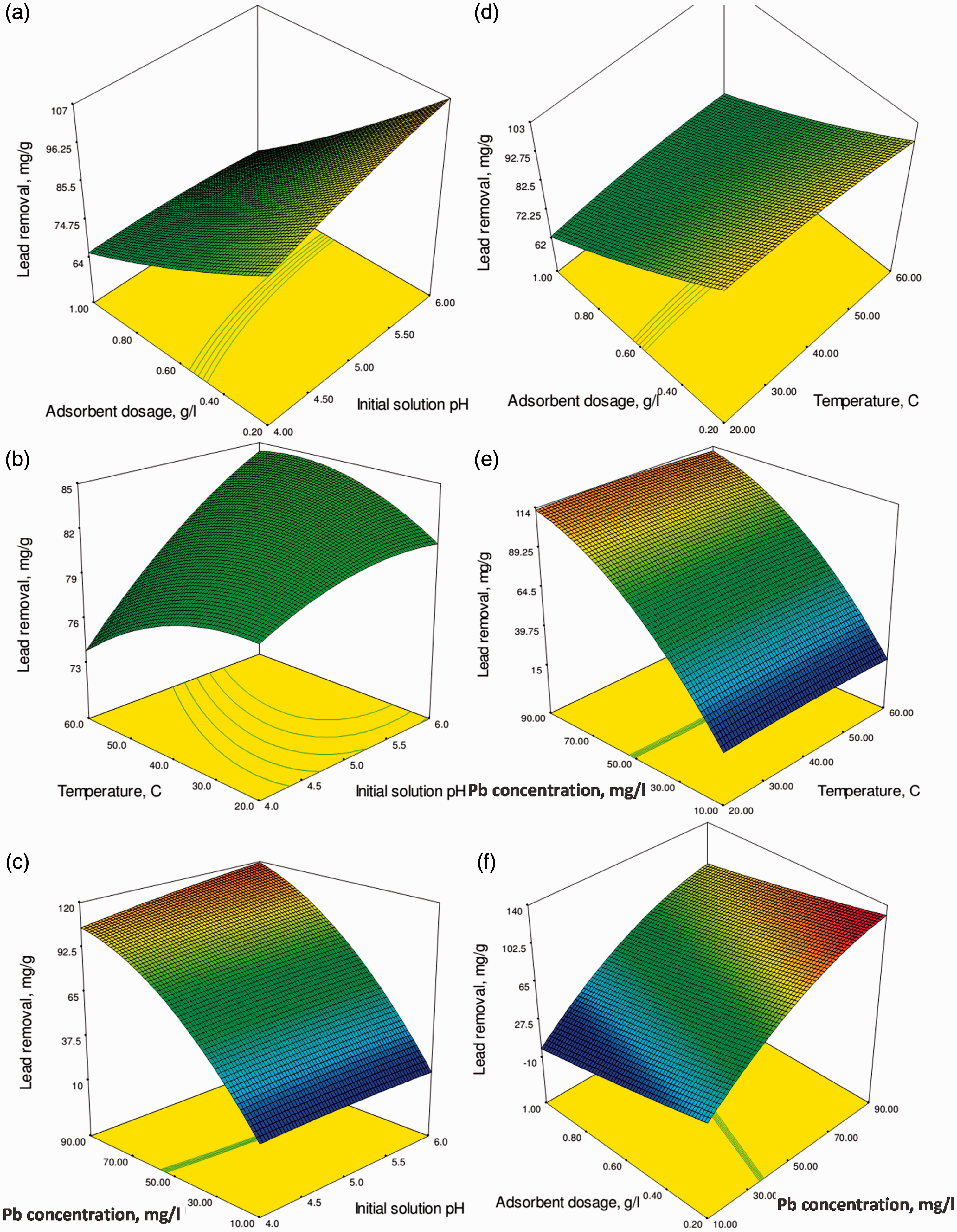
The effect of temperature and pH on the lead removal is shown in Figure 5(b). Increasing the temperature in a lower pH had a positive effect on the lead removal. The increased lead removal with increasing the temperature may be a result of the faster chemical precipitation rate of the lead hydroxide at higher temperatures (Ahmadi et al., 2014). The effect of the initial pH and the initial lead ion concentrations on the lead removal is shown in Figure 5(c). The lead removal increased with an increase in the initial lead ion concentration varying from 10 to 90 mg/L and with an increase in the initial solution pH varying from 4 to 6 in the higher lead ion concentrations.The solution pH affects both the adsorbent surface adsorption sites and the lead chemistry in solution. The functional groups such as amino groups and hydroxyl of Cs were found to be responsible in the adsorption of lead ions (Ngah et al., 2011). This proofs that the lead adsorption occurs through the ion exchange mechanism in witch, the lead ion binds to adsorption sites by replacing two acidic H+ at a low pH. However, the amount of the lead adsorbed is influenced by the properties of the adsorbent surface. An increase of the lead removal by increasing the initial lead concentration is a result of the increase in the driving force of the lead concentration gradient (Javanbakht et al., 2011). The removal of lead as a function of the adsorbent dosage and the temperature is shown in Figure 5(d). The results showed that the lead removal decreased with increasing the adsorbent dosage from 0.2 to 1.0 g/L. Thus, the effect of the adsorbent dosage is more significant than that of the temperature. Figure 5(e) shows the effects of the temperature and the initial lead concentration on the lead removal. The effect of the initial lead concentration is found to be more significant than that of the temperature. Figure 5(f) depicts the lead removal as a function of both the adsorbent dosage and the initial lead concentration.
Fitting the model
The analysis of variance (ANOVA) was used to evaluate the statistical significance of the quadratic model. It was found that the regression was statistically significant at the F-value of 204.42 and the values of prob > F (<0.0001). The determination coefficient (R2) was used to check the accuracy of the model. The determination coefficient (R2 = 0.9948) indicated that the model cannot explain only 0.52% of the total variable. The adjusted determination coefficient (R2adj = 0.9899) is also high, indicating a high significance of the model. The predicted R2 (R2pred) is also high enough to indicate the high significance of the model. The R2pred is 0.9707, revealed that it is in a reasonable agreement with the R2adj and showing that the model does not explain only 2.93% of the total variations. “Adeq Precision” gives the ratio of the signal to the noise. A ratio greater than 4 is desirable. Thus, the Adeq precision of 54.891 indicates an adequate signal. This model can be used to navigate the design space. A low value of the variation coefficient (CV = 3.50) shows reliability of the experiments.
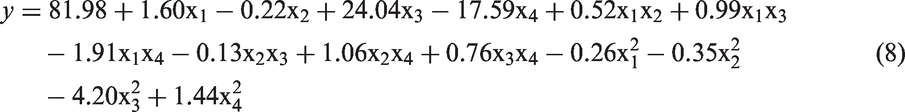
ANOVA for the response surface quadratic model.
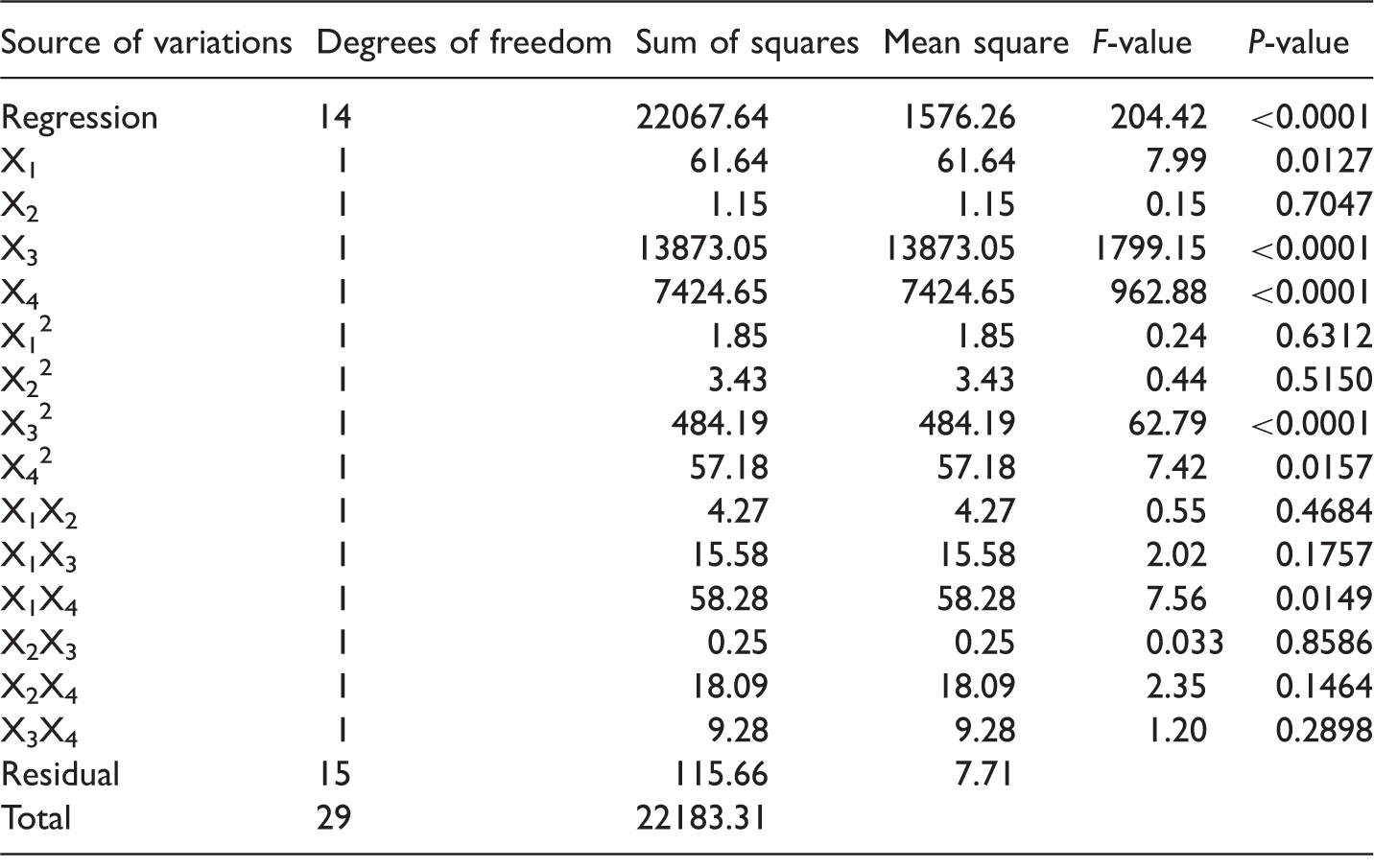
Note: R2 = 0.9948; R2adj = 0.9899; R2pred = 0.9707; CV = 3.50.
Regression analysis using the 24 factorial CCD.

Note: X1, X2 X3, and X4 are the main effects; X21, X22, X23, and X24 are the square effects; X1X2, X1X3, X1X4, X2X3, and X2X4 are the interaction effects.
In order to estimate the adequacy of the regression model, the diagnostic plots are given in Figure 6. The actual and the predicted removal capacity values are given in Figure 6(a). As can be seen, the model adequately explains the experimental studied range, and there are tendencies in the linear regression fit. The normal percentage probability plot is presented in Figure 6(b). The data points show that neither there is any apparent problem with normality nor the response transformation is required. The plot of the residuals versus the fits was used to examine the reliability of the model, as illustrated in Figure 6(c). As a result, no series of the increasing or decreasing points, of the patterns such as the increasing residuals with increasing fits and of a predominance of the positive or negative residuals should be found which is fortunately met in our work.
(a) The actual and the predicted lead removal capacity, (b) the studentized and the normal percentage probability plot of the lead removal capacity, and (c) the predicted removal and the studentized residual plot.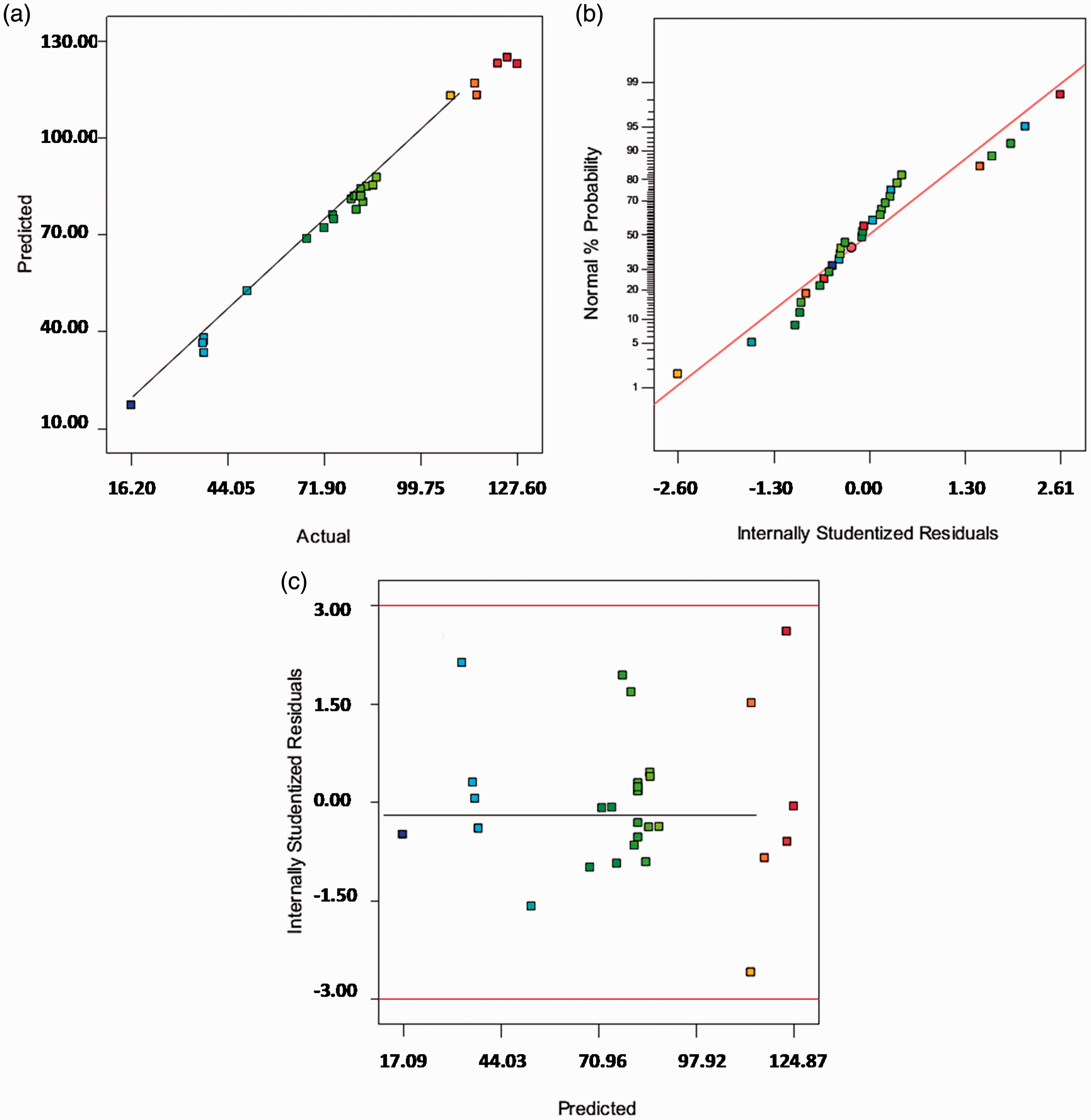
Optimization using the desirability function
For the numerical optimization, we consider the desired goal for each factor and the corresponding response (Batista et al., 2011). A weight can be assigned to each goal to adjust the shape of its particular desirability function. Our goals are the pH, temperature, initial lead concentration, adsorbent dosage, and the removal of lead. The overall desirability function, made of a combination of the goals, is an objective function ranging from zero outside of the limits to one at the goal. The function can be maximized by using a random starting point and then proceeding along the steepest slope up to a maximum. Starting from several points in the design space increases the chance of achieving the “best” maximum among all possible maxima of the function. A multiple response method was applied for optimizing any combination of our goals. The optimization resulted in a point that maximizes the desirability function. The maximum level of the initial lead concentration (90 mg/L), the minimum level of the adsorbent dosage (0.2 g/L), the level of the pH within the range of 4.0–6.0, the level of temperature within the range of 20.0–60.0℃, and the level of the removal capacity within the range of 16.2–127.6 (mg/g) were set for the maximum desirability. Figure 7 shows a ramp desirability made from 30 optimum points via the numerical optimization. By seeking 30 starting points in the response surface changes, the best local maximum was found to be at the pH of 5.49, the temperature of 60.0℃, the initial lead concentration of 89.08 mg/L, the adsorbent dosage of 0.48 g/L, the lead removal of 124.955 mg/g, and the desirability of 0.864. The desirability of 0.864 indicates that the estimated function may express the desired conditions and the experimental model.
Desirability ramp for the optimization of the response and the variables.
Comparison of adsorption capacity of various adsorbents for lead ions.

Conclusion
The results of this work show that the nanocomposite adsorbent is an efficient adsorbent of the lead in dilute solutions and has high adsorption yields for the treatment of wastewater containing lead ions. Adsorption processes were performed as a function of the pH, temperature, initial lead concentration, and the adsorbent dosage. The optimization of the lead adsorption by the RSM resulted in 124.955 mg/g. The levels of the following four variables were found to be optimal for the maximum lead removal: the pH of 5.49, the temperature of 60.0℃, the initial ion concentration of 89.08 mg/L, and the adsorbent dosage of 0.48 g/L. The scanning electron microscope and the energy dispersive X-rays results of the adsorbent show the presence of the chitosan covering the CPL/Fe3O4 nanocomposite surface. According to these observations, the nanocomposite revealed good magnetic properties in the magnetic field so that after the adsorption process is completed, the nanocomposite was completely attracted to the magnet.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Isfahan University of Technology (IUT).