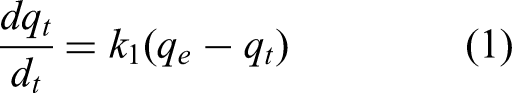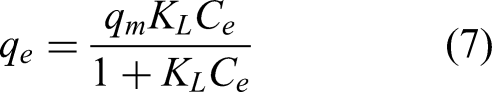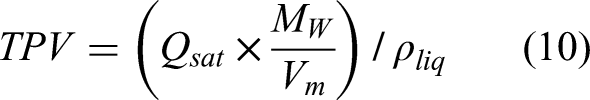Abstract
A Scottish wood biochar sample was investigated for water remediation against persistent organic pollutants as a potential renewable material for adsorption processes. Textural characterisation gave a high surface area (588 m2/g) and a mix of microporous and mesoporous nature with an average pore width of 4 nm. Morphological analysis revealed a layered carbon structure and spectroscopic analysis showed the presence of oxygen and nitrogen-based functionalities alongside 80% atomic carbon. The biochar had an average point of zero charge of 7.44 ± 0.2. 3,4-Dichloroaniline kinetic rates were rapid (<5 min), restricting kinetic analysis, while a pseudo-second-order kinetic model was best suited to represent the kinetic data for acetaminophen and carbamazepine, suggesting chemical control. The adsorption equilibria were most appropriately described by the Sips isotherm model, further supporting the chemical control theory for a multilayer system. Maximum adsorption capacity was 126 mg/g for acetaminophen removal, 40 mg/g for carbamazepine and 83 mg/g for 3,4-dichloroaniline. The biochar demonstrated good removal efficiency against all target species, showing potential as an adsorbent for water remediation.
Introduction
The demands on freshwater supplies to support growing populations and settlements is a key issue, particularly for communities located in water-stressed regions. Available freshwater amounts to 2.5% of total global water resources (International Energy Agency, 2017); in 2018, an estimated two billion people were living in countries experiencing water scarcity (United Nations Educational Scientific and Cultural Organization, 2021). Many also experience ‘economic’ water shortage, with ∼1.6 million people residing in areas where water is physically available but there is an acute shortage of the required infrastructure required to access those water resources (United Nations Educational Scientific and Cultural Organization, 2021). A significant disparity exists in gathering the required water quality data, due to a lack of monitoring and reporting, especially in developing nations.
Water quality has been severely affected by pollution via natural and anthropogenic factors. On a global scale, approximately 80% of industrial and municipal wastewater is discharged into the environment without undergoing any form of treatment beforehand. The risks associated with emerging pollutants, including micropollutants, have been recognised since the early 2000s (Bolong et al., 2009). Organic pollutants can be either synthetic compounds or oxygen-demanding wastes produced from household or industrial activities. They contain compounds such as hydrocarbons, detergents, insecticides and herbicides, lubricants, endocrine disrupting chemicals (EDCs), pharmaceuticals, etc. that have detrimental effects on human and environmental health (Burton and Pitt, 2001; Verla et al., 2019). Among these, EDCs and pharmaceutical compounds are of growing concern due to release into the environment through engineered and natural pathways, without adequate monitoring systems. Such chemicals can mimic hormones within the body and interfere with the endocrine system and adversely affect bodily functions (Crisp et al., 1998). Even at low concentrations, prolonged exposure can result in adverse health effects. An EDC of particular concern is 3,4-Dicholoroaniline (3,4-DCA), a derivative of aniline with high global annual production (Livingston and Willacy, 1991) intermediate in the production of urea herbicides (Luca Tasca and Fletcher, 2017) in the synthesis of fabric pigments, and as a paint precursor (Ibrahim et al., 2021; Nurul et al., 2021). Known to be toxic to aquatic organisms and humans, with the potential to cause long-term adverse effects in aquatic environments, the predicted no-effect concentration (PNEC) for 3,4-DCA is 0.2 µg/L. In addition, the detection of Active Pharmaceutical Ingredients (APIs) in various surface waters has sparked concerns regarding their potential impact on the environment and living organisms and common pollutants in this category include acetaminophen (APAP) and carbamazepine (CBZ). APAP, commonly known as paracetamol, is an analgesic used to mitigate moderate to severe pain (Rodriguez-Narvaez et al., 2017). Healthcare facilities, improper household disposal, inadequate treatment and veterinary treatment provided to livestock are pathways for water contamination. APAP can accumulate in the tissues of aquatic organisms, posing risks to higher trophic levels in the food chain. In humans, it has been reported to cause acute liver failure and is the second most frequent reason for liver transplantation in the USA (Larson et al., 2005). CBZ is extensively used worldwide as a multifunctional medication, serving both as an anticonvulsant and a pain-reliever (Décima et al., 2021) contamination pathways are similar to those for APAP, with a range of environmental and human health issues (Harvey, n.d.; Vaseashta, 2009) CBZ is among the emerging organic contaminants detected at the highest concentration with highest concentration (∼4.6 µg/L) (Loos et al., 2013). All of these pollutant species pose a challenge for microbial degradation or decomposition in the environment, and it is necessary to develop new methods of removal from water streams; this is increasingly complex when designing systems that can target a range of species.
Photocatalytic degradation processes using biochars can target most organic pollutants including EDCs. Photocatalytic processes were used for the remediation of antibiotics and organic dyes using bismuth-based nanostructured photocatalysts. The catalysts offer the advantage of narrow bandgaps and layered structures, however, are limited in application, due to their solubility and instability at low pH, as well as issues with recovery and toxicity (Oladipo and Mustafa, 2023). The process additionally raises concerns about the formation of intermediate species, as well as capital investment, despite the advantage of the high stability offered by biochar-based catalysts (Qiu et al., 2021). Photocatalysis of 3,4-DCA, using Ti-N and Ti-S catalysts, resulted in full degradation of 3,4-DCA in 2 h at an optimal pH of 6 for Ti-N (Ellappan and Miranda, 2014). Ti-N catalysts presented a higher surface area and lower particle size compared to Ti-S catalysts. The catalyst dosage above 0.1 g/L was observed to be inversely proportional to the reaction rate. Coagulation–flocculation systems are advantageous to remove lighter particles that do not easily settle in suspension. Certain inorganic metal salts are also readily available and cheap to add to these systems. Several coagulants, however, can cause oxidation–reduction reactions in suspension and there are also possibilities of corrosion, and the production of toxic sludge, while extensive polymer usage has attracted criticism from an environmental standpoint (Iwuozor, 2019). Cost analysis of antibiotic removal using Al and Fe anodes revealed an operational cost of 0.166 US$/m3 for a 200 mL reactor volume. Despite the process being reportedly economical, scaling up such a system, especially the fabrication of bigger reactors with higher pollutant loads would be challenging (Oladipo et al., 2022). To overcome such issues, adsorption systems using biochars have gained considerable interest. APAP removal using advanced oxidation processes has been widely studied over the years. Removal of APAP in such systems is generally instantaneous with efficiencies typically >75%. The conventional Fenton process can be modified into photo-based or electro-based processes. Both processes operate via the generation of hydroxyl radicals using UV radiation and electrodes, respectively. An investigation into the removal of 5 mM of APAP using UV radiation at 360 nm resulted in 99% removal in under 40 min (Su et al., 2013). Different electrode materials, such as Ti/boron-doped diamond and Pt sheets or gauzes have been tested for APAP removal and gave satisfactory results (He et al., 2015). Both processes can target high APAP concentrations; however, the operation of these processes requires a high capital investment due to electricity consumption (Phong Vo et al., 2019). For CBZ, the presence of electron-rich moieties makes it easier to react with strong hydroxyl radicals, and even ozone (Rizzo et al., 2019). However, the presence of other components, such as suspended solids and organic matter, can compete for ozone, reducing the reaction efficiency (Juárez et al., 2021; Rizzo et al., 2019). CBZ removal using Fenton processes was reported to achieve a mix of removal efficiencies (Klamerth et al., 2010; Li et al., 2012; Matta et al., 2011).
Adsorption is often used as an application for targeting persistent organic compounds owing to its ease of application and the availability of a range of materials as potential adsorbents. The choice of precursor is an important step in developing an effective adsorbent material. Biochar-based adsorbents can be derived from a range of precursors and are also versatile in their field of application. They are carbon-rich by-products obtained from the pyrolysis of biomass under an inert atmosphere. They are low-cost alternatives to conventional activated carbon materials and are flexible in their field of application (Jin et al., 2022). Biochars can be ideal adsorbent materials due to their stability, high carbon content and aromatic nature and can target a range of contaminants, such as organics, heavy metals, pesticides, dyes and pharmaceuticals. Without physical or chemical activation, biochars typically are ineffective against most contaminants, primarily owing to their low surface area. With recent developments in activation techniques, however, biochar application in water remediation has provided a low-cost, low-carbon footprint alternative (Qiu et al., 2022). There is also considerable influence of the operational parameters of the system as suggested by Jin et al. (2022), who investigated the removal of Pb (II) ions using peanut shell biochars.
Quirantes et al. (2017) investigated 3,4-DCA removal using biomass fly ash and concluded that removal is regulated by boundary layer diffusion, while strong adsorbate–adsorbent interactions have been observed (Bakhaeva et al., 2001; van Oss, 2007). APAP adsorption appears to be controlled by the hydroxyl and amino functional groups acting as electron donors, augmenting the electron density on the aromatic ring (Pauletto et al., 2021), which may be influenced by the surface groups present on the biochar surface (Tran et al., 2020). CBZ, on the other hand, is reported to interact through mechanisms including chemical bonding, π–π interaction, and Lewis acid–base interaction; and despite its weak hydrogen bonding, CBZ has been shown to chemical bond with biochar surfaces (Liang et al., 2020). Enrofloxacin removal was investigated using biochar derived from cow dung, modified under three different phosphoric acid concentrations (10%, 30% and 50%). Largest surface area was reported for 30% phosphoric acid modification, which also coincided with the maximum adsorption capacity of enrofloxacin at 63.61 mg/g (Jiang et al., 2024). This indicates that, despite all being aromatic molecules, there is a range of interactions between these target molecules and a potential adsorbent material.
Despite compelling concepts and innovation for the use of biochar, market demand and large-scale application require ample supply of feedstock and significant capital input for process application. A comprehensive production technique could lead to smaller profit margins providing a challenge in gaining capital investments. Companies and manufacturers often tend to evaluate these risk factors to ensure the accessibility of a final product that is reasonably priced and user-friendly. The aim of this work is to investigate the removal of 3,4-DCA, APAP and CBZ from an aqueous system using biochar derived from native Scottish woods, analysed using thermodynamic and kinetic studies. Locally procured raw material provides a considerable reduction in carbon footprint associated with supply and transport, offering the potential for circularity in the formation of biochar materials for possible applications.
Materials and methods
The precursor used for making the biochar was procured from Sustainable Thinking Scotland C.I.C. (Kinneil Estate, Bo’ness, Scotland). Wood samples are taken from deforested wood in the walled garden of the 200-acre estate. The sample mix was pure softwood including species such as Scots pine, western red cedar, and Sitka spruce.
Pyrolysis
Previous results (Jamal and Fletcher, 2023) indicated that, for the feedstock used, the optimum yield and surface area were obtained for a sample pyrolysed at 725 °C, with a hold time of 60 min and a heating rate of 15 °C/min (known as the optimised biochar). Before pyrolysis, wood samples were divided into small cubes, rinsed with DI water and oven-dried for 24 h at 100 °C. A precursor weight of 30 ± 0.1 g was distributed into three crucibles with a lid placed on top and pyrolysed inside a Thermconcept KLS 10/12/WS muffle furnace. An inert atmosphere was obtained inside the furnace using a CO2 flow rate of 250 mL/min. The furnace was switched on at the designated temperature and dwell time conditions and at the end of each run, the flow of gas was switched off and samples were allowed to cool overnight before analysis. Figure 1 shows a schematic diagram of the muffle furnace used in this study.

Schematic diagram of muffle furnace equipped with a weighing system (Licence number: 5501811254585) (Cao et al., 2021).
Biochar production was repeated twice to ensure the results fell within acceptable error margins.
Material characterisation
Porous structure characterisation
Biochar-specific surface area and pore size distribution were determined using Brunauer–Emmett–Teller (BET) (Brunauer et al., 1938) and Barrett–Joyner–Halenda (BJH) (Barrett et al., 1951) models. Samples were crushed to a powdered form before analysis. A Micromeritics ASAP 2420 system was used to perform nitrogen adsorption at −196 °C. Sample degassing was performed at 200 °C for 4 h at a heating rate of 10 °C/min. The adsorption branch of the isotherm consisted of 49 points, with 30 points on the desorption branch.
Fourier transform infrared spectroscopy (FTIR)
Attenuated Total Reflectance (ATR) was used to study functional groups on the biochar surface. A small amount of powdered biochar was analysed using an ABB IR Instrument MB3000 series. The acquisition mode was set to transmittance, with a detector gain of 80% for increased accuracy. A total of 32 scans were taken between wavenumbers 500 and 4000 cm−1, at 4 cm−1 resolution.
X-ray photoelectron spectroscopy (XPS)
XPS analysis was used to determine the surface chemistry of the biochar sample and was performed by the NEXUS facility. The biochar sample was crushed to a powdered form and analysed using a K-Alpha Photoelectron Spectrometer (Thermo Fisher) and electron detection using a hemispherical analyser. Measurements were taken with the flood gun on to lower charging with the beam energy at 40 eV and a step size of 0.05 eV. The generated results were analysed using the Fityk programme.
Point of zero charge (PZC)
PZC analysis was performed using a salt addition method (Bakatula et al., 2018). A 40 mL portion of a solution containing 0.1 M NaNO3 was modified to reach five different pH levels ranging from 3 to 11. To achieve the desired pH, solutions of 0.1 M NaOH and 0.1 M HCl were utilised. Approximately 0.2 g of powdered biochar was introduced into the beakers and stirred at a speed of 450 rpm for 24 h. The resulting mixture was filtered, and the pH of the filtered liquid was measured. By calculating the difference between the initial and final pH values of the samples and plotting the change in pH against the initial value, the PZC value was determined.
Scanning electron microscopy (SEM)
Scanning electron microscopy (SEM) of biochar samples was performed to examine surface structure and morphology. Surface imaging of biochars produced in this work was performed by clipping a small solid portion from the material and placing it into a Tungsten low-vacuum JEOL JSM-IT100 InTouchScope SEM. Images were captured at 10 µm with 1000× magnification. The beam current was kept constant at 35 with a voltage difference of 20 kV.
Analytical method
The experimental solutions were passed through Fisherbrand Grade 601 general-purpose filter papers (125 mm diameter). Two filter papers were used for each filtration step to ensure the removal of all suspended particles from the permeate. For the target species in this work, calibration curves were plotted prior to kinetic and adsorption experiments. The maximum adsorption wavelengths for 3,4-DCA, APAP and CBZ are 296, 243 and 285 nm, respectively. For calibration, absorbance obtained by passing light of monochromatic radiation was plotted against concentration and a liner fit provided the required parameters of slope and intercept. The generated equation was then used to identify residual concentrations from experimental runs.
Adsorption kinetics
Biochar samples (0.1 g) were added to 100 mL glass bottles and mixed with 50 mL of 100 mg/L solution of either APAP, CBZ or 3,4-DCA. The bottles were subsequently placed on an orbital shaker at 420 rpm for time steps 15, 30, 60, 120, 180, 240, 360 and 1440 min. Once completed, the solutions for each time step were double-filtered using cellulose acetate filter papers to obtain a clear solution free of suspended biochar particles. The supernatants were analysed using UV-Vis spectroscopy. The amount of target species adsorbed was calculated using calibration curves run prior to kinetic and isotherm measurements. It should be noted that there was an exception in the case of 3,4-DCA kinetic analysis. The speed of adsorption was observed to be extremely fast, not allowing sufficient time to measure and analyse multiple samples. Hence, a reduction in temperature was used, using an ice bath (∼2–3 °C) to slow the reaction process to obtain kinetic parameters. A series of kinetic models were applied to determine the most appropriate fit.
The pseudo-first-order (PFO) model was first proposed by Lagergren in 1898 (Svenska Vetenskapsakademiens, n.d.). The differential form of the model is given by Equation 1:
Ho and McKay (1998) proposed the expression for the pseudo-second-order (PSO) model by integrating Equation 4:
Adsorption isotherms
Batch adsorption experiments were carried out using 0.1 g biochar in 50 mL solution of the target species. For APAP and 3,4-DCA, the concentrations used were 25, 50, 75, 100, 150, 200, 250 and 300 mg/L. For CBZ, the maximum solubility is 125 mg/L and, hence, the isotherm points were taken between 10 and 100 mg/L at intervals of 10 mg/L. Based on the kinetic data, 3,4-DCA isotherm solutions were filtered after 10 min. Experiments for APAP were run for 6 h and CBZ experiments were allowed to run for 24 h to allow for full equilibration of the samples. The permeate collection procedure was similar to that used within the kinetic measurements, where the solutions were double-filtered using two cellulose acetate filter papers, and the absorbance was measured using UV-Vis spectroscopy. The collected isotherm data was analysed using three adsorption isotherm models.
The non-linear form of Langmuir model (Langmuir, 1916) is given in Equation 7:
The Freundlich model (Freundlich, 1907) is given in Equation 8:
The Sips model (Sips, 1948) is often referred to as the Langmuir–Freundlich isotherm model and was developed in 1948. The non-linear form is shown in Equation 9:
Results
Material characterisation
Figure 2 shows the adsorption–desorption isotherms obtained for the optimised biochar sample. The adsorption isotherm shows a high initial uptake at low relative pressures, followed by a plateau in the high relative pressure region, which can be attributed to the microporous and mesoporous nature of the material. This resembles a Type IV isotherm behaviour, as per the IUPAC classification of physisorption behaviour (Thommes et al., 2015). The desorption branch between the pressure range 0.85–0.4 shows the hysteresis loop which typically results from the presence of mesoporosity in porous materials. The difference in desorption branch can be linked to differences in evaporation mechanisms, cavitation and pore blocking.

Nitrogen adsorption and desorption isotherms on the optimised biochar sample.
The hysteresis loop resembles Type H4 where the adsorption branch is associated with micropore filling at low relative pressures as expected for microporous carbons (Thommes et al., 2015), this is further supported by the ratio of micropore to total pore volume for the biochar sample, which was 55%. The micropore volume was calculated using the t-plot method developed by Lippens and Boer (Lippens and de Boer, 1965). Total pore volume (TPV) was calculated using Equation 10:

Pore volume distribution of the optimised biochar sample using BJH method (Barrett et al., 1951).
The biochar surface area was calculated, using the BET method, to be 620 m2/g. BET analysis, however, is sensitive to the selected relative pressure region (Gómez-Gualdrón et al., 2016); specifically for microporous materials, the optimal range of relative pressure can be determined by applying the four consistency criteria proposed by Sing (2014). These criteria are as follows: (1) only the range where the product of the adsorbate loading rate and the difference between 1 and the relative pressure exhibits a monotonic increase with the relative pressure should be selected; (2) the value of the BET ‘C constant’ should be positive. The C constant represents the interactions between the adsorbent and adsorbate and is linked to the energetic aspects of the first adsorbed layer (Gómez-Gualdrón et al., 2016); (3) the linear region chosen should include the loading corresponding to the monolayer at the given relative pressure; (4) the relative pressure obtained from Criterion 3 should be within a 20% tolerance of the relative pressure calculated from BET theory that aligns with monolayer loading. Upon applying the correction to the data obtained for this biochar sample, the ‘corrected’ surface area was calculated to be 588 m2/g, which is larger than other wood-based biochars reported in previous studies (Idowu and Fletcher, 2020; Jindo et al., 2014; Kloss et al., 2012).
The FTIR spectra obtained for the wood feedstock and the optimised biochar are shown in Figure 4. The fingerprint region, observed between 600 and 1500 cm−1 shows the presence of vinyl terminals (Nandiyanto et al., 2019). At 1000 cm−1, there is evidence of a loss of the peak observed in the feedstock, due to heat treatment. The peak loss can be attributed to the removal of C-OH vibrations from dehydration during pyrolysis (Idowu and Fletcher, 2020). The process of heat treatment is essential in facilitating condensation of the carbonaceous skeleton and eliminating the hydroxyl groups found in the cellulosic compounds within the initial materials (Lee et al., 2010). Overall, the feedstock and biochar spectra look comparable with the presence of C = C bonds with symmetric and asymmetric vibrations between 1600 and 1800 cm−1.

FTIR spectra of wood feedstock and biochar.
The XPS spectra of the optimised biochar sample used are shown in Figure 5. The spectra show the presence of oxygen and nitrogen bonds, in addition to carbon with peaks at 532.5, 400 and 284.5 eV, respectively (Reddygunta et al., 2022). Peak deconvolution was achieved using the Fityk programme, and the Voigt function assisted in the identification of the heteroatoms present in the biochar. The corresponding parameters are provided in Table S1 (Supporting Information). C1s peaks were deconvoluted into three peaks at 284.5 eV, suggesting a graphene-like arrangement (C = C), at 285.6 eV indicating the presence of carbonyl bonds (C-O), and a third peak at 289.6 eV, which is ascribed to either carboxylic or pyridinic N bond functionalities (C = O/C-N) (Beamson and Briggs, 1992; Lhoest et al., 1995; Smith and Black, 1984; Wagener et al., 1989). The area under the C = C peak is the largest indicating the preservation of the graphene-like arrangement in the wood samples post-pyrolysis. Peak convolution of O1s showed the presence of both carbonyl, as well as carboxyl groups, with peaks at 531.1 and 532.9 eV (Beamson and Briggs, 1992; López et al., 1991). There were also trace amounts of nitrogen functionalities present in the sample and convolution of the N1s spectra suggested the presence of pyridinic (398.6 eV) as well as graphene N (400.2 eV) bonds in the sample (NIST X-ray Photoelectron Spectroscopy (XPS) Database, Version 3.5, n.d.). The incorporation of O and N-based functional groups into the carbon framework enhances its wettability (Reddygunta et al., 2022), which is further correlated with contact angle measurement presented in previous work (Jamal and Fletcher, 2023), where biochar samples proved to be extremely hydrophilic. The calculation of the elemental compositions of C, N and O were performed as suggested by Alexander G. Shard (Shard, 2020). Equation 11 works under the assumption that the sample is homogenous and a single phase within the penetration depth; where X represents the atomic fraction (in %), and IP/SP is the intensity divided by the sensitivity factor (SP = 1, 1.8 and 2.93 for C, N and O, respectively).
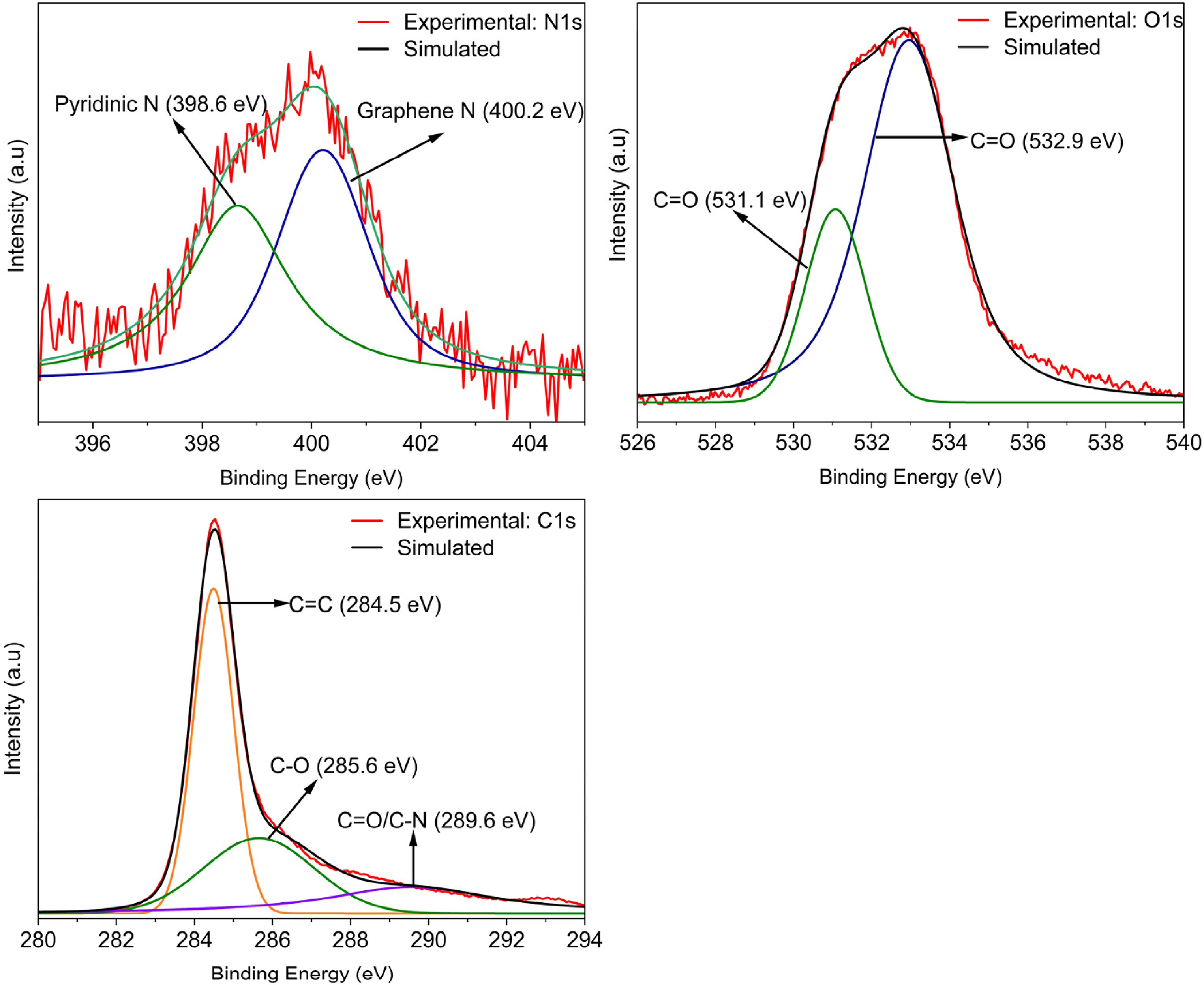
XPS spectra and Fityk simulated models showing N1s, O1s and C1s scans. Fityk models suggest possibilities of existing functional groups at specific binding energies that can be compared with the NIST database.
The biochar sample used consisted of 80% C, 13% O and 7% N fractions. The findings again supplement the results obtained in the previously reported study, where thermogravimetric analysis of the samples showed 80% fixed C (Jamal and Fletcher, 2023).
The PZC of the biochar was observed to be 7.44 ± 0.2. The results are comparable to the previous study on parameter optimisation, where the DOE biochars had similar average PZC values (Jamal and Fletcher, 2023). PZC values are temperature dependent, and high temperatures result in a loss of volatile matter including acidic functional groups such as phenols and carboxyl, causing the resulting biochar to have a more alkaline nature (Shaheen et al., 2019). PZC of wood pellet biochars was also reported to increase with increasing temperatures (Zhang et al., 2015); such alkaline/neutral PZC values make these biochars suitable for potential application in drinking water systems that operate naturally under slightly acidic conditions.
Figure 6(a)–(d) shows the SEM images recorded for lower ramp rate biochars and Figure 6(e)–(h) displays the observations for higher ramp rate biochars. There is evidence of a well-developed pore network in biochars produced at both ramp rates. The images at 10 µm and 1000× magnification suggest that the high pyrolysis temperatures used to create the biochars exposed the carbonaceous skeleton of the parent material encompassing an intricate network of pores (Angın, 2013). A pyrolysis temperature that is sufficiently high is necessary for the removal of the outer biochar layer. The open structure of pores could be attributed to a lower ash content, which reduces the potential for clogging. There is no apparent evidence of influence from different ramp rates on the pore networks developed in the biochars.

SEM images biochar material (T = 725 °C). (a, b) 60 min at 15 °C/min; (c, d) 20 min at 15 °C/min; (e, f) 60 min at 30 °C/min; (g, h) 20 min at 30 °C/min.
Adsorption kinetics
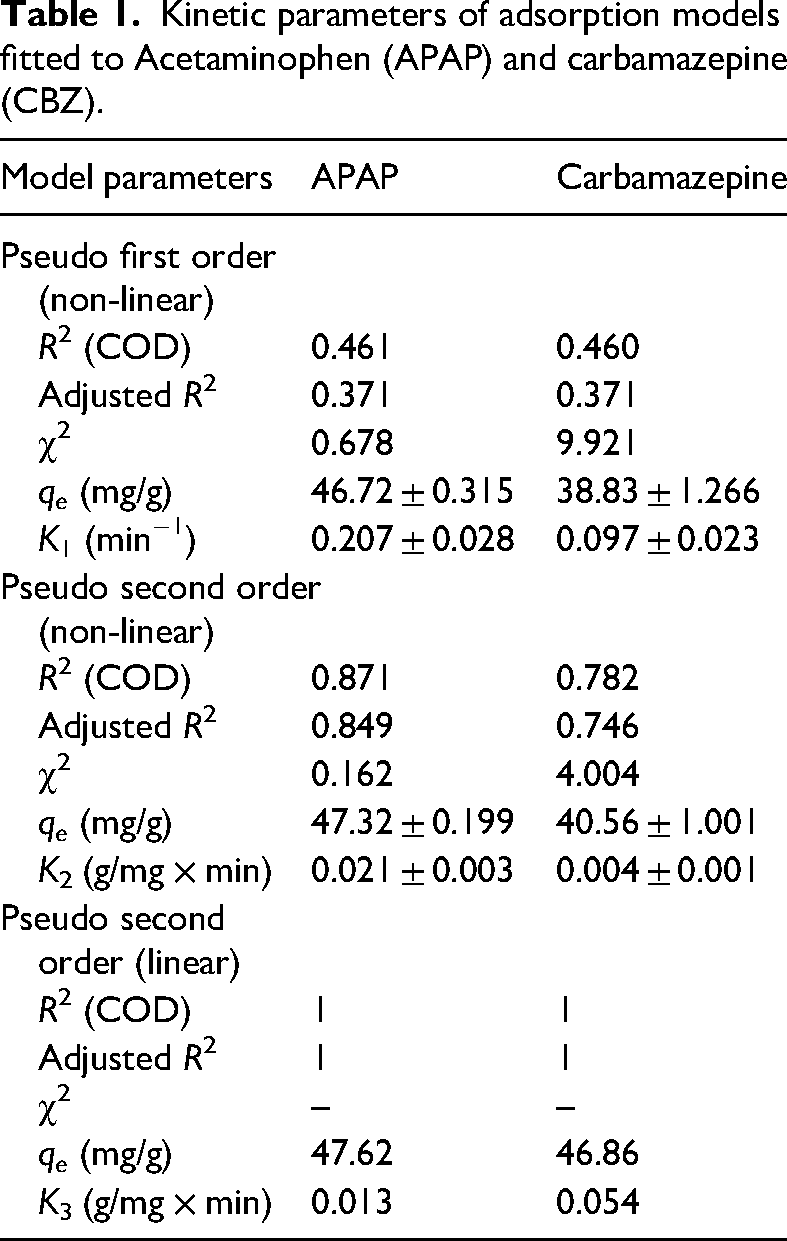
Table 1 shows the parameters of kinetic models fitted to the data obtained for adsorption of APAP and CBZ onto the optimised biochar. Kinetic analysis of 3,4-DCA revealed fast adsorption rates, which could not be fitted to any kinetic models.
Kinetic parameters of adsorption models fitted to Acetaminophen (APAP) and carbamazepine (CBZ).
Data was obtained for 3,4-DCA adsorption on the optimised biochars at room temperature, for an initial concentration of 100 mg/L. Removal was observed to be 90% after 15 min, with a plateau thereafter. Readings were, therefore, taken at shorter time intervals, and also at lower temperature in an attempt to slow the kinetic process. Table 2 shows the removal percentages of 3,4-DCA at room temperature and in the ice bath. The data obtained confirms the rapid adsorption of 3,4-DCA onto the optimised biochar, even at the lower temperature, hence, it was not possible to monitor the adsorption in order to determine the kinetic parameters for 3,4-DCA.
Biochar performance against 3,4-DCA at room temperature and in an ice bath.

The results indicate that the interaction between the biochar surface and 3,4-DCA molecule is almost instant. An investigation into solution pH also yielded similar results with overnight runs at pH 6 and 9 resulting in 93% and 91% removal, respectively. Previous research into 3,4-DCA removal using adsorption suggested that the data followed a pseudo-second-order kinetic model (Angioi et al., 2005). The observed reaction rates in this study are quicker than those previously reported. Experiments using kaolinite and montmorillonite to test the removal of chloroanilines, including 3,4-DCA, were reported to achieve equilibrium in under 4 days (Polati et al., 2006). Another study into 3,4-DCA removal from water using biomass fly ashes reported kinetic equilibrium at approximately 10 h (Quirantes et al., 2017). A selection of low-cost materials including corncob char, sugar beet pulp, perlite, and vermiculite were also tested against 3,4-DCA. The quickest reaction time to achieve maximum sorption percentage was 60 min using vermiculite (Huguenot et al., 2010). The rates observed here indicate that there is significantly quicker adsorption for the biochars created in this work.
Figure 7 shows the kinetic data obtained for the sorption of CBZ and APAP on the biochar sample. Sorption kinetics for APAP showed rapid uptake with 90% removal achieved in 15 min followed by a gradual increase in the uptake, and equilibrium achieved after approximately 2 h. For CBZ, the uptake was slower with just under 75% removal at 15 min and adsorption slowed considerably after 2 h, with equilibrium achieved after 24 h.
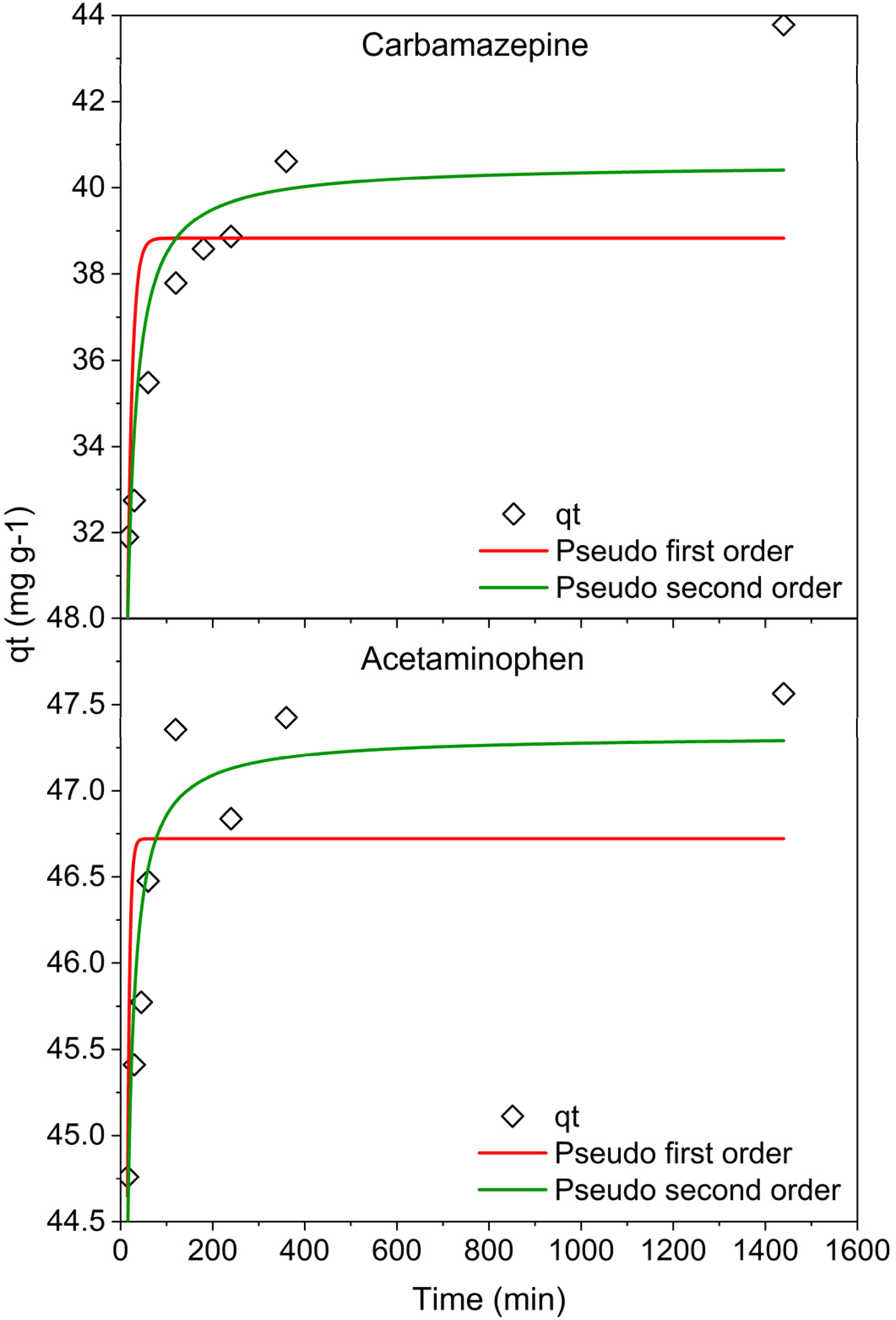
Non-linear kinetic model fittings for (top) carbamazepine and (bottom) acetaminophen on the optimised biochar showing fits for pseudo-first-order and pseudo-second-order models.
The linearised pseudo-second-order rate equation showed a better fit for the data for both species compared to pseudo-first-order and non-linear second-order kinetic models (Table 2). The adjusted R2 values for both species were≥0.99. The maximum adsorption capacity for APAP was 47.6 mg/g and 46.7 mg/g for CBZ. The second-order rate constants show a quicker uptake rate for APAP than CBZ with the adsorption rate being 0.013 g/mg × min. The high R2 values obtained for both species, with linear PSO model fitting, can be attributed to the availability of abundant vacant active sites in the physically activated biochar and the adsorption process being ruled by chemisorption (Abd et al., 2020; El Saied et al., n.d.; Wang and Guo, 2020b). The mechanism could likely be attributed to hydrogen bonding between the species. Additionally, π–π interactions between the benzene ring in APAP and CBZ and the aromatics in biochar can influence the adsorption process (Abd et al., 2020).
Further analysis of the kinetic data was performed using the intraparticle diffusion model proposed by Weber and Morris (Weber and Morris, 1963), to understand the rate-controlling step in the adsorption process. The model equation is given below:

Intraparticle diffusion model fittings for acetaminophen and carbamazepine adsorption show three steps of the diffusion mechanism.
The plots suggest that the adsorption process for both pharmaceutical species involved three diffusion steps. In both cases, liquid film diffusion was the dominating phase signifying rapid diffusion of molecules onto the active sites in the pores and voids of the biochar (Wang et al., 2021; Zhao et al., 2022). There is a hint of intraparticle diffusion in CBZ adsorption. This step involves the gradual diffusion of molecules into the micropores before the reaction proceeds to the equilibrium stage. For APAP however, the intraparticle diffusion phase is not as prevalent and the reaction appears to proceed rapidly to equilibrium after liquid phase diffusion. This also correlates with the longer time required by CBZ to reach equilibrium compared to APAP. The three target molecules have some similarities, allowing a comparison of their adsorption behaviour relative to their chemistry. All contain amine functionalities and benzene rings, with APAP/BBZ sharing carbonyl moieties and only 3,4-DCA containing halogenated species. The slower kinetics for CBZ could realistically be a consequence of the significantly larger relative size of the molecule (see Supporting Information), while the rapid adsorption of 3,4-DCA is likely a result of the comparatively increased electrostatic interactions, where the chlorine groups will be electron withdrawing from the benzene ring.
Adsorption isotherms
Adsorption isotherms are of crucial importance in determining the maximum adsorption capacity of the adsorbent and understanding the adsorption equilibrium. The results of isotherm fittings for the Langmuir, Freundlich and Sips models are given in Table 3, and isotherm plots for the target species are shown in Figure 9.

Adsorption isotherm models showing non-linear fits for (a) 3,4-DCA, (b) acetaminophen and (c) carbamazepine.
Adsorption isotherm parameters obtained for Acetaminophen (APAP), carbamazepine (CBZ) and 3,4-dicholoroaniline (3,4-DCA) on the optimised biochar sample.
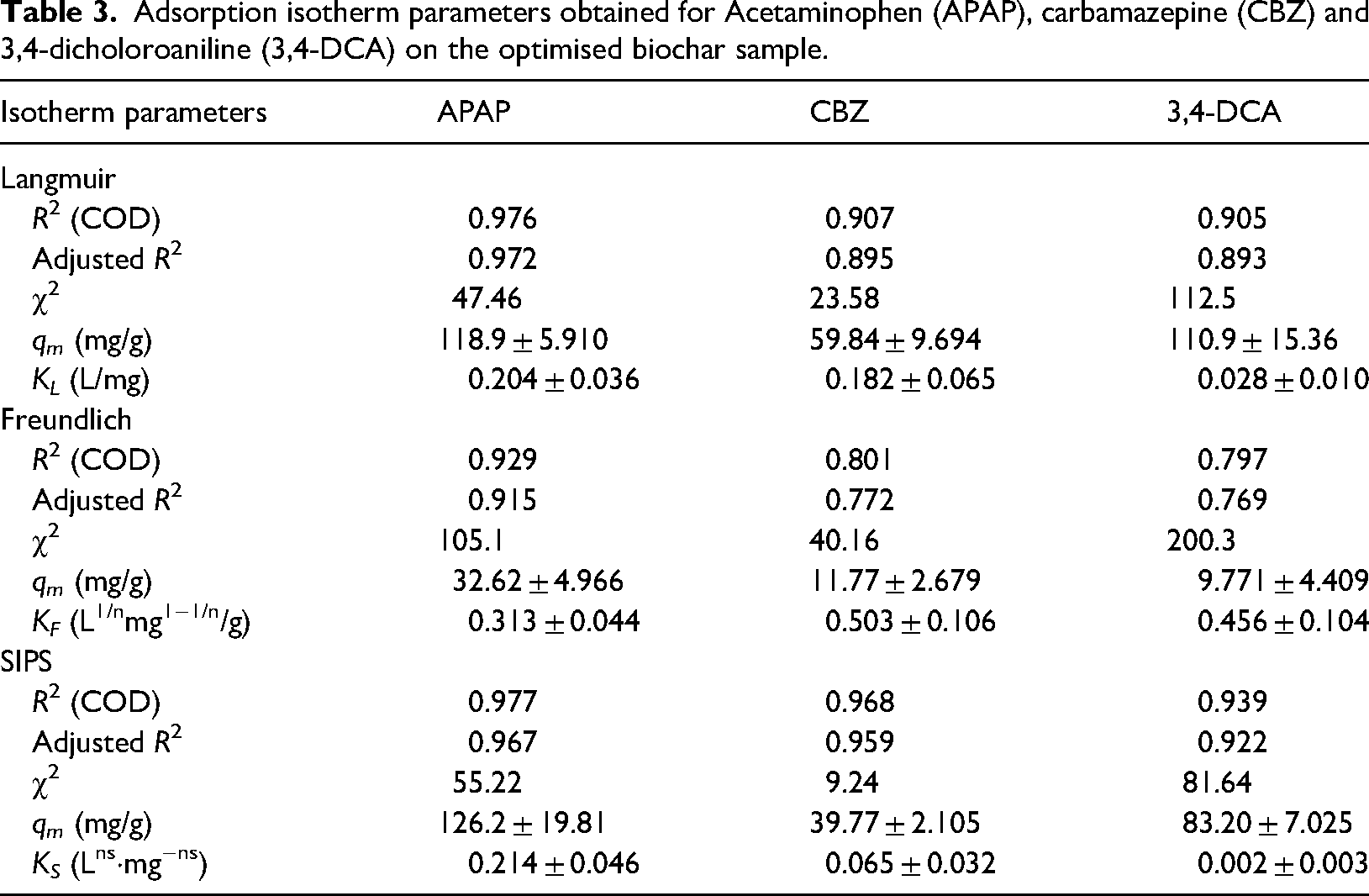
The results indicate that the adsorption uptake was significant at low pollutant concentrations. The data presented in Table 3 suggests that for CBZ and 3,4-DCA, the Sips model can be best used to describe the adsorption behaviour. This suggests that the adsorption process is a combination of physisorption at low concentrations and chemisorption at high initial Co values with the formation of a monolayer on the adsorbent material. The maximum adsorption capacities for CBZ and 3,4-DCA, suggested by the Sips model, were 39.8 mg/g and 83.2 mg/g, respectively. For APAP, both Langmuir and SIPS isotherm models demonstrated high R2 values with the chi-square value lower in the case of Langmuir isotherm. A good fit to the Langmuir model assumes monolayer adsorption of the molecules on the biochar surface, with almost identical activation energies, albeit with the possibility of multilayer formation (El Saied et al., n.d.). The maximum adsorption capacity for APAP is determined as 126 mg/g, much higher than the other two species, which may be reduced as a consequence of molecular size affecting packing in the case of CBZ and electrostatic repulsion in the case of 3,4-DCA due to overlapping of the electron clouds of the molecule within the pore structure (Weber, 1974).
Since the two models exhibited a good fit to the experimental data for APAP, the value of the separation factor (RL) suggested by Webber and Chakkravorti (Weber and Chakravorti, 1974) was calculated to further verify the favourability of the Langmuir adsorption isotherm (see Supporting Information). The separation factor is determined from Equation 13:
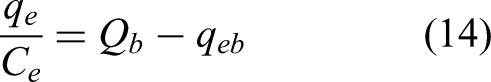
Figure 10 shows the Scatchard plots obtained for the adsorption of target species. It is evident from the plots that the adsorption behaviour deviates from linearity. The high and low binding affinities can be attributed to the presence of more than one type of binding site, offering strong and weak interactions, respectively. The high affinity binding sites can be attributed to chemical interactions between the biochar surface and the target compounds whereas the low affinity sites indicate weak physical bonds. The plots further supplement the observations from Sips isotherm indicating a heterogeneous material with multiple binding sites.

Scatchard plots for (a) APAP, (b) CBZ and (c) 3,4-DCA to determine the nature of binding sites on the biochar surface. Graph shows clear distinction between affinity zones suggesting heterogeneous surface.
Discussion
The results obtained suggest potential for application of the optimised biochar in water remediation, targeting persistent organic pollutants. The biochar surface area was higher than those reported for many wood-based biochars reported in the literature (Bataillou et al., 2022; Idowu and Fletcher, 2020). The sample presents a mixed microporous–mesoporous structure, as observed from the nitrogen isotherms with an average pore width of 4 nm, indicating that the mesopores present in the sample are narrow. Biochars pyrolysed at higher temperatures are linked with alkaline surface character (Shaheen et al., 2019), which fits with the PZC value obtained for the biochar produced at 725 °C (7.44 ± 0.2). FTIR analysis suggested a layered carbon structure with the presence of oxygen and nitrogen chemical moieties. These findings were further assisted by XPS analysis presenting a graphene-like arrangement, and 80% atomic carbon fraction. Additionally, the presence of oxygen and nitrogen-based functionalities was confirmed from XPS data. These chemical moieties assist the hydrophilic nature of the biochar and provide a potential avenue for application in water remediation.
The application of biochar against 3,4-DCA, APAP and carbamazepine resulted in efficient removal of the species from an aqueous system. 3,4-DCA removal was characterised with fast removal rates and a very short equilibrium time of around 6 min and a maximum adsorption capacity of 83 mg/g. Although the maximum removal capacity was lower than some activated carbons reported in the literature, adsorption rates were far superior in achieving acceptable removal overall (Bakhaeva et al., 2001). Adsorption of APAP and CBZ was best explained by the linearised pseudo-second-order model indicating the formation of chemical bonds as the rate-limiting step. The maximum adsorption capacities for APAP and CBZ were 126 and 40 mg/g, respectively, and these were superior or on par with those reported in Table 4. The presence of nitrogen and oxygen functionalities on the surface of the biochars presents possible hydrogen bonding or π–π interactions between the benzene rings in the target elements and the biochar surface, as a possible removal mechanism. Overall, the optimised biochar sample selected for application from the design of experiments optimisation study conducted previously (Jamal and Fletcher, 2023), provided fast adsorption kinetics and high adsorption capacities against the identified target molecules. The results provide an attractive avenue for biochar application for water remediation targeting a range of pollutants in aqueous media.
Maximum uptake capacities of different adsorbents for Acetaminophen (APAP), carbamazepine (CBZ) and 3,4-DCA.

Conclusions
The optimised biochar produced from Scottish softwood showed good performance against selected pollutant species. The material was characterised with a high surface area and mixed microporous/mesoporous nature. Point of zero charge analysis indicated a neutral surface charge and X-ray photoelectron spectroscopy data suggested a hydrophilic nature and potential for application in water remediation. The material showed great performance against 3,4-dichloroaniline with rapid equilibrium proving difficult to perform a kinetic analysis. For acetaminophen and carbamazepine, adsorption followed a pseudo-second-order kinetic model. Adsorption isotherms were best explained by the Sips model for all three species suggesting the formation of an initial monolayer with chemical bonding as the rate-limiting step. The highest adsorption capacity was noted in the case of acetaminophen. Scatchard plots suggested a heterogeneous surface with multiple binding sites. Ultimately, the results indicate a potential application of softwood biochar as a renewable adsorbent in water remediation.
Supplemental Material
sj-docx-1-adt-10.1177_02636174241256854 - Supplemental material for Scottish softwood biochar for water remediation targeting selected persistent organic pollutants
Supplemental material, sj-docx-1-adt-10.1177_02636174241256854 for Scottish softwood biochar for water remediation targeting selected persistent organic pollutants by Mohammad Umair Jamal, Ashleigh Fletcher, Alan Baby, Isaac Maso and Lidija Šiller in Adsorption Science & Technology
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
All data generated in this study are included in this article. No AIF-related data were generated in this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.