Abstract
Magnetic nanoparticles are amongst the most promising adsorption materials for oil spill clean-up due to their high surface area, ease of functionalization with high oil affinity and facile separation after the cleaning process with an external magnetic field. In this work, we successfully synthesized magnetic cobalt ferrite nanoparticles (CoFe2O4 NPs) that were electrostatically stabilized and functionalized with various alkoxysilanes for effective oil adsorption and oil spill removal. Additionally, the adsorption capacity of CoFe2O4 NPs was determined, and the possibility of their reuse assessed. Prepared samples showed high oil adsorption capacities between 2.6 and 3.5 g of oil per g of nanoparticles and were successfully collected with an external magnet. Furthermore, the samples showed excellent properties after regeneration, as their adsorption capacity decreased by less than 3% after reuse. All the prepared samples were thoroughly characterized to better understand their behaviour and the differences in the use of various silanes were highlighted.
Introduction
Crude oil is an indispensable raw material for humanity and remains our crucial source of energy. Despite the preventative measures in the industry of oil drilling and transportation, quite some quantities are discharged in the ecosystem and approximately 0.1% of all oil is accidentally spilled into the oceans or the seas each year (Al-Majed et al., 2012, 2014; Bayat et al., 2005; Farias et al., 2015; Lin et al., 2012). In other words, out of 5 million tons of oil transported by sea, 500 tons end up getting spilled, which causes grave and irreparable damage to the marine and coastal environment (Annunciado et al., 2005; Liu et al., 2019). It is thus important to develop quick and efficient oil removal methods.
The ideal oil spill cleaning material must be hydrophobic, lipophilic (oleophilic), selective, stable, with high adsorption capacity and the ability to adsorb thick and thin layers of oil quickly and efficiently. Moreover, the material must be sustainable, environmentally friendly, and enabling immediate usage, whilst procedures for the synthesis of such material must be simple and economical (Chen et al., 2013; Gu et al., 2014). Since the material must be re-used, it needs to allow quick, simple and inexpensive separation from the contaminated water and subsequent regeneration (Tang and Lo, 2013; Zhang and Lei, 2012). Therefore, the development of an ideal oil cleaning agent remains a challenge and each oil spill removal requires an individual approach for which adsorption agents are considered to be the most economical, sustainable and efficient solution (Singh et al., 2014).
Researchers have developed various nanomaterials to clean oil spills such as aerogels, nanodispersants, nanocomposites, membranes, foams and nets, filters, carbon nanostructures, and micro- and nano-titanium oxide for photocatalytic degradation of oil (Kharisov et al., 2014). These materials have excellent adsorption capacity, but show poor regeneration yield. Also, they are not biodegradable and can, therefore, cause secondary pollution (Xu et al., 2012). Nanoparticles possess high surface area, are affordable, efficient and present an environmentally friendly solution for the removal of pollutants from water (Kharisov et al., 2014). The attempt to produce hydrophobic surfaces for oil spill removal led to studying of nanoparticles based on silicon dioxide, titanium dioxide, zinc oxide, alumina and magnetic nanoparticles based on iron oxide (magnetite and maghemite). The latter is of great interest in different fields of environmental protection, especially in adsorption of pollutants such as heavy metal ions and oil due to their superparamagnetic properties (Tang and Lo, 2013; Zhang and Lei, 2012), and ease of removal from the treated water by using the external magnetic field (Kharissova et al., 2015; Thinh et al., 2016).
Superparamagnetic iron oxide nanoparticles (SPIONs) can be synthesized by co-precipitation, microemulsion, thermal decomposition, hydrothermal or solvothermal synthesis, sonochemical synthesis, microwave-assisted synthesis, chemical evaporation method, incineration method, chemical reduction, laser pyrolysis and other methods (Faraji et al., 2010; Lu et al., 2007). Coprecipitation is simple and highly effective for the synthesis of SPIONs of controlled size, shape and chemical composition. Various iron oxides can be prepared using this method, such as magnetite (Fe3O4), maghemite (γ-Fe2O4), or cobalt ferrite (CoFe2O4).
Herein, the preparation of electrostatically stabilized and surface functionalized CoFe2O4 NPs and their successful use for adsorption of mineral oil from water is reported. Particles were functionalized using different alkoxysilanes – tetraethoxysilane (TEOS), TEOS in combination with methyltrimethoxysilane (MTMS), and TEOS with
Materials and methods
Materials
Cobalt (II) chloride hexahydrate (CoCl2·6H2O), iron (III) chloride hexahydrate (FeCl3·6H2O), sodium hydroxide (NaOH, 99%), tetraethylorthosilicate (Si(OC2H5)4 or TEOS, 99%), nitric acid (HNO3, 65%), ammonium hydroxide (NH4OH, 25%),
Synthesis of CoFe2O4 NPs
CoFe2O4 NPs were prepared by co-precipitation of an aqueous solution of CoCl2·6H2O and FeCl3·6H2O salts with molar ratio of Co2+/Fe3+ being 2:1 in an alkaline medium (NaOH) at pH value of 10.5. Typically, 75 mL of 1 M salts solution was added to 400 mL of 1 M NaOH. The reaction described below as equation (1) was carried out for 1 h at a temperature between 85°C and 90°C

Stabilization of CoFe2O4 NPs
The prepared CoFe2O4 NPs were saturated on a magnet. The residual liquid was flushed, and the nanoparticles were electrostatically stabilized with 50 mL of 2 M HNO3 solution at room temperature with moderate agitation for 1 h. The particles were separated with the external magnet, the excess liquid was removed, and the sediment was treated with a fresh aqueous solution of Co2+/Fe3+ (1:2) described in previous paragraph for 1 h at 85°C. After saturating the particles on the magnet, the upper super-fluidic liquid phase was removed, and the particles were washed with 50 mL of 1 M HNO3. In the last phase, the particles were saturated on a magnet, washed with acetone, centrifuged at 3500 r/min for 10 min and dispersed in 50 mL of distilled water.
Functionalization of stabilized CoFe2O4 NPs – Formation of core-shell structure
The dispersed CoFe2O4 NPs were surface functionalized with the selected methoxy silanes (MTMS and PTMS) in combination with TEOS or with TEOS alone according to the modified Stöber method. Table 1 shows the chemical and structural formula of the selected silanes.
Chemical and structural formula of the selected silanes.

MTMS: methyltrimethoxysilane; PTMS:
Functionalization was carried out in a closed reaction vessel using 2-propanol, 25% NH4OH as catalyst and water. The pH of the medium was adjusted to 10. The alcoholic mixture was stirred with 6 mL of an aqueous dispersion of magnetic nanoparticles with an average mass concentration of magnetic phase being 0.0914 g/mL and with a corresponding amount of TEOS precursors and selected alkoxysilane under the conditions shown in Table 2. The molar ratio between water and TEOS (
Composition of reaction mixture where molar ratio

MTMS: methyltrimethoxysilane; PTMS:
Characterization
The phase composition of prepared samples of CoFe2O4 NPs was determined by X-ray powder diffraction (XRD) on Bruker D4 Endeavor apparatus with Topas software in the range of 20°–80° with 10° step. Observed diffraction peaks were also compared with standard JCPDS database (JCPDS card No. 22-1086). Particle size (
Particle size, particle size distribution, morphology and core-shell structure of prepared NPs was observed by transmission electron microscopy (TEM) on Jeol TEM 2100 apparatus. The NP samples were placed on copper mesh and observed at 200 keV under different magnifications. TEM micrographs of nonfunctionalized CoFe2O4 NPs, as well as PTMS and MTMS functionalized CoFe2O4 NPs were obtained and compared.
The infrared spectra of the samples for determination of successful formation of functionalized silica coatings were measured between wavenumbers 4000 and 600 cm−1 using an attenuated total reflection Fourier transformed infra-red (FT-IR) spectrophotometer model GX FT-IR. The spectra of functionalized samples, prepared with TEOS, a combination of TEOS with MTMS or PTMS at different molar ratios were compared with pure precursor spectra.
The specific surface area was determined by Brunauerr–Emmet–Teller (BET) method that predicts multi-layered or infinite adsorption of gases on the surface of the particles and no interactions between the layers of the adsorbent. Obtained BET isotherms provided information of adsorbed gas on dried NPs surface of a given mass. From the results of the specific surface area (
Magnetic nature of adsorbents used for oil spills removal facilitates the process of their removal from water media after the clean-up via the use of the external magnetic field. These particles are afterwards regenerated and can be re-used in the adsorption process. Achieving the sufficient magnetic response of adsorption materials is therefore crucial in ensuring the quality removal of oils from the water media. Magnetic properties of nonfunctionalized and functionalized CoFe2O4 NPs, such as saturation magnetization (
To determine the hydrophilicity or hydrophobicity of the prepared NPs surfaces, the water contact angle measurements were conducted on goniometer Dataphysics OCA35. Measurements of the static contact angle were carried out by Zisman model by spreading a uniform amount of the NPs sample onto the carrier and applying a drop of water of a certain volume to the surface of the samples using a metal tube on the gauge. By image analysis, the changes in height, width, volume and diameter of the drop in a certain time state were monitored (Černe et al., 2008).
Oil adsorption capacity and evaluation of oil removal efficiency of prepared NPs
The oil adsorption capacity of functionalized magnetic CoFe2O4 NPs was evaluated by used motor oil as testing material. Used motor oil was chosen because it is already intensely coloured to ease the visual observation without the need to add colourings that could negatively affect the adsorption. Due to its less volatile nature than crude oil, petrol or diesel fuel, it is also safer to handle and finally, it represents an actual environmental pollutant.
The oil adsorption capacity was determined gravimetrically in two parallel tests. Oil removal testing is described below and depicted in Figure 1. The functionalized magnetic NPs (

Procedure of oil removal from water using functionalized magnetic NPs.
The adsorption capacity of each prepared material was calculated according to equation (2) (Chen et al., 2013; de Souza et al., 2010; Ge et al., 2014; Liu et al., 2014; Zhu et al., 2010)

Two samples with the highest oil adsorption capacity results were also evaluated for regeneration, reuse and its effectiveness.
Results and discussion
X-ray powder diffraction of CoFe2O4 NPs
During the co-precipitation reaction in alkali media the Co2+ and Fe3+ ions firstly precipitate in the form of hydroxides (Cornell and Schwertmann, 2003) and secondly, oxidize to CoFe2O4 NPs. XRD pattern of prepared CoFe2O4 NPs is shown in Figure 2. Diffraction peaks at (220), (311), (222), (400), (422), (511), and (440) were compared with the standard data for CoFe2O4 (JCPDS card No. 22-1086). The observed peaks are characteristic for cobalt ferrite and belong to cubic spinel crystal structure.

X-ray powder diffraction pattern of CoFe2O4 NPs.
The average particle size (
Using the Bragg equation (equation (4)) and the position of the individual diffraction peaks and Miller indices (

Obtained
Transmission electron microscopy of nonfunctionalized and functionalized CoFe2O4 NPs
A TEM micrograph of nonfunctionalized CoFe2O4 NPs (Figure 3(a)) shows their spherical morphology and narrow size distribution of 11.5 ± 1.9 nm. The estimated particle size coincides with the calculated

(a) TEM image of CoFe2O4 NPs, (b) diffraction image of CoFe2O4 NPs, (c) HRTEM image of CoFe2O4 NPs, (d) TEM image of SiO2@CoFe2O4 NPs and CoFe2O4 NPs functionalized with TEOS in combination with silane PTMS at molar ratios
Figure 3(d) shows functionalized CoFe2O4 NPs coated with a layer of silicon dioxide (SiO2@CoFe2O4), which contributed to an increase in the particle size to the average value of 13.1 nm. Since organic coatings can be sensitive to ionizing radiation present in conventional TEM microscopes and are known to have poor electronegative contrast of the image, SiO2 coatings around NPs are poorly seen (Egerton et al., 2004; Gontard et al., 2014). The average thickness of the CoFe2O4 surface coating of the NPs prepared in combination with the selected alkoxysilanes, estimated based on TEM images, occupies values between 1.5 and 6 nm. Figure 3(e) to (h) shows TEM images of SiO2@CoFe2O4 in combination with silanes (
FT-IR spectroscopy
FT-IR spectra of pure TEOS and TEOS functionalized magnetic NPs are shown in Figure 4. Typical precursor peaks are between 2975 and 2880 cm−1 and represent C–H stretching in the ester groups (Yong et al., 2014). The peaks between 1443 and 1296 cm−1 can be attributed to the asymmetric bending or wagging of C–H bonds. Peaks at 1168 and 968 cm−1 belong to C–H rocking. The ethoxy group bound to the silicon atom (Si–OCH2CH3) is characterized by peaks around 1100 cm−1 (Viart and Rehspringer, 1996;Ypenburg and Gerding, 1972). SiO2@CoFe2O4 NPs show absorption peaks at 3388 cm−1, which is characteristic for the O–H bond, whilst the peaks at 1100 and 1050 cm−1 correspond to Si–O–Si bonds (Heidari et al., 2009). The absence of a peak at 2975 cm−1, reduction of the 968 cm−1 peak and its shift to 962 cm−1 confirm the complete conversion of TEOS ester groups to silanols, the complete hydrolysis (Bauman, 2010) and successful functionalization of CoFe2O4 NPs.
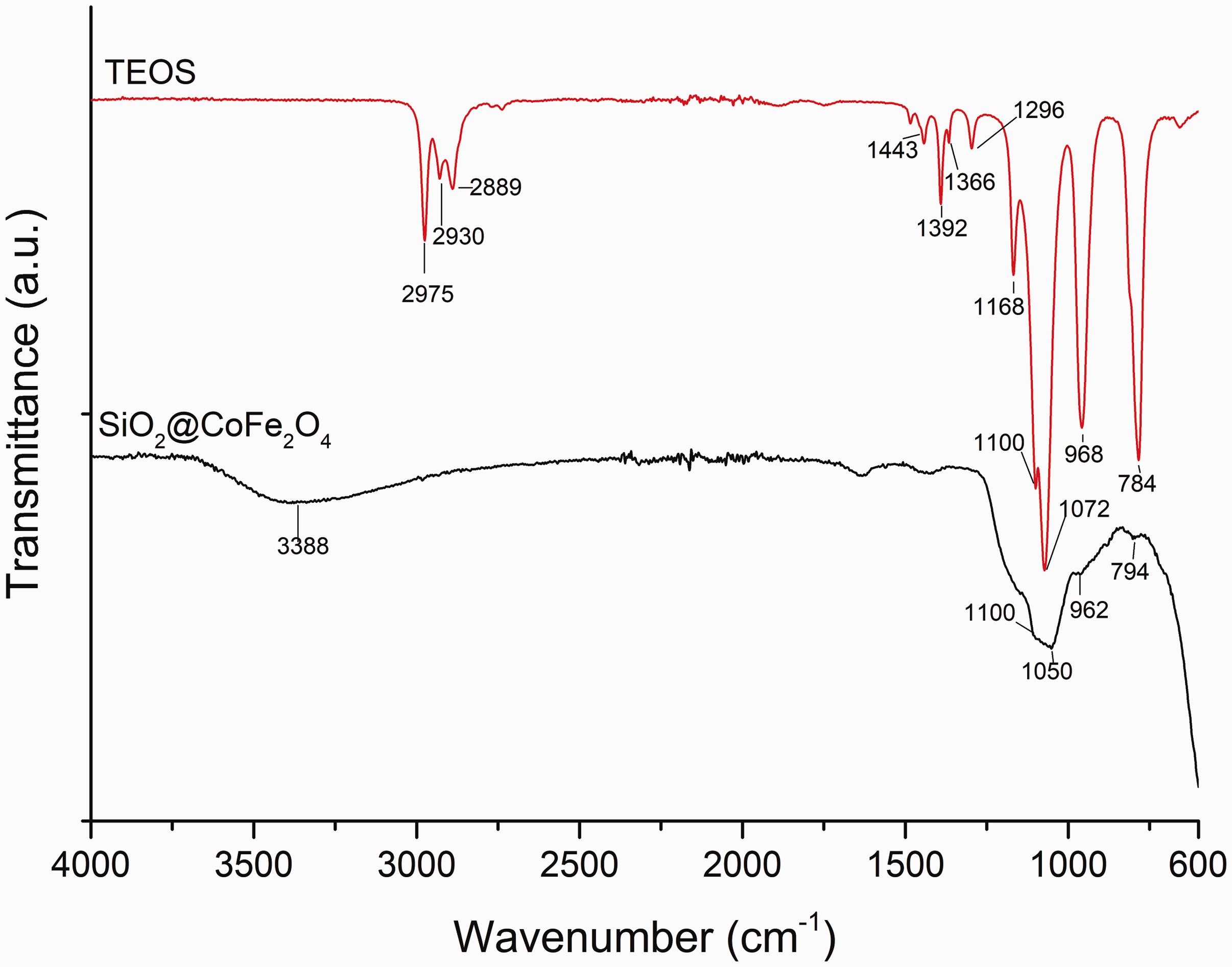
FT-IR spectra of pure precursor TEOS and SiO2@CoFe2O4 NPs.
Figure 5 shows the FT-IR spectra of functionalized samples, prepared with precursor TEOS with MTMS (Figure 5(a)), and TEOS with PTMS (Figure 5(b)) at molar ratios

FT-IR spectra of (a) MTMS@SiO2@CoFe2O4 and (b) PTMS@SiO2@CoFe2O4 NPs.
In Figure 5(a), FT-IR spectra of precursor MTMS, CoFe2O4 NPs functionalized with TEOS and MTMS in different molar ratios (
Figure 5(b) shows that PTMS absorbs infrared waves between 2957 and 2872 cm−1, which was ascribed to the C–H stretching (Mansur et al., 2002). A pronounced narrow peak at 2840 cm−1 is characteristic of the Si–O–CH3 group (Bellamy, 1975; Montgomery, 1997; Sibeaud et al., 2016). The peak at 1215 cm−1 was attributed to the Si–CH2CH2CH3 group, whilst the peaks at 1190 and 1080 cm−1 can be attributed to the Si–O–CH3 group. The Si–O bond was characterized by the peak at 1003 cm−1, Si–C bond by the peaks between 896 and 774 cm−1, and –CH2– bond by the peak at 752 cm−1 (Coates, 2000; Lee Smith, 1960; Montgomery, 1997). The O–H bond from the water adsorbed onto the surface of the material can be seen in the FT-IR spectra of NPs functionalized with TEOS and PTMS (peaks between 3401 and 3382 cm−1, and between 1632 and 1628 cm−1). Peaks at around 1220 cm−1 in samples
Specific surface area and calculated diameter of CoFe2O4 NPs and functionalized CoFe2O4 NPs using BET method
The results of specific surface area measurements of the nanoparticle samples are shown in Table 3.
Specific surface area of CoFe2O4 NPs prepared in combination of TEOS with alkoxysilanes in various molar ratios.

NPs: nanoparticles; MTMS: methyltrimethoxysilane; PTMS:
From the data of the NPs specific surface area determined by the BET method, the average diameter of nanoparticles (
Average diameter of CoFe2O4 NPs prepared in combination of TEOS with alkoxysilanes in various molar ratios.
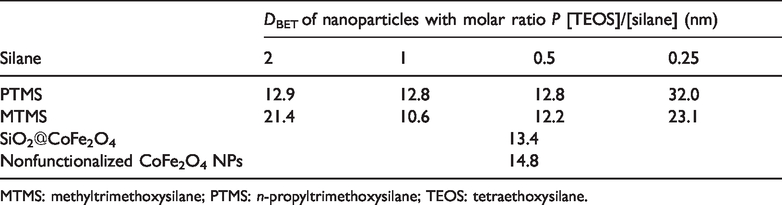
MTMS: methyltrimethoxysilane; PTMS:
The particle size of CoFe2O4 NPs determined with the measurements of the specific surface area takes a slightly higher value (14.8 nm) than the particle size determined by X-ray powder diffractometry (11.4 nm) and electron microscopy (11.5 nm), which can be attributed to the agglomeration of the particles.
Calculated
We expected that the specific surface area of the particles will decrease with the increasing length of the aliphatic chain from methyl (MTMS) to propyl (PTMS), and that consequently the size of the particles will increase. As the length of the aliphatic chain of the precursor increases, the interactions between the alkyl parts in the structure begin to dominate, leading to an increase in steric obstruction between silane molecules. Compared to long aliphatic chains, short aliphatic chains allow molecules of silanes a less obstructed approximation to the surface, which is reflected in a greater proportion of the surface coverage of the particles, a lower specific surface and a larger particle size. In the case of selected silanes, the specific surface follows the findings and, on average, decreases from silane with the short chain (MTMS) to the longer chain silane (PTMS; Figure 6) for the molar ratios
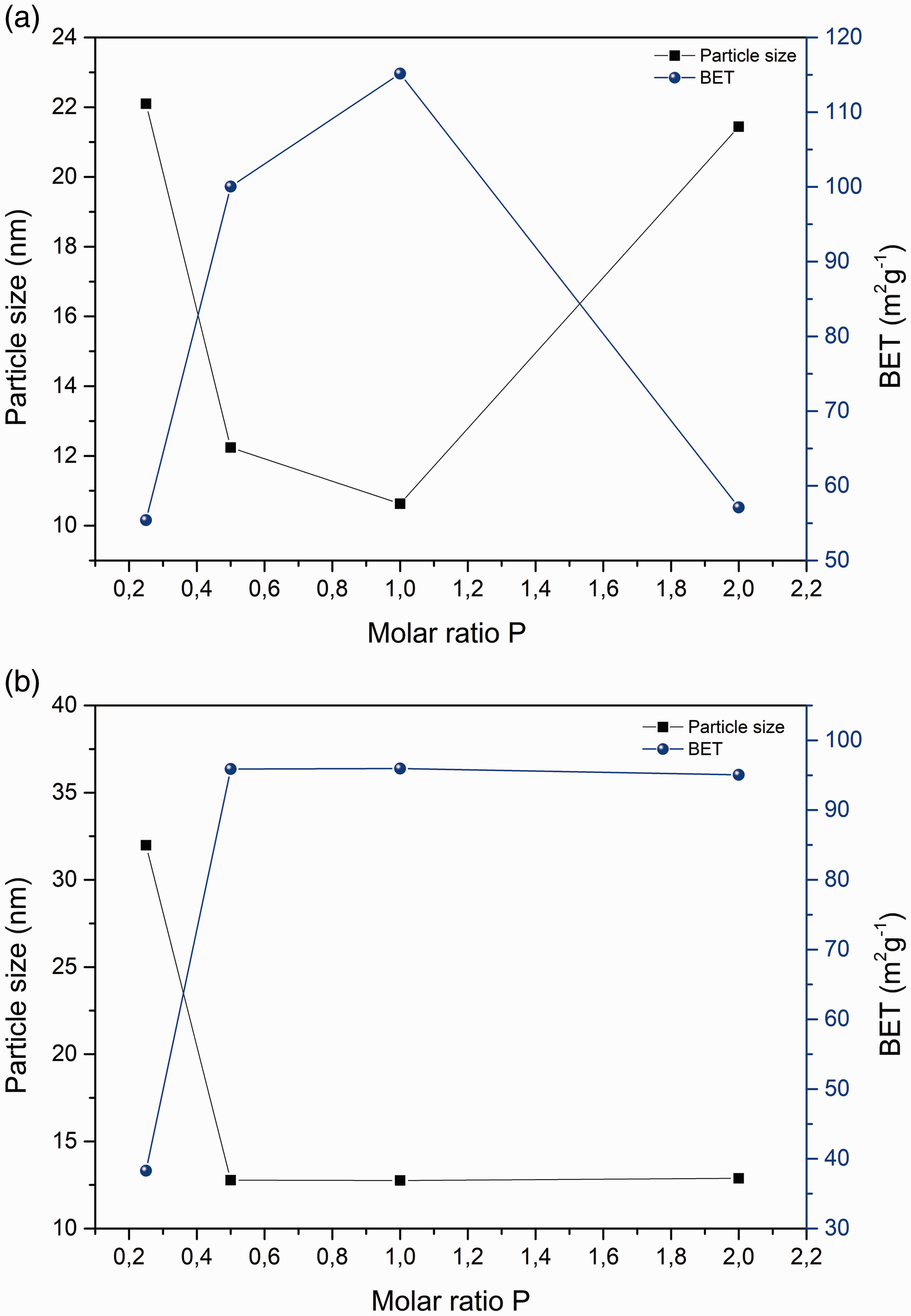
Dependence of specific surface area and diameter on molar ratio
Depending on the amount of silane addition, we found that the largest diameter (
According to the results, it can be concluded that the length of the aliphatic chain affects the specific surface of functionalized NPs (
VSM characterization
Figure 7(a) shows the hysteresis curve of the nonfunctionalized CoFe2O4 NPs sample. CoFe2O4 NPs reach a saturated magnetic response of 52.00 emu g−1 at the magnetic field strength

Magnetic properties of (a) CoFe2O4 and (b) SiO2@CoFe2O4 NPs.
Figure 7(b) shows the hysteresis curve of surface-treated CoFe2O4 NPs sample (SiO2@CoFe2O4). The specific magnetization of this sample is lower compared to the nonfunctionalized CoFe2O4 NPs sample. That can be contributed to the non-magnetic contribution of the amorphous phase of silicon dioxide (SiO2) covering the surface of CoFe2O4 NPs.
It is therefore expected that the further functionalization of SiO2@CoFe2O4 NPs with the molecules of selected alkoxysilanes will further reduce the specific magnetization of the samples due to the increased contribution of non-magnetic alkoxysilanes to the total magnetization.
The magnetic properties of materials are largely influenced by their intrinsic properties and particle size, which is determined by the high ratio between surface and volume. The particle size affects both the value of the specific magnetization (
Determining the crucial diameter of synthesized magnetic NPs is of utmost importance due to its influence on magnetic properties and to understand the response of prepared nanoparticles to the external magnet. For the CoFe2O4 NPs, the reported values of critical diameter vary from 23 nm (Maaz et al., 2007) up to 70 nm or more (Berkowitz and Schuele, 1959). With the presumption that the prepared CoFe2O4 NPs have a spherical shape, we used equation (6) to determine the critical diameter


With the literature data (Mathew and Juang, 2007; Rondinone et al., 2000) and obtained experimental values critical diameter for prepared nonfunctionalized nanoparticles is 512 nm. Because the prepared nonfunctionalized nanoparticles have much lower values than calculated critical diameter confirmed by BET and TEM analysis, we concluded that all prepared functionalized nanoparticles are single-domain particles.
Figure 8 shows the magnetic properties of sample SiO2@CoFe2O4 NPs prepared in combination with alkoxysilanes MTMS and PTMS in molar ratio

Magnetic properties of (a) PTMS@SiO2@CoFe2O4, (b) MTMS@SiO2@CoFe2O4 samples at molar ratio
It is known that the specific magnetization
The magnetic properties of prepared NPs are influenced by the NPs size (
Comparison of the average particles size (

NPs: nanoparticles; MTMS: methyltrimethoxysilane; PTMS:
Water contact angle measurements
Measurements of water contact angle for nonfunctionalized NPs showed that water droplet immediately wetted the surface of CoFe2O4 NPs firmly pressed on the adhesive substrate, which gives the value of contact angle of 0°. According to the literature of hydrophilic/hydrophobic theory the contact angles below 90° represent hydrophilic surfaces and contact angles above 90° represent hydrophobic surfaces (Arkles, 2011). SiO2-coated CoFe2O4 NPs have also shown hydrophilic surface, although the water droplet took 4 s to wet the surface.
PTMS functionalized magnetic NPs showed somewhat less hydrophilic character for molar ratios of

Water contact images of PTMS functionalized magnetic NPs with molar ratio (a)
MTMS functionalized magnetic NPs had for all molar ratios hydrophilic character, but the time until the water droplet fully wetted the surface was longer than observed for nonfunctionalized NPs, showing that the functionalization with MTMS lowers their hydrophilic character.
According to the results, the alkyl chain length influences the hydrophilicity. With increased chain length, the hydrophilicity decreases and the hydrophobicity increases. Methyl chains present in MTMS give more hydrophilic character to the functionalized magnetic NPs, whereas the propyl chains of PTMS give the NPs a hydrophobic character, as was observed for the sample PTMS@SiO2@CoFe2O4 with molar ratio
Evaluation of oil removal capacity, oil removal efficiency and recovery of prepared magnetic NPs adsorbents
According to the procedure described in ‘Materials and methods’ section the obtained adsorption capacities of PTMS and MTMS functionalized magnetic NPs are summarized in Table 6.
Adsorption capacity values obtained for PTMS and MTMS functionalized magnetic NPs.

MTMS: methyltrimethoxysilane; PTMS:
The results show very good used motor oil adsorption capacity for prepared functionalized magnetic NPs with values between 2.6 and 3.6 g of oil for 1 g of NPs, which is roughly three times the value of their own weight. Both silanes used for functionalization of magnetic NPs have the best adsorption capacity when their molar ratio is
Higher adsorption capacity of PTMS functionalized NPs can be attributed to longer aliphatic chain of propyl, compared to shorter methyl chains of MTMS. Higher C atom presence in longer aliphatic chains presumably have more Van der Waals attractive forces on the NPs surface that can interact with oil molecules, giving the particles higher adsorption capabilities. Measured water contact angles also showed that PTMS functionalized NPs had slightly more hydrophobic character than MTMS functionalized NPs. Hydrophobicity is also an important property of oil spill adsorbent material, so that it adsorbs mostly oil and not the water.
Regeneration of prepared oil adsorbent magnetic NPs was conducted with facile ethanol rinsing in the ultrasound bath for 5 min in three repetitions, described in other work (Chen et al., 2013; Zhu et al., 2010). Regenerated NPs were used again for removal of oil from water and evaluation of specific adsorption. PTMS (
For the adsorption capacities comparison with other published work, similar researches were chosen in terms of using functionalized magnetic NPs for adsorption of oil types – new and used lubricating oils, such as motor oil. The adsorption capacity of here reported functionalized magnetic NPs were amongst the highest with the comparable research shown in Table 7 and their almost unchanged adsorption capacities after regeneration are the highest amongst reported, thus showing a great potential for their application in real case scenarios.
Comparison of adsorption capacity of lubricating oils using functionalized magnetic NPs and their regeneration.
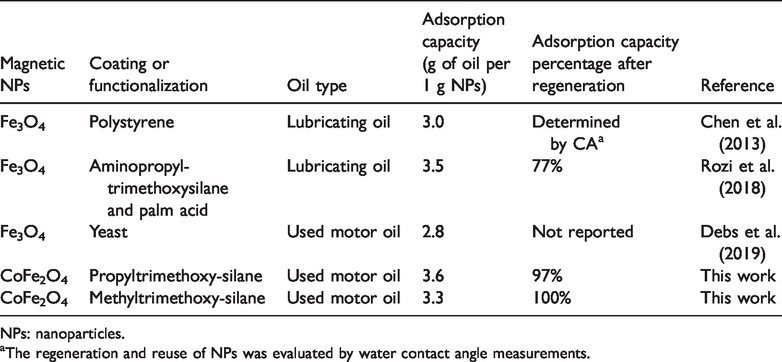
NPs: nanoparticles.
aThe regeneration and reuse of NPs was evaluated by water contact angle measurements.
Conclusions
In the presented research work, we have successfully synthesized monodisperse CoFe2O4 magnetic NPs with spinel crystal structure with diameters of 11.5 ± 1.9 nm and electrostatically stabilized them to obtain a stable colloidal solution. The magnetic NPs were functionalized with TEOS in combination with two different chain length silanes (PTMS and MTMS) in various molar ratios. Extensive characterization of nonfunctionalized and functionalized magnetic NPs was conducted in order to understand their behaviour when used as oil adsorbents. Silicon layer increased the particles size from 11.4 to 13.1 nm. The specific surface area that is crucial for good adsorbent material was determined with BET method for all prepared NPs and was the highest for MTMS and PTMS functionalized NPs at molar ratio
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant for Young Researches (No. 1000–16-0552) from the Slovenian Research Agency (ARRS).
