Abstract
Development of new materials includes establishing relationships between their different components and the physical and chemical properties pertinent to a particular application. In this work, the relationship between several magnetic nanoproperties of cobalt–zinc ferrite as functions of the contents of sodium hydroxide (NaOH) and gadolinium (Gd) are studied using statistically designed experiments. The results point towards the possibility of optimizing the material’s coercivity while leaving the rest of the properties statistically unaffected. This study helps to establish the material’s feasibility to be used in magneto-caloric applications.
Introduction
The application of design of experiments to determine laboratory synthesis settings can be useful to study the effect of controllable variables on material properties, while reducing costs and material usage. When cobalt–zinc ferrite is doped with a rare earth element it results in an attractive candidate for magneto-caloric applications.1–3 In this particular work, industrial refrigeration systems are of interest. A promising rare earth element for this application is Gadolinium (Gd) owing to its proven beneficial effect on magnetic properties. The possibility of developing a magneto-caloric pump for refrigeration systems using accessible magnetic materials is an attractive solution to reduce maintenance costs since it does not require the usage of mechanical parts. 1 The material developed must achieve low demagnetization temperature (Td), high magnetization (Mmax), low coercivity (Hci), and high pyromagnetic coefficient.
Design of experiments can be applied to meet such specifications, as presented in this work. For the experiment described here, Co0.7Zn0.3Fe2-yGdyO4 was synthesized by co-precipitation and the magnetic properties were measured using a vibrating sample magnetometer, while the structural properties were analyzed by X-ray diffraction (XRD) analysis. We applied the, so-called, central composite statistical design (CCD) to study the effect of sodium hydroxide (NaOH) and gadolinium (Gd), on the expected structural and magnetic properties of cobalt–zinc ferrite.
Background
Central composite design
The main purpose of this article is to study the effect of the molarity of sodium hydroxide and the concentration of gadolinium (controllable variables) on: Td, Mmax, Hci, and pyromagnetic coefficient. The ferrite nanocrystals were synthesized by co-precipitation; where NaOH is a precipitating agent that promotes the ferrite’s growth, making its molarity critical. The controllable variable levels chosen were previously studied varying in range [0.24–0.72 (M)] for the sodium hydroxide and [0.01–0.03] for gadolinium, in the formation of Co0.7Zn0.3Fe2-yGdyO4 ferrite.
Designing experiments, in general, consists of three phases: planning, execution, and analysis. The use of designing tools, such as the one proposed for this article, provides the smallest number of runs aiming to obtain the most information. Research laboratories can greatly benefit from these characteristics owing to their usual scarcity of resource. Design of experiments are also useful for subsequent system optimization, as shown in Garcia-Loera et al. 4 and Hernandez et al. 5 The design chosen for this work was the central composite design for various reasons:
it is capable of estimating curvature;
it is modular;
it is economic.
The CCD, in short, studies the effect of each of multiple factors and their interactions, usually up to a second order. The design for this work consists of corner, central, and axial points with four replicates in the center, as seen in Figure 1. Statistical characterization of the magnetic and structural properties was performed using a significance of α = 0.05. The coded values were used to perform every analysis throughout the experiment. For the practitioner, it is important to note that the CCD can be manipulated to sample each factor at three levels, which results in the more typical configuration of a 32 factorial experiment.
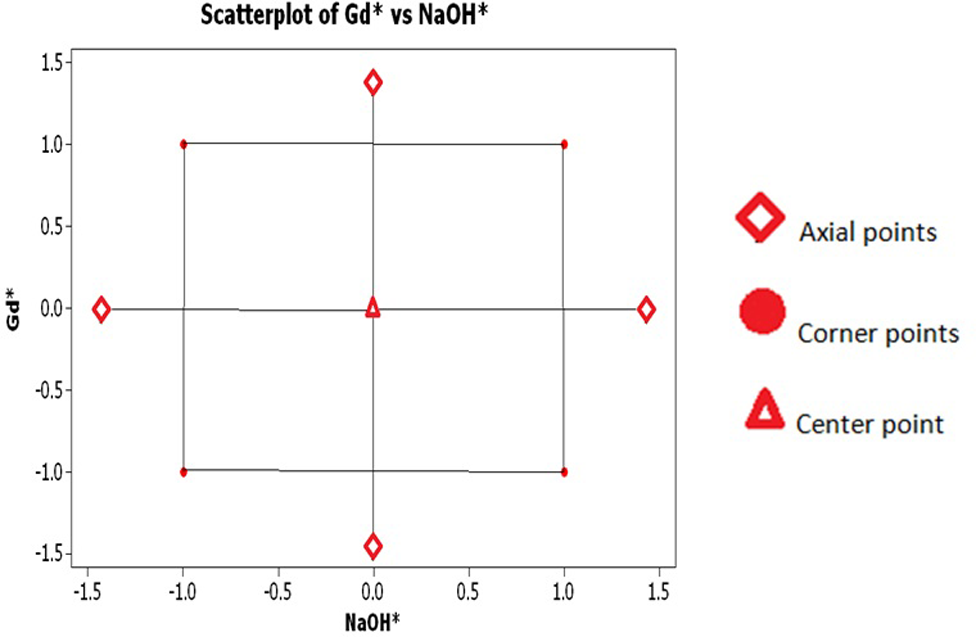
Central composite design with coded values.
Results
Statistical characterization
The transformation of the natural variables (NaOH molarity and Gd-concentration) to the coded variables for each factor was carried out using the coordinates of the central composite design. For each combination of factors, the ferrite nanoparticles were synthesized by the conventional co-precipitation method. The corresponding structural analysis of the powdered samples was carried out on a Siemens D5000 X-ray diffractometer. The magnetic properties Mmax (emu/g) and Hci (Oe) were obtained on a LakeShore 7400 Series vibrating sample magnetometer. Magnetization-Applied magnetic field (M-H) measurements were performed at room temperature in fields up to 22 kOe. Results for the properties measured are displayed in Table 1. All equipment and software used in this work were available at UPRM facilities.
Results for the structural and magnetic properties of the ferrite.
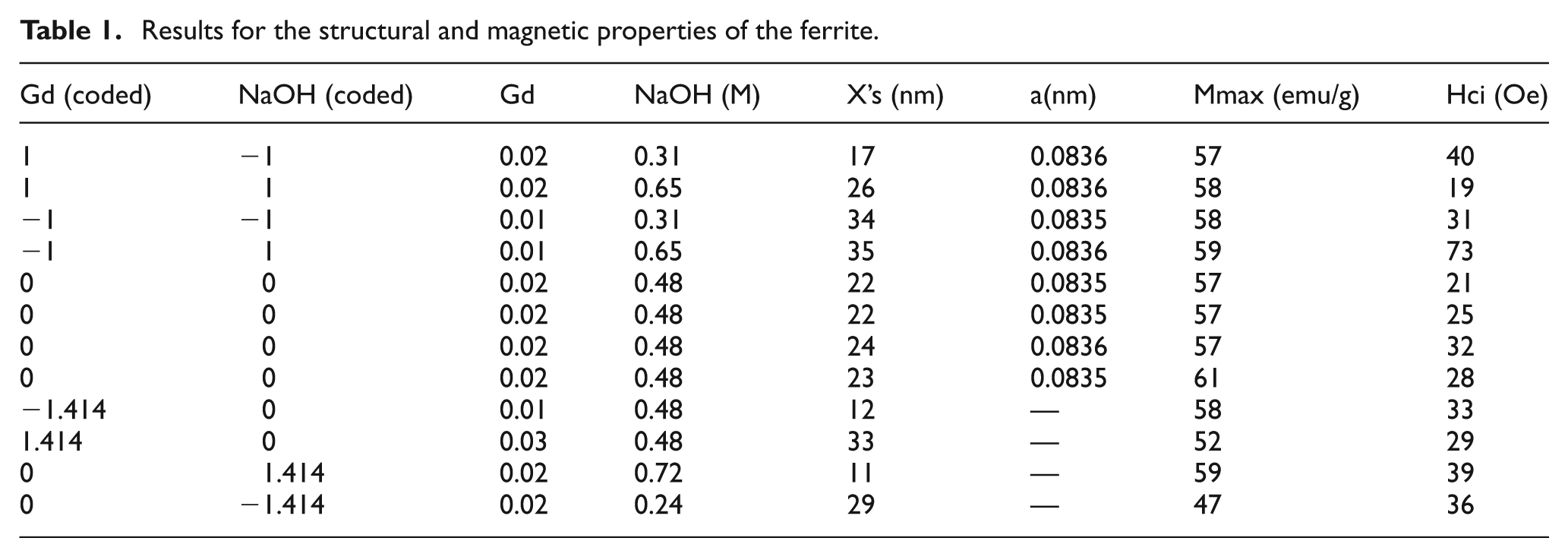
The structural properties of the Co0.7Zn0.3Fe2-yGdyO4 ferrite are studied since X (nm) is related to the maximum magnetization and the formation of a ferrofluid suspension. The XRD patterns confirm the formation of the ferrite phases for the corner points as seen in Figure 2.
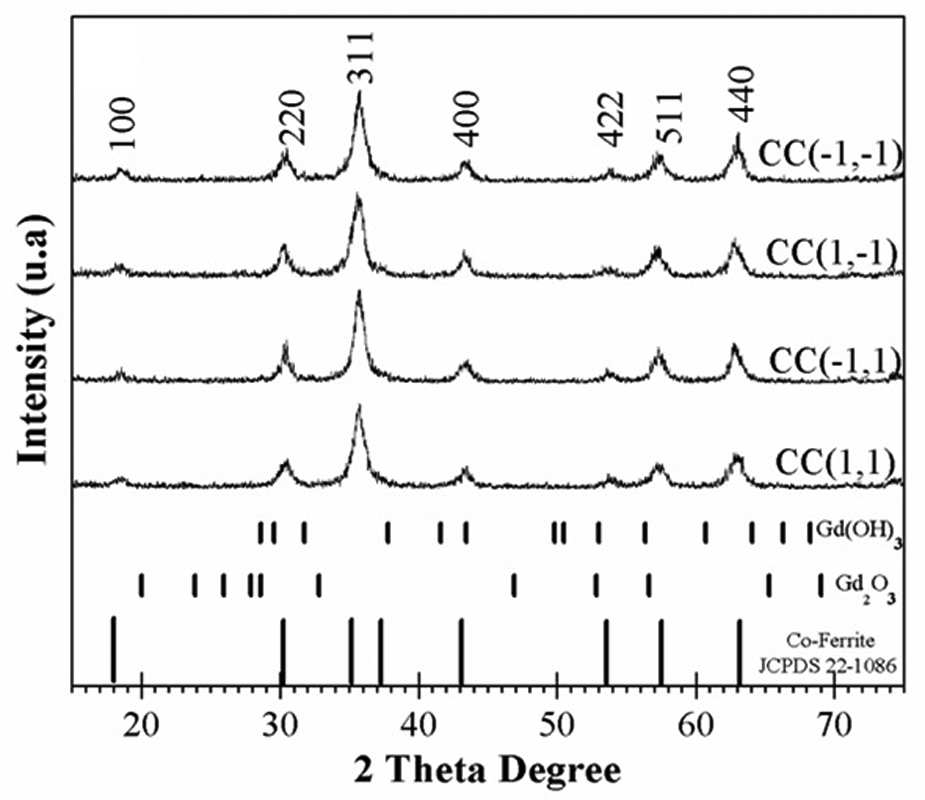
X-ray diffraction analysis for corner points.
The average size of the crystal X(nm) and lattice parameter (structural properties) were analyzed statistically using MINITAB16, as well as for the remaining properties. The analysis of variance for the structural properties, average size of the crystal, and lattice parameter showed no linear effect, interaction, or curvature as functions of the desired variables, thus neither property can be manipulated through variation of gadolinium or sodium hydroxide. The statistical analysis of Mmax (emu/g) revealed that neither sodium hydroxide nor gadolinium had a significant effect on magnetization, which assumes values of between 57 and 61 emu/g. This slight variation is within the experimental error. In turn, the analysis of variance for coercivity (Oe) suggested an interaction between the controllable variables with a p-value of 0.009 and a negative coefficient. The results are displayed in Table 2 and Figure 3.
Analysis of variance for coercivity (hci).
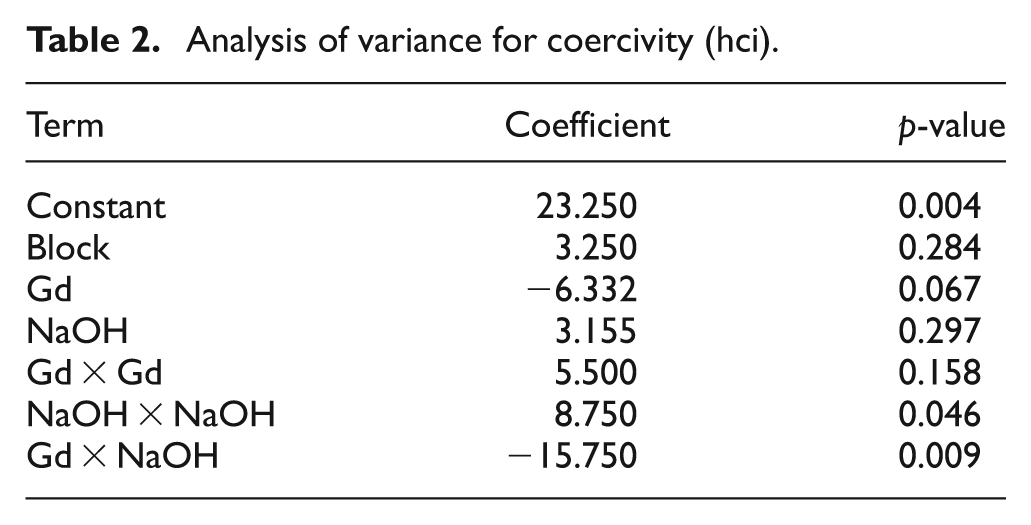
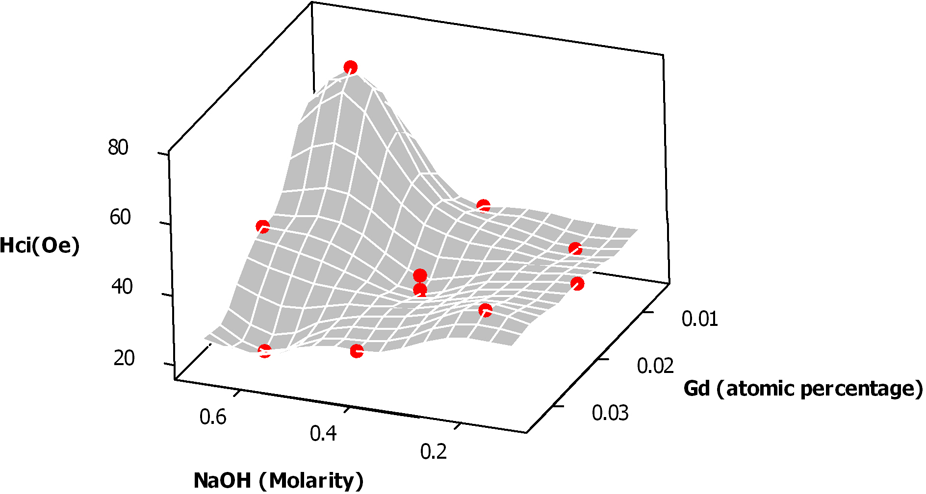
Surface plot for coercivity (Hci) as a function of NaOH molarity and Gd atomic percentage.
Our results suggested that an increase in the atomic concentration of gadolinium-ions in the ferrite lattice would cause coercivity to decrease. This decreasing trend in coercivity can be attributed to the null contribution of Gd3+ (4f7) ions to the angular momentum, which is conducive to vanish the spin–orbit interaction associated with coercivity. 6 This behavior has been previously reported in Urcia-Romero. 7
Figure 3 graphically shows the interaction detected by statistical means. This figure shows the variation of coercivity as a function of both, gadolinium-ion atomic percentage and sodium hydroxide molarity. In this case, the response surface suggests an enhancement of coercivity in the sodium hydroxide and gadolinium doping ranges of 0.625–0.675 M and 0.010–0.012%, respectively. The corresponding average crystal size was around 35 nm. This rising trend in coercivity can be explained in terms of the enlargement of the crystal within the single magnetic domain region8–9 and the eventual variation in the ionic distribution in the ferrite lattice. This hypothesis will be corroborated by Mössbauer analyses and the results will be reported elsewhere in the future.
Conclusion
In terms of characterization, our results show that only coercivity (Hci) can be manipulated with the variation of sodium hydroxide and gadolinium. This one is, indeed, a conclusion that is statistically based. This result means that this property can be controlled while leaving the rest of the properties statistically untouched. The value where coercivity is the minimum, is when both the concentrations of gadolinium and sodium hydroxide are maximum in the specified experimental region. This work supports experimental design as a tool that can be used in laboratories to characterize and model different systems to facilitate and drive discovery.
Footnotes
Acknowledgements
This work was supported by the Applied Optimization Group at the University of Puerto Rico – Mayaguez campus and by the Center for Research Excellence in Science and Technology (CREST) at the University of Puerto Rico at Mayagüez.
Funding
This work was funded by the National Science Foundation (NSF) [grant number 0833112].
