Abstract
The calcium silicate hydrate synthesized from electrolytic manganese residue was investigated as adsorbents to remove the phosphate from aqueous solutions. The effects of different experimental parameters, including contact time, initial phosphate concentration, solution pH, and temperature on the phosphate adsorption were investigated. The results showed that the phosphate adsorption was highly pH-dependent, with removal being more efficient under the pH range of 10.0–11.0. The Freundlich and Langmuir models were used to simulate the sorption equilibrium, and the results indicate that the adsorption data fitted well to the Langmuir model with which the maximum phosphate adsorption capacity was estimated to be 65.79–85.47 mg/g at 25–35°C. Meanwhile, the kinetic data confirmed that the sorption of phosphorus onto electrolytic manganese residue–calcium silicate hydrate can be best described by the pseudo-second-order kinetic model, suggesting that the adsorption process might be chemical sorption. Furthermore, thermodynamic studies illustrated that the adsorption process was endothermic and spontaneous in nature.
Introduction
Phosphorus, as an essential resource and material, is widely used in the areas of agriculture and industry. However, large quantities of phosphate present in wastewater are one of the main causes of eutrophication that leads to short- and long-term environmental and aesthetic problems in lakes, coastal areas, and other confined water bodies (Biswas et al., 2008; Kawasaki et al., 2010). Accordingly, the removal of phosphates from wastewater discharges to water bodies is an important component of eutrophication control strategies.
To date, various technologies including chemical precipitation, biological treatment, and adsorption method have been employed for the phosphate removal from wastewater (Mino et al., 1998). The chemical precipitation and biological treatment methods have proved to be either poor operational stability, strict control of operating conditions, or slow treatment speed, requiring considerable infrastructure investment (Ning et al., 2008). Therefore, among them, adsorption method using solid adsorbents has been a promising method for treating wastewater, owing to its advantages such as high efficiency, relatively inexpensive operation, and potential for saving and recycling phosphate resources in a sufficiently large-scale operation (Seida and Nakano, 2002). In recent years, considerable attention has been paid to the development of effective and low-cost adsorbents used for the phosphate removal including fly ash (Agyei et al., 2000), red mud (Li et al., 2006), slag (Xue et al., 2009), sand (Ou et al., 2007), aluminum compounds (Kawasaki et al., 2010), iron oxide (Zeng et al., 2004), calcium-based adsorbents (Karaca et al., 2006), niobium oxide (Rodrigues and da Silva, 2010), zeolite (Wang et al., 2011), and other waste materials (Zhang et al., 2009). As reported in the above literatures, it was well known that the adsorption capacity and efficiency were greatly dependent on the materials of adsorbents. That is to say, if inexpensively, environmentally friendly, and effectively alternative adsorbents can be developed, it would be beneficial to the environment and have attractive commercial value due to the risk of phosphate in water and more stringent effluent standards imposed.
By 2012, the production capacity of electrolytic manganese metal (EMM) in China had exceeded 2.11 Mt/a, accounting for 98% of the total global annual production. Electrolytic manganese residue (EMR) is discharged from the electrolytic manganese industry, and it is a by-product of making electrolytic manganese powder (Hagelstein, 2009). In the current EMM industry, with the grade of manganese ore decreasing, about 10–12 tons of EMR is discharged into the environment per ton of produced EMM. As EMR contains some toxic and hazardous substances, it is therefore highly questionable if the EMR generated is managed properly. Currently, almost all EMR in China is dumped without any treatment into landfill sites near the plants. Hence, with the long-term weathering, pollutants in EMR can migrate and transfer continuously, which can cause soil contamination, river, or groundwater pollution and some other serious environmental pollution (Li et al., 2014). On the other hand, disposal of tons of EMR resulting from large-scale industrial activities is an ongoing financial burden for EMM manufacturers due to the high costs for long-term maintenance and new disposal sites. Thus, for environmental protection and sustainable development, it is urgent to continuously develop new and advanced recycling techniques for the utilization of EMR.
Recently, our research group developed a novel conversion process for the production of calcium silicate hydrate from EMR. Calcium silicate hydrate (generally called C–S–H) is one of the components found in cement minerals and includes many amorphous and metastable structures (Richardson, 2008). On the basis of previous studies, C–S-H generally shows a high surface area and contains surface hydroxyl groups and hydrated Ca2+ ions to which various chemical entities can be attached or ion-exchanged, thereby being effective in adsorbing metal ions and phosphate ions (H
The purpose of this work is to examine the feasibility of using EMR-made C–S–H (EMR–CSH) as adsorbents for phosphate removal. And, the adsorption properties of EMR–CSH in phosphate removal process from aqueous solution under various experimental conditions, including pH value, contact time, and initial phosphate concentration, have been investigated. Moreover, kinetic and equilibrium models to fit experimental data were used to understand the adsorption process.
Methodology
Materials
The EMR investigated in this study was collected from an electrolytic manganese company, situated in western of Hunan province, China. The chemical properties of the EMR used are shown in Table 1. Meanwhile, the chemical composition of several other EMR has also been listed in Table 1. The results in Table 1 show that this chemical composition is fairly common in EMR produced in China’s EMM industry. Stock solutions of phosphate ions with concentration at 1000 mg/L were prepared by dissolving given amounts of Na2HPO4·12H2O in distilled water. The experimental solution was prepared by diluting the stock solution with distilled water when necessary.
Chemical composition of EMR.

aThe EMR used in this study was collected from Hunan, China.
bThe EMR was collected from Guangxi, China.
cThe EMR was collected from Chongqing, China.
dOther components may include biomass and some small amounts of other elements (<0.1% each), such as Co, Se, Cr, As, etc.
Synthesis of EMR–CSH from EMR
Based on our previous studies (Li et al., 2015), a hydrothermal reaction process was adopted for the synthesis of EMR–CSH in this research. First, the Ca-rich filtrate was collected by filtration during the MnO2 recovery process described in our previous studies. EMR–CSH samples were prepared by precipitation in gradually adding the obtained Ca-rich filtrate into sodium silicate solution under stirring, which was pre-dissolved with PEG400 in distilled water (PEG400 concentration was 0.6 mL/L). Initial Ca/Si ratio was kept at 1.0 in this study according to Kuwahara et al. (2013). To the blended solutions, 1 mol/L NaOH aqueous solution was added dropwise, and the pH value was adjusted to 12.0 ± 0.1, followed by stirring for 2 h at 100°C and aging at the same temperature for another 10 h. At the end of the process, the thus-obtained solid product was separated by filtration and washed with a copious amount of distilled water to remove sodium ions and any residual polymer. The waste water obtained during the distilled water washing process can be reused as solvent in the leaching process prior to synthesis of EMR–CSH (for details, please see Li et al. (2015)). The precipitates were then washed with ethanol and dried at 100°C in an oven for 7 h for further work. As for the effluent collected from the ethanol washing process, the valuable component, namely ethanol, can be recovered from the effluent using a convenient distillation process for the next use. The synthesized EMR–CSH were analyzed by X-ray powder diffraction (XRD; D8 Discover, Bruker, Germany), FE–SEM (Mira3, Tescan, Czech Republic), and specific surface area (SSA; ASAP2020, Micromeritics, USA) using the same methods described in a previous paper (Li et al., 2015)
Adsorption studies
In this study, phosphate removal for EMR–CSH was carried out through batch experiments. The experimental procedure was as follows: 0.1 g of EMR–CSH was placed in 250 mL volumetric flasks and mixed with 100 mL phosphate solution of different concentrations ranging from 40 to 120 mg/L. The solution pH was adjusted from 4.0 to 11.0 by using 0.5 mol/L HCl and/or NaOH. The mixture was then stirred at 200 r/min in a constant temperature shaker bath at different temperatures (25, 30, and 35°C). During each run, a portion of the solution was withdrawn at regular intervals of time, diluted, and centrifuged for 2 min at 5000 r/min. The residual phosphate concentration was then analyzed according to national standards (GB/T 6913–2008) using Shimadzu UV–vis spectroscopy at 710 nm. The equilibrium adsorption capacity qe (mg g−1) was defined as

Meanwhile, it is worth to note that the aim of this work is to remove phosphate from drinking water and water bodies such as rivers and lakes, where pH value is generally close to neutral. Therefore, the kinetic study at pH 6.0 is useful in some of practical applications.
Results and discussion
Characteristics of the prepared EMR–CSH
The XRD pattern of the prepared EMR–CSH was demonstrated in Figure 1. It was observed that the synthesized EMR–CSH sample particularly exhibited clear diffraction peaks at 16.6°, 29.2°, 31.9°, 49.5°, and 54.9° which are well consistent with those of reported in Kuwahara et al. (2013), suggesting a successful formation of C–S–H. Meanwhile, the morphological analyses of synthesized EMR–CSH were carried out using FE–SEM under different resolution ratio, and the results are shown in Figure 2. As seen in Figure 2(b), EMR–CSH comprises nano-size plates, where the plates with a diameter of approximately 200 nm are fused into an open framework structure with a “desert rose”-like surface morphology (Li et al., 2015). And, the open framework structure means that it has a large surface area and pore volume to which adsorbates are readily accessible. The observed morphological feature agrees well with that for pure C–S–H previously reported by Johnston and Small (2011), again elucidating the successful formation of C–S–H. Brunauer–Emmet–Teller (BET) surface area measurement was also adopted to determine the SSA of the synthesized EMR–CSH. And the results show that the EMR–CSH material has a high BET-specific surface area of 205 m2/g with a pore volume of 0.68 cm3/g, which is generally desirable for different applications such as adsorbent usages.
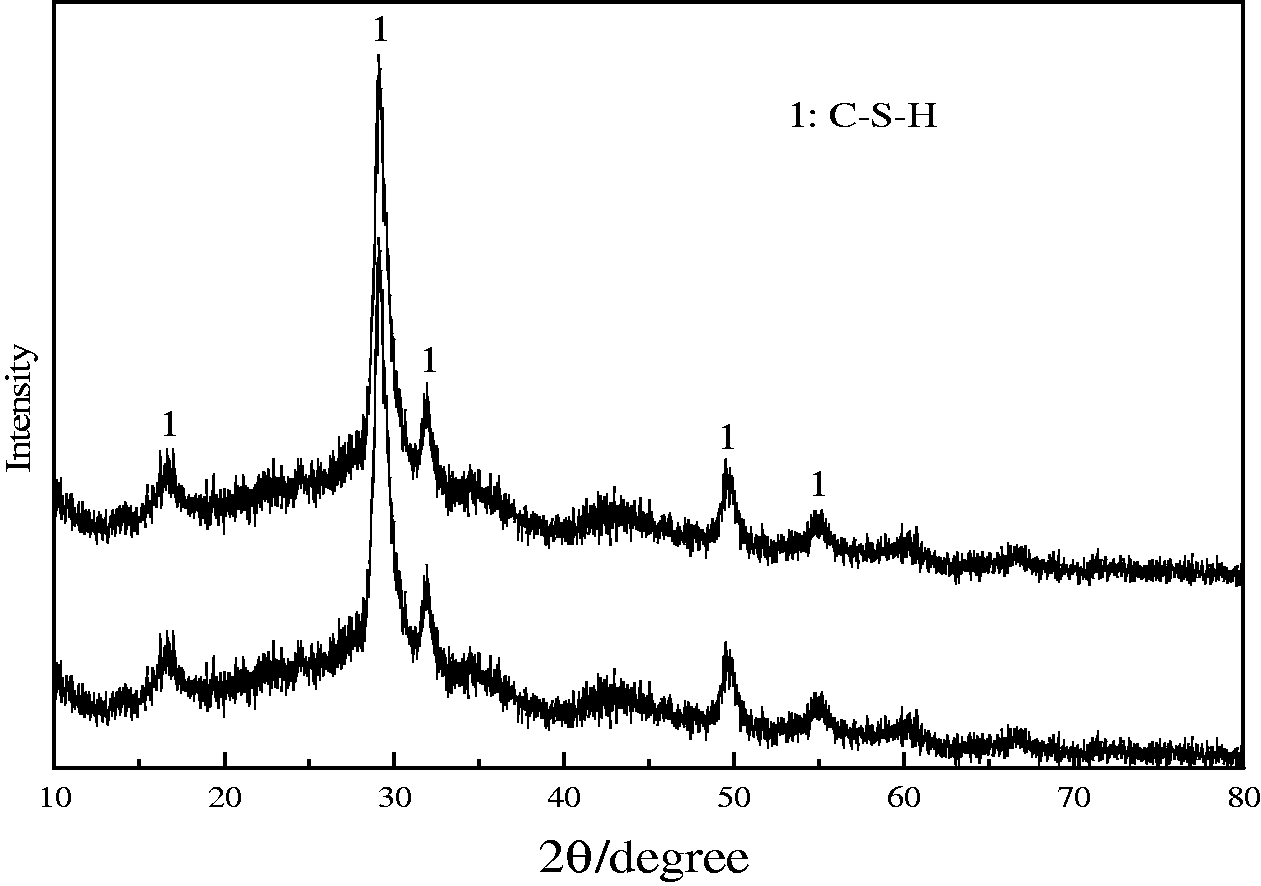
XRD patterns of the synthesized EMR–CSH before phosphate adsorption (a) and after phosphate adsorption (b).

FE–SEM images of synthesized EMR–CSH: (a) resolution ratio ×10,000; (b) resolution ratio ×20,000).
Effect of contact time
The contact time required for adsorption equilibrium usually depends on the initial phosphate concentration. In this work, the experimental results on adsorption of phosphate onto EMR–CSH at various concentrations (40, 60, 80, 100, and 120 mg/L) with contact time are illustrated in Figure 3. It was seen from Figure 3 that all curves have the similar shape, and the amount of phosphate adsorbed increased with the increase of both initial concentration of phosphate and contact times. And the most of the phosphate uptake occurs within 165 min, ranging between 82 and 100% of the totally adsorbed. The contact time needed for phosphate removal by EMR–CSH to reach equilibrium was established at 465 min for all the concentrations studied. The unit adsorption for phosphate was increased from 18.64 to 46.93 mg/g as the phosphate concentration increased from 40 to 120 mg/L. The effective phosphate adsorption by the prepared EMR–CSH can be contributed to the fact that EMR–CSH is composed of porous structure with large internal surface area (205 m2/g). Meanwhile, the higher uptake at higher concentrations was obviously observed, which can be attributed to a greater driving force by a higher concentration gradient pressure. Also as seen from Figure 3, it is obvious that the adsorption process of phosphate consisted of two phases: an initial rapid phase and a slower second phase. The amount of phosphate adsorbed increased rapidly in the beginning 100 min. For periods greater than 100 min, the adsorption capacity increased only a little with progress of adsorption. The initial rapid phase may be due to greater accessibility to pores and increasing the number of vacant sites available at the initial stage, which lead to the enhancement of mass transfer driving force (Zhang et al., 2009). However, at approaching equilibrium, adsorbate concentration is much reduced, and the saturation of adsorption sites is achieving, and the rate of adsorption therefore will be much slow.
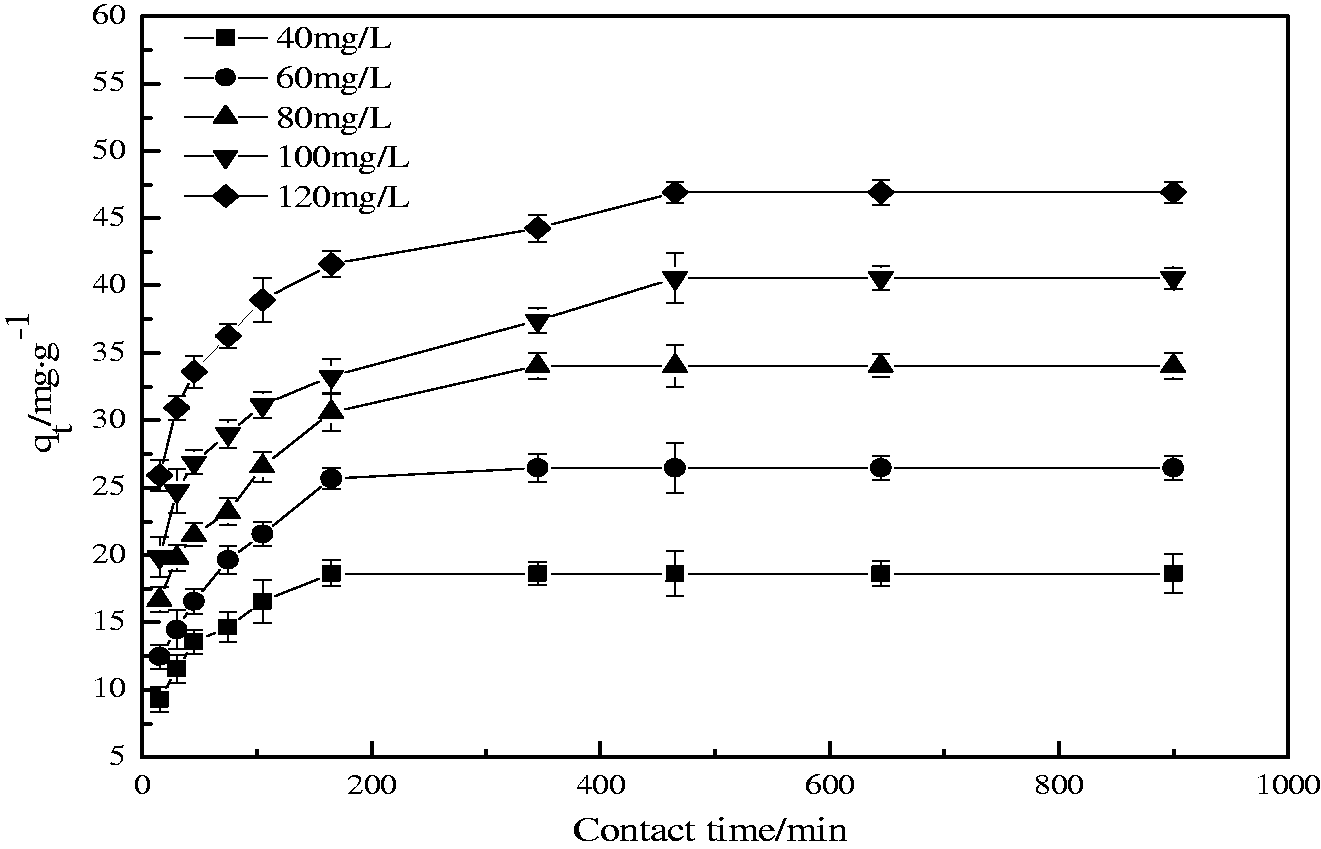
Effect of contact time on phosphate adsorption (adsorbent dose = 1.0 g/L; pH value = 7.0; temperature = 30°C).
Effect of pH
Since the adsorption process is usually influenced by the pH value of the solution, it is important to assess the adsorption behavior of phosphate on EMR–CSH in solutions with different pH values. And phosphate adsorption onto EMR–CSH in solutions with different pH values is shown in Figure 4. As seen from Figure 4, the removal of phosphate strictly increases as the pH increases. This variation is quite similar to the previous reports (Wang et al., 2011). It has been found that the pH is an important factor regarding phosphate adsorption onto EMR–CSH. The phosphate uptake is comparatively lower in the acid pH region (4.0–7.0), and it increases for higher pH values, and the greatest adsorption occurs under the pH range of 10.0–11.0. The remarkable observation from the pH tests can be probably attributed to the following reasons. In the higher pH range, the concentration of hydroxide groups is high, which is favorable for the precipitation of calcium phosphate. That is to say, the calcium phosphate precipitation could be a predominate mechanism for the phosphate removal under higher pH values. It is well known that the calcium-based materials have great potential for adsorbing phosphate ions from water owing to the strong affinity with phosphate ions to form insoluble calcium phosphate species (Kaasik et al., 2008; Yin et al., 2011; Zhao et al., 2009]. Also, this observation is consistent with the findings of Johansson and Gustafsson (2000) and ; Agyei et al. (2002), who suggested that the adsorption of phosphate was mainly due to the formation of calcium phosphate. Meanwhile, the lower adsorption of phosphate at pH range of 4.0–7.0 is probably due to the fact that EMR–CSH crystals begin to collapse or dissolve with decreasing pH in aqueous solutions (Lothenbach and Nonat, 2015).

Effect of pH value on phosphate adsorption (initial phosphate concentration = 120 mg L−1; adsorbent dose = 1.0 g/L; contact time = 465 min; temperature = 30°C).
Further analysis of the adsorbent and test solution recovered after the equilibrium adsorption under the pH range of 10.0–11.0 was performed by XRD, FTIR (Nicolet 6700, Thermo Fisher, USA), and ICP (Optima 5300DV, Perkin-Elmer, USA) to investigate the phosphate adsorption process. And it can been seen from the XRD and ICP analysis that the treatment with phosphate adsorption neither changes the crystalline phase nor leads to any new crystalline phases (for details, please see Figure 1), and the leaching amount of Ca2+ performed by ICP (Optima 5300DV, Perkin-Elmer, USA) analysis in the test solution recovered after equilibrium adsorption was negligible. These results indicate that the phosphate anions (HPO42– and PO43– are the dominant at pH 10.0–11.0) are adsorbed onto Ca2+ sites, probably via an electrostatic attraction between the positively charged Ca2+ sites and negatively charged HPO42–/PO43– anions, forming stable and dispersed calcium phosphate species on the adsorbent surfaces.
Moreover, the FTIR analysis was also conducted to investigate the change of the structure of EMR–CSH samples before and after the equilibrium adsorption, and the results were shown in Figure 5. The silicate vibration regions of C–S–H spectra generally resemble those of 1.4 nm tobermorite, and all contain a characteristic set of bands centered at about 970 cm−1. And in the FTIR spectrum of EMR–CSH samples before adsorption, a strong and broad band in the 3600–3300 cm−1 region (stretching vibrations of O–H groups in H2O or hydroxyls with a wide range of hydrogen bond strengths) and a band at 1632 cm−1 (H–O–H bending vibration of molecular H2O) indicated the presence of coordinated water molecules, with the peak at 1484 cm−1 (O–H bending vibration) indicating the presence of surface hydroxyl on the EMR–CSH surface. Compared with spectrum of EMR–CSH before phosphate adsorption (see Figure 5(a)), the peaks of 1632 cm−1 (H–O–H bending vibration of molecular H2O) and 1484 cm−1 (O–H bending vibration) after phosphate adsorption (see Figure 5(b)) were weakened dramatically and completely disappeared, respectively, while a new peak appeared at 1025 cm−1. The decreasing tendency of these two peaks indicated that the surface hydroxyl groups of EMR–CSH were replaced by adsorbed HPO42–/PO43– anions. And the new band at 1025 cm−1 could be assigned to the asymmetry vibration of P–O (Liu et al., 2008; Long et al., 2011; Persson et al., 1996). These results proved that phosphate was successfully adsorbed on the surface of EMR–CSH adsorbent, and the hydroxyl of EMR–CSH adsorbent was disappeared simultaneously. Therefore, it was again suggested that the specific adsorption could occur at the aqueous phosphate/EMR–CSH interface, and the replacement of surface hydroxyl groups by the phosphate species to form some stable and dispersed calcium phosphate species on the adsorbent surfaces is the main adsorption mechanism. Meanwhile, the band appears at 454–469 cm−1, which can be assigned to the deformation of TO4 tetrahedra (T = Si, Al, Mg, etc.) (Kuwahara et al., 2013).
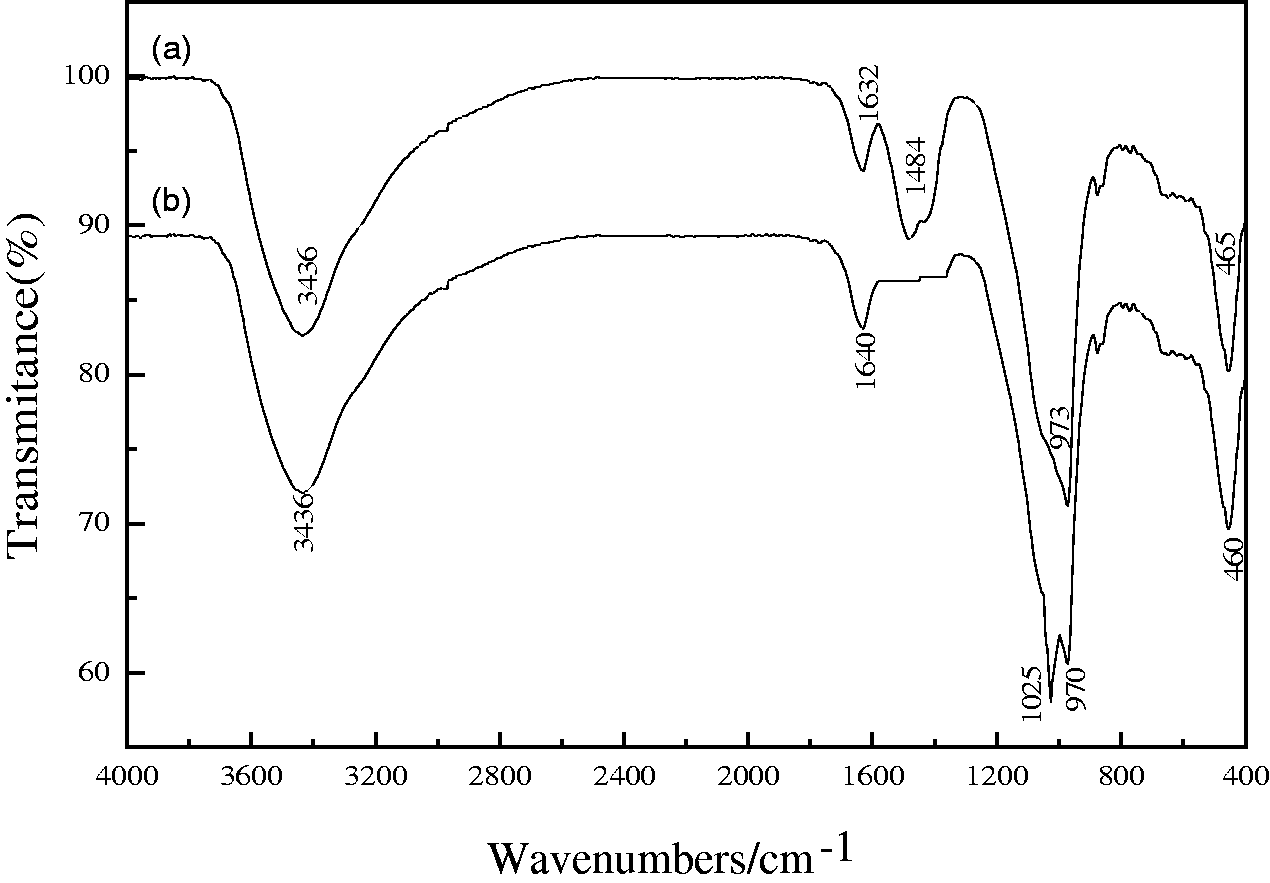
FTIR of the synthesized EMR–CSH before phosphate adsorption (a) and after phosphate adsorption (b).
Effect of initial phosphate concentration
The initial concentration of adsorbate in solution provides an important driving force in overcoming mass transfer resistance between the aqueous and the solid phases. The effect of the initial phosphate concentration in the solutions on adsorption at 25, 30, and 35°C was shown in Figure 6. As seen from Figure 6, equilibrium uptake increased with the increasing of initial phosphate concentration at the range studied, which is due to the fact that the adsorbent has sufficient active sites competent for relatively high concentration of phosphate (Karaca et al., 2006). Meanwhile, Figure 6 also illustrates that the equilibrium adsorption capacity is clearly affected by temperature, with the amount of phosphate adsorbed increasing from 46.35 to 59.38 mg g−1 initially at 120 mg L−1 when the temperature was raised from 25 to 35°C. The phosphate adsorption on the EMR–CSH powder was definitely endothermic in nature requiring some amount of activation. And the increases of adsorption with an increasing temperature have been widely observed in many studies (Liu et al., 2011; Zeng et al., 2004). Generally, the temperature has two major effects on the adsorption process. Increasing the temperature can not only increase the rate of diffusion of the adsorbate molecules across the external boundary layer and in the internal pores of the adsorbent particles, but also alter the equilibrium capacity of the adsorbent. Therefore, the high temperatures favor phosphate removal by adsorption onto EMR–CSH which may be due to the enhanced rate of intraparticle diffusion of the adsorbate, as diffusion is an endothermic process (Liu et al., 2011). On the other hand, the mobility of molecules increases generally with a rise in temperature, thereby facilitating the mass transport process by increasing the driving force of phosphate ions onto EMR–CSH and reducing the energy barrier of reaction between the adsorbent and the adsorbate.
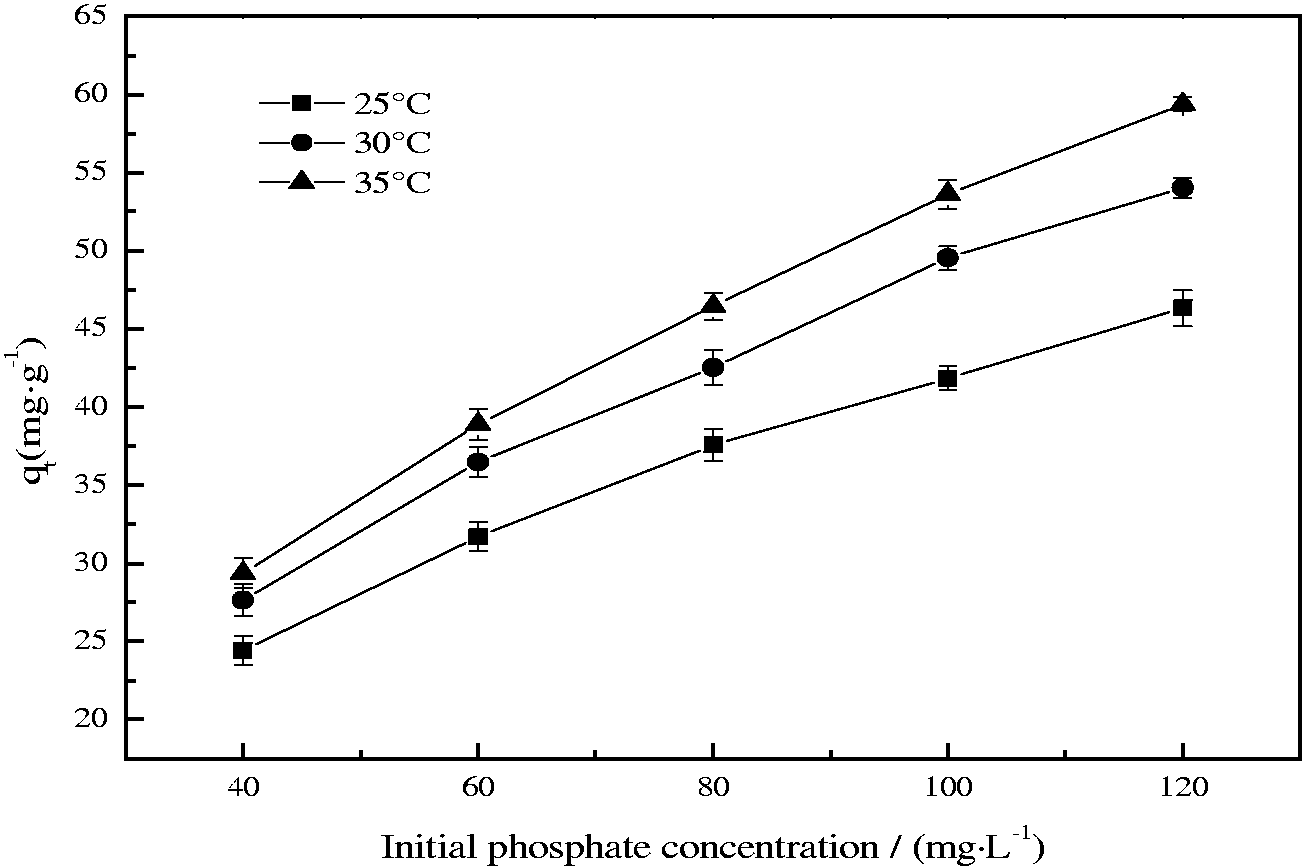
Effect of initial phosphate concentration on phosphate adsorption (pH value =10.0; adsorbent dose = 1.0 g/L; contact time = 465 min).
Meanwhile, in order to figure out whether the any toxic things can be produced in the adsorption process, elemental analyses by ICP (Optima 5300DV, Perkin-Elmer, USA) of the test solution recovered after the equilibrium adsorption revealed that leaching of the slag-derived heavy metals into solution was negligible under the conditions we examined, thereby demonstrating that the EMR–CSH can be used as an active, easy-to-handle, and environment friendly adsorbent for water purification applications.
Influence of Cl– ion on the adsorption of phosphate
As we know, identification of the rate-limiting step involved in a solid–liquid adsorption process is an important when hetero-ions (e.g. chloride ion) that have binding affinity to adsorbent are present. According to Figure 4, it can be found that the phosphate adsorption tends to increase with the increase of pH value. That is to say, the phosphate uptake amount considerably decreased with increase of the concentration of Cl– ion (the solution pH was adjusted under acidic condition by using 0.5 mol/L HCl), indicating that the presence of Cl– ion increased the effect of intra-particle diffusion on the adsorption of phosphate onto EMR–CSH. A probable explanation was that Cl– ions would reduce the tunnel size of the sorbent, resulting in slower diffusion that may hinder the access of phosphates ions to the active sites (Liu et al., 2011; Mezenner and Bensmaili, 2009).
Adsorption kinetics
In order to further understand the characteristics of the absorption process, two kinetic models were used to fit the experimental data, namely, pseudo-first-order and pseudo-second-order models (Biswas et al., 2008; Zhang et al., 2009). The best-fit model was selected based on both linear regression correlation coefficient (R2) and the calculated qe values. The pseudo first-order and second-order models were given as equations (2) and (3), respectively


Linear lines obtained from the variation of ln(qe − qt) versus t and t/qt versus t, according to the pseudo-first-order and pseudo-second-order kinetic model, have been given in Figure 7. And, the rate constants and correlation coefficients from the slope and intercept have been calculated and listed in Table 2. As shown in Table 2, the adsorption of phosphate onto EMR–CSH was not fitted to a first-order reaction, for the whole regression coefficients obtained at various phosphate concentrations are less than the ones obtained from the second-order kinetics. Besides, the qe value calculated from the second-order model is much more comparable to the experimental qe value than that from the first-order model, again suggesting the better fit of the second-order kinetics.

Plots of kinetics of phosphate adsorption onto EMR–CSH at different initial phosphate concentration (adsorbent dose = 1.0 g/L; pH value = 6.0; temperature = 30°C).
The constants and correlation coefficients of pseudo-first order and pseudo-second order kinetic models for adsorption of phosphate onto EMR–CSH.
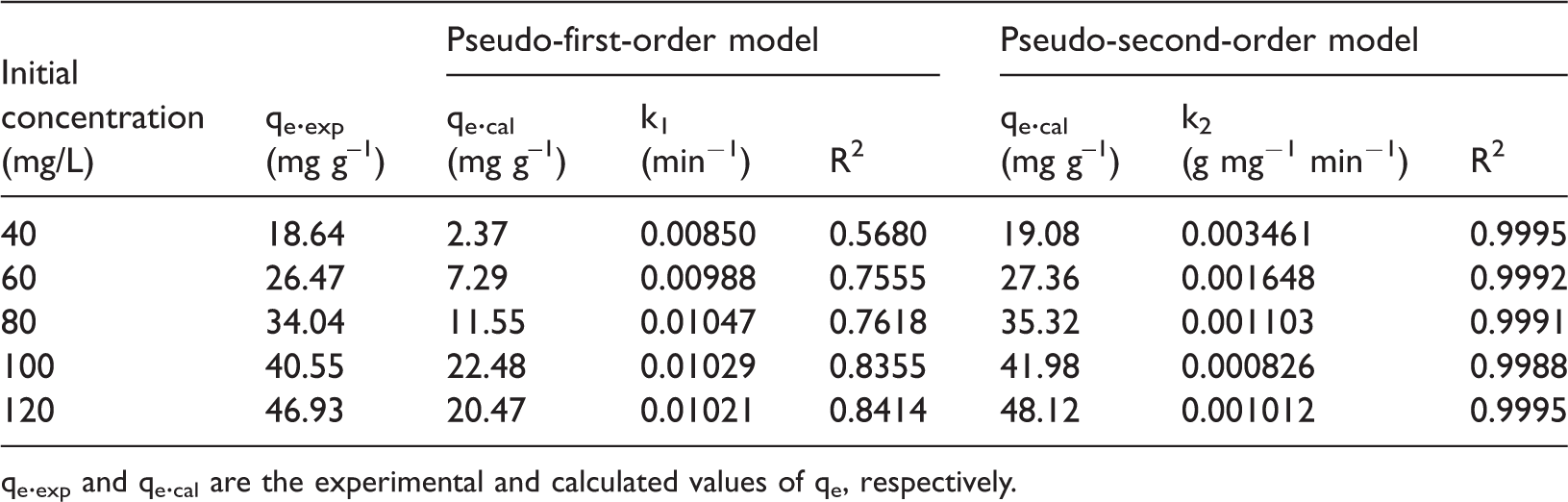
qe.exp and qe.cal are the experimental and calculated values of qe, respectively.
Adsorption isotherm
Langmuir and Freundlich models were used to describe the adsorption capacity of EMR–CSH for phosphate. The Langmuir model is based on the assumption of a structurally homogeneous adsorbent where all adsorption sites are identical and energetically equivalent. The linear form of Langmuir’s isotherm model is given by the following equation

The Freundlich model is applied to describe a heterogeneous system characterized by a heterogeneity factor of 1/
The Langmuir and Freundlich models were all tried to fit the experimental data. While the Langmuir isotherm parameters q0 and KL were obtained by plotting Ce/qe versus Ce, the Freundlich isotherm parameters were obtained by plotting ln qe versus ln Ce. The adsorption isotherms of phosphate on EMR–CSH at different temperatures and the fitted curves for Langmuir and Freundlich models are listed in Figure 8. The curve-fitting and statistical analyses were performed with OriginPro 8.5 software, and the parameters obtained from the isotherms are given in Table 3. As seen from Table 3, the R2 values of the Langmuir adsorption isotherms for phosphate are all much higher than those obtained for the Freundlich adsorption isotherms at the three different temperatures. Namely, the equilibrium data was well represented by the Langmuir isotherm equation when compared to the Freundlich equation. Meanwhile, the R2 values obtained for the Langmuir isotherms were both above 0.99, indicating the monolayer coverage of phosphate ions on EMR–CSH particles. In addition, the q0 from the Langmuir isotherm indicated that the sorption capacity of phosphate increased with an increase of temperature, indicating an endothermic nature of the adsorption. These results again confirm the endothermic nature of phosphate uptake by the synthesized EMR–CSH.

(a) Linearized form plot of Langmuir isotherm for phosphate adsorption onto EMR–CSH; (b) linearized form plot of Freundlich isotherm for phosphate adsorption onto EMR–CSH.
Langmuir and Freundlich isotherm constants for phosphate adsorption onto EMR–CSH at different temperature.

On the other hand, the Langmuir parameters can be used to predict the affinity between the sorbate and sorbent using the dimensionless separation factor RL, and RL is defined as follows

Table 4 presents a comparison of adsorption capacity of EMR–CSH with other various adsorbents for phosphate adsorption. Although direct comparison on the adsorption capacity of EMR–CSH via the Langmuir, q0 value with other adsorbent materials is difficult because of different applied experimental conditions, it was found, in general, that it could still be useful in comparing alternative materials for wastewater treatment (Cheung and Venkitachalam, 2000). In general, although very few materials such as MG@La (Rashidi et al., 2017) and ferric hydroxide-coated cellulose nanofibers (Cui et al., 2016) show higher adsorption capacity, still, the prepared EMR–CSH in the present work is superior to many other adsorbents reported in the literature (see Table 4). Thus, these results indicate that EMR–CSH has relatively high potential to be used as a commercial adsorbent for the removal and recovery of phosphate from aqueous solutions.
Comparison of maximum adsorption capacity of phosphate on various adsorbents.

On the other hand, because the EMR is waste materials, the cost to make EMR–CSH should be very low. Therefore, our synthesis pathway can replace the existing inorganic adsorbent synthesis route that requires costly, multiple pure metal-containing chemical sources, and would meet the strong demand for the effective utilization of waste slags in the EMM industry. In addition, when used as an adsorbent to reclaim phosphate from water, the exhausted EMR–CSH can be directly applied to agricultural fields as a fertilizer to improve soil fertility because the P-loaded EMR–CSH contains abundance of valuable nutrients. That is to say, there is no need for further treatment of the EMR–CSH adsorbed-phosphate product, which can greatly reduce the post-treatment fee compared with the other adsorbent. Hence, making use of the EMR–CSH is also a promising and low cost candidate for bulk wastewater treatment.
Adsorption thermodynamics
According to the Van’t Hoff equation, thermodynamic parameters, that change in free energy (ΔG), enthalpy (ΔH), and entropy (ΔS), can be calculated using equations (7) and (8). And all the thermodynamic parameters are listed in Table 5


Thermodynamic parameters for the adsorption of phosphate onto EMR–CSH.

Using the results in Table 3, ΔH and ΔS can be calculated from the slope and intercept of the plot of ln KL versus 1/T using equation (7) which is shown in Figure 9. The value of ΔH during the adsorption of phosphate was found to be 15.76 KJ/mol, suggesting that the adsorption of phosphate onto EMR–CSH was endothermic. The positive value of ΔS clearly states that the randomness increased at the solid-solution interface during the phosphate adsorption onto EMR–CSH. The values of ΔG decreased from –16.23 to –17.30 KJ/mol with increasing temperature (from 25 to 35°C), indicating that the adsorption of phosphate ions is a spontaneous process and more favorable at higher temperature.
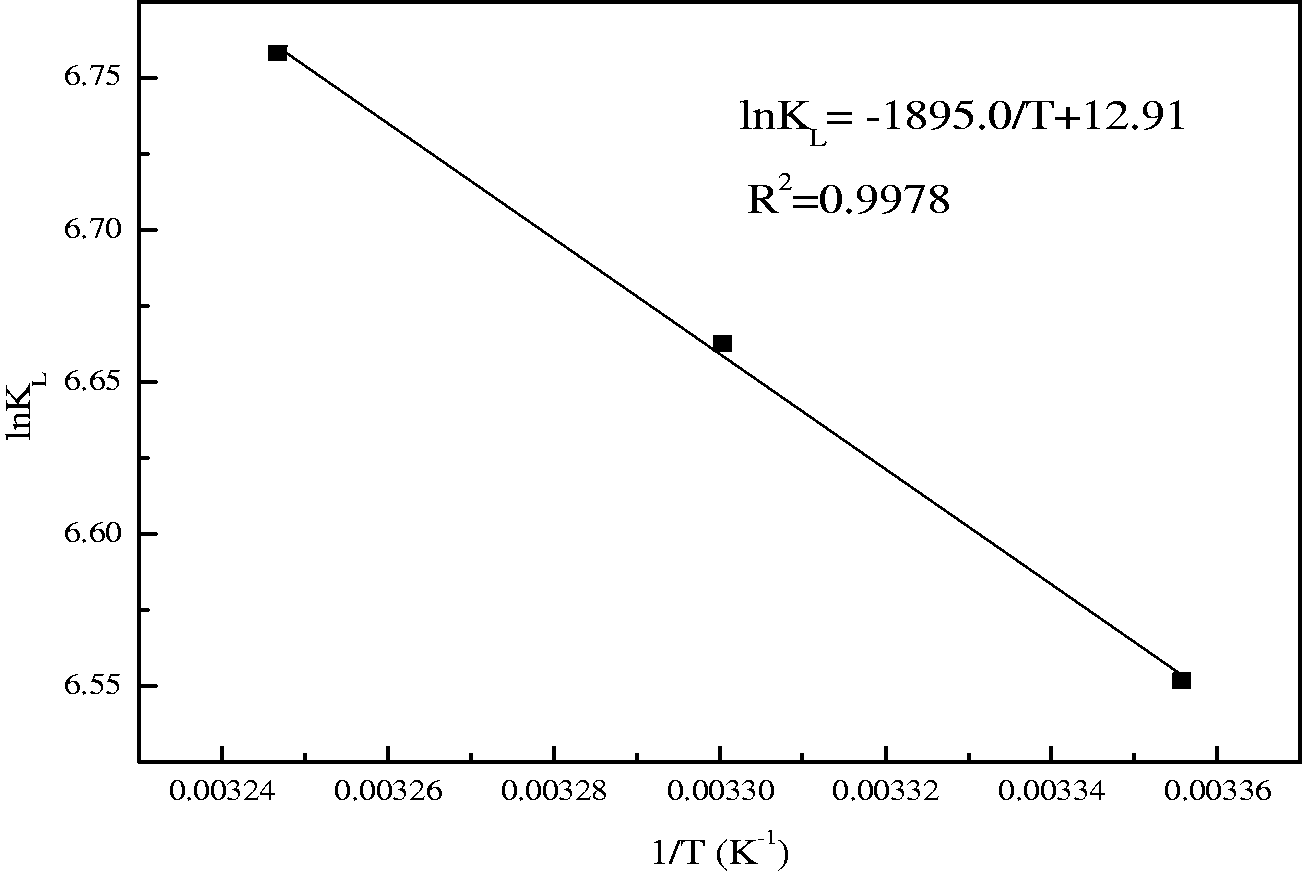
Plots of ln KL versus 1/T.
Conclusions
In this study, the EMR–CSH prepared from EMR has been investigated for phosphate adsorption. The results of this study indicate that EMR–CSH is an effective adsorbent for the removal of phosphate in aqueous solutions, which exhibits much higher adsorption capacity than other low-cost adsorbents. And the phosphate adsorption capacity of EMR–CSH was found greatly dependent on pH value, initial phosphate ions concentrations, temperature, and contact time. The adsorbed amount of phosphate increased with increasing initial concentration, contact time, and temperature. And the maximum adsorption capacity of phosphate was achieved at around pH 10.0–11.0. Analysis of kinetic data showed that the uptake of phosphate on EMR–CSH follows pseudo-second-order model. Correlation coefficients indicate that the Langmuir isotherm model was applied more accurately than the Freundlich model, which confirms the monolayer sorption of phosphate onto the EMR–CSH particles. And the Langmuir isotherm model indicated that the maximum sorption capacity of phosphate by the synthesized EMR–CSH was 65.79–85.47 mg/g at 25–35°C. Moreover, according to the thermodynamic studies, negative ΔG (–17.30 to –16.23 KJ mol−1) and positive ΔH (15.76 KJ mol−1) indicate that the adsorption phosphate onto EMR–CSH is a spontaneous, endothermic process. The synthesized EMR–CSH, with large adsorption capacity and low cost, is a very promising and potentially attractive adsorbent for treating wastewater contaminated with inorganic phosphate and consequently provides a step toward a sustainable society. Overall, it is demonstrated that the synthesized EMR–CSH is an effective adsorbent for phosphate removal. Due to their low cost and high capability, the synthesized EMR–CSH is a very promising and potentially attractive adsorbent for treating wastewater contaminated with inorganic phosphate and consequently provides a step toward a sustainable society.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: China Postdoctoral Science Foundation (No. 2017M611799), Hunan Provincial Science and Technology Plan Project, China (No. 2016TP1007), and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
