Abstract
Herein active manganese dioxide was prepared by different methods using KMnO4 and MnSO4·H2O as inorganic precursors. The impact of preparation methods on morphology and adsorption performance of the synthesized products was investigated. The experimental results show that the reaction temperature and pressure had a great effect on the morphology and adsorption performance of active manganese dioxide. The shape of active manganese dioxide prepared at room temperature and pressure was short rod-like while active manganese dioxide synthesized by hydrothermal method was mesoporous fibers and had better adsorption performance. The adsorption behavior of 2,4-dinitrophenol on mesoporous manganese dioxide was well described by Langmuir and Freundlich isotherm equation (
Introduction
Manganese dioxide, for its well-known adsorption properties, catalytic activity and ion exchange ability capacity compared with other adsorbents, such as zeolite and activated carbon, has attracted more attention in environmental field application (Lisha et al., 2010; Liu et al., 2010). Moreover, owing to the high oxidation potential (
Manganese dioxide can be synthesized by physical and chemical methods such as ultrasonic process (Bastami and Entezari, 2012; Sun et al., 2011), chemical synthesis (Piumetti et al., 2015), solid-phase synthesis (Lee and Jung, 2012), hydrothermal method (Lu et al., 2012; Xu et al., 2013), sol–gel methods (Gupta et al., 2011; Yi et al., 2006), and supercritical technology (Nugroho and Kim, 2014). Studies of manganese oxide have indicated that the adsorption capacity of the manganese oxide is dependent on the preparation methods due to the variable manganese oxidation state. Previous studies have focused on preparation and characterization of manganese oxide by a single method or technique (Hu et al., 2016; Ryu et al., 2015; Samarghandi et al., 2015; Verma et al., 2016; Xu et al., 2015). However, the influence of preparation methods on surface morphology and adsorption properties of manganese oxide was neglected. It is still a great demand to investigate the relationship of preparation methods and adsorption properties of manganese oxide.
2,4-Dinitrophenol (DNP) is widely applied to production of organic synthesis (Mackedonski and Hadjiolov, 1970), then a great deal of DNP wastewater was produced in all above manufacturing process. DNP is strictly limited by every nation for discharges because of high toxicity, bad biodegradability, and environmental hormone activity. The ingestion of DNP causes harmful effects on human beings (Lu et al., 2011). Several treatment techniques for DNP wastewater have been reported and these include solid phase extraction (Luo et al., 2008), biochemical (Hirooka et al., 2006), chemical oxidation (Pillai and Gupta, 2015), and activated carbon absorption (Cao and Shiraishi, 2010; Carvajal-Bernal et al., 2015). Although these traditional treatments had mature application, they had many limitations, such as high cost, source waste, and second pollutions.
From the above-mentioned facts, active manganese dioxide (MnO2) adsorbent was prepared using KMnO4 and MnSO4·H2O as inorganic precursors under hydrothermal conditions and ambient pressure conditions, respectively. The structure and morphology of the obtained samples were systematically characterized by X-ray diffraction (XRD), high resolution transmission electron microscopy (HR-TEM), and N2 adsorption–desorption. The impact of preparation methods on structure, morphology, and adsorption performance of active manganese dioxide was investigated. The adsorption behavior of DNP on active manganese dioxide was studied. The effect of initial solution pH on adsorption performance was investigated, and possible mechanisms of adsorption were also discussed.
Materials and methods
Sample preparation
All reagents for synthesis active manganese dioxide were analytic grade, potassium permanganate (KMnO4) (KeBang Chemical Products Co., Ltd., Tianjin, China; 99.99% purity), manganese sulfate (MnSO4·H2O) (KeBang Chemical Products Co., Ltd., Tianjin, China; 99.99% purity), DNP (Aladdin Industrial Corporation, Shanghai, China; 99.99% purity). All reagents without additional purification and deionized (DI) water were used in all processes.
Firstly, active manganese dioxide was prepared at ambient pressure (denoted as sample 1). 5.0708 g of MnSO4·H2O and 3.1598 g of KMnO4 were dissolved in 60 mL DI water, respectively. Then KMnO4 solution was gradually added into MnSO4 solution under magnetic stirring. A dark brown precipitate formed immediately. After reaction, the precipitation was separated by filtration, washed with ethanol and DI water, and dried at 120°C for 12 h.
For comparison of the impact of preparation method on structure and adsorption performance of active manganese dioxide, active manganese dioxide was also synthesized by hydrothermal method (denoted as sample 2). Similarly, 5.0708 g of MnSO4·H2O and 3.1598 g of KMnO4 were dissolved in 60 mL DI water, respectively. Then KMnO4 solution was gradually added into MnSO4 solution under magnetic stirring. The mixture was stirred for another 30 min, then was transferred to a 100 mL Teflon-lined autoclave (φ 50 mm × 100 mm, Senlong experimental apparatus Co., Ltd., Beijing, China) and heated at 100°C for 2 h. Active manganese dioxide was formed at high temperatures and pressures. After reaction, the precipitation was separated by filtration, washed with ethanol and DI water, and dried at 120°C for 12 h.
Sample characterization
Phase composition of active manganese dioxide was studied by XRD (Bruker D8 Focus, in 10–70° 2θ range with a step of 0.02° and a residence time of 0.5 s), diffraction pattern was analyzed using International Centre of Diffraction Data (ICDD, PDF4) in EVA (Bruker) software. The microstructure of active manganese dioxide was assessed by HR-TEM (JEM-2100F, JEOL, Japan). Quantachrome Nova Station A (Quantachrome, NOVA 2200e) was used to obtain N2 adsorption–desorption isotherms at 77.3 K. The samples were out gassed at 300°C for 6 h before nitrogen adsorption. The pore-size distributions were calculated based on the Barrett–Joyner–Halenda (BJH) method. Specific surface areas were determined by the BET method (Monosorb Analyzer MS-13, Quanta Chrome, USA), using liquid N2 as adsorbate.
pH value at the point of zero charge (pHpzc) has important effects on adsorption capacity of mesoporous manganese dioxide and was determined by ζ potentiometer (zetaplus4 instrument Brookheaven, USA). The surface ζ potential of mesoporous manganese dioxide is negative when pH value is greater than 2.4, however, the surface ζ potential is positive. So the pHpzc of mesoporous manganese dioxide is 2.4.
Evolution of adsorption performance
To determine the adsorption performance of DNP on active manganese dioxide, experiments were carried out using batch equilibration techniques at room temperature and atmosphere pressure. The Erlenmeyer flasks, containing DNP solution of 20 mg/L and adsorbent, were placed in an electronic temperature-controlled shaker (Taihua HZQ-F160, Taichang, China), and shaken at 150 r/min. After adsorption at 25°C for 12 h, the concentration of residual DNP was determine by UV spectrophotometry (UV2300IIUV spectrophotometry, Techcomp Limited, Shanghai, China). The solution pH was measured using pH meter (Leici PHS-3C pH meter, Shanghai, China). All experiments described above were performed in duplicate and the averaged values were recorded.
Results and discussion
Active manganese dioxides prepared by different methods were characterized by XRD (Figure 1) to study the variety of the position and intensity of diffraction peak. According to the ICDD, powder diffraction file (PDF) standard card, active manganese dioxide prepared at ambient pressure(sample 1) matches a typical XRD of δ-MnO2, and active manganese dioxide prepared under hydrothermal conditions (sample 2) matches a typical XRD of α-MnO2. Result of XRD indicates that crystalline morphological of active manganese dioxide depends on reaction temperature and pressure.

XRD pattern of active manganese dioxide. XRD: X-ray diffraction.
The structure of active manganese dioxide can be observed from HR-TEM images (Figure 2(a–c)). As shown in Figure 2, preparation method has a prominent influence on the surface morphology of the samples. The shape of active manganese dioxide prepared at ambient pressure (sample 1) appears to be short rod-like (Figure 2(a)) structure. Active manganese dioxides prepared under hydrothermal conditions (sample 2) exhibit a uniform fibrillar morphology with length from 0.2 to 1 μm (Figure 2(b)), and possessed well-defined mesoporous structure according to the HR-TEM images (Figure 2(c)). The morphology identification indicates that mesoporous manganese dioxide (sample 2) with good crystal structure could be achieved by the hydrothermal method.

HR-TEM micrographs of sample 1 (a) and sample 2 (b and c). HR-TEM: high resolution transmission electron microscopy.
Although the N2 adsorption–desorption isotherm of sample 1 and sample 2 all belong to Langmuir type IV, the hysteresis loop of active manganese dioxides are entirely different (Figure 3(a)). The hysteresis loop of sample 1 belongs to type H3, it illustrates that there are slit pores due to conglomeration of active manganese dioxides particles. The hysteresis loop of sample 2 belongs to type H2, and the adsorption isotherm is relatively far from desorption isotherm at high pressure proving the mesoporous structure of the sample 2. Type H2 hysteresis loop usually results from a complex, interconnected pore network. The H2 hysteresis indicates that relatively uniform pores are present in the sample 2. The broad hysteresis loop centered at higher

Nitrogen adsorption–desorption isothermal (a) and BJH pore size distribution (b) of active manganese dioxides. BJH: Barrett–Joyner–Halenda.
The pore size distribution of sample 1 and sample 2 show narrow distributions centered at 5.4 nm and 7.5 nm (Figure 3(b)) and pore volumes of 0.216 and 0.237 cm3/g (Table 1), respectively. According to IUPAC classification, mesoporous solids contain pores with a pore diameter between 2 and 50 nm (Xi et al., 2009).
Mesoporous parameters and adsorption ability of active manganese dioxide.

From the BET result, the surface area of sample 1 was found to be 198.149 m2/g and that of sample 2 is 378.149 m2/g (Table 1). The surface area, pore volumes and pore size of sample 2 are obviously bigger than those of sample 1, which is ascribed to the mesoporous structure of sample 2.
By carrying out the batch adsorption experiments, adsorption performance of active manganese dioxides preparation at different methods was compared. 100 mL of 20 mg/L DNP solution were added into the Erlenmeyer flask containing 0.8000 g of each adsorbent. The Erlenmeyer flasks were placed in an electronic temperature-controlled shaker, and shaken at 150 r/min. After adsorption at 25°C for 12 h, the absorbance of DNP at

UV-vis spectra of different concentrations of 2,4-dinitrophenol and work curve of 2,4-dinitrophenol.
Table 1 shows that the adsorption effect of mesoporous manganese dioxide (sample 2) are better than that of sample 1 due to the mesoporous structure of sample 2. UV-Vis spectra of different concentrations of DNP and work curve were showed in Figure 4. The absorbance of DNP at
The influence of various initial quantity of mesoporous manganese dioxide on adsorption performance was investigated (Figure 5). The concentration of DNP was kept constant (20 mg/L) while the quantity of mesoporous manganese dioxide varied between 2 and 15 g/L. The Freundlich (

The effect of the quantity of mesoporous manganese dioxide on UV-vis spectra of 2,4-dinitrophenol.

Freundlich and Langmuir adsorption isotherm of mesoporous manganese dioxide on 2,4-dinitrophenol.
Here,
The parameter 
Here, 
The approach was usually applied to distinguish the physical and chemical adsorption with its mean free energy, if the value is between 1 and 8 kJ/mol, then the adsorption is physical in nature, and if it is between 8 and 20 kJ/mol, then the adsorption is chemisorption.
The results show the characteristic absorbance peak of DNP decreases with increasing quantity of mesoporous manganese dioxide (Figure 5). B band of DNP exhibits a blue-shift in the UV-vis adsorption spectra during the adsorption process, which illustrate that DNP was partly oxidized accompanied by adsorption process.
The adsorption isotherms of Freundlich, Langmuir, and Dubinin-Radushkevich models for DNP removal are shown in Figure 6. Freundlich isotherm yielded linear plots and the values of the coefficients (
From the linear plot of Dubinin-Radushkevich models,
The adsorption capacity of DNP on mesoporous manganese dioxide versus contact time was also studied. The concentration of DNP was 20 mg/L while the quantity of mesoporous manganese dioxide was 8.000 g/L. The results are depicted in Figure 7. As shown, the characteristic absorbance peak of DNP decreased with time. The intraparticle diffusion model (

The effect of adsorption time on UV-vis spectra of 2,4-dinitrophenol.
Here,
Figure 8 shows that the kinetics process for adsorption of DNP on the mesoporous manganese dioxide was closely consistent with the characteristics of the intrapartide diffusion and pseudo-second order reaction. The pseudo-second-order kinetics model is based on the postulate that the adsorption rate is controlled by chemical adsorption. The kinetics analysis showed the intraparticle diffusion and chemical adsorption control the adsorption together, which are in good agreement with the analysis conclusion of Dubinin-Radushkevich models.

Adsorption kinetic of 2,4-dinitrophenol by mesoporous manganese dioxide.
The influence of the pH on adsorption performance was investigated (Figure 9). The concentration of DNP was 20 mg/L, the quantity of mesoporous manganese dioxide was 8.000 g/L and the contact time was 12 h while the initial solution pH was in the range of 2–8. The pH of these DNP solutions was adjusted by using NaOH or H2SO4.
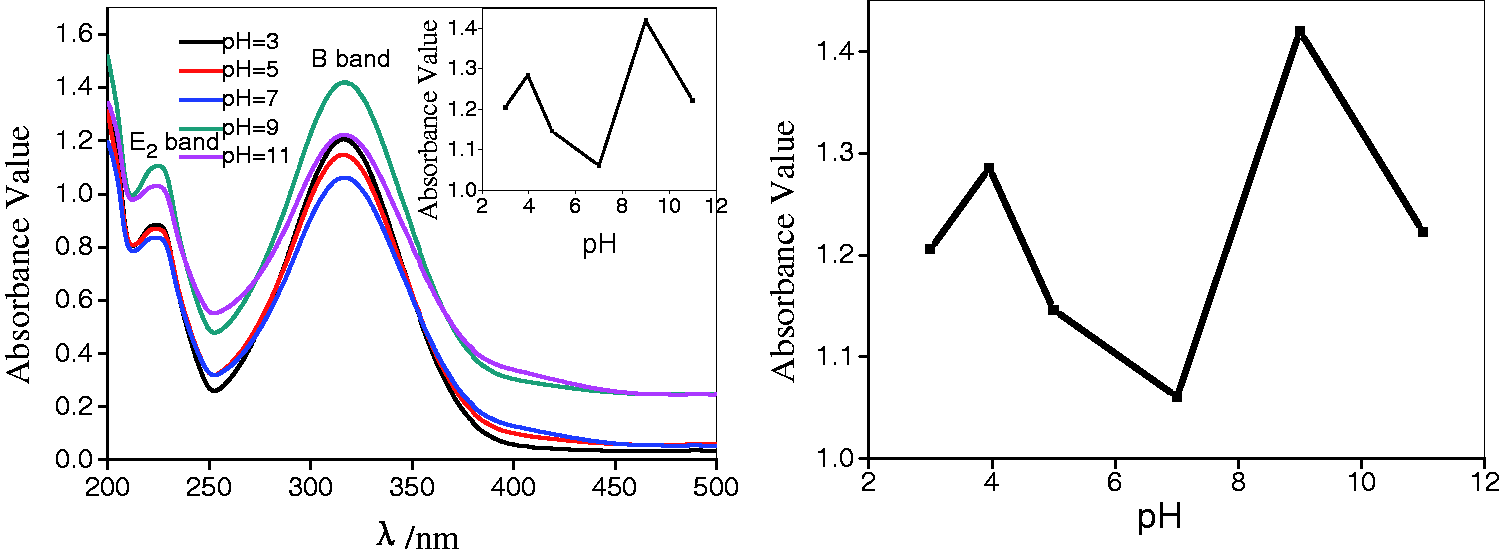
The effect of pH on adsorption efficiency of mesoporous manganese dioxide.
Figure 9 shows that the pH has an apparent influence on adsorption performance of mesoporous manganese dioxide. The pKa value of DNP is 4.09 (Liu et al., 2010). The pHpzc of the mesoporous manganese dioxide is 2.4, which was determined by ζ potentiometer, and the particles of mesoporous manganese dioxide were negatively charged when pH value is greater than 2.4. There existed competitive adsorption between DNP and H+ ions on the particle surface of mesoporous manganese dioxide under acidic conditions. The levels of H+ ions dropped and the removal efficiency of DNP increased with pH increase from 3 to 7. The surface of mesoporous manganese dioxide is rich in hydroxyl groups which have excellent adsorbed properties for DNP, and the quantity of hydroxyl groups increased with increasing pH value. So the removal efficiency of DNP increased with pH increase from 9 to 11.
Conclusions
Active manganese dioxide was prepared by different methods using KMnO4 and MnSO4·H2O as inorganic precursors. The shape of active manganese dioxide prepared at room temperature and pressure is short rod-like. Active manganese dioxide synthesized by hydrothermal method is mesoporous fibers and has better adsorption performance. The adsorption behavior of DNP on mesoporous manganese dioxide is well described by Langmuir, Freundlich isotherm equation ( The pH of solution has significant effect on the adsorption effect of DNP on mesoporous manganese dioxide, and the adsorption capacity is 2.539 mg/g in the condition of pH = 7.
Footnotes
Authors′ contributions
H Chen carried out the experiments. H Chen and W Tian participated in the preparation and characterization of active manganese dioxide. H Chen and W Ding interpreted the results and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors is greatly indebted to the National Natural Science Foundation of China for the financial support (Project No 51279073).
