Abstract
An ordinary silica gel (SG-1) was chosen as the raw material and hydrophobically modified with trimethylchlorosilane under different microwave exposure. The orthogonal experiment was designed to screen the optimized modification method. The hydrophobic properties modified silica gel (SG-2) was analyzed by comparing the difference changes of surface chemistries. Then, the adsorption capacities of SG-1 and SG-2 to the n-hexane vapor and water vapor, respectively, were investigated and compared with the capacities of activated carbon and SG-3 (hydrophobically modified using the same composite modifier without microwave exposure). Meanwhile, there cyclable adsorptions of SG-2 to the two vapors were conducted while the desorptions of vacuum and heat was observed. The results showed that the effect of hydrophobic modification to SG-1 under microwave exposure was obvious and SG-2 also had a very good effect of adsorption for the n-hexane vapor. The modified silica gel shows great potential and effect for the recovery of volatile organic compounds with high content of water.
Introduction
Recently, environmental protection has drawn increasing attentions because of the more deteriorating pollution problems, including solid waste, water resource pollution, and air contamination (Pandea et al., 2012). Therein, air pollution, which is closely related to the leakage and diffusion of volatile organic compounds (VOCs) during oil storage, transportation, and sale, becomes one of the reasons to induce some diseases. Adsorption technology has been widely adopted to recover VOCs because of some advantages of adsorption, such as easy operation, high adsorption efficiency, remarkable economic benefit, and wide application range (Swetha et al., 2017). Appropriate adsorbent should be first considered for the adsorption feasibility and be selected to improve the recovery efficiency of VOCs, and the ideal adsorbents should possess high adsorbing capacity, strong hydrophobicity, and high stability.
Compared with activated carbon (AC) (as a traditional adsorbent), silica gel is nonflammable, which endows a higher safety for the VOCs adsorption and avoids the spontaneous combustion as a result of high temperature (Blocki, 1993; Ushiki et al., 2015). However, silica gel has high hydrophilicity and is easy to adsorb the water vapor (WV), and then the rigid structure of silica gel is damaged easily because of the adsorption of WV. Meanwhile, these will also cause competitive adsorption between WV and VOCs and influence VOC’s recovery efficiency (Duan et al., 2016; Mahadik et al., 2011). Therefore, it is difficult to achieve the high efficient adsorption and separation of VOCs from the polluted air with high water content by the ordinary silica gel. So, the modification of silica gel is very necessary with high hydrophobicity, high VOCs adsorption capacity, and high thermal stability.
Preparation of hydrophobic silica gel with different methods attracted widely the researchers’ interests. Chung’s group showed that commercial silica particle was hydrophobically modified with octyltriethoxy-silane (OTES) in an aerosol spray reactor, and the conclusions were that the hydrophobicity was increased as increasing the concentration of OTES and had the optimum point with the reaction temperature (Chung et al., 2004). Pradip’s group applied acid–alkali two-step catalysis technology to prepare silica gel with sphere morphology. The resulting samples showed high hydrophobicity with the adsorbing capacity for oil vapor of 3.6 mL/g and WV of 0.12 mL/g (Sarawade et al., 2010a). And his group also reported that the hydrophobic mesoporous silica powder was obtained by surface modification of wet-gel slurry with trimethylchlorosilane (TMCS), and the results demonstrated that exploiting a low-cost silica source (sodium silicate) and spray-drying technique was suitable for economic and large-scale industrial production of silica powders (Sarawade et al., 2010b). Lee and Cha (2007) used sodium silicate, TMCS, and xylene solution as raw materials to fabricate functional silica gel, and the hydrophobicity of the resulting sample was enhanced significantly.
From the above reviews, hydrophobically modification with silane modifiers (such as TMCS) is the main method (Lee and Cha, 2007; Sarawade et al., 2010a, 2010b; Wang et al., 2014; Wu et al., 2016). Further, the preparation method of hydrophobic silica gel was improved in this study. In other words, an ordinary silica gel (SG-1) was chosen as the raw material and hydrophobically modified with a composite modifier which contains TMCS especially under microwave exposure, and then the adsorption performance of the modified silica gel to VOCs from the polluted air with high water content was evaluated.
Experimental
Materials
SG-1 was purchased from Qingdao Guichuang Company of China, AC was purchased from Fujian Xingsen Company of China and TMCS, ethyl alcohol, n-heptane, n-hexane, distilled water, etc. were purchased from Changzhou Runyou Company of China.
Modification
Different solutions were prepared as the composite modifiers, according to the volume ratio of n-heptane, ethyl alcohol, and TMCS of 12–14:1:1. SG-1 (8–10 g) was put into vacuum drying oven at 70–80℃ for 4–6 h and then was immerged into the modifier for 1 h at room temperature. Afterward, the mixture of the modifier and SG-1 was put into microwave oven under the exposure conditions which the microwave power was 500–700 W according to the designed oven temperature of 60℃ and the exposure time was 10–30 min. After another 10–12 h at room temperature for the mixture, the modified silica gel was filtered and washed several times from the mixture and was dried at 60–80℃ until its weight was invariable.
In the above-mentioned modification processes, the orthogonal experiments were designed to screen the optimized modification method.
Characterization
Scanning electron microscopy (SEM) images were recorded on a Philips XL30 electron microscope (Netherlands) operating at 15 kV. Fourier transform infrared (FT-IR) spectra images were collected on Nicolet Fourier spectrophotometer, using KBr pellets of the samples. Nitrogen sorption isotherms were measured at 77 K with Autosorb-IQ2-MP-XR-VP analyzer. The Brunauer–Emmett–Teller (BET) method was utilized to calculate the specific surface areas (SBET) using the adsorption data in a relative pressure range from 0.05 to 0.30. By the Barrett–Joyner–Halenda model, the pore volume and pore size distribution were derived from the adsorption branches of isotherms, and the total pore volumes (Vt) were estimated from the adsorbed amount at a relative pressure p/p0 of 0.95. The micropore volumes (Vm) and micropore surface areas (Sm) were calculated from the t-plot method, and the average pore diameter (D) calculation can be found in the Tabular Data portion of the software. The hydrophobic properties were analyzed by comparing the difference changes of surface chemistries which was measured by X-ray photoelectron spectroscopy (XPS).
Static adsorption and desorption
The n-hexane vapor (HV) was adopted as the adsorbate in the experiment, based on the component of VOCs from the polluted air. The samples (i.e. adsorbents) were put in a desiccator with n-hexane or distilled water under a constant temperature (20℃) water bath. The adsorption amounts of the samples to HV or WV were measured with an analytical balance. The desiccator was placed in a constant-temperature water container and equipped with an analytical balance. The weight of samples was tested every hour until the adsorption saturation. The desorption procedure was conducted by heating the samples to 80℃ in vacuum drying oven under the vacuum of 0.095 MPa until the mass of the samples did not change any more. According to the mass change of the samples before and after the adsorption and desorption procedure, the adsorption capacity and desorption percentage of the samples could be calculated as follows:
Dynamic adsorption and desorption of HV
An experiment unit for the dynamic adsorption–desorption measure was designed by Huang et al. (2015). Before the experiment, the samples need to be pretreated by heating to 80℃ in vacuum drying oven under the vacuum of 0.095 MPa for 2 h to remove the water molecules and small organic impurities adsorbed in the pores of the samples. The adsorbent was then filled into the adsorption tower with adsorption bed hight of 24 cm, and two temperature sensors of AT4320 temperature auto-recorder were put into the center of the adsorbent at the distances of 16 and 8 cm from the adsorbent top, respectively (The uniform distribution of temperature sensors makes it easy to study to study the adsorption heat of different positions). The HV concentrations in the inlet and outlet of the adsorption tower were monitored by GC-2010 plus gas chromatograph. In the experiment process, nitrogen was taken as a carrier gas for HV and adjusted to keep the flow rate at 2(±0.2) L/min, and the HV concentration was controlled at 0.8 g/L. After the flow rate and concentration of the mixed gas were steady, the mixed gas was loaded into adsorption tower and the adsorption procedure of HV was carried on. The adsorption experiment was finally stopped when the HV concentration in the outlet was almost equal to the inlet concentration and become steady, and then the desorption step for the adsorption tower was continued with vacuum pump for 2 h at the vacuum pressure of 0.09 MPa.
Furthermore, the dynamic adsorption breakthrough curves of HV can be simulated by Logistic model of Origin based on the experiment data. The equations of Logistic and the saturated adsorption capacity (q, g/g) of the adsorbent are as follows (Wang et al., 2015; Zhang et al., 2017):
Results and discussions
Orthogonal experiment
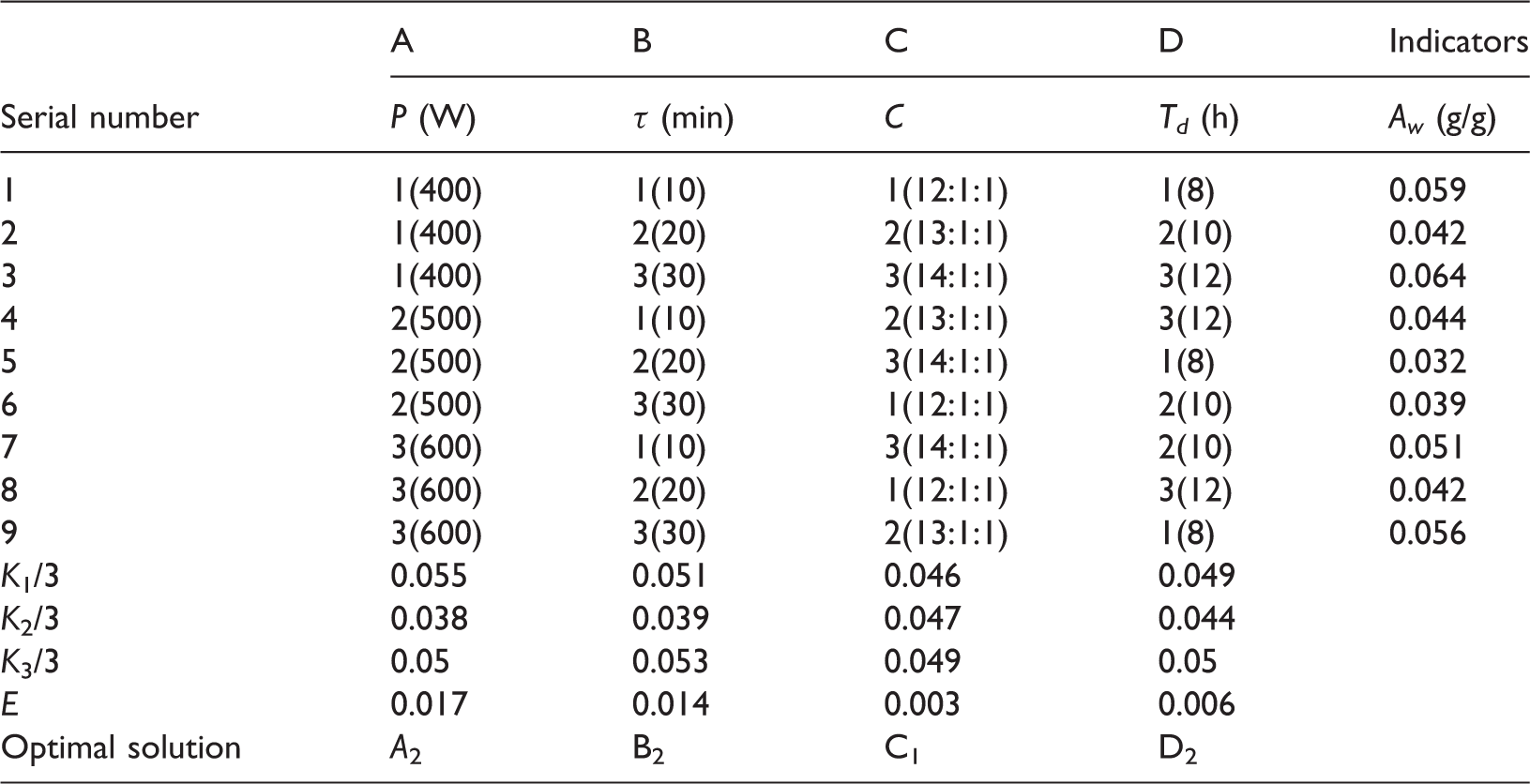
Table 1 shows the arrangement and results of the orthogonal experiment. The orthogonal experiment with four factors and three levels was designed and carried out to obtain the optimized modification factors; the four factors were the power (P) and time (τ) of microwave exposure, the volume ratio (C) of n-heptane, ethyl alcohol, and TMCS, and the standing time (Td) of the mixture after microwave exposure; the three levels represented that there were three modified conditions under each of the factors. The static adsorption capacity for WV (Aw) was the indicator of screening (where E represents the Range, i represents the number of the levels, Ki represents the sum of Aw at the i level for the certain factor).
The arrangement and results of orthogonal experiment.
Morphology analysis
SEM images of AC, SG-1, SG-2, and SG-3 are shown in Figure 1. Compared with rough surface of AC and SG-2, the surface of SG-1and SG-3 is relatively smooth. And some clusters can be seen on the surface of SG-2, which may reduce BET area of the samples (more details will be discussed in the following). After modifying microwave assisted with TMCS technology, the surface of SG-2 became rough and the arrangement of surface structure was changed tightly, and the presence of interval between granules could be observed clearly which is fit for adsorbing VOCs. However, the average pore size of the resulting sample SG-2 decreased slightly, which manifests that the additional microwave measurement makes the modifying effect of TMCS evenly.
SEM images of AC, SG-1, SG-2, and SG-3. AC: activated carbon; SEM: scanning electron microscopy; SG: silica gel.
FT-IR analysis
The infrared patterns of SG-1 and SG-2 are shown in Figure 2. The signals located at 3448 and 1654 cm−1 are assigned to antisymmetry vibration of Si–OH (Horváth et al., 2005). The corresponding intensity weakened significantly for the SG-2 compared with that of SG-1, indicating the removal of –OH by the additional treatment (Kokunesoski et al., 2010). After modifying to SG-1, new signs at 1096 and 468 cm−1 which were caused by Si–O–Si antisymmetric stretching vibration can be seen. Moreover, Si–O stretching vibration (802 cm−1) and C–C vibration peak (968 cm−1) appeared (Ghoshal and Manjare, 2003; Sarawade et al., 2011, 2013), manifesting the enhanced hydrophobicity of SG-2.
FT-IR patterns of SG-1 and SG-2. FT-IR: Fourier transform infrared; SG: silica gel.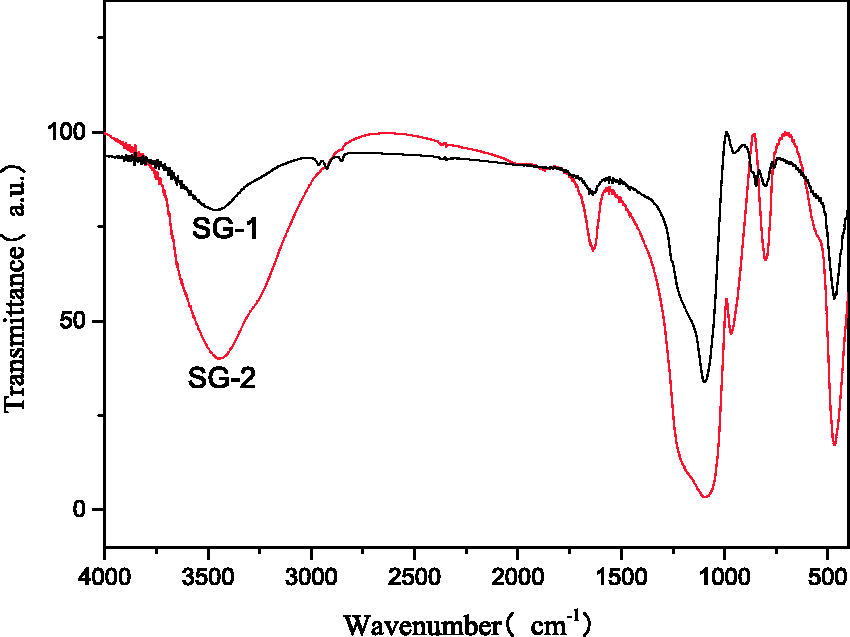
Micro-morphology
Surface structure parameters of adsorbents.

AC: activated carbon; BET: Brunauer–Emmett–Teller; SG: silica gel.
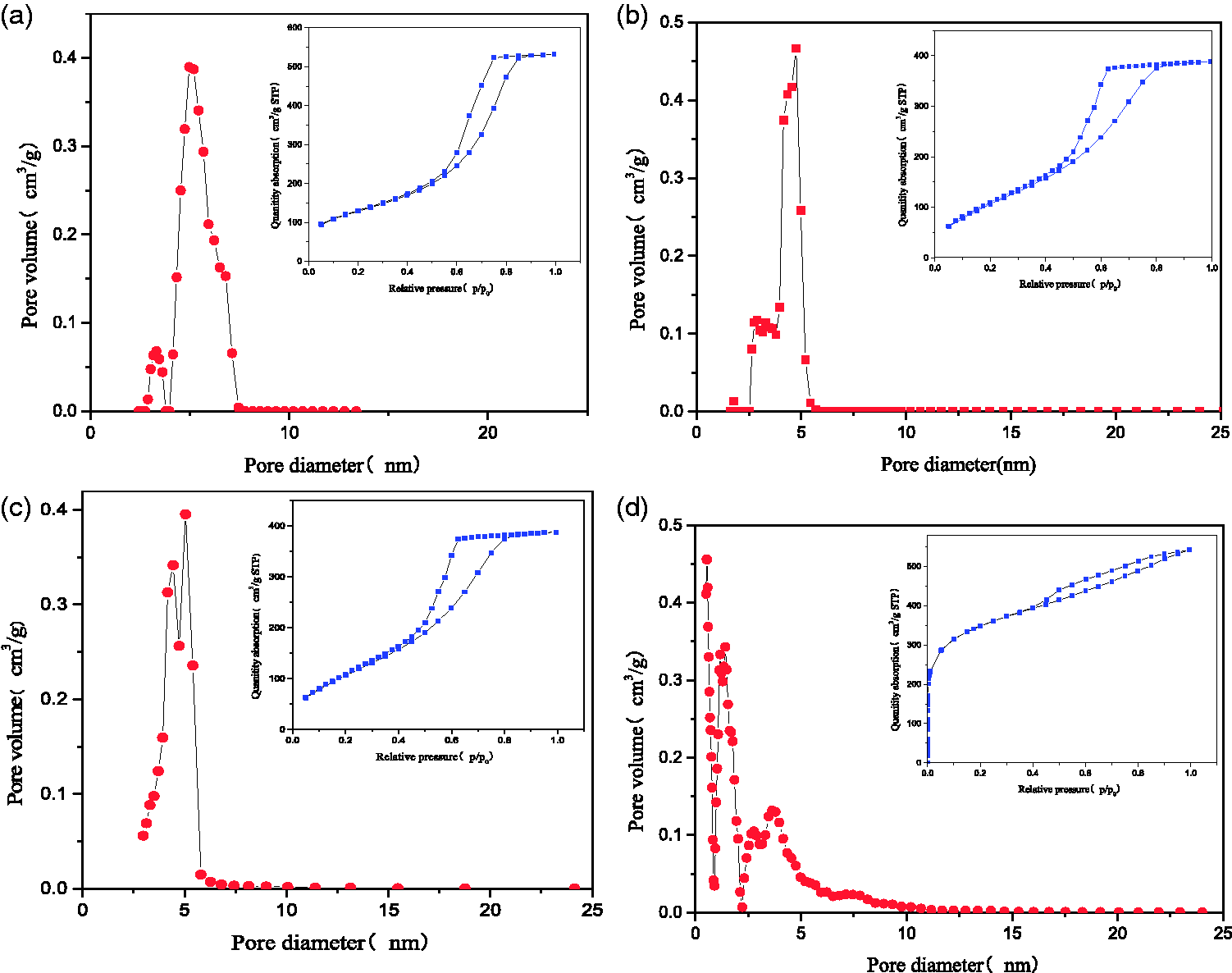
Pore size distribution curve and N2 adsorption–desorption isotherms of adsorbents. (a) SG-1, (b) SG-2, (c) SG-3, and (d) AC. AC: activated carbon; SG: silica gel.
Adsorption properties
Hydrophobicity properties
Figure 4 shows the photos of SG-1 and SG-2 in the distilled water, and the obvious diversity could be seen. Herein, the bulk densities of SG-1 and SG-2 are 0.73 and 0.65 g/cm3, respectively. SG-1 samples could sink to the bottom in a relatively short time because of the poor hydrophobicity, while SG-2 samples could float for a long time, and only few SG-2 samples sink to the bottom 2 h later, indicating the better hydrophobicity of the SG-2.
Hydrophobic property of SG-1 and SG-2. SG: silica gel.
Static adsorption properties
According to equations (1) and (2), the sample SG-2 were selected to estimate the static adsorption–desorption properties for WV and HV, comparing with SG-1, AC, and SG-3.The static adsorption curves for WV and HV by SG-1, SG-2, SG-3, and AC are presented in Figure 5.
Adsorption curve for HV and WV at 20℃. For HV: (a) AC, (b) SG-1, (c) SG-2, (d) SG-3; for WV: (e) AC, (f) SG-1, (g) SG-3, (h) SG-2. AC: activated carbon; HV: n-hexane vapor; SG: silica gel; WV: water vapor.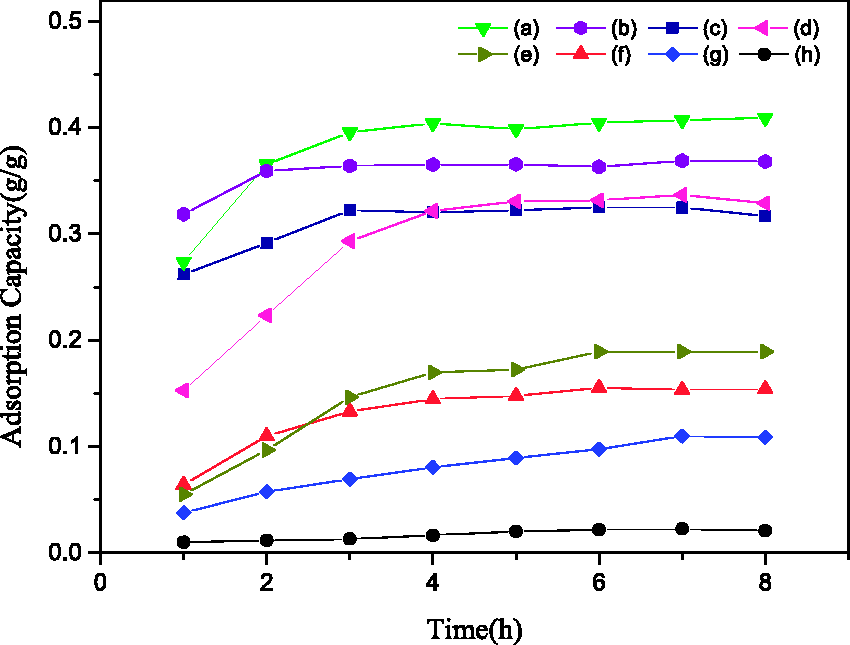
The WV adsorption curve of SG-2 was almost a horizontal line, which presented that SG-2 reached the saturated state in the shortest time, and with the increase of adsorption time, the adsorption capacity for WV also remained a stable state owing to the high hydrophobicity. The saturated adsorption capacity for WV by SG-2 was 0.022 g/g, which was much less than that of SG-1 or AC (0.154 or 0.189 g/g, respectively). Meanwhile, the saturated adsorption capacity of SG-2 for WV was also less than that of SG-3 (0.109 g/g), indicating that the modification effect is better under microwave exposure. Moreover, the adsorption capacity for HV by SG-1, SG-2, SG-3, and AC were 0.369, 0.322, 0.329, and 0.409 g/g at 20℃, respectively. Thus, SG-2 showed the better hydrophobicity and higher adsorption capacity for HV.
To seek out the reasons of high hydrophobicity of SG-2, the C, O, and Si elements of SG-1 and SG-2 were tested by the instrument of XPS, and the results are shown in Figure 6. The peak position of C element in SG-1 signed at 284.11, 285.86, and 287.85 eV, and the signs of peak position represented zero-valent carbon element, carboxyl and hydroxyl groups of carbon in SG-1, respectively. After modifying, the signs of carboxyl and hydroxyl groups of carbon in SG-2 were almost disappeared, but the proportion of zero-valent carbon element was significantly improved. Meanwhile, the peak shift of O and Si element were around 0.22 and 0.23 eV, respectively, and the results could be considered as reasons for the instrument error. From the above analysis results, the functional groups of O and Si element in SG-2 had no change, comparing with the functional groups of O and Si element in SG-1 (Demchenko et al., 2017; Lakshmi et al., 2017). In addition, the primary cause of hydrophobicity was determined that the hydroxyl and carboxyl of carbon atoms had been removed after modification.
XPS images of C, O, and Si element in SG-1 and SG-2. SG: silica gel; XPS: X-ray photoelectron spectroscopy.
Reutilization performance
The static reutilization performance of SG-2 and AC was tested and the corresponding results are shown in Figure 7. After seven times cycles, the adsorption amount of SG-2 and AC for HV decreased from 0.322 g/g and 0.409 to 0.302 g/g and 0.361 g/g, respectively, i.e. the seventh adsorption amount of SG-2 was about 93% of the first adsorption amount. Meanwhile, the adsorption capacity of SG-2 for WV increased from 0.022 to 0.037 g/g. The adsorption capacity of AC for WV increased from 0.189 to 0.224 g/g. From the results of cyclic adsorption experiment, there was no obvious increase or decrease in adsorption capacity of SG-2 after seven cycles.
Static curves for reutilization performance of SG-2 and AC. For HV: (a) AC, (b) SG-2; for WV: (c) AC, (d) SG-2. AC: activated carbon; HV: n-hexane vapor; SG: silica gel; WV: water vapor.
The dynamic reutilization performance of SG-2 and AC was also tested; the corresponding results in Figure 8 showed that there was also no obvious increase or decrease in adsorption capacity of SG-2 after seven cycles. The results of static and dynamic adsorption capacities for HV and WV after seven cycles indicated that the performance of SG-2 was stable for the repeated uses, and the resulting adsorbents could possess a long life time for the VOCs recovery.
Dynamic curves for reutilization performance of SG-2 and AC. For HV: (a) AC, (b) SG-2; for WV: (c) AC, (d) SG-2. AC: activated carbon; HV: n-hexane vapor; SG: silica gel; WV: water vapor.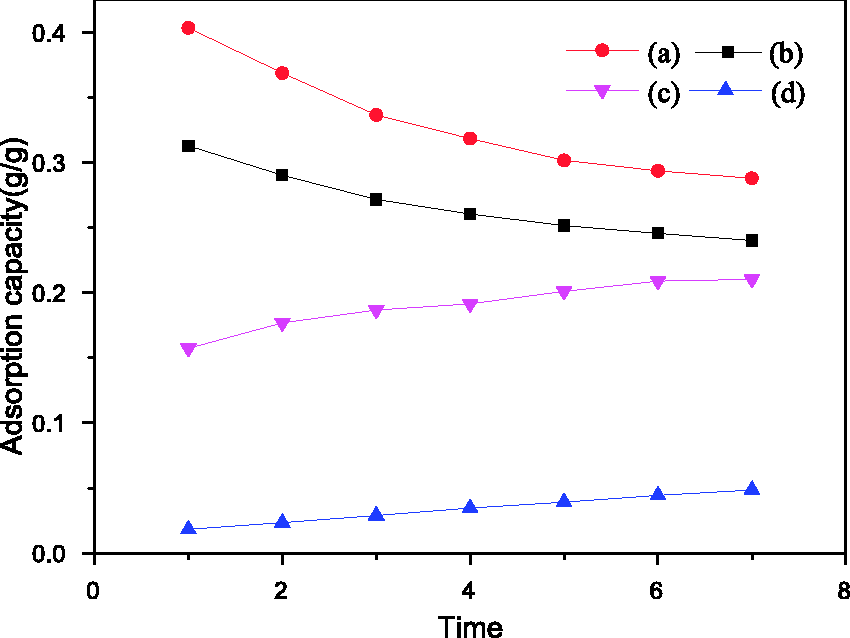
Dynamic adsorption properties
Dynamic adsorption performance is very important for various industrial applications of VOCs adsorption. The breakthrough curves of SG-1, SG-2, and AC for HV are shown in Figure 9. Therein, the solid lines are the breakthrough fitting curves by the Logistic model; the points are the experimental results.
Adsorption breakthrough curves of HV by SG-1, SG-2, and AC. AC: activated carbon; HV: n-hexane vapor; SG: silica gel. Adsorption heat in the process of SG-2 and AC adsorbs HV. AC: activated carbon; HV: n-hexane vapor; SG: silica gel. Desorption heat of SG-2 and AC for HV. AC: activated carbon; HV: n-hexane vapor; SG: silica gel.


Fitting parameters of Logistic model.

AC: activated carbon; SG: silica gel.
Adsorption–desorption heat
Nitrogen was taken as a carrier gas for HV and adjusted to keep a mean flow rate of 2 L/min, and the concentration of HV was controlled at 0.8 g/L at room temperature and the results of the adsorption heat are shown in Figure 10. Under the steady conditions, the maximum adsorbent temperature variation for WV was 15.4℃ at 16 cm for AC, while the adsorbent temperature variation decreased to 7.2℃ for SG-2. Meanwhile, the maximum adsorbent temperature variation of AC and SG-2 at 16 cm was slightly higher than that of at 8 cm owing to more amount of HV was gathered in the bottom of adsorption tower. Moreover, the adsorption curve showed as drum form, which was closely to the adsorption amount (higher adsorption amount and slower temperature drop bring about the drum shape), manifesting the high adsorption amount of the SG-2.
The adsorption experiment was finally stopped when the HV concentration in the outlet was almost equal to the inlet concentration and become steady, and then the desorption step for the adsorption tower was continued with vacuum pump for 2 h at the vacuum pressure of 0.09 MPa. The results of the desorption heat are shown in Figure 11.
During the desorption process, the maximum adsorbent temperature variation was −14.1℃ for AC, and the value was −6.7℃ for SG-2. Thus, compared with that of AC, SG-2 displayed higher thermal stability, which brought about the better safety when it is adopted for the VOC’s recovery application.
Conclusions
An ordinary SG-1 was chosen as the raw material and hydrophobically modified with TMCS under different microwave exposure, the n-heptane and ethanol are the solvents of TMCS. The orthogonal experiment was designed to screen the optimized modification method. The physical properties and hydrophobic properties of the modified silica gel (SG-2) were analyzed by various analytical instruments; some conclusions can be drawn as follows:
SG-2 can be optimally prepared in the conditions of 500 W (the microwave exposure power), 20 min (the microwave exposure power time), 12:1:1 (the volume ratio of n-heptane, ethyl alcohol, and TMCS), and 10 h (the standing time of the mixture after microwave exposure). Compared with SG-1, the surface of SG-2 became rough, and the average pore size of SG-2 increased from 5.57 to 7.13 nm. Moreover, SG-2 possessed better hydrophobicity and lipophilicity because of reduced hydroxyl and carboxyl of carbon atoms on its surface. The results of static adsorption experiments showed that the saturated adsorption amount for WV by SG-2 was 0.022 g/g, which was much less than that of SG-1 or AC (0.154 or 0.189 g/g, respectively). Meanwhile, the saturated adsorption capacity of SG-2 for WV was also less than that of SG-3 (0.109 g/g), indicating that the modification effect is better under microwave exposure. Then, after seven times adsorption cycles, the adsorption amounts of SG-2 for HV decreased from 0.322 to 0.302 g/g, i.e. the seventh adsorption amount was about 93% of the first adsorption amount. Meanwhile, the adsorption amount for WV increased from 0.022 to 0.037 g/g. Thus, SG-2 showed the better hydrophobicity and more stable for the repeated uses. The dynamic adsorption of SG-2 for HV was tested and the saturated adsorption amounts of SG-2, SG-1, and AC for HV were calculated as 0.31, 0.36, and 0.40 g/g, respectively. The breakthrough curve of SG-2 for HV can be fitted better with the Logistic model of Origin software. The thermal effects of the adsorption and desorption of SG-2 and AC for HV were measured and the maximum temperature variation were 7.1 and 15.4℃ for the adsorption and −6.7 and −14.1℃ for the desorption, respectively. So, the modified silica gel will bring about the better safety for the industrial applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial supports from the National Natural Science Foundation of China (No. 51574044), the Qing Lan Project of Jiangsu Province (No. SCZ1409700002), the Special Foundation of Sci-Tech achievements Transformation of Jiangsu Province (No. BA2015166), and the Sci-Tech Support Plan of Changzhou City (NO. CE20150085) are gratefully acknowledged.




