Abstract
The adsorptive removal of Ni2+ and Cd2+ at concentrations of approximately 50 mg L−1 in wastewater is investigated using an agricultural adsorbent, longan hull, and the adsorptive mechanism is characterized. The maximum adsorption capacity of approximately 4.19 mg g−1 Cd2+ was obtained under the optimized conditions of room temperature, pH 5.0, and a solid-to-liquid ratio of 1:30 in approximately 15 min. For Ni2+, the maximum adsorption capacity of approximately 3.96 mg g−1 was obtained at pH 4.7 in approximately 20 min. The adsorption kinetics for both metal ions on the longan hull can be described by a pseudo second-order rate model and are well fitted to the Langmuir adsorption isotherm. The adsorption mechanism of the longan hull to Ni2+ and Cd2+ ions is shown to be a monolayer adsorption of metal ions onto the absorbent surface. Thereinto, the longan hull adsorbent contains N–H, C–H, C=O, and C=C functional groups that can form ligands when loaded with Ni2+ and Cd2+, which reduces the fluorescence of the dried longan hull material.
Introduction
Pollution caused by metal ions in wastewater is a common problem faced by many countries in recent years. Ni2+ and Cd2+ ions in wastewater, which mainly originate from mining and their application in various production activities, have been especially recognized as key contaminants that damage the environment and adversely affect human health. Possible methods for the removal of Ni2+ and Cd2+ from wastewater include chemical precipitation, filtration, coagulation, solvent extraction, electrolysis, ion exchange, and adsorption techniques (Raval et al., 2016; Tan et al., 2015; Zhang et al., 2016). Adsorption techniques have been rapidly gaining prominence as an economical and effective treatment process for wastewater containing Ni2+ or Cd2+, and a number of adsorbents, such as activated carbon, industrial waste, and agricultural products have been used for the removal of Ni2+ and Cd2+ in wastewaters.
Compared with activated carbon, agricultural adsorbents are often superior in terms of adsorptive capacity and cost. Saced et al. (2005) reported that black gram husk could remove heavy metal ions at 1000 mg L−1 concentration efficiently from aqueous solutions with a selectivity order of Pb2+ > Cd2+ > Zn2+ > Cu2+ > Ni2+. The maximum amount of heavy metals adsorbed at equilibrium was 49.97, 39.99, 33.81, 25.73, and 19.56 mg g−1, respectively, using black gram husk biomass, and the biosorption equilibriums were described well by Langmuir and Freundlich adsorption isotherms. Benaissa (2006) reported a content of adsorbed Cd2+ of 1000 mg L−1 using pea, broad bean, and medlar peels and fig leaves, yielding maximum adsorption capacities of approximately 147.71 mg g−1 for broad bean peel, 118.91 mg g−1 for pea peel, 103.09 mg g−1 for fig leaves, and 98.14 mg g−1 for medlar peel. Martinez et al. (2006) reported a maximum adsorption capacity for grape stalk of 0.241 mmol g−1 and 0.248 mmol g−1 for Pb2+ and Cd2+, respectively, in the batch experiments at an initial pH of approximately 5.5. Pb2+ could be desorbed from the grape stalks completely with HCl or EDTA solution, while an approximate 65% desorption yield was obtained for Cd2+. Chand et al. (2015) introduced xanthate groups onto apple pomace for the removal of Cd(II), Ni(II), and Pb(II) from aqueous solutions; the maximum adsorption capacities were 112, 51, and 178.57 mg g−1 for Cd(II), Ni(II), and Pb(II), respectively. Garba et al. (2016) investigated the adsorption of Ni(II) and Cd(II) ions from aqueous solution onto modified plantain peel. Adsorption capacities of 77.52 and 70.92 mg g−1 for Ni(II) and Cd(II), respectively, were obtained under the optimized adsorption conditions. Similar results were also reported when apple residues (Lee et al., 1998), wheat bran (Farajzadeh and Monji, 2004), rice husk ash (Srivastava et al., 2006), and maize straw (Guo et al., 2015) were used as adsorbents, showing the potential of agricultural adsorbents for the adsorptive removal of Ni2+ and Cd2+ in wastewater.
Although the complexity of the composition of agricultural adsorbents has presented a challenge, the sorption of metal ions has usually been attributed to the presence of carboxyl, hydroxyl, sulfate, phosphate, and amino groups on these adsorbents. It is obvious that some empirical or deductive factors are involved in these attributions. Moreover, because the adsorptive capacity of adsorbents is closely related with the surface composition of the adsorbent and because in practice, plants regularly come into contact with wastewater containing metal ions, it is essential to understand the impact of the adsorption of metal ions on the plants, not only for the identification of suitable adsorbents but also for the evaluation of the potential of metal ions to harm agricultural plants. In this study, an agricultural adsorbent, i.e. longan, grown largely in subtropical regions, was used to remove Ni2+ and Cd2+ from wastewater, and the adsorption conditions and kinetics of Ni2+ and Cd2+ on longan hull powders, as well as the reaction mechanism, were investigated.
Experimental
Materials and chemicals
Longan, grown in the Pearl River Delta of South China, was used as the absorbent. After removing its pulp, the longan hull was washed with distilled water, dried under vacuum condition at 80℃ for over 48 h, mechanically crushed to particles and sieved through a 100 mesh sifter. The simulated Ni2+ and Cd2+ wastewater, with each ion at 50 mg L−1, was prepared by dissolving Cd(NO3)2•4H2O and NiSO4•6H2O (analytical reagents) in distilled water. The solution pH was adjusted by adding 0.1 mol L−1 HNO3 or 0.1 mol L−1 NaOH.
Adsorption procedure
It is well known that the adsorption process is mainly related with the reaction time, solution pH, solid-to-liquid ratio (ratio of the weight of the solid adsorbent to the volume of liquid solution or S/L ratio), solution concentration, and temperature. Based on their importance in the adsorption process, these four parameters were optimized. During optimization, one parameter was varied, while the other parameters were held constant. The temperature and solution volume were kept at 25 ± 1℃ and 30 ml, respectively. To determine the individual adsorption characteristics of Ni2+ and Cd2+, their adsorption–desorption processes were individually investigated. The adsorption experiments were carried out in glass beakers with magnetic stirring at the set temperature. The absorbent was removed from the solution by centrifugation after the adsorption treatment. All adsorption experiments were carried out in triplicate, and the results were recorded as the average values.
Analysis methods
The concentrations Ni2+ and Cd2+ in the initial and final solutions were all determined by using atomic absorption spectrophotometer (Model AA320, ReDian, USA). The pH values of the aqueous solutions were measured with a pH/mV meter (Model pHSJ-3F, LeiCi, China). The morphologies of the dried Longan hull powders were characterized using scanning electron microscopy (SEM, ZEISS Ultra 55). Fourier Transform Infrared (FTIR) Spectrophotometer (Model Prestige-21, DaoJin, Japan) was used to characterize the main functional groups that might be involved in the adsorption process. The fluorescence of the dried Longan hull with Ni2+ and Cd2+ was performed with the Fluorescence Microscope (Model LV-LH50PC, Nikon, Japan).
Computation of adsorption capacity
The metal ion concentrations adsorbed on Longan hull particles were calculated from the difference between the initial C0 and final Ce ion concentrations in the solution. The following equations (Martinez et al., 2006) were used to compute the adsorption ratio %S and the adsorption capacity (q) of the adsorbent, respectively:
Results and discussion
Adsorption conditions
The adsorption parameters were optimized by changing one parameter while the other parameters remained fixed and the temperature maintained at room temperature (25 ± 1℃). First, the reaction time was investigated using the prepared solution with constant pH. The pH of the cadmium and nickel solutions were monitored with the pH/mV meter and were held constant at approximately 6.0. Figure 1 shows the effect of reaction time on the adsorption capacity for cadmium and nickel ions using the dried longan hull as an adsorbent. The initial concentrations of Ni2+ and Cd2+ were 50 mg L−1, the absorbent quantity was 0.5 g, and the initial pH of the solution was 6.0.
Effect of reaction time on the adsorption capacity of Ni2+ and Cd2+ using the longan hull adsorbent.
From Figure 1, it can be seen that with of the addition of adsorbent at constant pH, over time, more cadmium ions could be removed from the solution compared to nickel ions, with maximum adsorption achieved at approximately 20 min and 15 min, respectively. The adsorption equilibrium of Ni2+ and Cd2+ was reached faster using the longan hull adsorbent than when using the peels of peas or broad beans or fig leaves (Benaissa, 2006). The increase in the adsorption capacity at the initial stage was attributed to the adsorption of Ni2+ and Cd2+ at the vacant surface sites on the longan hull adsorbent. The small decrease in adsorption capacity observed after the maximum adsorption was reached may have been due to the weak force between the adsorbent and metal ions, resulting in the desorption of some adsorbed metal ions during the magnetic stirring process.
The effect of pH on the removal of Ni2+ and Cd2+ using the dried longan hull is shown in Figure 2. The initial concentrations of Ni2+ and Cd2+ were 50 mg L−1, and the absorbent quantity was 0.5 g. The adsorption capacity of Ni2+ and Cd2+ increased in the pH range from 1 to 5, and the maximum adsorption of both metal ions occurred at pH 4.5–5.5. When the pH was increased above 7.0, the cadmium and nickel hydroxides became insoluble, and the adsorption capacity of Ni2+ and Cd2+ decreased dramatically. The adsorption capabilities of Ni2+ and Cd2+ at pH 1–5.5 can be explained by the fact that the metal ions compete with hydrogen ions for the binding sites on the surface of the dried longan hull. In addition, when the pH increases, there is an increase in the negative surface charge, which results in a lower electrostatic repulsion between the positively charged metal ions and the surface of the longan hull, which favors the adsorption process. From the data presented in Figure 2, it is clear that no adjustment was needed for the pH of Ni2+ and Cd2+ solutions used in the subsequent experiments with the dried longan hull adsorbents. In addition, although there was a small difference in the adsorption, the adsorptive removal of Ni2+ and Cd2+ can be carried out in the pH range of 3.5–6.
Effect of pH on the adsorption capacity of Ni2+ and Cd2+ using the longan hull adsorbent.
The effect of adsorbent quantity on the adsorption ratio of Ni2+ and Cd2+ with the dried longan shell is shown in Figure 3. The initial concentrations of Ni2+ and Cd2+ were 50 mg L−1. When the adsorbent quantity was increased to approximately 1.0 g, the adsorption ratio of Ni2+ and Cd2+ was enhanced to 90% and 93%, respectively, suggesting an optimized solid-to-liquid (S/L) ratio of 1:30 for a solution with 50 mg L−1 concentration. In particular, a higher adsorption ratio was achieved for Cd2+ than for Ni2+ when the S/L ratio was more than 1:60. This shows that a greater mass of Cd2+ can be removed when the quantity of the adsorbent is increased, while there were no visible effects for the Ni2+ solution. In further experiments, a solution with a concentration of 0.7 mg L−1 Cd2+ was obtained after two repeated adsorption treatments, reflecting the potential applied value of the longan hull in the treatment of wastewater that contains Cd2+. In contrast, for Ni2+ solutions, it was difficult to achieve Ni2+ concentrations less than 1.65 mg L−1, suggesting a lower efficiency of longan hull in the treatment of Ni2+ solutions compared to Cd2+. These results are consistent with adsorption experiments using black gram husk as the adsorbent (Saced et al., 2005).
Effect of adsorbent quantity on the adsorption ratio of Ni2+ and Cd2+ using the longan hull adsorbent.
The effect of the initial Ni2+ and Cd2+ concentrations on the adsorption capacity of dried longan hull is shown in Figure 4. The adsorbent quantity used was 0.5 g. It can be seen that the amount of adsorbed ions increased with the increase in the initial concentrations of Ni2+ and Cd2+. The increase in the initial concentrations of the metal ions provided the necessary driving force to overcome the resistance to the mass transfer of Ni2+ and Cd2+ between the aqueous and solid phases, and the interaction between the metal ions and the longan hull was also enhanced. This suggests that the initial ion concentrations are a key factor related with the adsorption capacity of longan hull adsorbent. Moreover, the difference in adsorptive capacity between the longan hull and the other agricultural adsorbents, such as the peels of peas, broad beans, and medlar and fig leaves can be ascribed to differences in the concentrations of metal ions in the wastewater (Martinez et al., 2006). It was also determined that the adsorbed Ni2+ and Cd2+ can be removed from the adsorbents using the following process: submerge the adsorbents with adsorbed metal ions in 1 mol L−1 HCL with stirring for 3 h and then wash with distilled water until a constant pH is achieved.
Effect of the initial Ni2+ and Cd2+ concentrations on their adsorption capacity using the longan hull adsorbent.
Adsorption model
The adsorption kinetics of Cd2+ on rice husk has been investigated by Kumar and Bandyopadhyay (2006), and a pseudo second-order kinetic model was reported as follows:
Plots of Ni2+ and Cd2+ on the longan hull adsorbent using the pseudo second-order kinetic model based on the data presented in Figure 1.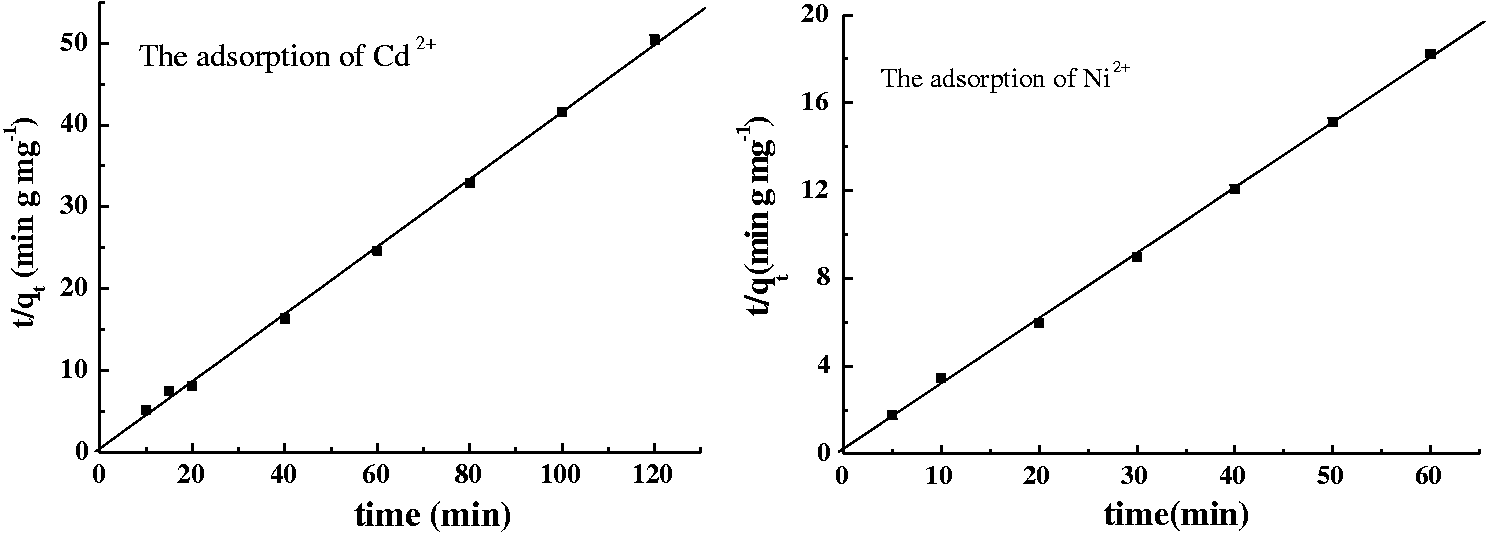
It can be seen that the above model adequately describes the kinetics of Ni2+ and Cd2+ adsorption with high correlation coefficients (R2 > 0.99), suggesting that the adsorption of Ni2+ and Cd2+ on longan hull can be described well by the pseudo second-order rate model. These experiments demonstrate the connection between the final concentration of metal ions and the adsorption capacity of adsorbents.
To further illustrate the adsorption parameters of Ni2+ and Cd2+ on longan hull adsorbent, their Langmuir and Freundlich isotherms are presented in Figures 6 and 7. The following formulae, (4) and (5), represent the Langmuir and Freundlich equations, respectively (Huang et al., 1991).
Plots of the Langmuir isotherm for the adsorption of Ni2+ and Cd2+ on longan hull adsorbent. Plots of the Freundlich isotherm for the adsorption of Ni2+ and Cd2+ on longan hull adsorbent. The adsorption parameters of Ni2+ and Cd2+ on longan hull adsorbent.


It is known that the Langmuir model is valid for monolayer adsorption onto a surface containing a finite number of identical sorption sites, while the Freundlich model is purely empirical and is based on the sorption of metal ions on a heterogeneous surface. By comparing the correlation coefficients obtained, it appears that the Langmuir model provides the best correlation for the adsorption of Ni2+ and Cd2+ on longan hull adsorbents. From the values of qm obtained with the Langmuir model, the higher adsorptive capability on longan hull for Cd2+ compared to Ni2+ is similar to the results obtained using grape stalk (Saced et al., 2005), although the comparison is not strict due to the varying experimental conditions used in the different studies. In addition, this association can also be primarily validated using a glancing collision calculation based on the specific area of longan hull adsorbent and the lower ion concentration used in the experiments.
Adsorption mechanism
The morphologies of longan hull powder were characterized by SEM to illustrate the surface structure. As shown in Figure 8(a), the powder generally has a rough appearance with sheet-like nanostructures. These nanosheets have a smooth surface layer composed of micropores. In other experiments, the Brunauer–Emmett–Teller surface areas and average pore volume were measured by adsorption–desorption isotherms. The results showed that longan hull powders exhibit a specific area of only 1.6 m2 g−1 and do not exhibit abundant nanopores or mesopores, as do most well-known adsorbents (Srivastava et al., 2006; Zhang et al., 2016). Therefore, it is understandable why longan hull exhibits a lower adsorption capacity for the removal of Ni2+ and Cd2+ from wastewater compared to other agricultural adsorbents. That is, the low specific area and lack of nanopores and mesopores are likely the main causes of the low adsorption capacity. Based on the observed particle appearance and elemental composition, it is possible that the specific area and adsorption capacity of longan hull powders could be enhanced by carbonization, which has been mostly validated.
(a) SEM of longan hull powders and (b) FT-IR spectra of longan hull powders (a) loaded with Cd2+, (b) loaded with Ni2+, (c) treated with HCl, and (d) no treatment.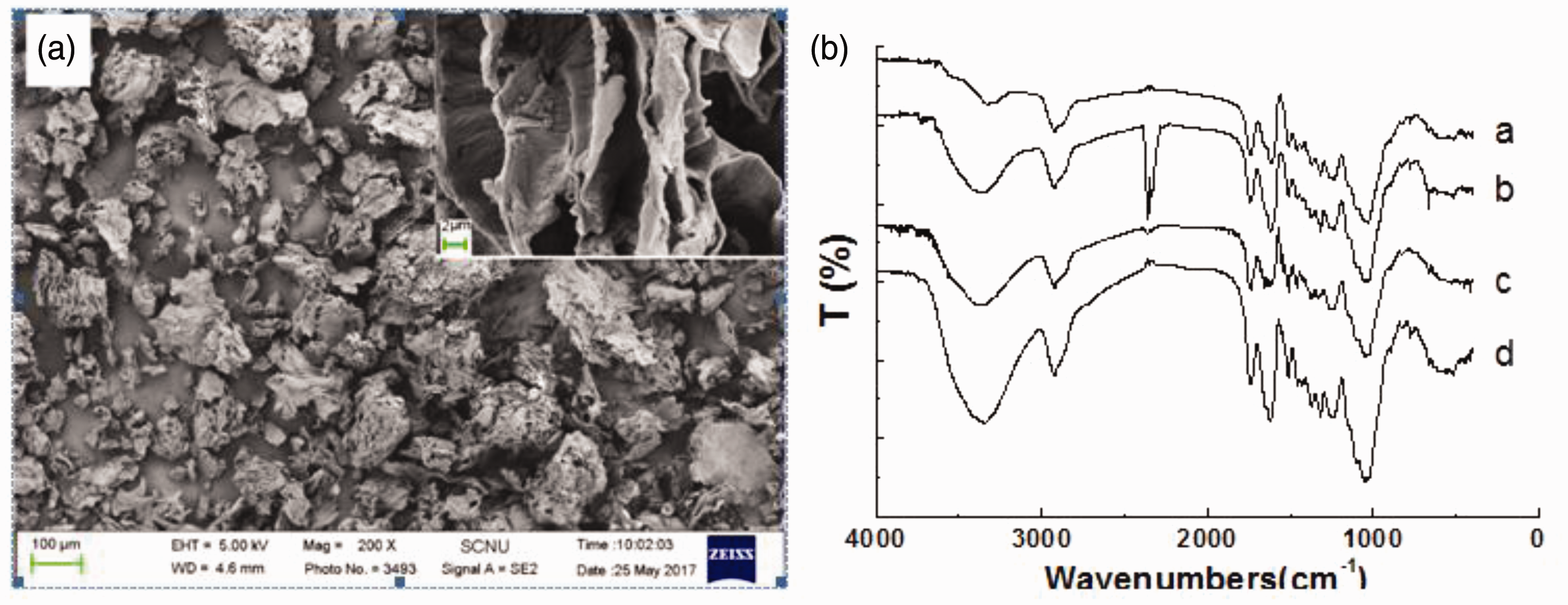
It is generally considered that the adsorption of metal ions by agricultural adsorbents that have carboxyl, hydroxyl, sulfate, phosphate, and amino functional groups in their protein, carbohydrate, and phenolic compounds might be attributed to these binding and adsorbing functional groups (Al-Asheh and Duvnjak, 1998; Meunier et al., 2003; Mohan et al., 2006). However, as indicated above, little evidence exists to elucidate the adsorption mechanisms. Consequently, several spectral techniques were used to characterize the functional groups in the longan hull powders. The FT-IR spectra of fine longan hull powders under different conditions are shown in Figure 8(b).
The characteristic adsorption peaks at 3592 cm−1, 2924 cm−1, 1745 cm−1, 1622 cm−1, and 1508 cm−1 related to the vibration or stretching of the N–H, C–H, C=O, and C=C chemical bonds could be detected in the IR spectra for the different longan hull powders. In addition, some differences between the dried longan hull powders and the treated samples were also found. The region between 1610 and 1500 cm−1 is associated with C=C stretching in aromatic rings. The peaks appearing at 1622 and 1508 cm−1 can be attributed to the vibration of the dried longan hull powders, while a shift to lower frequencies, i.e. 1620 cm−1, occurred when the longan hull was loaded with Cd2+ and Ni2+ and treated with acid. The peak at 3592 cm−1 is associated with N–H groups. A small decrease in its intensity was observed after treatment with Ni2+ and acidic solution, and a large decrease was observed after treatment with Cd2+ solution. The stronger binding force of Cd2+ with hydroxyl groups may have led to this difference. Carboxyl and hydroxyl groups on the longan hull powders serve as the adsorption sites for metal ions, at which organic-metal ligands can be formed, and this adsorption integrates the metal ions.
Although no visible differences could be detected in the SEM images, there were distinct changes in the fluorescence images of the longan hull powders after the adsorption of Ni2+ and Cd2+. Figure 9 shows the fluorescence images. Obviously, the dried longan shell is an intensely fluorescent material. It is suggested that the protein composing the cell wall of dried longan shell produces the fluorescence, which is consistent with the FT-IR results. The intensity of fluorescence decreased after the adsorption of Cd2+ and Ni2+ via their functional groups and decreased slightly more after treatment with HCl. All proteins contain tryptophan and tyrosine, which are the main fluorescent groups in proteins, but treatments such as heat, acid, base, and heavy metal salt can obviously change the fluorescence intensity of a protein. This is because acids, such as HCl, and heavy metal salts containing Hg2+, Pb2+, Ag+, Ni2+, Cd2+, and other metals with high atomic weights can disrupt the salt bridges in proteins, and salt bridges partially result from the amines on the side chains of proteins. A type of reaction occurs where the positive ions in the added acid combine with the negative ions in the salt bridges. The reaction of cadmium and nickel salts with proteins can form ligands with the amino acids, i.e. tryptophan and tyrosine. This will reduce the fluorescence intensity of the longan hull after the adsorption of Ni2+ and Cd2+ and treatment with HCl.
The fluorescence images of the longan hull powders. (a) No treatment; (b) treatment with HCl; (c) loaded with Cd2+; and (d) loaded with Ni2+.
Conclusions
It was demonstrated that longan hull powders can be used as an agricultural adsorbent to adsorb Ni2+ and Cd2+ from wastewater at concentrations of approximately 50 mg L−1. (1) The adsorption process was relatively fast, and the maximum adsorption capacities for Cd2+ and Ni2+ were approximately 4.19 mg g−1 and 3.96 mg g−1, respectively, under the optimized conditions. (2) The adsorption kinetics of both metal ions on the longan hull powders can be described by a pseudo second-order rate model and are well described by the Langmuir model. (3) The longan hull has N–H, C–H, C=O, and C=C functional groups, which serve as the adsorption sites for the metal ions. Additional work will be required to increase the adsorption capacity of this adsorbent and to identify the different functional groups responsible for the metal ion binding.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.





