Abstract
This work presents a comparison between the biosorption of Hg (II) by raw almond shell and activated almond shell. Almond shell based activated carbon has been obtained by physicochemical activation. Batch biosorption results confirmed that, activating condition has a strong influence on the final biosorption process. The biosorbent was characterized using scanning electron microscopy and Fourier transform infrared spectroscopy. To optimize the biosorption conditions pH, adsorbent dose, initial concentration, contact time, stirring speed, and temperature on Hg (II) removal were studied. The optimum conditions for maximum Hg (II) was achieved at 20 and 10 min for raw almond shell and activated almond shell, respectively. The equilibrium data were described well by Langmuir, Freundlich, Dubinin–Radushkevich isotherm models and appling a test of model fitness. Best fit of Langmuir and Freundlich models were found for experimental data, which reveal the homogenous surface of raw almond shell and the heterogeneity of activated almond shell surface. The kinetic data had been divided into either pseudo first order or second order on the basis of the best fit obtained from calculations, confirmed by a test of kinetic validity. An industrial application was examined to improve high biosorption capacity of raw and activated almond shells toward Hg (II).
Introduction
Due to toxic metals in industrial wastewaters, water pollution has become a major issue worldwide. Heavy metal ions are toxic to aquatic flora and fauna even in relatively low concentrations. The presence of them over the permissible levels in the environment is a severe public health problem. Thus, governments have established environmental restrictions with regard the quality of wastewater, forcing industries to remove metals from their effluents before discharging. Hg and its compounds have drawn much attention due to persistence, bioaccumulation, nonbiodegradability and can bind with organic and inorganic matter and form various composites, which limit water use. It is present in the environment from both natural sources and anthropogentic activities and the latter are believed to be responsible for the elevated mercury levels in the environment. Mercury released to the environment can be further converted to monomethylmercury (MMHg), a substance much more toxic than inorganic mercury (Cui et al., 2013; Harmayani and Anwar, 2016; Li et al., 2013; Ronda et al., 2013; Sun et al., 2014; Wang et al., 2013; Zhang et al., 2014).
To solve this issue (Önnby et al., 2010) various physical and chemical strategies like precipitation, ion exchange, evaporation, oxidation, and membrane filtration were applied. However, application of such technologies is restricted because of the technical or economical limitations. Biosorption (Farooq et al., 2010; Sud et al., 2008) as an alternative and effective technology has been widely used because of its wide range of target pollutants, high sorption capacity, excellent performance, eco-friendly nature, low operating cost, and energy use. The term, biosorption is used to describe a method that utilizes materials of biological origin (biosorbents formulated from nonliving biomass) for the removal of target substances from aqueous solutions (Ronda et al., 2013; Sun et al., 2014; Zhang et al., 2014).
Lignocellulosic biomass derived from agricultural by-products has proven to be a promising type of raw material for producing activated carbon (AC), which is widely used for pollutant removal. Studies have reported that almond shell-based material could be activated by various methods to prepare activated carbons. The performance of AC can be highly improved under appropriate condition. AC is produced by three different processes: physical activation, chemical activation, and physicochemical activation. The activation process leads to production of AC with high porosity, large surface area, and high pore volume (Foo and Hameed, 2010; González and Pliego-Cuervo, 2014; Nor et al., 2013).
Also, we can focus on the importance of almond shells: above the almond hulls there is a hard shell that further protects the almonds. Depending on the variety of almond, the hardness of the almond shell varies. The majority of almond shells are sold to co-generation plants to be used as a fuel source. Another use is that they are incorporated in some feed rations for livestock. They are also used as a landscaping material similar to the use of wood chips. Additionally, metal ion removal, phenol removal, pollutant removal, horticulture, agriculture, farming and more can be done with the use of almond waste and almond co-products (Chayande et al., 2013).
The ultimate goal of the this study is to compare the characteristics of almond shell before and after activation by physicochemical method in order to produce AC that can be used as a potential biosorbents for removal of Hg (II) from waste water. The structure of the resulted biosorbents was studied with the help of scanning electron microscopy (SEM) and Fourier-transform infrared spectroscopy (FTIR) analysis. The effect of solution pH, adsorption time, initial ion concentration, effective dose, stirring speed, and temperature were investigated in the removal of Hg (II) by raw almond shell (RAS) and compared with the activated almond shell. Adsorption kinetics and thermodynamics were fitted to the experimental data and the biosorption mechanism was proposed. Finally, we studied an application case from the environment.
Experimental
Raw almond shell
Almond nut shell (
Preparation of activated almond shell
RAS was chosen for the preparation of AC as lignocellulosic biomass. The product was passed through sieves to obtain samples with different particle sizes. The physicochemical activation was carried out by carbonizing samples in a furnace in a nitrogen atmosphere at 500℃ for 1 h. These conditions were selected from the literature (Hasar, 2003; Pang et al., 2011). The obtained char was cooled at room temperature and then mixed with a solution of ortho-phosphoric acid (85 w%), in a ratio of 2:1 (H3PO4 solution /char, W/W). This ratio was selected from previous studies (Arrigada et al., 1997), which show that the adsorption capacity increases remarkably with increasing H3PO4 /char ratio up to 2:1. The mixture was dehydrated at 250℃ for 3 h and subsequently activated at 800℃ in the absence of air for 1 h. It was also reported that, higher temperatures (900℃) favor the production of an adsorbent with larger adsorption capacity due to an increase in the micro pore size. The products formed were thoroughly washed with deionized water and dried in oven at 100℃ for 2 h. The prepared activated carbons were stored in a dessicator for later studies (Pragya et al., 2013).
Metal solution
Standard stock solution (1000 mg/L) for Hg (II) was prepared by dissolving desired amount of HgCl2 (Merck), in deionized water (Millipore, Milli-Q). Then, rest of solutions of different concentrations were prepared by appropriate dilution of above stock solution using deionized water. Also, the pH was adjusted using 0.1M HNO3 and 0.1M NaOH solution.
Experimental procedure
The biosorption tests were done by using thermostated magnetic stirrer (AGIMATIC–N). Concentration of Hg (II) ions was obtained using a mercury analyzer (Model No. Nippon RA-3000). PH-meter (Model No. HI-2212) was used to adjust pH of solutions.
Batch experiments
Synthetic waste water
All experimental equipments and glassware were soaked in an acid bath (HNO3 (10% v/v)) for 24 h, rinsed thoroughly with tap water, then rinsed 3 to 4 times with deionized water. Afterwards, batch adsorption experiments were carried out and solutions of the metal ion containing the adsorbent were placed in Erlenmeyer flasks and stirred using thermostatted magnetic stirrer. As a preliminary kinetic test revealed that biosorption equilibrium of mercury on the RAS or AAS achieved within approximately 60 min under the experimental conditions, an equilibrium time of 60 min was adopted for all biosorption testing. Batch biosorption experiments were conducted to investigate Hg (II) adsorption as a function of pH, contact time, adsorbent dose, initial metal ion concentration, temperature, and stirring speed.
Real industrial waste water
In order to investigate the efficiency of almond shell, industrial wastewater effluent collected from Alexandria fertilizers (Alexfert) company, Alexandria, Egypt was also treated by RAS and AAS with different masses. The samples were filtered using aWhitman filter paper to remove the suspended particles and then the samples were spiked by 10 mg/L Hg2+ standard solution. The samples were analyzed before and after treatment for various parameters like pH, chemical oxygen demand (COD), biochemical oxygen demand (BOD), total dissolved solids (TDS) and total suspended solids (TDS) using standard methods (APHA, 1998). Different masses of RAS (0.5, 0.7, and 1.0 g) and (0.03, 0.05, and 0.07 g) for AAS were suspended in 100 mL of spiked wastewater effluent at pH of 5.0, temperature of 298 K, stirring speed of 150 r/min, and contact time of 60 min.
Surface characterization
A JEOL scanning electron microscope (Model no. JSM-5300) was used to study the surface morphology of RAS and AAS. For FTIR, samples of almond shells were investigated before and after biosorption processes for functional groups by Perkin-Elmer Spectrum BX FTIR. Spectra were recorded in the range of 4000–400 cm−1. For all samples, the amount of the samples were scanned and KBr were kept constant to investigate the changes in the intensities and shifting of the characteristic peaks as a result of the structural changes.
Analytical method
At specific time intervals, solution aliquots were removed and filtered using 0.45 µm Whatman filter paper to separate the adsorbent, which was left to dry in oven at 65℃ for 8 h. The filtrate was diluted and its concentration was determined using a mercury analyzer (RA-3000). The data were used to calculate the metal percentage removal according the following equation

The adsorption capacity at time 
The adsorption capacity at equilibrium, 
Biosorption experiments
Effect of pH
Batch biosorption experiments were conducted by varying the initial pH value from 2.0 to 7.0. The initial pH of the Hg (II) solution was adjusted with 0.1M HNO3 and 0.1M NaOH. At a temperature of 298 K, 100 mL of 25 mg/L Hg (II) solution was stirred with 3.0 g for RAS, 0.3 g for AAS at 150 r/min for a contact time of 60 min. The solution was then filtered using 0.45 µm Whatman filter paper and its concentration was determined using a mercury analyzer (RA-3000).
Effect of contact time
The effect of contact time was studied at optimum pH 5.0, constant Hg (II) concentration of 25 mg/L, adsorbent dose (3.0 g for RAS and 0.3 g for AAS) /100 mL of Hg (II) solution at 298 K. The samples were stirred using a magnetic stirrer at 150 r/min, then the samples were taken after 1, 3, 5, 7, 10, 15, 20, 30, 45 and 60 min.
Effect of adsorbent dose
The biosorption was carried out by adding different amounts of adsorbent (0.5, 1.0, 2.0, 3.0, and 5.0 g for RAS and 0.05, 0.1, 0.3, 0.5, and 0.7 g for AAS) to 100 mL of 25 mg/L of Hg (II) solution. The batch experiments were carried out under continuous shaking at 150 r/min for 60 min at 298 K and optimum pH 5.0.
Effect of initial concentration
The initial concentration of Hg (II) solution varied from 25 to 100 mg/L at pH 5.0, temperature of 298 K, adsorbent dose of 3.0 g for RAS and 0.3 g for AAS. The samples were stirred for 60 min at 150 r/min.
Effect of stirring speed
To obtain the data of the effect of stirring speed, measurements were conducted in a series of solutions under different stirring speeds, ranging from 75 to 500 r/min at pH 5.0, contact time 60 min, and adsorbent dose of 3.0 g for RAS and 0.3 g for AAS /100 mL of 25 mg/L Hg(II) solution.
Effect of temperature
The temperature properties were investigated at pH 5.0 by varying the temperature from 298 to 318 K at biosorbent dose of 3.0 g for RAS and 0.3 g for AAS /100 mL of 25 mg/L Hg (II) solution for 60 min at 150 r/min.
Results and discussion
Characterization of raw and activated almond shell
Fourier-transform infrared spectroscopy
The surface chemistry of both RAS and AAS was performed before and after Hg (II) biosorption by FTIR analysis in order to understand the metal binding mechanism onto biomass and provide valuable information about chemical compositions of the materials. The FTIR spectrum point the presence of some typical bands of lignocellulosic materials belonging to the functional groups such as hydroxyl, ethers, esters, aldehydes, and Si–H bonds. RAS consists of several functional groups, which include amines, hydroxyl groups, aldehydes, various unsaturated hydrocarbons, carbonyl compounds, phosphates, and sulfonates. whereas the spectrum of AAS is very simple, consisting of a considerable number of peaks or functional groups.
In case of RAS, the wide band at 3432.41 cm−1 is ascribable to O–H stretch vibrations in the hydroxyl groups. It was possible to identify the presence of methylene groups by the detection of υ (C–H) and δ (C–H) absorption bands. These bands are connected to C–H aromatic and aliphatic asymmetric stretch (2924.96 cm−1) and C–H bend vibrations (1461.48 and 1377.15 cm−1), respectively. The band at 1742.75 and 1636.50 cm−1 is caused by carbonyl (C = O) stretching vibrations. The band at 1508.39 and 1429.84 cm−1 reflects the aromatic C = C stretch vibration and the band at 1329.69 cm−1 is ascribable to δ (O–H). The attributed O–H vibrations belonging to primary, secondary, tertiary alcohol, or phenol can be evaluated by the relative position of the C–O stretching frequency. The bands centered in 1161.14, 1110.41, and 1052.87 cm−1 belong to υ (C–O) vibration in tertiary, secondary, and primary alcohol, respectively. The band at 1247.72 cm−1 may be related to esters (e.g. CH3–CO–O–), epoxides, as well as with acyclic C–O–C groups conjugated with carbon–carbon double bonds (C = C–O–C) in olefinic or aromatic structures. The band at 608.85 cm−1 may correspond to SiO–H stretch vibration. The presence of hemicelluloses and cellulose is probably responsible for the appearance of these bonds. In fact, data from literature refers those compounds and lignin as the main constituent of almond shells (Figure 1).
FTIR spectrum of RAS: (a) before Hg(II) adsorption and (b) after Hg(II) adsorption.
After the biosorption of Hg (II), the υ (O–H) band shifts from 3432.41 to 3431.11 cm−1. The stretch vibration of C–H has shifts from 2924.96 to 2916.61 cm−1 and also the deformation (C–H) band shifts from 1377.15 to 1376.57 cm−1. The C = O stretching vibration band at 1742.75 and 1636.50 cm−1 shifts to 1742.30 and 1635.49 cm−1, respectively. The aromatic and aliphatic C = C stretching vibration band at 1508.39 and 1429.84 cm−1 shifts to 1506.79 and 1428.82 cm−1. The bending vibration of the O–H band shifts from 1329.69 to 1328.16 cm−1. The C―O stretching vibration band (related to esters, epoxide, and cyclic C–O–C groups conjugated with carbon–carbon double bond) shifts from 1247.72 to 1249.97 cm−1 and also the band of υ (C–O) at 1161.14 and 1052.87 (related to tertiary and primary alcohol) shifts to 1162.55 and 1039.27 cm−1, respectively. Also, the SiO–H stretch vibration band shifts from 608.85 to 608.26 cm−1.
In case of AAS, the FTIR analysis reveals a simple spectrum compared to that of the raw material. The AAS displays biosorption bands at 3437.88, 1720.05, 1585.43, and 1203.98 cm−1, assigned O–H, C = O, C = C, and C–O bond stretching modes, respectively. The band at 3782.62 cm−1 is ascribable to stretch vibration of free (nonbonded) OH groups (Figure 2). After Hg (II) adsorption, the stretch vibration modes of O–H, C = O, C = C, and C–O shifts from 3437.88, 1720.05, 1585.43, and 1203.98 cm−1 to 3433.02, 1727.79, 1588.52, and 1166.48 cm−1. The υ (O–H) band at 3782.62 cm−1 was disappeared after adsorption of Hg (II), which attributed to change in nature of binding after interaction with Hg (II) ions. So the above IR analysis shows the presence of different oxygen groups, mainly carbonyl, alcohol, ethers, and esters characteristic of these types of waste and makes them good biosorbent. It was observed that the biosorption of Hg (II) ions onto RAS and AAS through the functional groups of almond shell has changed both the position and the intensities of their IR bands. These changes reflect the interaction between the functional groups of almond shell and Hg (II) ions (Chayande et al., 2013; Mohan et al., 2011; Sun et al., 2014; Wang et al., 2013).
FTIR spectrum of AAS: (a) before Hg(II) adsorption and (b) after Hg(II) adsorption.
Scanning electron microscopic analysis
The SEM images show the surface of the biosorbent prior and after biosorption process. The SEM photographs of the original almond shells show that the whole shells have a rough surface with almost non-compact structure. As shown in Figure 3(a), (c), and (d) it is obvious that both RAS and AAS are composed of irregular and porous particles, mainly macropores, which indicated high surface area and consequently, appropriate conditions exist for Hg (II) ions to be trapped and adsorbed into these pores.
SEM images of RAS: (a) without biosorption, (b) after Hg(II) biosorption: magnification 5000× and for AAS: (c, d) without adsorption, (e, f) after Hg(II) ions adsorption (magnification: 2000× and 3500×).
It was observed that a considerable number of pores with different structures were formed due to H3PO4 chemical activation of lignocellulosic almond shell (Figure 3(c) and (d)). The pore formation may be due to that the addition of H3PO4 causes the material to swell resulting in the opening of the surface structure. Phosphoric acid is a strong dehydrating agent that accelerates the bond cleavage reactions leading to the early evolution of volatiles at below 300℃ and the generation of pore spaces. At high temperatures, the reactive sites leave a hard aromatic porous carbon structure for biosorption. Also, it can be observed that the surface of AAS shows a lot of grooves, cracks, and crevices in the matrix after activation. The macropores are highly developed deep inside the surface, which indicates that the precursor material and method for activation is adequate for the purpose.
As shown in
Effect of initial pH on biosorption
The pH factor plays a vital role in the biosorption of cations from aqueous solutions because; it affects the solubility of adsorbate, concentration of counter ions on the functional groups of the adsorbent, metal chemistry in solution, and the ionization state of the functional groups of the sorbent, which affects the availability of active sites (Afkhami et al., 2010; Heiddari et al., 2009; Ngomsik et al., 2009). At lower pH values, HgCl2 (Hg2+) species are predominant in the solution and the concentration of H+ ions is high, therefore the active sites of the adsorbent are less available for Hg2+ ions due to the protonation of the surface functional groups of the adsorbent. By increasing the pH value, the percent of Hg(Cl)OH and Hg(OH)2 species increases, which are considered easier to be adsorbed than HgCl2. Also the degree of dissociation of carboxylic acid groups (equation (4)) increased too, resulting in an increase in the adsorption of mercury ions (Anagnostopoulos et al., 2012; Yu et al., 2008). As shown in Figure 4, the two curves present similar trends with apparent suppression in highly acidic solution. The biosorption capacity of RAS and AAS was strongly increased by increasing the pH value from 2.0 to 5.0 reaching a maximum at pH 5.0. At pH higher than 5.0, only slight decrease in Hg (II) biosorption was observed. We consider that at pH 5, there is no competition between the hydrogen and Hg (II) for the available sorption sites on the RAS and AAS achieving a relative maximum biosorption capacity (González and Pliego-Cuervo, 2014). Therefore, in the subsequent work, experiments were carried out at fixed optimum pH of 5.0. It is believed that at a pH range from 3.0 to 6.0, both ion exchange and surface complexation (equations (5) and (6)) are the major mechanisms for the removal of mercury ions. The biosorption of Hg (II) ions on the surface of RAS and AAS is considered to occur via different mechanisms such as electrostatic forces (electrostatic attraction between positively charged metal ions (Hg2+ and Hg(OH)+) and negatively charged functional groups of the adsorbent (R–COO−), which act as anionic chelating agent), ion exchange, and chemical complexation (Anirudhan et al., 2012).
Effect of initial pH on adsorption capacity at equilibrium of Hg(II) ions onto: (a) RAS and (b) AAS (the initial concentration = 25 mg/L, adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, 

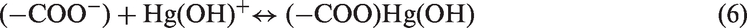

Adsorbent dose influence
Adsorbent dosage is an important parameter because it determines the capacity of an adsorbent for a given adsorbate initial concentration. It was observed that in case of RAS, the percentage removal of Hg (II) ions increased from 82.91% to 99.93% by increasing the adsorbent dose from 0.5 g to 5.0 g and in case of AAS, the percentage removal increased from 72.52% to 97.03% by increasing the adsorbent dose from 0.05 g to 0.7 g. This may be due to that increase in the adsorbent dose results in more adsorptive sites being available in the same solution volume. Also, an increase in the solid concentration increases the surface area of the adsorbent, which in turn increases the number of binding sites for the same liquid volume and thus the total amount of metal that is removed increases (Figure 5). It was also observed that, for both RAS and AAS, the amount of adsorbed Hg (II) ions per unit mass of adsorbent decreased with increasing the adsorbent dose. This may be due to the large adsorbent amount effectively reduces the unsaturation of the adsorption sites and correspondingly, the number of such sites per unit mass comes down resulting in comparatively less adsorption at higher adsorbent amount. Also, the higher adsorbent dosage creates particle aggregation, resulting in a decrease in the total surface area and increase in the diffusion path length, which lead to decrease in the amount of adsorbed Hg (II) ions per unit mass of adsorbent (Gupta and Bhattacharyya, 2008; Viera et al., 2010).
Effect of adsorbent dose on the % removal and adsorption capacity at equilibrium of Hg(II) ions onto: (a) RAS and (b) AAS (the initial concentration = 25 mg/L, stirring speed = 150 r/min, 
Effect of contact time and temperature
Biosorption of Hg (II) ions onto RAS and AAS was studied in the range of 1–60 min. As shown in Figure 6, it was observed that in case of RAS, the adsorption efficiency of Hg (II) ions increased rapidly during the first 10 min and remained nearly constant after 20 min of adsorption while in case of AAS, the adsorption efficiency increased rapidly during the first 5 min and remained nearly constant after 10 min. This was probably due to the availability of so many adsorptive sites at the beginning of the experiment and as the biosorption proceeds, the free binding sites become rarer causing a slower biosorption rate until approaching the equilibrium due to the saturation of active sites with Hg (II) ions. It was also observed that, the Hg (II) adsorption onto AAS attained equilibrium faster than that onto RAS, which was due to the large surface area of AAS that suggest more adsorptive sites (Al-Rmalli et al., 2008).
Effect of contact time on the adsorption of Hg(II) ions onto: (a) RAS and (b) AAS at different initial concentrations (the adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), pH = 5.0, 
Figure 7 shows the experimental results obtained from Hg (II) ions removal by RAS and AAS by increasing the temperature from 298 K to 328 K reaching a maximum removal efficiency at 328 K. An increase in the temperature involves increasing the mobility of Hg (II) ions and decreasing the retarding force acting on the diffusing ions, which result in the enhancement in the sorptive capacity of the adsorbent, increasing the chemical interaction between adsorbate–adsorbent and creation of active surface centers or by an enhanced rate of intra-particle diffusion of Hg (II) ions into the pores of the adsorbent at higher temperature (Bouhamed et al., 2012; Kalavathy and Miranda, 2012).
Effect of temperature on the perecnatge removal of Hg(II) ions onto: (a) RAS and (b) AAS (the initial concentration = 25 mg/L, adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, pH = 5.0 and contact time = 60 min).
The effect of temperature on the biosorption process is associated with changes in several thermodynamic parameters. The adsorption equilibrium data obtained for different temperatures were used to calculate the thermodynamic parameters such as standard Gibbs free energy (ΔG°), standard enthalpy change (ΔH°), and standard entropy change (ΔS°). These parameters were estimated using the following equations (Fultalan et al., 2011)
Influence of temperature on the thermodynamic behaviour of adsorption of Hg(II) ions onto: (a) RAS and (b) AAS. Thermodynamic parameters for adsorption of Hg(II) ions onto raw and activated almond shell.




In case of RAS, it could be observed that at a temperature of 298 K, the value of
Kinetics of Hg (II) biosorption and effect of initial concentration
Initial Hg (II) concentration was adjusted in ranges of 25–100 mg/L for biosorption on RAS and AAS. It is necessary to highlight that the adsorption capacity, Effect of initial concentration on the percentage removal and adsorption capacity at equilibrium of Hg (II) ions onto RAS and AAS (adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, 
The kinetics rate of Hg (II) ions sorption onto RAS and AAS were analyzed using pseudo-first-order, pseudo-second-order, and intra-particle diffusion model, which are the most commonly used models. The conformity between the experimental data and the model-predicted values was expressed by the regression coefficients (
The pseudo-first-order kinetic model
The integral form of the pseudo-first-order model is generally expressed as follows

The model is based on the assumption that the rate is proportional to the number of free sites. If the pseudo-first-order kinetics is applicable, a plot of ln( Pseudo-first-order kinetic fit for adsorption of Hg(II) ions onto: (a) RAS and (b) AAS at different initial concentrations (adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, 
Adsorption kinetic models and other parameters for the biosorption of Hg(II) ions onto (a) RAS and (b) AAS at different initial concentrations.

The pseudo-second-order kinetic model
The pseudo-second-order model is based on the assumption that biosorption follows a second-order mechanism. This means that the sorption rate would be proportional to the metal concentration and the square of the number of free sites, which corresponds to the term (
The linearized pseudo-second-order plot of Pseudo-second-order kinetic fit for adsorption of Hg(II) ions onto RAS and AAS at different initial concentrations (adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, 

It was also observed that the values of the rate constant
The half-life data of adsorption (Table 2) (
The results showed that the half-life of adsorption reduces with decreasing initial metal concentration. This means that, as the initial metal concentration decreases, less time is needed to reduce the initial concentration to half its original value (Senthilkumar et al., 2011).
Intra-particle diffusion
A detailed understanding of the adsorption mechanisms facilitates a determination of the rate-limiting step. This information can then be used to optimize the design of adsorbents and adsorption conditions. The overall rate of adsorption can be described by the following three steps: (1) film or surface diffusion where the sorbate is transported from the bulk solution to the external surface of sorbent, (2) intra-particle or pore diffusion, where sorbate molecules move into the interior of sorbent particles, and (3) adsorption on the interior sites of the sorbent. Since the adsorption step is very rapid, it is assumed that it does not influence the overall kinetics. The overall rate of adsorption process, therefore, will be controlled by either surface diffusion or intra-particle diffusion. The Weber–Morris intra-particle diffusion model has often been used to determine if intra-particle diffusion is the rate-limiting step and can be expressed as

The intra-particle diffusion rate constant
According to this model, a plot of Intra-particle diffusion kinetic fit for adsorption of Hg(II) ions onto (a) raw and (b) activated almond shell at different initial concentrations (adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), stirring speed = 150 r/min, 
Test of kinetic validity
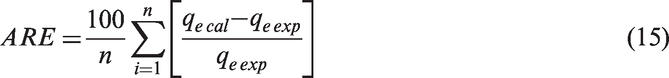
The goodness of fitting the kinetic data was evaluated by the average relative error (ARE) function calculated by the following equation
Effect of stirring speed
This effect is shown in Figure 13. It was observed that by increasing the stirring speed from 75 r/min to 500 r/min, the percentage removal of Hg (II) ions increased for both biosorbents. This may be due to that increasing the stirring speed decreases the boundary layer and the film resistance to mass transfer surrounding the adsorbent particles. Also, when the system was well-mixed under higher stirring speed, the mass transfer effect became insignificant.
Effect of stirring speed on the percentage removal and adsorption capacity at equilibrium of Hg(II) ions onto RAS and AAS (the initial concentration = 25 mg/L, adsorbent dose = 3.0 g (RAS) and = 0.3 g (AAS), pH = 5.0 and 
On the other hand, the percentage removal of Hg (II) decreased with decreasing stirring speed. This can be explained by the fact that for a relatively lower mixing rate, the system was incompletely agitated and hence the RAS and AAS were poorly dispersed in the solution. This may have resulted in a part of the surface area of the adsorbent being exposed for the adsorption of Hg (II) ions.
Adsorption isotherm study



Fitted isotherm models for the biosorption of Hg(II) ions onto RAS and AAS at different initial concentrations.

The Langmuir model is valid for monolayer sorption on a surface with a finite number of identical sites (Farghali et al., 2013). The slope and intercept of the linear plots of
Comparison of Langmuir biosorption capacity of RAS and AAS for Hg(II) with that of different biosorbents.

RAS: raw almond shell; AAS: activated almond shell.
Another important parameter, 
The lower
It was observed that the values of regression coefficient (
The empirical Freundlich model is based on a multilayer adsorption with nonuniform distribution of adsorption heat and affinities over the heterogeneous surface (Foo and Hameed, 2010).
The applicability of Freundlich adsorption isotherm was analyzed by plotting log
The D–R isotherm (equation (18)) is generally applied to express the adsorption mechanism with a Gausian energy distribution onto a heterogeneous surface (Foo and Hameed, 2010).
Meanwhile, the Polanyi potential, 
The parameters of the D–R isotherm, for both raw and activated almond shell, are given in Table 3. The results show that the
Test of model fitness
In order to find out the best fitting isotherm model, the data were verified by nonlinear residual root-mean squared error (
The isotherm parameters obtained at equilibrium obtained at different initial concentrations for the two isotherm models along with
Application case
Removal percent of Hg(II) ions from spiked wastewater effluent (

RAS: raw almond shell; AAS: activated almond shell.
Analysis of wastewater effluent before and after treatment.

COD: chemical oxygen demand; BOD: biological oxygen demand; TDS: total dissolved solids; TSS: total suspended solids; RAS: raw almond shell; AAS: activated almond shell.
Conclusion
This work explains a comparative study between RAS and AAS for the removal of Hg (II) ions. Batch experiments were carried out in order to study the effect of different parameters such as contact time, initial metal ion concentration, stirring speed, pH, adsorbent dose, and temperature. The optimum pH was found to be 5.0 for both raw and activated almond shell. Biosorption kinetics was studied using pseudo-first-order, pseudo-second-order, and intra-particle diffusion models at various initial concentrations of metal ion. Langmuir, Freundlich, and Dubinin–Rdushkevich adsorption isotherms were applied on data at different initial concentrations. The maximum biosorption capacity was found to be (
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
