Abstract
During the processes occurring at a solid surface, the changes in the surface free energy take place. The knowledge about surface free energy is very helpful for understanding the processes taking place on the surface. However, experimental determination of solid surface free energy is still not a fully solved problem. In this paper, some problems dealing with calculation of solid surface free energy from contact angle are discussed based on literature values of advancing and receding contact angles measured on four different fluoropolymers surface. The four approaches most often used for the calculation are described and especial focus on the approach in which both the advancing and receding contact angles is paid. It is concluded that using probing liquids the absolute value of solid surface free energy cannot be determined. However, the determined apparent values of the energy are very helpful to understand the conditions necessary for a given process to occur.
Introduction
The processes occurring on a solid surface are accompanying with the surface free energy changes. Some important surface processes are wetting, cleaning, washing, anticorrosion coverages, painting, chemical plant protection, mineral flotation, adsorption, etc. The knowledge about surface free energy is very helpful for understanding the processes taking place on the surface. However, while there are several good methods for determination of surface free energy of a liquid (surface tension of liquid), so experimental determination of surface free energy of a solid is still not a fully solved problem (Bormashenko, 2013; Chibowski, 2007; Chibowski and Terpiłowski, 2009; Della Volpe and Siboni, 2008; Etzler, 2003; Marmur, 2006, 2009; Marmur et al., 2017, and the references therein).
The most often used method relies on measurement of wetting contact angles and then application of the existing theoretical approaches to calculate the energy, although inverse gas chromatography technique is sometimes applied too (Mohammadi-Jam and Waters, 2014; Yao et al., 2015). If behind the liquid droplet the solid surface is bare, the contact angle formed by the droplet is termed as the advancing contact angle θa. Then, when the three-phase line has retreated, e.g. by sucking a volume of the liquid drop into the syringe, the contact angle of this “new equilibrium” is smaller, and it is called receding contact angle θr. The difference between the advancing and receding contact angles is defined as contact angle hysteresis (CAH), H (Chibowski and Jurak, 2013; Kuchin and Starov, 2015a, 2015b). Actually, there are also other definitions of appearing contact angles (Marmur, 2006, 2009; Marmur et al., 2017). Most often used probe liquids for contact angle measurement are polar water and formamide and apolar diiodomethane. The theoretical models of solid surface free energy are in fact based on Young’s equation (Young, 1805)

There are different theoretical approaches and the most often applied for determination of a solid surface free energy are the following:
1. Lifshitz–van der Waals/acid base which was proposed by Van Oss et al. (1986, 1988). In this approach, the total surface free energy consists of the apolar Lifshitz–van der Waals component 
And the interfacial free energy is expressed

The apolar 
To determine the solid surface free energy components, one has to solve simultaneously three equations of type equation (4) using the advancing contact angle of three different probe liquids whose surface tension components are known and at least two of them are polar. From the energy components total surface free energy can be calculated.
2. Owens–Wendt (O–W) (Owens and Wendt, 1969) based on Fowkes’ (1963) pioneering ideas assumed that the surface free energy can be expressed as a sum of two components: London dispersion interactions 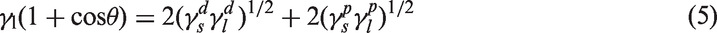
3. Neumann’s equation of state (Li and Neumann, 1990, 1992; Neumann et al., 1974; Tavana and Neumann, 2007) allows calculation of a solid total surface free energy using measured advancing contact angle and numerical solution of equation (6) in which the coefficient β (0.0001247 m2/mJ) was determined experimentally
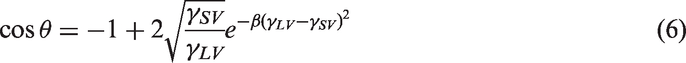
4. Measuring the advancing 
This is the only approach in which both the advancing and receding contact angles are applied in calculation of the free energy and the privilege is that only one liquid is needed to evaluate the energy. It should be stressed that determination of surface free energy of solids is not a fully solved problem. The values determined by different approaches, even using the same probing liquids, may differ more or less depending on the kind of solid surface. Therefore, application of different approaches allows verification of the apparent surface free energy of investigated surface (Chibowski and Jurak, 2013; Chibowski and Terpiłowski, 2009). It should be stressed that all the above-mentioned methods are debatable and were criticized in the numerous published papers which would be difficult to cite here. Most of the problems are discussed in the above cited papers and in the references therein quoted. However, because there is not any other method for solid surface free energy determination, the existing ones are useful for better understanding of the processes occurring at interfaces. Because the purpose of this paper was not a general critical review of the existing problems, therefore it is mainly focused on application of CAH for solid surface free energy determination and to point out importance of orientation of the probe liquid molecules for the calculated values of solid surface free energy. For this purpose using the literature data of advancing and receding contact angles of homological series of
Surface free energy of some fluoropolymers
Tavana et al. (2007) published a very precisely measured advancing and receding contact angles of above-mentioned high purity liquids measured on four different fluoropolymer films deposited on silicon surface by dip coating whose repeat units are shown in Figure 1. As is seen the polymers differ in their structure and number of flour atoms present in the units. This reflects in the advancing and receding contact angles which are presented in Table 1 for The polymer units: (a) poly[4,5-difluoro-2,2bis (trifluoromethyl)-1,3-dioxole- Advancing and receding (extrapolated) contact angles (degrees) of EGC-1700: poly(2,2,3,3,4,4,4-heptafluoro-butyl methacrylate); ETMF: poly(ethene-alt- Advancing and receding (extrapolated) contact angles (degrees) of probing liquids on the polymers in degrees and the liquids surface tension (mN/m) (adapted from Tavana et al. (2007)), and their density (g/cm3). CDDT: 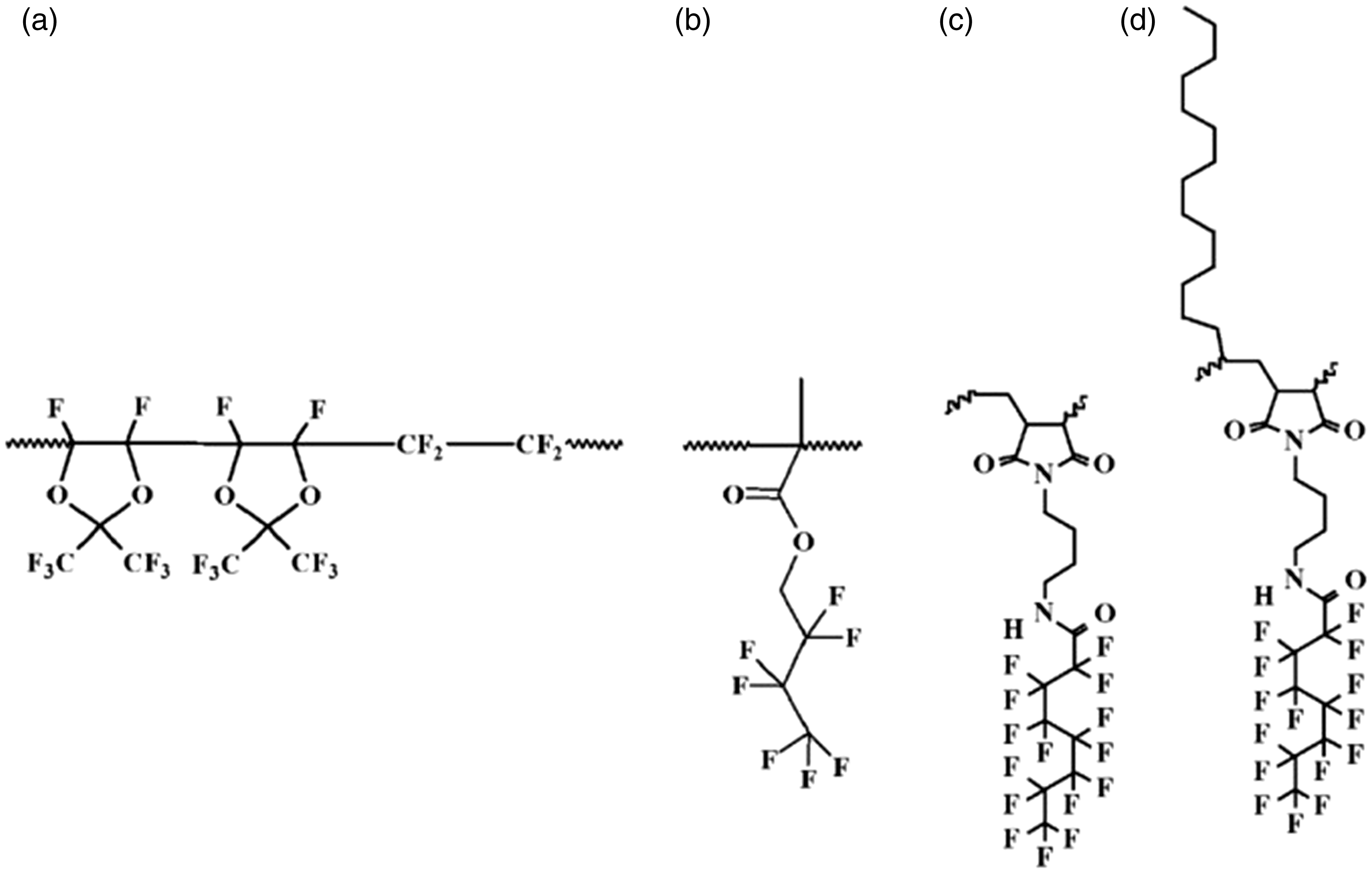

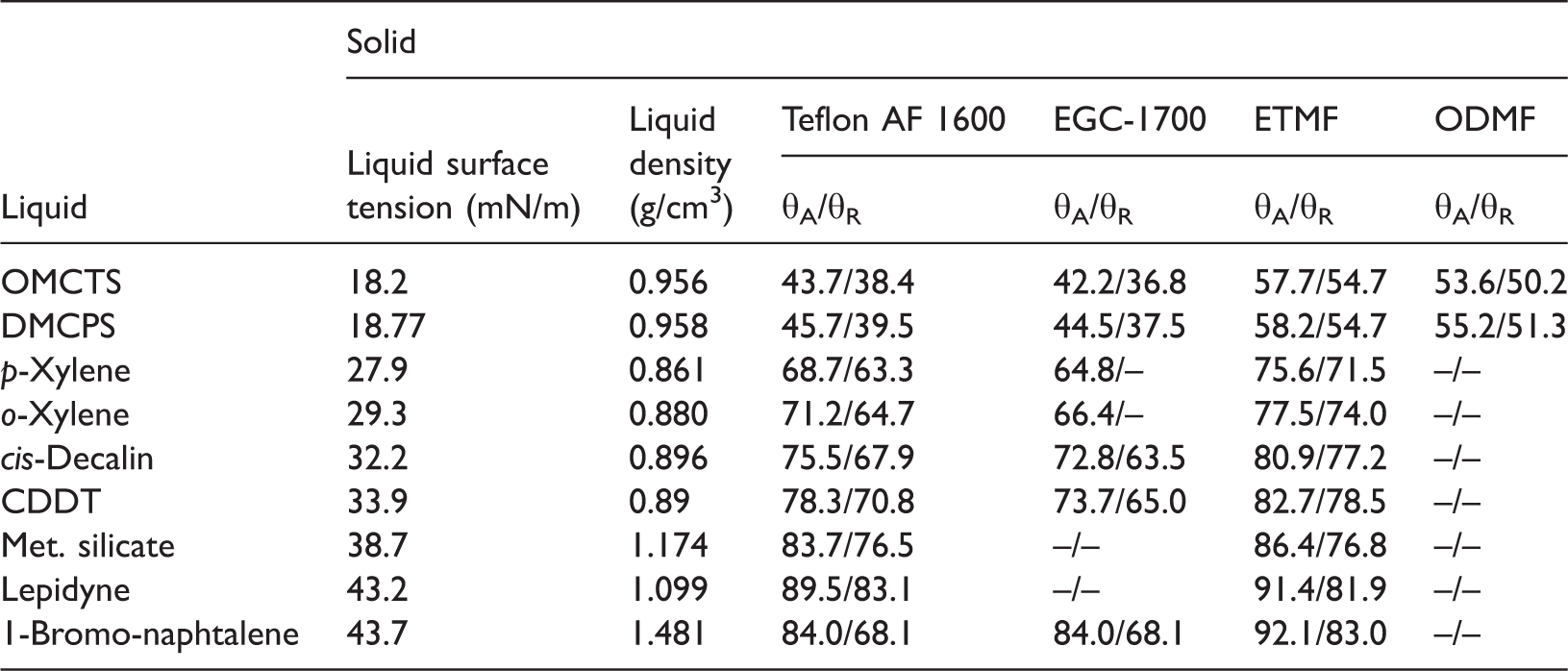
The contact angles were measured by sessile drop method. The advancing angles were determined at a very slowly advancing three-phase contact line (0.5 mm/min) and using drop shape analysis profile. Although the advancing contact angles were constant, the receding ones, measured in a similar manner but during the line receding, appeared to decrease on timescale. The decrease depended on the kind of polymer and liquid. Therefore, the authors extrapolated the receding contact angles values to zero time when the three-phase line started to recede. The detailed description of the method and discussion of the reasons of the observed behavior can be found in Tavana et al. (2007). Because purpose of this paper is to analyze the polymer surface free energy, therefore the contact angles values are used as published in the paper but rounded to decimal of degree. First from the contact angles of the liquids the surface free energy values of the polymers were calculated by CAH approach (equation (7)) and they are plotted in Figure 2 versus the surface tension of the probing liquids.
Surface free energy of fluoropolymers (see Figure 1) calculated from contact angle hysteresis of the liquids whose advancing and receding contact angles were measured on the polymers versus the liquid surface tension.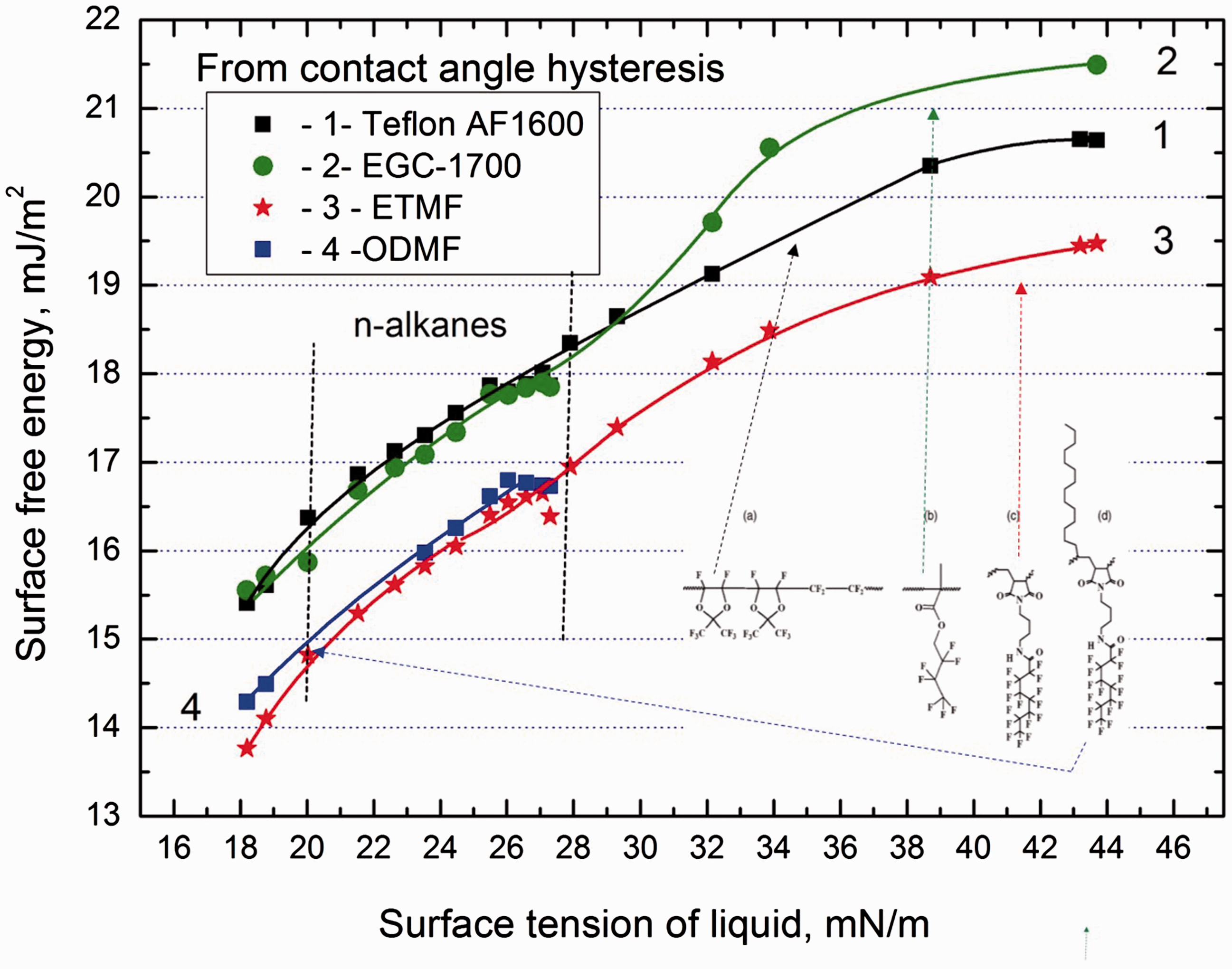
Analyzing the results in Figure 2 first of all it is seen that the calculated values of the energy increase with increasing surface tension of the probing liquid even up to 6–7 mmJ/m2. The surface free energy of the polymers depends also on the number of flour atoms and their arrangements in the unit and therefore Teflon AF 1600 and EGC-1700 possess bigger free energy than ETMF and ODMF polymers, which is completely understandable (see also discussion in Tavana et al. (2007)). However, an interesting question is why the values of energy calculated by CAH model increase with increasing surface tension of the probing liquids. Moreover, even for Surface free energy of Teflon AF 1600 calculated from CAH approach and from advancing and receding contact angles of 
Although the fluoropolymers have low surface free energy indeed, the values determined from θA seem to be evidently too small. More appropriate are the values calculated from CAH (Figure 3). The literature values for Teflon PTFE (–CF2–CF2–)n range from 18.0 to 25.8 mJ/m2 (Surface Energy Data for PTFE, http://www.accudynetest.com/polymer_surface_data/ptfe.pdf). The question is still open why the values increase or decrease monotonically with increasing the chain length of
Figure 4 presents original and CMO surface tension of Original and corrected for correlated molecular orientation surface tension of Correlating factor for 

In the case of solid hydrocarbons their surface free energy is lower if the chains are oriented normal than parallel to the surface. This weaker interaction is due to the lower surface concentration of interacting groups than in case of parallel orientation. The interacting potential of CH3 groups is 82% greater than for CH2 and the ratio of the work of adhesion of the two orientation amounts 3.23 (Fowkes, 1969).
Taking into account the above data it was possible to recalculate the surface free energy of Teflon AF 1600 using only the “active” part of the Surface free energy of Teflon AF 1600 calculated with total surface tension and CMO surface tension of 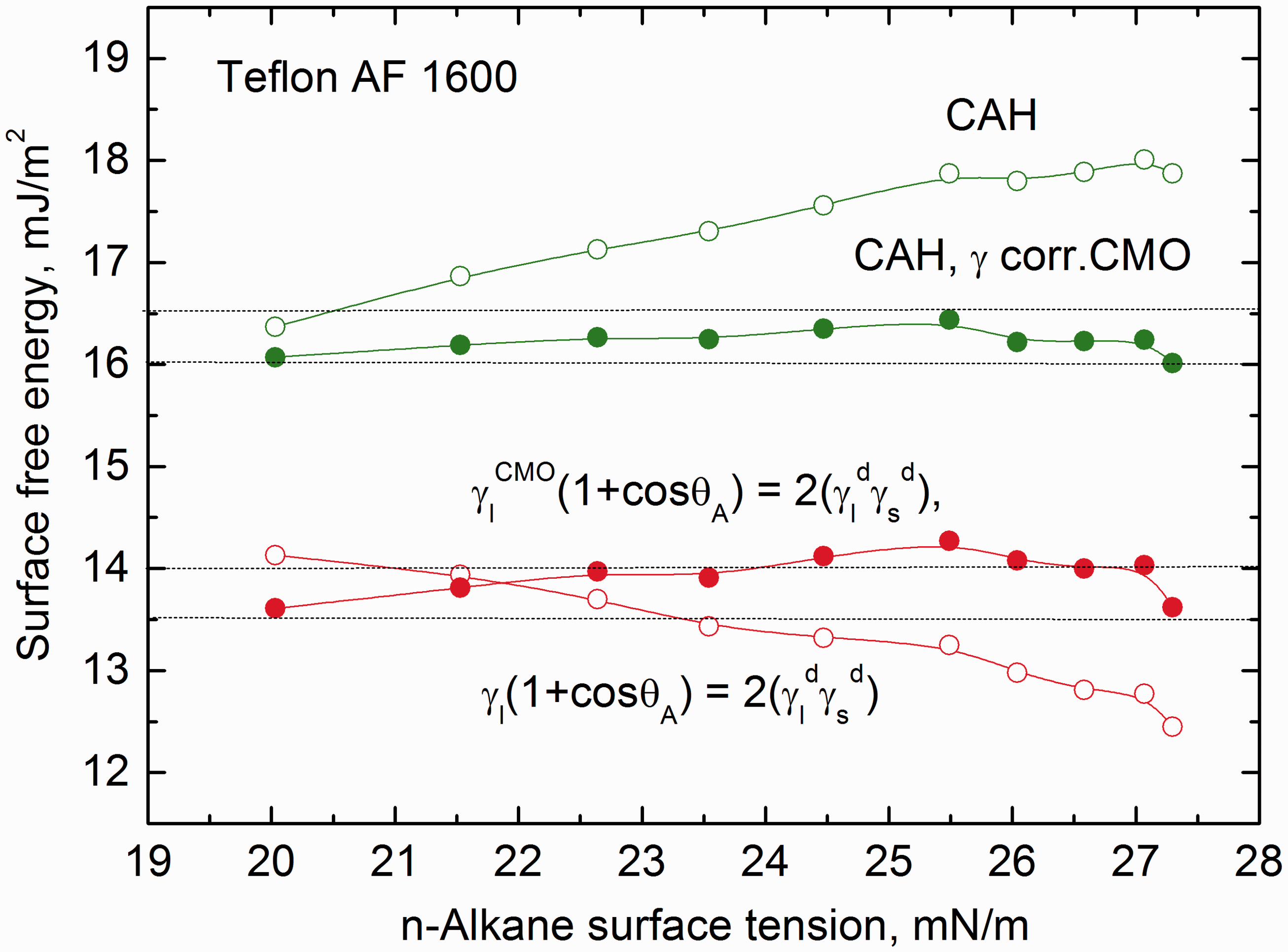
To better depict the surface free energy of Teflon AF 1600 in Figure 7 are plotted the values, which were calculated by different approaches and with or without the correlation of Surface free energy of Teflon AF 1600 calculated using total and CMO surface tension of 
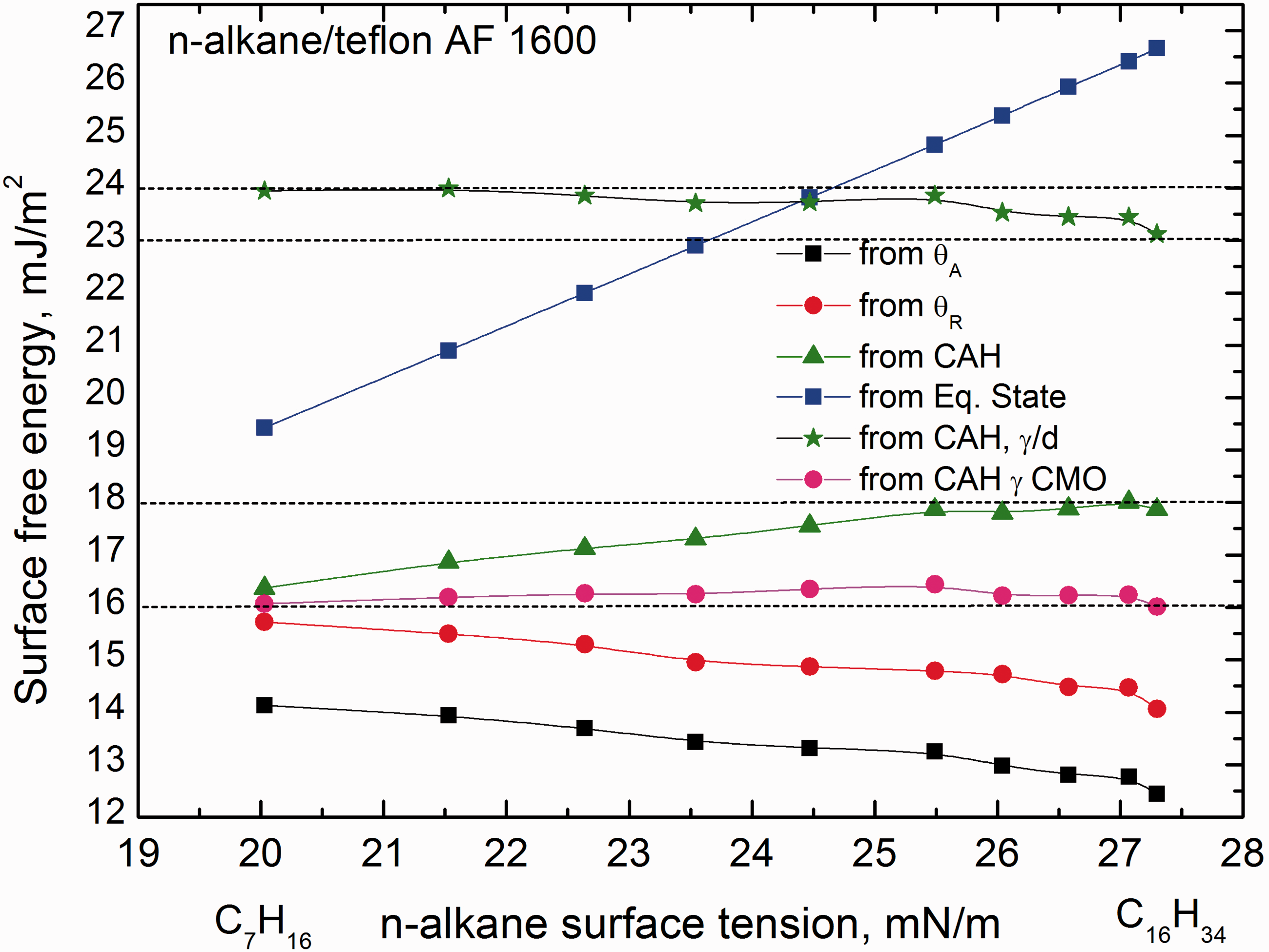
Because for rest probing liquids no correlation of molecular orientation data could be found, contrary to their densities, therefore to find whether calculated values of the Teflon surface free energy could be more coherent, the surface tension values of the liquids from Table 2 were corrected with their density, i.e. divided by density of respective Surface free energy of Teflon AF 1600 calculated by CAH approach using “corrected” with density surface tension of the probing liquids γl/d. CAH: contact angle hysteresis.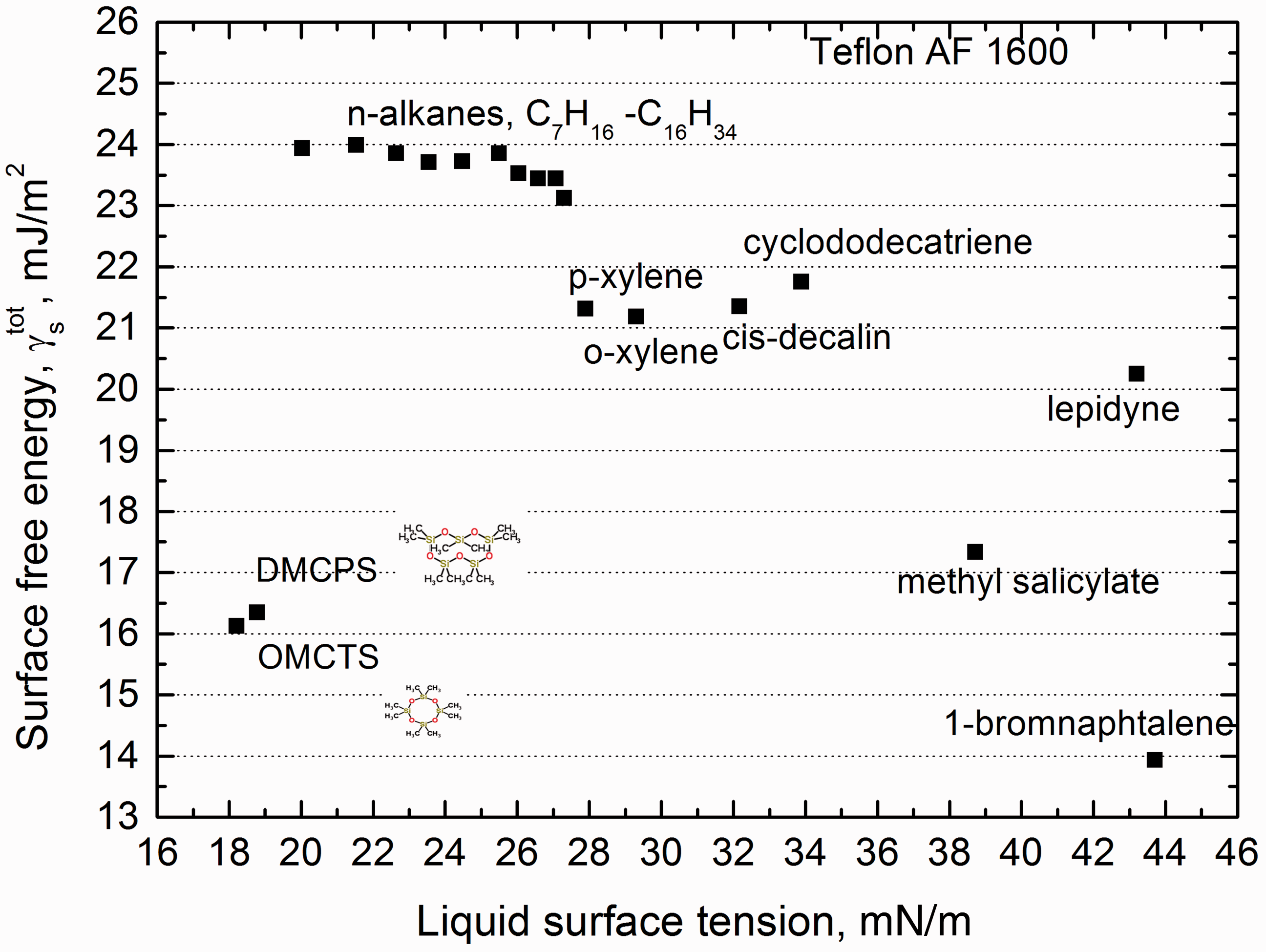
Summary and conclusion
In the light of the above results, it can be concluded that the experimentally determined surface free energy of solids depend to some extent on the probing liquid used for the contact angle measurement and the theoretical approach applied for calculation. It means that thus determined values are not absolute but apparent ones.
The total surface tension of a probing liquid is not appropriate one for determination of real value of a solid surface free energy. This is also true even in the case of such relatively simple systems like nonpolar polymer/apolar
One may conclude that no absolute value of solid surface free energy can be determined because its value depends on the kind and strength of forces acting on the solid surface from the probing liquid phase, and the distance of interacting moieties (groups or atoms), and/or the molecule orientation. Nevertheless, these apparent values of the energy are useful for better understanding of processes occurring at the surface (interface).
CAH method gives comparable results of the surface free energy to those obtained by other approaches and possesses convenience that the energy can be calculated from CAH and surface tension of only one liquid.
Footnotes
Acknowledgements
This work has been partially carried out in the frame of Marie Curie Initial Training Network “Complex Wetting Phenomena” (Project number 607861).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
