Abstract
Hydrophobic poly(methyl methacrylate)/silica layers on the glass support activated with plasma were obtained. Wettability was investigated from the contact angle measurements and surface free energy estimation by means of using contact angle hysteresis approach. The thermal analysis of poly(methyl methacrylate)/silica films was conducted on a
Introduction
In order to protect constructive materials from harmful atmospheric factors, for example glass used in solar panels or photovoltaic cells, it is necessary to cover them with special protective layers (Akamatsu et al., 2000; Kim et al., 2012). The lens surfaces with the reflective layer get dirty easily and have to be very often cleaned. The solution of such a problem seems to be a deposition of hydrophobic layer, so-called clean coat. Glass surface becomes smoother and more hydrophobic because water creates drops easier to remove and contamination embeds more difficult. Hydrophobic coats are widely used also on the concrete surfaces, polymers, or painted surfaces as protective layers for water and frost (Akamatsu et al., 2000). Unfortunately, the support coverage with a hydrophobic layer is very often problematic due to low adhesion. Because of that using plasma technique for surface modification is reasonable (Jung et al., 2011; Kang et al., 2010).
Using silica as a filler in polymers very often leads to mechanical properties improvement of organic–inorganic nanocomposites (Zhou et al., 2004). Yamano and Kozuka (2010) investigated thin poly(methyl methacrylate) (PMMA)/silica films on the glass. They have characterized the obtained films using scanning electron microscopy (SEM), wettability, and FT-IR spectroscopy. PMMA/silica films were optically transparent and exhibited slightly hydrophobic nature with the contact angle 54°. The addition of the PMMA resulted in photochromic changes in color and high chemical durability as well as mechanical hardness. Such films cannot be obtained by the conventional sol–gel method.
Tsai et al. (2011) obtained polyimide (PI)/silica layers on the glass surface. It was found that the addition of silica (from 10 to 40%) to PI increased hydrophobicity which ranged from 84 to 98° depending on the silica contents. On the other hand, transparency of such hybrid films is associated with improved compatibility between the polymer and silica. Hydrogen bonding between PI and silica leads to better dispersion of silica in the film. Pipatchanchai and Srikulkit (2007) investigated hydrophobicity modification of the cotton fabric surface with hexadecyltrimethoxysilane (HDTMS) and silica. The prepared coatings changed surface property from hydrophilic to hydrophobic. The confirmation of obtaining hydrophobic surface can be clearly seen in the contact angle values, which changed from 0° to even over 120° depending on the silica and HDTMS contents.
Experimental
Plasma treatment of glass surface
Air plasma treatment of glass surface was performed using a low-pressure plasma (
Modification of initial silica
Properties of HMDS-modified silica.

HMDS: hexamethyldisilazane; TMS: trimethylsilyl
Deposition of polymer/silica layers on the glass surface
PMMA used in the experiments was from
PMMA (0.2 g/100 cm3) (Chibowski et al., 2006) in chloroform was prepared and the determined amount of silanized silica with different surface fraction coverages with TMS groups (Θ) from 0.53 to 1.0 (origin of silica is given in Table 1) was dispersed (0.16 g) in the PMMA solution with a sonicator. Then, 0.5 ml of the PMMA solution with a filler was poured out in the middle of a circular glass plate and rotated in the spin coater (
Contact angle measurements using the sessile drop method
For the contact angle measurements by the sessile drop method
Surface free energy calculation
Apparent surface free energy 
SEM
Morphology observation was made using a high distributive field emission scanning electron-ion microscope (
Thermal analysis
Thermal analysis was made on a
Results and discussion
Nowadays glass is a very widely used type of support in everyday life and industry. From our previous investigation it can be concluded that the optimal air plasma treatment time of glass surface is 30 s (Terpiłowski and Rymuszka, 2016). Activation with plasma leads to improvement of the adhesion between the deposited layer and glass support. It was observed that modification leads to contact angle decrease about nine times and surface free energy increase (Terpiłowski et al., 2015). The calculated work of adhesion for the glass surface treated by air plasma amounts 145.60 ± 13.33 mJ/m2 and is higher than for the unmodified surface (133.44 ± 12.84 mJ/m2). Therefore, it can be concluded that plasma treatment leads to adhesion improvement between the deposited layer and the glass surface.
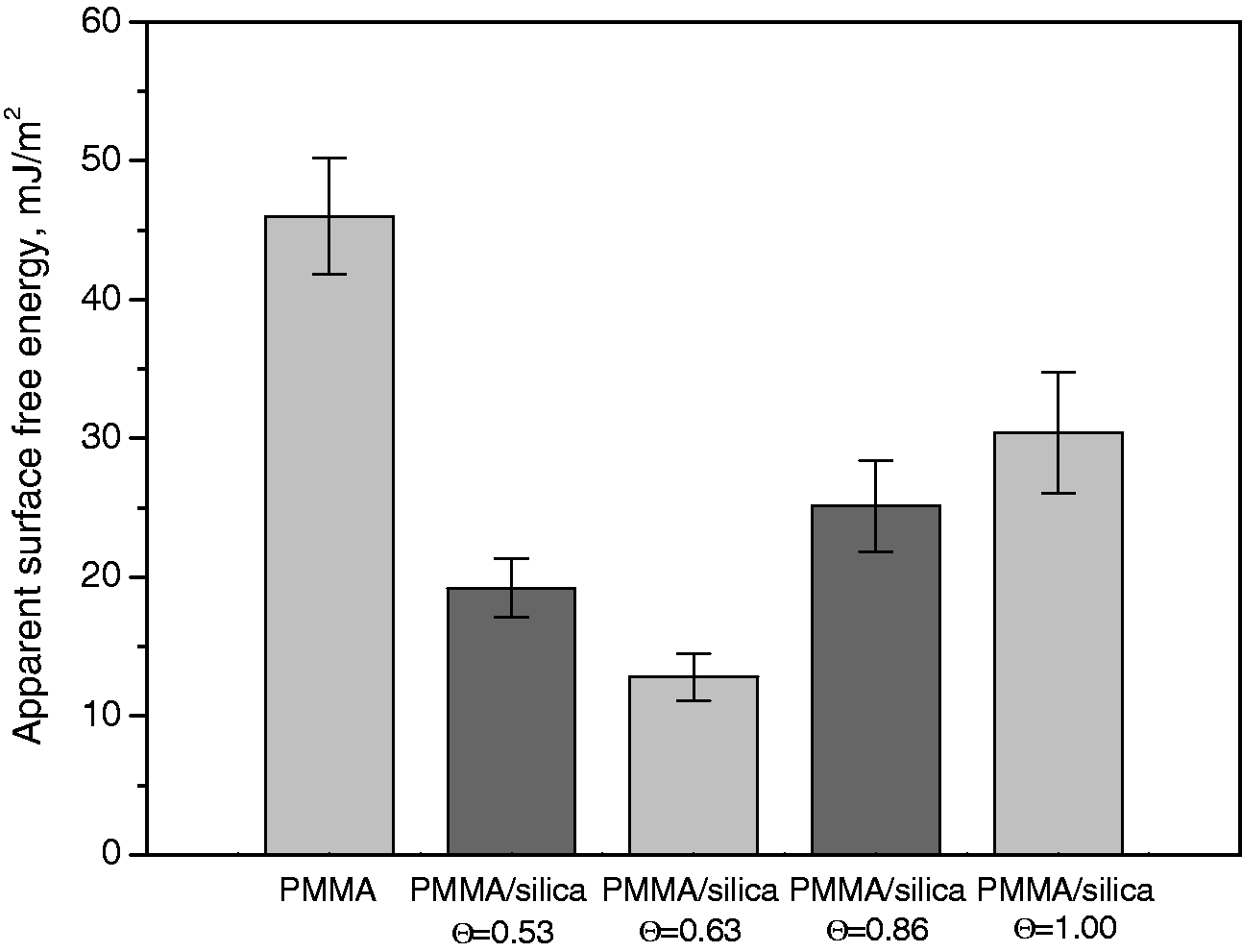
Wettability and surface free energy
Figure 1 presents the water contact angle values measured for the PMMA/silica films deposited on the glass surface with a different surface coverage (Θ: Advancing and receding contact angles of water measured on the PMMA/silica films deposited on the glass surface. PMMA: poly(methyl methacrylate). Apparent surface free energy calculated from CAH approach of PMMA/silica films deposited on the glass support. CAH: contact angle hysteresis; PMMA: poly(methyl methacrylate).
SEM analysis
The SEM micrographs of the films of PMMA and different content silica are shown in Figure 3. Angular structures of modified silica were observed in all samples. The modified silica particles are homogeneously dispersed in the PMMA/chloroform solution and the presence of big silica aggregations was not observed. Increasing of the hydrophobic group’s number on the silica surface can prevent the aggregation of the silica particles and leads to obtaining better surface properties of hydrophobic nanocomposites.
SEM images of PMMA/silica films deposited on the glass support and modified silica (Θ = 
Thermal analysis
Thermal durability of PMMA coverage of silica powders was investigated by TG, derivative thermogravimetry (DTG), and DSC. In Figure 4 the TG, DTG, and DSC curves measured for the silica nanocomposites loaded with PMMA are presented. Analyzing these dependences in the range 30–900℃ indicates that thermo-oxidation of the all samples proceeds in two main stages. The first small weight loss (about 0.2%) at the temperatures below 100℃ corresponds to removal of physically adsorbed water from hydrophobic nanoparticles. The second exothermic peaks from 120 to 950℃ are due to the decomposition of organic molecules. According to the literature (Landau et al., 2002; Yariv, 2004) the oxidation process at 200–900℃ for organic compounds generally can be divided into a few main stages: defragmentation, oxidation, and burning carbonized residue. The TGA curves of silica nanocomposites reveal a major weight loss in the temperature range from 120 to 500℃. Calculations indicate that this degradation step results in weight loss of 4.11, 3.42, 3.23, and 3.46% for PMMA/silica (Θ = TG (a), DTG (b), and DSC (c) curves for the silica nanocomposites: PMMA/silica (Θ = Tonset, Tmax, Tend and the total mass loss of the thermal processes occurred for examined silica nanocomposites. PMMA: poly(methyl methacrylate).
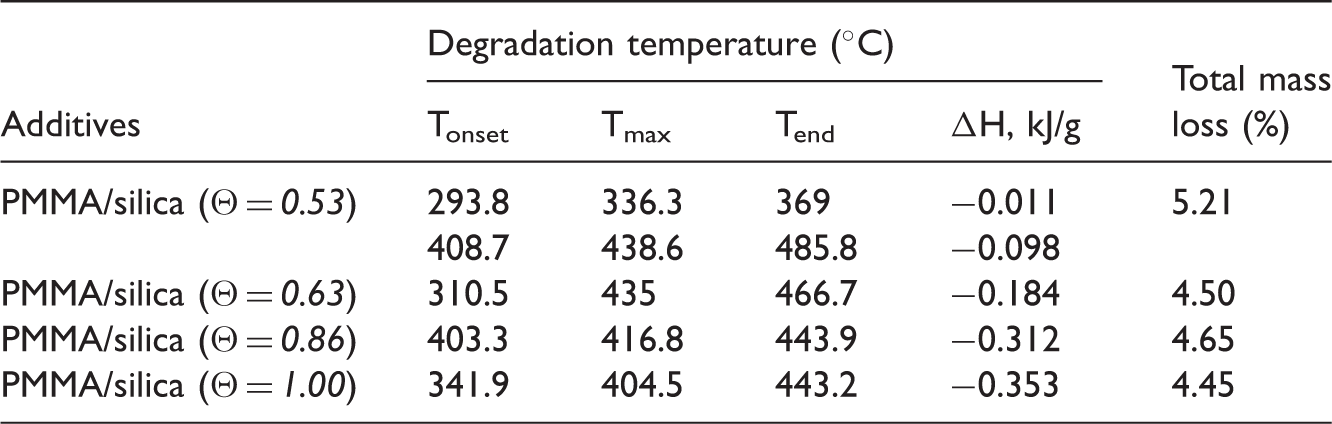
The biggest changes on the DTG curves took place for the samples modified in the presence of the highest TMS amount. In this case above 120℃ two peaks can be observed on the DTG curves. The first one with the minima at 257 and 272℃ (PMMA/silica (Θ =
For better illustration of the processes taking place during the thermal degradation of the examined samples, the measurements of the presence of gaseous products by mass spectrometry were performed. The intensity profiles of main decomposition products of PMMA (H2O (18), CO2 (44), the monomer after the loss of the entire side chain –COOCH3 (41), the monomer after the loss of –OCH3 (69), the entire monomer (100)) (Tatro et al., 2003) are presented in Figure 5. The analysis of M/Z = 41, 69, and 100 indicates that at low concentrations of PMMA (black curve) mainly defragmentation of polymer molecules is the dominant step in the temperature range 250–400℃. Due to strong interactions of polymer fragments with the silica surface, the process takes place at lower temperatures. In the case of gaseous profiles characteristic of CO2 and H2O, the appearance of distinct peaks in the temperature range 200–750℃ (with the maximum at about 300 and 450℃ for PMMA/silica (Θ = MS profiles of gaseous products characteristic of PMMA decomposition (m/z:18 (H2O), 41 (the monomer after the loss of the entire side chain (–COOCH3)), 44 (CO2), 69 (the monomer after the loss of –OCH3), 100 (the entire monomer) versus the temperature for the systems: PMMA/silica (Θ = 
Conclusions
Modification of glass surface by air plasma causes improvement of adhesion between the polymer/silica layers and the glass support which is related to the polar groups introduced on the glass surface. The contact angle values over 90° measured for hydrophobic PMMA/silica coatings and lower surface free energy calculated from the CAH approach allow to conclude that the modified silica addition leads to obtaining films with more hydrophobic properties than pure polymer coatings. From the SEM images it can be observed that silica particles are well dispersed in the PMMA matrix and aggregates are not formed.
These TG/MS data support the conclusion that the amount of monomer in the gaseous products decreases and thermal stability decreases with the increased amount of PMMA in the sample. With the increasing concentration of PMMA process in thermal degradation of the obtained composites, there are two predominant processes: defragmentation and combustion of the polymer. Moreover, the increase in enthalpy of the reaction suggests that polymer combustion is the predominant process in thermal degradation with the increasing concentration of PMMA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research has been funded by the intentional grant of Ministry of Science and Higher Education to carry out research as well as do the related tasks, contributing to the development of junior researchers and doctoral students at the Faculty of Chemistry of the Maria Curie-Skłodowska University, project titled:
