Abstract
The thermal stability and characteristics of the surface of the Fe82Nb2B14RE2 (RE = Y, Gd, Tb, Dy) amorphous alloys were studied by differential scanning calorimetry and atomic force microscopy methods. Alloying of the Fe84Nb2B14 alloy by 2 at.% of RE results to increase of nanocrystallization temperature on 110 K for amorphous Fe82Nb2B14RE2 (RE = Y, Gd, Tb, Dy). Values of the roughness increased after thermal treatment for 1 h in air due to nucleation and growth of nanocrystals in bulk and surface oxidation of the amorphous Fe82Nb2B14RE2 alloys. The nanocrystallization of Fe82Nb2B14RE2 amorphous alloys after thermal treatment increases the catalytic activity for hydrogen evolution reaction.
Introduction
The Fe-based amorphous alloys possess some outstanding physical and chemical properties (high specific strength, elastic modulus, excellent corrosion resistance and low cost), which are different from the appropriate characteristics of the conventional crystalline alloys (Botta et al., 2013; Koga et al., 2014).
The hydrogen evolution reaction (HER) has been extensively investigated using both wide scale electrode materials and solutions (Navarro-Flores et al., 2005). Different metals and alloys are used as electrode materials for HER. They exhibit high catalytic activity, chemical and corrosion stability and low cost for their production (Safizadeh et al., 2014). HERs have been widely investigated in alkaline solutions on various types of electrodes in order to get electrocatalytic materials that are effective and stable (Sequeira et al., 2011). The characteristics of the amorphous and nanocrystalline metallic electrodes can be modified by changing the elements’ composition and geometric surface area (Domínguez-Crespo et al., 2005).
The aim of this study was to investigate the morphology and electrocatalytic activity of the Fe82Nb2B14RE2 (RE = Y, Gd, Tb, Dy) amorphous and nanostructured samples for HER in alkaline solution. The alloy Fe84Nb2B14 was used as the reference alloy.
Materials and methods
Іnvestigations were carried out for five amorphous alloys in the form of strips (thickness: 20–25 µm and width: 3 mm): Fe84Nb2B14, Fe82Nb2B14Y2, Fe82Nb2B14Gd2, Fe82Nb2B14Tb2 and Fe82Nb2B14Dy2. The alloys were prepared by method melt spinning on a copper wheel with circular velocity 30 m/s at helium atmosphere were received the investigated alloys. The melts were prepared from pure Fe and B and the binary REFe2 and NbFe2 compounds. Purity (wt. %) of the initial elements was as follows: Fe – 99.99, Y – 99.96, Gd – 99.96, Tb – 99.96, Dy – 99.96 and B – 99.96.
The thermal stability of the amorphous alloys was studied by the differential scanning calorimetry (NETZSCH DSC 404) method at a scan rate of 10 K/min.
The hydrogen evolution investigations on the as-cast and annealed AMA were carried out in a three-electrode cell using potentiostatic mode method. These measurements were carried out in 1 M KOH solution, using AUTOLAB electrochemical system. The auxiliary electrode was a platinum mesh and the reference electrode was of the type Hg/HgO/1 M KOH. The hydrogen evolution was performed for 1 h. The hydrogen evolution study on the AMA was carried out five times.
The topography and morphology of the as-cast and annealed samples of Fe82Nb2B14RE2 (RE = Y, Gd, Tb, Dy) amorphous alloys were analysed using the atomic force microscopy (AFM) and the scanning probe microscope SolverProM.
Results and discussion
Figure 1 shows the DSC curves vs. temperature determined for all the examined alloys at a heating rate of 10 K/min. DSC curves exhibit two peaks indicating the two-stage crystallization process in the alloys. In Chrobak et al. (2011), it was shown that the crystallization of the Fe82Nb2B14RE2 (RE = Y, Gd, Tb and Dy) amorphous alloys proceeds in two stages. Table 1 shows the temperatures of the first (TX1) and the second (TX2) crystallization peaks for the examined alloys. The substitution of Fe atoms in the amorphous Fe84Nb2B14 alloy by 2 at.% RE leads to increase of nanocrystallization temperature on 110 K and accelerates nucleation nanocrystals of a-Fe and Fe23B6 phases with 15–20 nm in size.
DSC thermograms of the alloys: 1: Fe84Nb2B14, 2: Fe82Nb2B14Y2, 3: Fe82Nb2B14Gd2, 4: Fe82Nb2B14Tb2, 5: Fe82Nb2B14Dy2 (heating rate 10 K/min). Temperatures (±5 K) of the first (TХ1) and the second (TX2) stages of crystallization of alloys at heating rate 10 K/min.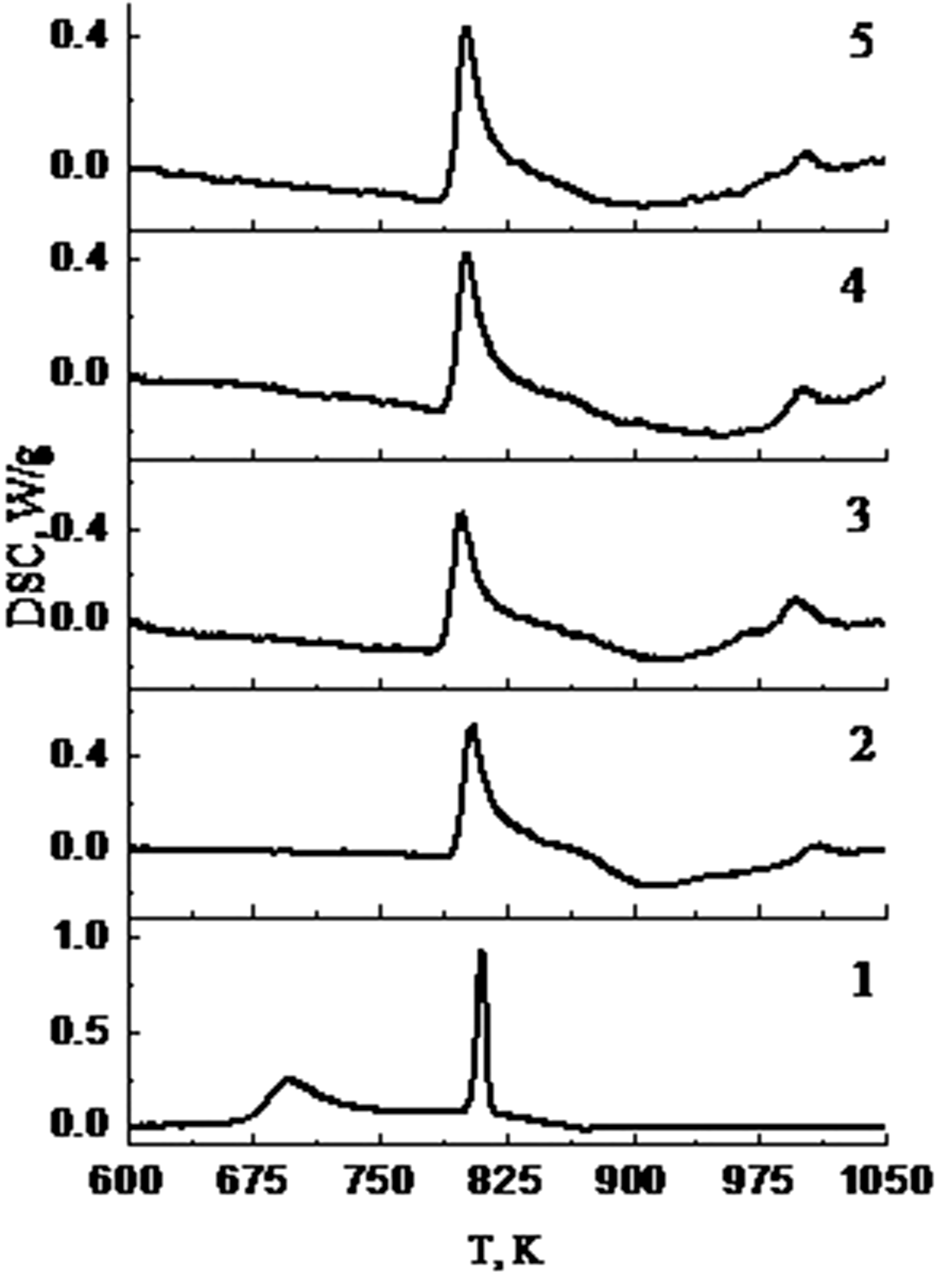

The surface relief of the initial and annealed alloys for 1 h at TX1 temperature (see Table 1) was studied by AFM. Thermal treatment of the amorphous ribbons changed the characteristics of the surface. All AMAs have amorphous structure (Chrobak et al., 2011). 2D AFM-images of the initial and the annealed Fe82Nb2B14Dy2 samples are presented in Figure 2. Figure 3 shows the distribution of the structural units vs. their dimensions of the initial and the annealed samples. The size and counts of the structural units changed on the surface of alloys after their heat treatment. For example, globules of the initial AMA Fe82Nb2B14Y2 (maximum counts of the structural units ∼540) were mostly ranging in size from 20 to 23 nm and the globules of the annealed AMA Fe82Nb2B14Y2 (maximum counts of the structural units ∼400) were mostly ranging in size from 23 to 25 nm (Figure 3).
2D AFM-images of the initial (a) and the annealed (b) Fe82Nb2B14Dy2 samples. Distribution of the structural units vs. dimensions of the initial (a) and the annealed samples (b): 1: Fe84Nb2B14, 2: Fe82Nb2B14Y2, 3: Fe82Nb2B14Gd2, 4: Fe82Nb2B14Tb2, 5: Fe82Nb2B14Dy2.

The adhesion work on phase boundaries of Fe84Nb2B14 alloy/distilled water is 88.5 mJ/m2. Alloying of Fe84Nb2B14 alloy by RE leads to increased the values of the work of adhesion. The highest values of the adhesion work observed in a contact of as-cast Fe84Nb2B14, Fe82Nb2B14Tb2 and Fe82Nb2B14Dy2 alloys with 1 M aqueous KOH solution (Figure 4).
The work of adhesion on the interface of the initial AMA – distilled water (a); initial AMA – 1 M aqueous KOH (b); annealed AMA – 1 M aqueous KOH (c). AMAs: 1: Fe84Nb2B14, 2: Fe82Nb2B14Y2, 3: Fe82Nb2B14Gd2, 4: Fe82Nb2B14Tb2, 5: Fe82Nb2B14Dy2.
Volume of evolved hydrogen for the initial and the annealed amorphous alloys.


The average surface roughness of the alloys: 1: Fe84Nb2B14, 2: Fe82Nb2B14Y2, 3: Fe82Nb2B14Gd2, 4: Fe82Nb2B14Tb2, 5: Fe82Nb2B14Dy2. The light grey and grey columns are characteristics of the initial and the annealed samples, respectively.
Conclusions
A two-stage crystallization process was observed for Fe82Nb2B14RE2 (RE = Y, Gd, Tb, Dy) and Fe84Nb2B14 amorphous alloys. Alloying of the reference alloy Fe84Nb2B14 by 2 at. % RE increases the crystallization temperatures of Fe82Nb2B14RE2 alloys for ∼110 K and their roughness as well.
Thermal treatment and alloying lead to increasing volume of evolved hydrogen for the Fe82Nb2B14RE2 amorphous alloys due to the enhancement of roughness of the electrode surface.
Footnotes
Acknowledgement
This paper was first presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
