Abstract
The influence of metal oxide type on the adsorption affinity of anionic polyacrylamide for the solid surface was studied. There were selected three metal oxides: chromium(III) oxide, zirconium(IV) oxide and aluminium(III) oxide. The adsorption behaviour of high-molecular weight polymer containing 30% of carboxyl groups was determined based on the results of spectrophotometric, electrokinetic and turbidimetric measurements. The anionic polymer exhibits the greatest adsorption on the surface of chromium(III) oxide whereas PAM affinity for the aluminium(III) oxide is the lowest. The polymer presence has a pronounced impact on the electrokinetic properties of examined suspensions manifesting in changes in pHpzc and pHiep values of PAM-containing systems compared to those without polyacrylamide. The anionic PAM addition at pH 6 has the greatest effect on the stability of metal oxide aqueous suspensions.
Keywords
Introduction
Metal oxides are widely used in many technological processes due to their good mechanical, electrical, optical and physicochemical properties. They find application in the microelectronics to create sensors and piezoelectric devices, in the fuel cells production, in anticorrosion coatings preparation and in catalysis – as catalysts and catalysts carriers (Kantorova and Vesely, 2013; She and Flytzani-Stephanopoulos, 2006; Yu et al., 2016). Additionally, metal oxides, besides activated carbons (Nowicki, 2016; Nowicki et al., 2016) and zeolites (Franus, 2012; Franus et al., 2014), play an important role in adsorptive removal of a great variety of noxious substances – both toxic gases and liquid compounds (Wawrzkiewicz et al., 2015). Another application area of these solids includes production of cosmetics, pharmaceuticals and mineral pigments (Simpson et al., 2011; Vargas-Reus et al., 2012). Due to very good biocompatibility, high mechanical strength and chemical inertness of some metal oxides (especially zirconia) they can be used in implantology in the processes of tissues regeneration (Salinas and Vallet-Regi, 2007; Silva-Bermudez and Rodil, 2013).
The above-mentioned practical applications of metal oxides require often modification of their surfaces by the adsorption process of a specific substance. Macromolecular compounds (polymers) proved to be an excellent tool to achieve the desired surface properties of different solids. Due to the fact that long polymeric chains composed of numerous segments can adopt specific conformations in the adsorption layer formed on the solid surface, a great variety of different hybrid materials can be obtained. There are a lot of parameters which affect macromolecule conformation at the solid–liquid interface (Chibowski and Krupa, 2000; Chibowski and Wiśniewska, 2001; Wiśniewska, 2011). Besides the kind of polymer (ionic, nonionic) and type of metal oxide, the most important are temperature, solution pH, supporting electrolyte presence and its concentration, addition of other low-molecular substances (surfactants, amino acids, dyes) and magnetic field action. Understanding the adsorption mechanism in such systems makes it possible to obtain the desired surface properties of the metal oxide coated by an appropriately selected polymer.
On the other hand, polymeric chains present in the adsorption layers around the solid particles determine the stability of aqueous suspensions of metal oxides (Wiśniewska, 2013). This property is essential for the colloidal systems and is the most important parameter in their characteristics. Ionic macromolecules bounded with the solid surface affect system stability through both steric and electric forces. The steric interactions are a result of spatial hindrance which prevents particles aggregation. Additionally, the charges of functional groups in the adsorbed macromolecules can cause two opposite effects: solid surface charge neutralization (resulting in the suspension stability decrease) or electrosteric stabilization of solid particles.
In the present paper, the comparison of adsorption behaviour of anionic polyacrylamide (PAM) in the aqueous suspensions of three metal oxides was presented and discussed. There were applied the following metal oxides: chromium(III) oxide – Cr2O3, zirconium(IV) oxide – ZrO2 (zirconia) and aluminium(III) oxide – Al2O3 (alumina). The adsorption and electrokinetic data enabled not only determination of mechanism of PAM binding with the solid hydroxyl groups of amphoteric character but also explanation of solid suspension stability in the polymer presence. The adsorption properties of other metal oxides in relation to PAM were also studied, for example in the systems containing Fe2O3 (Chibowski and Wiśniewska, 2002), MnO2 (Chibowski et al., 2002) and SiO2 (Wiśniewska, 2012).
Experimental
Physicochemical characteristics of applied metal oxides.

The anionic PAM (
All measurements were made in the pH range 3–9 at 25℃ in the presence of NaCl with the concentration 1·10−2 mole/dm3.
Adsorption measurements were performed using the static method. The reaction of PAM with hyamine proposed by Crummet and Hummel (1963) was applied to determine the PAM concentration in the solution after the adsorption process. The PAM solution turbidity was measured using the UV–VIS spectrophotometer (
The potentiometric titration method (Janusz, 1999) was used for the solid surface charge density determination. The special program Titr_v3 (author W. Janusz) was also applied to this end. The appropriate mass of metal oxide was added into the thermostated (
The electrophoretic mobility of metal oxide particles (without and covered with polymer) was measured using the Zetasizer Nano ZS with the universal dip cell and MPT-2 titrator (
The stability measurements of metal oxide suspensions in the absence and presence of PAM were performed using the turbidimetry method (Terpiłowski et al., 2015). For this purpose the apparatus Turbiscan LabExpert with the cooling module TLAb cooler (
Results and discussion
The analysis of the data presented in Figure 1 indicated that anionic PAM exhibits the greater adsorption on the chromium(III) oxide surface and the lowest on the aluminium(III) oxide surface. Additionally, for all examined metal oxides the adsorbed amounts of the polymer decrease with the solution pH increase. Such behaviour in the aqueous solution is the result of changes in the sign and value of the solid surface charge with the pH rise and dissociation of the PAM carboxyl groups.
Adsorbed amounts of PAM on the surfaces of chosen metal oxides at pH 3, 6 and 9.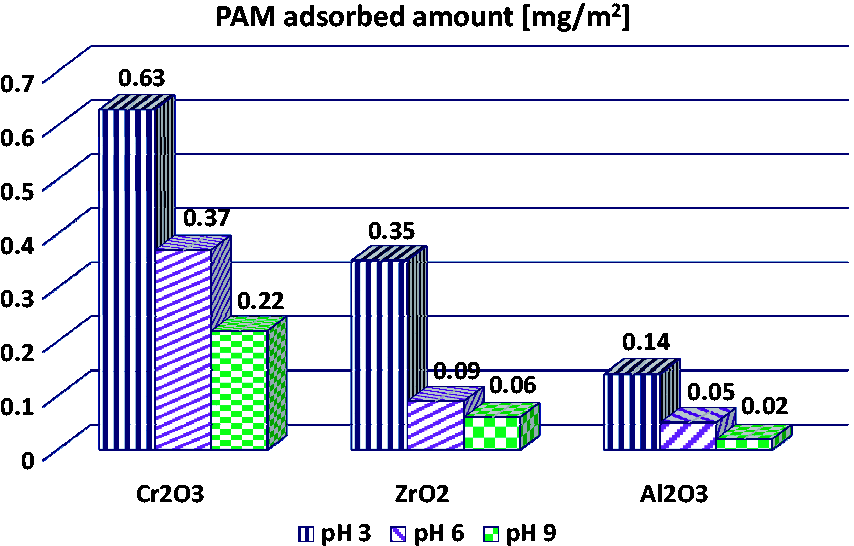
The source of metal oxide charge is their hydroxyl surface groups ≡ MeOH of the amphoteric character (where Me is the metal atom: Cr, Zr, Al). H+ ions were considered as potential-determining ions because they affect the formation of the solid surface charge according to the following acid–base reactions
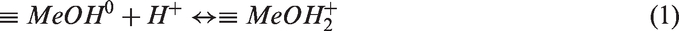

Moreover, in the solution of supporting electrolyte the reactions of the surface hydroxyl groups with Na+ and Cl− ions are also possible. According to the site-binding model (Janusz, 1999), the surface complexes can be formed in the following way


The presence of anionic PAM in the aqueous suspension of metal oxide particles affects also the solid surface charge density according to the reaction (Wiśniewska et al., 2016)

The total charge of the solid is the sum of all contributions of the above processes and at specific pH for each metal oxide it assumes the value zero (point of zero charge – pzc). The determined values of pHpzc points for all examined systems are presented in Figure 2.
Values of pHpzc for the metal oxide systems without and with the ionic polymer.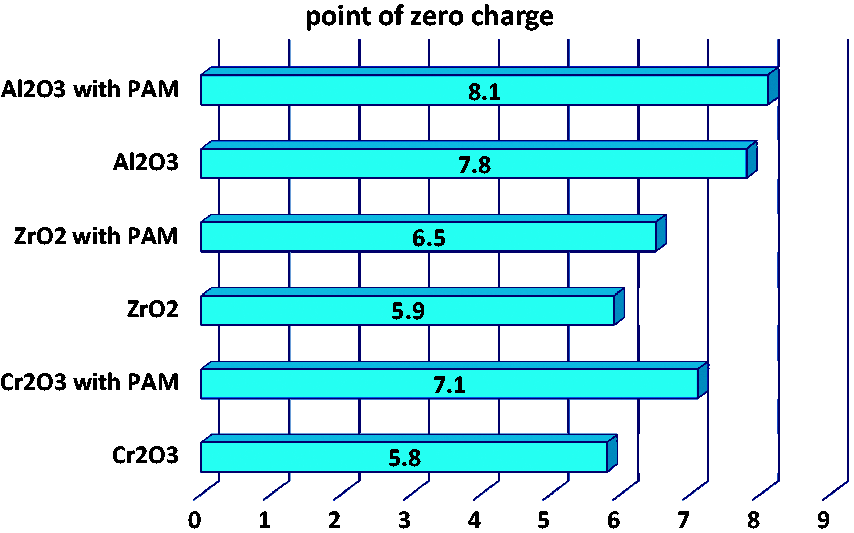
As can be seen in Figure 2 the points of zero charges for Cr2O3, ZrO2 and Al2O3 oxides are equal to 5.8, 5.9 and 7.8, respectively. Thus, the greatest adsorption level of anionic PAM at pH 3 is a result of favourable electrostatic attractions between the positively charged solid surface and the slightly dissociated polymeric chains (assuming more coiled conformation at the solid–liquid interface). The PAM adsorption is the lowest at pH 9 due to the repulsion between the totally ionized PAM macromolecules and the negative surface of the solid. Under these disadvantageous conditions the existence of hydrogen bonds between the adsorbent and the adsorbate is demonstrated. Although the charged active solid sites can contribute to formation of these bonds, the neutral ones are mainly responsible for it. This results from the fact that ≡MeOH0 groups are much more numerous than ≡MeOH2+ and ≡MeO− ones (Chibowski and Wiśniewska, 2001). The presence of hydrogen bonds in the metal oxide–polymer (containing carboxyl groups) system was proved by our previous studies (Wiśniewska et al., 2009) concerning desorption measurements of radioactive ions caused by macromolecules, as well as calculations from Pradip equation based on zeta potential data and from Langmuir equation based on adsorption data.
The greatest adsorption of anionic PAM on the chromium(III) oxide surface is probably caused by the greatest concentration of solid hydroxyl groups per unit surface area of the metal oxide. In such situation the considerable number of polymeric segments can interact directly with the solid hydroxyl groups and as a consequence, the adsorbed amounts of PAM are the largest on the Cr2O3 surface.
Such adsorption mechanism is confirmed by the greatest difference in the pHpzc values obtained for the Cr2O3 suspension in the PAM presence in comparison to the system without polymer (Figure 2). This difference for Cr2O3 equals 1.3 (for ZrO2 it is 0.6 and for Al2O3 only 0.3).
The adsorption of anionic polymer containing negatively charged functional groups at the interface enforces the formation of an additional number of positive surface sites (according to reaction (5)). As a consequence, the solid surface charge density of metal oxide increases and the pHpzc point is shifted towards the higher pH values (Wiśniewska et al., 2015). This shift is the greatest in the case of chromium(III) oxide due to the highest adsorption of anionic PAM on its surface, which is related to more numerous polymeric segments bonded directly with the Cr2O3 hydroxyl groups (in train-type structures of adsorbed PAM chains).
The electrokinetic (or zeta) potential is a very important parameter characterizing the solid particle interaction in the aqueous media related to such systems stability. The absolute value of ζ potential is decisive for the about suspension aggregation or stabilization (Jesionowski, 2003; Klapiszewski et al., 2013). As can be seen in Figure 3 the isoelectric points (iep) of metal oxides without the polymer are located at pH values 6, 6.4 and 8.4 for Cr2O3, ZrO2 and Al2O3 adsorbents, respectively. Some differences between the pHpzc and pHiep values of applied oxides are a result of overlapping of the electrical double layers formed on the mesopores surface existing in the solids structure (Skwarek, 2014, 2015).
Values of pHiep for the metal oxide systems without and with the ionic polymer.
The presence of anionic polymer layer around the solid particles results in a significant decrease of the zeta potential values and the considerable shift of the pHiep to lower pH values. The differences between the pHiep values obtained for the solid suspension in the PAM presence in comparison to the system without polymer are as follows: for Cr2O3 – 2.5, for ZrO2 – 2.7 and for Al2O3 – 5.1. This is a completely opposite tendency compared to that obtained for the surface charge density characteristics and pHpzc location. The main reason for zeta potential lowering in the anionic polymer presence is the occurrence of negatively charged carboxyl groups of the adsorbed polymer macromolecules in the area of slipping plane around the solid particle (M’Pandou and Siffert, 1987). These groups belong to the polymeric segments located in the loop and tail structures of the adsorbed PAM chains. For this reason these groups are not directly bound with the solid surface and exist in the border of the stiff and diffusion parts of the electrical double layers formed on the solid particle surfaces. The shift of slipping plane due to the polymer adsorption can also play an important role in the zeta potential decrease in the systems under consideration. The more extended conformation (towards the bulk solution) is assumed by the adsorbed macromolecules, the greater shift of slipping plane is observed. The greatest difference between the pHiep points location (suspension with PAM in comparison to the system without PAM) is obtained for the aluminium(III) oxide. This is another evidence for correctness of the assumed mechanism of anionic PAM adsorption. The lowest polymer adsorption level on the Al2O3 surface is associated with more extended conformation of PAM macromolecules at the solid–liquid interface which results from a lower concentration of the hydroxyl groups on the alumina surface. As a consequence, the shift of slipping plane is considerable and the probability of negatively charged PAM carboxyl groups occurrence in this plane area is greater. It leads to the most pronounced modification of the electrokinetic properties of alumina suspension after the anionic PAM addition.
The polymer presence at pH 6 has the greatest impact on the metal oxide suspension stability. The main reason is the fact that at the pH value, which is close or equal to the isoelectric points of all examined solids, the metal oxide particles dispersed in the aqueous media (without PAM) show a strong tendency towards aggregation which is evidenced by high values of TSI stability coefficients (Figure 4) changed from 61 for Cr2O3 to 71.6 for Al2O3.
Values of TSI stability coefficients for the metal oxide suspension without and with polyacrylamide at pH 6. TSI: turbiscan stability index.
The anionic PAM addition causes considerable improvement of metal oxide suspension stability and this effect is the greatest for the alumina-containing system – the TSI decrease by 57.8 units (for the Cr2O3 system the reduction of TSI is 27.8 and for the ZrO2 system - 33.2). The adsorption of completely dissociated polymer chains on the solid surface at pH 6 results in appearance of electrosteric repulsion of PAM adsorption layers covered by metal oxide particles. The most extended conformations of the PAM macromolecules on the alumina particles surface provide the most efficient repulsion of solid particles preventing successfully their aggregation.
Conclusions
High-molecular anionic PAM exhibits the greatest adsorption affinity for the surface of chromium(III) oxide. This is probably caused by the greatest concentration of the amphoteric hydroxyl groups on the Cr2O3 surface. The evidence of this is the biggest difference in the position of pHpzc points between the Cr2O3 suspension without and with PAM. The polymeric segments (occurring in train structures) directly bonded with numerous ≡CrOH0 groups are responsible for the pronounced increase of the chromium(III) oxide surface charge density (pHpzc shift towards the higher pH values). On the other hand, the more extended conformation of the adsorbed PAM chains in the aluminium(III) oxide system (the lowest adsorption affinity of the polymer for ≡AlOH0 surface groups) results in considerable reduction of the suspension zeta potential and shift of pHiep point towards the more acidic pH values. This is due to both pronounced displacement of slipping plane towards the bulk solution and occurrence of the greatest number of negatively charged PAM carboxyl groups (belonging to the loop and tail structures) in this plane area. The PAM addition to the aluminium(III) oxide aqueous suspension at pH 6 has the greatest impact of the solid suspension stability. The lowest PAM adsorption on the Al2O3 surface and the most developed conformation of macromolecules at the solid–liquid interface provide the most efficient electrosteric repulsion between the metal oxide particles covered with the polymeric layers.
Footnotes
Acknowledgements
First presented at the 15th Ukrainian-Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and Their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
