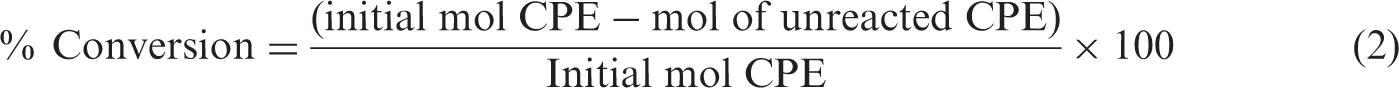Abstract
The system hydrogen peroxide/sodium bicarbonate/manganese sulfate was used for the first time to epoxidize cyclopentene. Effects of parameters such as type and amount of solvent, ratio of hydrogen peroxide and manganese sulfate to cyclopentene, presence of additives, and reaction time and temperature on the selectivity to cyclopentene oxide were evaluated. Gas chromatography was used to quantify residual cyclopentene and produced cyclopentene oxide using the internal standard method. Type and amount of solvent, addition method, and temperature were important factors to increase the selectivity to cyclopentene oxide. Unlike previous reports on epoxidation of different substrates, additives like sodium acetate and salicylic acid did not improve the selectivity to cyclopentene oxide. One time, single-step addition of hydrogen peroxide/sodium bicarbonate to the solution of cyclopentene/solvent/manganese sulfate produced more cyclopentene oxide than stepwise addition. The maximum selectivity obtained was 56%, possibly due to the high reactivity of cyclopentene that causes the formation of oxidation products different to cyclopentene oxide, which were not detected in the analyzed phase.
Keywords
Introduction
Oxidation reactions are important in the petrochemical and fine chemicals industry, and in processes to clean the environment (Wang and Yang, 2015). Epoxides are examples of oxidation products used as intermediates in the synthesis of fine chemicals (Corma and García, 2002; Goodman, 2008). Epoxidation reactions have been performed under a wide variety of conditions such as methods developed by Sharpless, Katsuki-Jacobsen, and Shi (Fingerhut et al., 2015) for the enantioselective epoxidation of allylic alcohols or olefins. In the Sharpless–Katsuki method, chiral epoxides of allylic alcohols could be obtained with a catalytic system that contained diethyltartrate, titanium tetraisopropoxide, and terbutylhydroperoxide as oxidant (Sharpless and Katsuki, 1980).Katsuki and Jacobsen used Mn III (salen) complexes as catalyst in epoxidation processes of olefins (Adam et al., 2000). The epoxidation system developed by Shi (Wang et al., 1997) was based on a fructose-derived ketone as catalyst and peroxomonosulfate as oxidant.
In other epoxidation systems, hydrogen peroxide is frequently used as a terminal oxidant; it is consider environmentally benign compared to other reagents (Lane and Burgess, 2003), since water is one of the by-products. Tungstic acid and its derivatives, zeolites, hydrotalcites, polyoxometalates, manganese complexes, and its salts have been used as epoxidation catalysts (Lane and Burgess, 2003).
Yao and Richardson (2000) reported an epoxidation method in which bicarbonate and hydrogen peroxide were used at almost neutral pH. The bicarbonate-activated peroxide system was used to epoxidize water-soluble alkenes with acetonitrile as solvent. The reaction time was from 3 to 48 h.
Lane and Burgess (2001) and Lane et al. (2002) achieved the selective epoxidation of slightly water-soluble alkenes using a combination of manganese (II) as catalyst, sodium bicarbonate buffer, hydrogen peroxide as oxidant, and N,N-dimethylformamide (DMF) or t-Butanol (t-BuOH) as solvent. They also used additives like sodium acetate or salicylic acid, in order to increase the selectivity toward the epoxides. Qi et al. (2010) used a similar system to epoxidize alkenes, but instead of a water-soluble Mn(II) salt, they used Mn-exchanged zeolites as catalysts.
Cyclopentene (CPE) can be oxidized under different conditions to obtain products such as glutaraldehyde, cis and trans 1,2-cyclopentenediol, cyclopentene oxide (CPO) (Xue et al., 2010), cyclopentanone, 1-cyclopenten-1-ol, 2-cyclopenten-1-ol (Zhang and Du, 2013), penta-1,4-dien-3-ol, and carbon dioxide and water under total oxidation conditions (Kluson et al., 2005). The oxidation products of CPE may be applied not only in the fine chemicals industry but also as bactericides, tanning agents, and in protein crosslinking (Yuan and Chen, 2011). The selective oxidation of CPE to each product is important to reduce separation processes. CPE was not among the alkenes used as substrate in the bicarbonate-activated hydrogen peroxide system, with or without manganese (II) reported by Lane and Burgess (2001, 2003) and Lane et al. (2002) or by Yao and Richardson (2000).
Methods to epoxidize CPE using conditions with more contaminant or expensive reagents than NaHCO3/Mn(II) have been reported. CPE epoxidation and pH-controlled hydrolysis of CPO to produce 1,2-cyclopentanediol was achieved using xylene as solvent, hydrogen peroxide, sodium tungstate, phosphoric acid, and a phase transfer catalyst (Meshechkina et al., 2015). CPE epoxidation was also performed using cobalt (II) perchlorate as catalyst and 3-chloroperoxybenzoic acid as oxidant (Hyun et al., 2012).
A challenge in the oxidation process of CPE is to obtain a high selectivity toward the epoxide at a high CPE conversion. The reports of the epoxidation of CPE to CPO using titanium-based catalysts like Ti-MWW, TS-1, Ti Beta, and TiO2 showed selectivity to the epoxide of 92–98% at CPE conversions around 25%, and at conversion of 87%, the selectivity was around 21%. The maximum selectivity achieved at a conversion of 96% was around 49% in a gas phase reaction (Hulea et al., 1998; Kluson et al., 2005; Wu et al., 2006).
We report the epoxidation of CPE with sodium bicarbonate/hydrogen peroxide/manganese (II), a less expensive and environmentally benign system, which has not been reported in the epoxidation of this substrate. We also evaluated the effect of factors such as temperature, time, molar ratio of reactants, type of solvent, additives like sodium acetate or salicylic acid, buffer, and addition method on selectivity toward CPO and conversion of CPE. Evidence of the manganese oxidation state change during the reaction was obtained by visible spectroscopy.
Experimental
General conditions used in the experiments are presented in this section. Details are specified in the “Results and discussion” section.
Solution 1 was prepared by stirring a certain amount of solvent (DMF or t-BuOH—Carlo Erba 99.5%), CPE (97% Alfa Aesar), and catalyst (manganese sulfate monohydrated, ACS reagent Merck) in a 25 ml double-neck round-bottom flask adapted to a condenser. A water bath was used to control the temperature to the desired range (3–5, 13–15, or 20–22℃).
Solution 2 was prepared in a different flask. A defined volume of hydrogen peroxide (30 wt% Carlo Erba, 1.1 g/ml) was added to a 0.2 M sodium bicarbonate buffer solution at 1℃. A measured volume of solution 2 was added either slowly or in one step to solution 1. Different conditions of temperature (3–5, 13–15, or 20–22℃) and reaction time were tested (from 0.25 to 6 h). After the reaction time was completed, the sample was evaluated by gas chromatography analysis.
A gas chromatograph, equipped with a flame ionization detector (GC-FID) (GC-8610C, SRI Instruments, California, USA) was used to quantify the unreacted CPE and CPO obtained during the catalytic reaction. Toluene (99.5% PS Panreac), internal standard, was added to the round-bottom flask, then 5 ml of a 10% w/v sodium chloride solution was added and the mixture was transferred to a separatory funnel and mixed with ethyl ether (ACS JT Baker). The aqueous phase was separated and the organic phase was dried with sodium sulfate. Then 0.5 ml of the organic phase was mixed with 5 ml of acetone (Suprasolv for GC) for GC analysis.
GC-FID analyses used an on-column injection, MXT-1 (30 m, 0.530 mm, 3 µm, 3.5″ coil) column, with a temperature program as follows: 36℃, 2 min, 30℃/min; 100℃, 2 min, 30℃/min; 140℃, 5 min. The detector temperature was held at 160℃. Calibration curves in t-BuOH or DMF were prepared at three different concentration levels. The same extraction procedure for the samples was applied to the standards. The percentage of conversion of CPE and the percentage of selectivity toward CPO were calculated using equations (1) and (2)
Results represent the average of three analyses.
Genesys 10 S UV–Vis was used to run spectra of samples of the reaction mixture taken at different time intervals (composition 51 mmol DMF: 1.6 mmol CPE: 0.024 mmol MnSO4: 0.1 mmol NaHCO3: 4.35 mmol H2O2. Temperature 3–5℃).
Results and discussion
Effect of solvent
Effect of solvents in epoxidation of cyclopentene.

CPE: cyclopentene; DMF: N,N-dimethylformamide; t-BuOH: t-Butanol.
Solution 1: 4 ml of solvent; 0.35 g CPE; and 0.009 g MnSO4·H2O. Solution 2: 5 ml of buffer 0.2 M NaHCO3 and 2 ml 30 wt% H2O2. 2 ml of solution 2 was added to solution 1 in a 30 min period. The total reaction time was 1 h at 20℃.
CPE was not epoxidized when t-BuOH was used as solvent under the reaction conditions of experiment 2 (Table 1). However, CPE was oxidized in the presence of t-BuOH after 24 h of reaction at 20℃ (Table 1, Experiment 3). Two phases were observed when t-BuOH was used as solvent; CPE is a slightly polar molecule (μ: 0.9 D) (Beyer et al., 2000) and does not form hydrogen bonds as t-BuOH. The low solubility of CPE in t-BuOH made the reaction slower (Hulea et al., 1998).
The homogeneity of the reaction system in DMF improved conversion and selectivity to CPO. DMF is nonhydrogen bonded, highly polar, aprotic, protophilic, a potentially basic medium, miscible with water, alcohols, ethers, aromatic hydrocarbons, with a strong electron pair donating and accepting ability, suitable solvent for salts, and with ability to form complexes (Venkatesu, 2010). These properties make DMF a suitable solvent for this reaction system that contains a slightly polar substrate, aqueous oxidant, manganese salt, and sodium bicarbonate buffer. In a homogeneous system the catalysts are more active and selective than in heterogeneous systems (Farnetti and DiMonte, 2009). Solvents like dimethylsulfoxide, with properties similar to DMF, could be tested in epoxidation of CPE. But, not only solubility but also interactions between solvent and species in the reactant media may affect the conversion of CPE and selectivity to CPO.
CPE reacted in the bicarbonate/peroxide/manganese system under the conditions of experiments 1 and 3, however the selectivity to CPO was low. Epoxides in the presence of water may suffer ring opening to give the corresponding diol (McMurry, 2008).
Effect of buffer
Experiments of CPE epoxidation were conducted under the following reaction conditions (molar ratio: H2O2/CPE 1.1; MnSO4/CPE: 0.01; solvent DMF; temperature 20℃, time 1 h) using two different buffer solutions: a 0.2 M NaHCO3/7.4 × 10−5 M CH3COONa pH 7.8 solution and a 0.2 M NaHCO3/9.4 × 10−4 M Na2CO3 pH 7.3. Conversion and selectivity were 72 and 27%, respectively, in both cases. There was not a significant effect of the presence of sodium carbonate or sodium acetate on the selectivity to the epoxide. The low concentration of sodium acetate or sodium carbonate does not affect conversion and selectivity. There is not a significant effect on selectivity toward the epoxide by using solutions of 0.2 M of sodium bicarbonate with pH adjusted to values ranging from 6.7 to 8 (results not shown here). This result is consistent with previous reports where the selectivity to epoxide is almost constant when pH of the sodium bicarbonate solution is less than 8 and drops drastically at pH values from 8.2 to 9 (Lane et al., 2002).
Effect of molar ratio of reagents
Effect of H2O2/CPE molar ratio
To evaluate the effect of the molar ratio of hydrogen peroxide to cyclopentene (H2O2/CPE) on the selectivity to CPO and conversion of CPE, different volume of hydrogen peroxide was added to solution 2. The results are presented in Figure 1.
Effect of molar ratio of H2O2 to CPE on conversion and selectivity. Solution 1: 2 ml of DMF, 0.1 g CPE, 0.004 g MnSO4·H2O, stirred at a temperature between 3 and 5℃. To this mixture, 1 ml of solution 2 (prepared by adding a volume of 1, 3, 4, or 5 ml of hydrogen peroxide 30 wt% to 5 ml of buffer 0.2 M NaHCO3/sodium acetate pH 7.8 at 1℃) was added in a single step. Molar ratio H2O2/CPE: from 1 to 3.1; MnSO4/CPE: 0.015; NaHCO3/Solvent: 0.005. Reaction time: 1.25 h. CPE: cyclopentene; DMF: N,N-dimethylformamide.
Increasing the molar ratio of H2O2 to CPE from 1 to 2.5 increases the conversion of CPE from around 90 to 100%, CPE can be oxidized under the conditions studied and the selectivity to CPO increases from 20 to 48.5%. Further increases in the molar ratio H2O2/CPE caused a decrease in selectivity and conversion remains around 100%. The reduction in selectivity to CPO is associated with the oxidation of CPE to compounds different from CPO. The oxidation of CPE with hydrogen peroxide yields a complex mixture of products such as CPO, 2-cyclopentene-1-one, 1,2-cyclopentanediol, a monoether, and glutaraldehyde (Lu et al., 2010). CPO and traces of cyclopentenone and formamide N-formyl N-methyl were detected by GC–MS analysis of the ether extract of experiment performed with molar ratio H2O2/CPE around 2.9.
A mixture of sodium carbonate and hydrogen peroxide was prepared in solution 2. The volume of sodium carbonate buffer solution was kept constant at 5 ml while the volume of hydrogen peroxide was varied from 1 to 5 ml. The equilibrium reaction between hydrogen peroxide and sodium bicarbonate forms sodium peroxymonocarbonate (Bakhmutova-Albert et al., 2010), active species in the epoxidation of CPE. Increasing the volume of hydrogen peroxide increases the concentration of peroxymonocarbonate ion available for epoxidation and maximum selectivity to CPO was obtained. Further increases in hydrogen peroxide at constant concentration of sodium bicarbonate cause a decrease in selectivity, maybe there is excess hydrogen peroxide that generates free radicals, which react to form oxidation products different from CPO.
Effect of molar ratio MnSO4·H2O/CPE
The effect of molar ratio of MnSO4·H2O to CPE was evaluated by adding different amounts of manganese sulfate to the reaction mixture. The results are presented in Figure 2.
Effect of molar ratio MnSO4/CPE on the cyclopentene epoxidation. Solution 1: 2 ml of DMF; 0.1 g CPE; 0; 0.004; 0.007 or 0.01 g of MnSO4·H2O, stirred at a temperature between 3 and 5℃. To this mixture, 1 ml of solution 2 (4 ml hydrogen peroxide to 5 ml buffer NaHCO3/sodium acetate pH 7.8) was added in one single step. Molar ratio H2O2/CPE: 2.6; MnSO4/CPE: 0–0.036; NaHCO3/solvent: 0.004. Reaction time 1.25 h, Temperature 3–5℃. CPE: cyclopentene; DMF: N,N-dimethylformamide.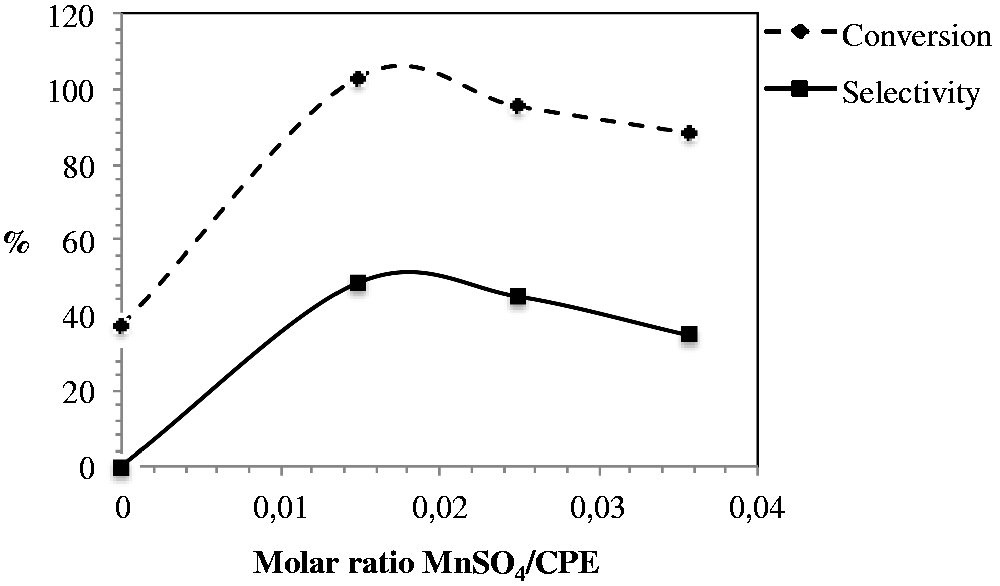
Maximum conversion of CPE and selectivity to CPO was achieved when a 0.015 molar ratio MnSO4/CPE was used. Under the reaction conditions used, a ratio of MnSO4/CPE smaller or larger than 0.015 causes a decrease in conversion and selectivity. In the absence of manganese sulfate, CPO was not detected and a 40% conversion of CPE was obtained.
CPE was transformed even when manganese was not present in the reaction media but at a low conversion. This may be due to the high reactivity of CPE, the most reactive among the cycloalkenes (Suresh et al., 2000). CPO was obtained with maximum selectivity when a 0.015 MnSO4/CPE molar ratio was used. According to Quiñonero et al. (2009), a complex Mn(II) peroxymonocarbonate is formed in the system Mn(II)/hydrogen peroxide/sodium bicarbonate. This complex is involved in the mechanism of olefin epoxidation by a one-electron transfer and oxygen atom transfer. During the oxidation process, Mn changes from Mn(II) to Mn(IV) and Mn(III) and at the end of the reaction returns to Mn(II).
Increasing the amount of manganese sulfate beyond a 0.015 MnSO4/CPE molar ratio showed a decrease in selectivity to epoxide. Manganese (II) or (IV) compounds may behave as Lewis acids, and increasing their concentration may increase the tendency of the Lewis acid to catalyze the ring opening of sensitive epoxides to diols (Lane et al., 2002). The conversion of CPE decreases as the amount of manganese in the media is increased. This may be due to the increased rate of decomposition of hydrogen peroxide that is associated with the increased concentration of manganese (Do et al., 2009).
Effect of additives
The conversion of CPE and selectivity to CPO in presence of salicylic acid or sodium acetate as additives did not improve. Lane et al. (2002) used 4 mol% of salicylic acid in epoxidation systems with H2O2/NaHCO3 in DMF to increase the epoxide yield of several substrates. In our case, when 4 mol% of salicylic acid was added to the reaction mixture in DMF, the conversion of CPE was 40% and CPO was not detected. Reactions performed under the same conditions but without salicylic acid produced CPO with a CPE conversion of 76%. In the presence of salicylic acid, the conversion and selectivity were similar to those obtained when Mn was not added. This result might be explained by the possible formation of complexes of manganese–salicylic acid (Lal et al., 2006), making manganese ions unavailable to participate in the epoxidation reaction.
Sodium acetate effect on epoxidation of CPE.
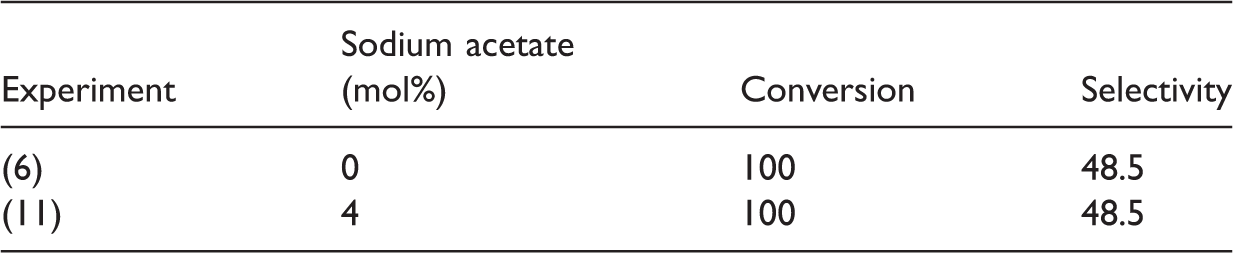
CPE: cyclopentene.
Molar ratio of reagents; H2O2/CPE: 2.6; MnSO4/CPE: 0.01, NaHCO3/solvent (DMF): 0.004, temperature 3–5℃, time 1.25 h.
An activation route of hydrogen peroxide in presence of acetic acid involves the formation of peroxyacids like peroxyacetic acid (Drago et al., 1996). The fact that there was not an effect after addition of sodium acetate on the 4 mol% on conversion of CPE and selectivity to CPO might be due to the pH of the media, since in basic media there is no formation of the peroxyacetic species or the concentration of the additive was not sufficient, since peroxyacids have been used as stoichiometric oxidants (Drago et al., 1996). Lane et al. (2002) explained the role of sodium acetate and salicylic acid as carboxylates that may form bridged dimer or oligomer catalytic species.
Effect of ratio CPE/solvent
The effect of the concentration of the system is shown in Figure 3. Reduction of the amount of solvent decreases the conversion and selectivity to CPO.
CPE concentration effect on conversion and selectivity. Solution 1: 1, 2, 4, or 6 ml of DMF; 0.1 g CPE; 0.004 g of MnSO4·H2O, stirred at a temperature between 3 and 5℃. To this mixture, 1 ml of solution 2 (4 ml hydrogen peroxide added to 5 ml buffer NaHCO3/acetate pH 7.8) was added in a single step. The reaction time was 1.25 h except for the experiment with a 2.9 molar ratio CPE/DMF with a reaction time of 2 h. Molar ratio H2O2/CPE: 2.6; MnSO4/CPE: 0.015; NaHCO3/solvent: 0.002–0.009. CPE: cyclopentene; DMF: N,N-dimethylformamide.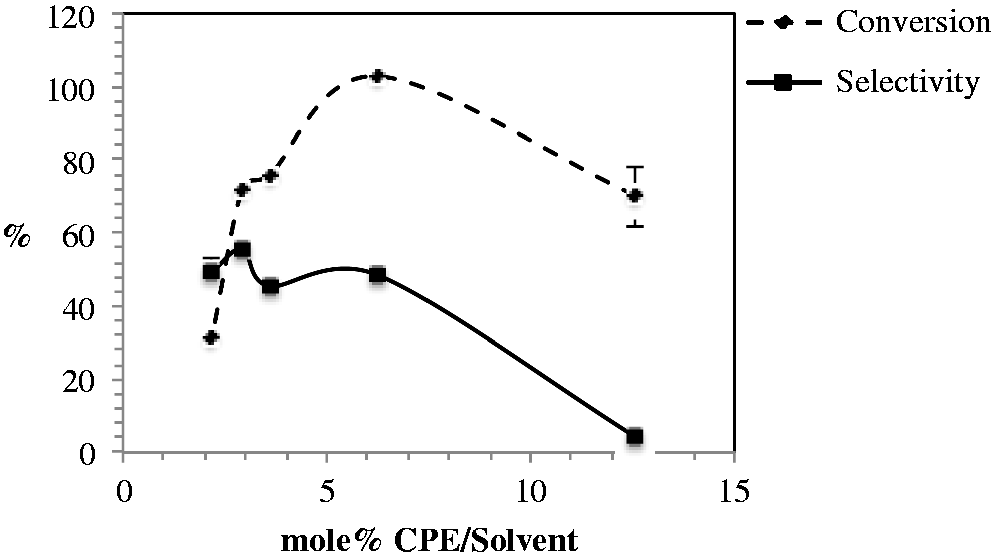
Experiments were run under the same conditions of temperature, time, and amount of reagents but changing the volume of DMF. CPE was soluble in all the experiments. At a 2.1 mol% CPE/DMF, the conversion of CPE was 30% and selectivity to CPO was 49% (in an experiment not shown here, with a 1.5 mol% CPE/DMF there was no conversion of CPE). In more concentrated systems, with 2.9 and 3.6 mol% CPE/DMF, the conversion of CPE increases from 76 to 100%. As the volume of DMF is reduced, the concentration of reagents increases and its interaction also increases favoring the oxidation of CPE. Further increase in concentration to 12.6 mol% CPE/DMF, by reduction of the volume of DMF, decreases the conversion of CPE and reduces significantly the selectivity to CPO. Since selectivity to epoxides has been previously associated to the presence of the peroxymonocarbonate, the reduction in selectivity to CPO may be explained by a decreased solubility of the complex Mn-peroxymonocarbonate. Lane and Burgess (2001) observed that solubility problems reduced the epoxide yield.
Effect of reaction time
Figure 4 presents the effect of reaction time on the conversion of CPE and selectivity to CPO at two different molar ratios CPE/solvent (3.1 mol% CPE/DMF and 6.2 mol% CPE/DMF). Each data point corresponds to a reaction run during the reaction time established. The time was measured from the addition of solution 2 to the reactor until the reaction solution was mixed with sodium chloride and ethyl ether for extraction. Increasing reaction time increases conversion and selectivity until a maximum is reached. The experiment performed at a 6.2 mol% CPE/DMF with a reaction time of 15 min had a conversion of 91% and selectivity of 17%. At a reaction time of 1.25 h the maximum selectivity of 48% was achieved (Figure 4(a)). In a more diluted reaction system with a 3.1 mol% CPE/DMF, the maximum selectivity of 56% with a conversion of 72% was obtained after 2 h of reaction (Figure 4(b)).
Effect of time on conversion of CPE and selectivity to CPO. (a) 25.6 mmol DMF: 1.6 mmol CPE: 0.024 mmol MnSO4: 0.1 mmol NaHCO3: 4.35 mmol H2O2. (b) 51 mmol DMF: 1.6 mmol CPE: 0.024 mmol MnSO4: 0.1 mmol NaHCO3: 4.35 mmol H2O2. Temperature 3–5℃. Solution 1: (a) 2 ml DMF or (b) 4 ml DMF; 0.1 g CPE; 0.004 g MnSO4·H2O, temperature 3–5℃, add 1 ml of solution 2 (4 ml of hydrogen peroxide added to 5 ml 0.2 M NaHCO3/CH3COONa pH 7.8) in a single step to the reactor. CPE: cyclopentene; DMF: N,N-dimethylformamide.
The conversion of CPE increases with reaction time in experiments performed with two different mol% CPE/DMF. The reaction rate increased when the mol% CPE/DMF increased from 3.1 to 6.2, but the selectivity to CPO decreased. The selectivity to CPO was 48% at 6.2 mol% CPE/DMF and 56% at a 3.1 mol% CPE/DMF. The selectivity was favored in a more diluted system. Selective epoxidations are obtained in systems where the heterolytic activation of hydrogen peroxide predominates over the homolytic activation. Free radicals oxidations are fast and not selective (Neimann and Neumann, 2000). Free radicals predominate in the reactions run with a 6.2 mol% CPE/DMF.
DMF may reduce the reactivity by strong interaction with reagents. According to previous reports, DMF not only has a role as solvent in epoxidation reactions but also interacts with cations of transition metals. Suzuki et al. (2013) reported the formation of the complex cation [Mn(DMF)6]2+ when a manganese(II) acetate was dissolved in DMF. DMF also participates in the formation of active species in the transition state (Lu et al., 2015; Opre et al., 2007).
Dilution reduces the rate of the reaction, decreasing the possibility of side reactions that affect the selectivity to the epoxide.
Effect of temperature
Experiments were performed at three different temperature ranges, 3–5, 13–15, and 20–22℃. Low temperature favors selectivity to CPO (Figure 5). At 20℃ the conversion was 100% but the lowest selectivity was obtained (27%). When the reaction was performed at temperatures between 3 and 5℃, 97% conversion and 44% selectivity were obtained.
Variation of conversion and selectivity with reaction temperature, 25.3 mmol DMF: 1.6 mmol CPE: 0.02 mmol MnSO4: 0.11 mmol NaHCO3: 4.3 mmol H2O2, reaction time: 1 h. Solution 2 (4 ml H2O2, 5 ml 0.2 M NaHCO3 pH 7.8) added in one step to solution 1. CPE: cyclopentene; DMF: N,N-dimethylformamide.
The epoxidation reaction is exothermic. Low reaction temperature may help the production of CPO (Lane and Burgess, 2001; Quiñonero et al., 2009) and decrease the ring opening reaction avoiding the formation of other compounds.
Effect of addition method
Addition method and conversion and selectivity to CPO.

CPE: cyclopentene; CPO: cyclopentene oxide; DMF: N,N-dimethylformamide.
1 ml of solution 2 (4 ml of H2O2 30 wt% mixed with 5 ml of 0.2 M NaHCO3/acetate buffer pH 7.8 at 1℃) added to solution 1 (DMF, CPE, and 0.004 g MnSO4·H2O) either in one step or at a rate of 33 µl/min; temperature: 3–5℃, reaction time: 2 h.
Different reports suggest a stepwise addition of the solution hydrogen peroxide/sodium bicarbonate to minimize side reactions like the decomposition of hydrogen peroxide and to avoid inadequate heat transfer, since the reaction is exothermic. Also, stepwise addition reduces the concentration of active species like
Contrary to those reports, an improvement in selectivity was obtained by adding the mixture in a single step. The selectivity toward CPO was 30% when the mixture NaHCO3/H2O2 was added to solution 2 at a rate of 33 µl/min and increased to 44% when solution 2 was added at once. The effect of heat of reaction on selectivity was counteracted by keeping the temperature of the system at 3–5℃, instead of 20–25℃, which is the temperature reported by Lane et al. (2002).
The color of the reaction mixture changed during the course of reaction, from colorless to light brown, dark brown, then light brown, finally light yellow or colorless. Higher selectivity was obtained when the brown color was more intense. When the final mixture was colorless, CPE was 100% consumed during the reaction. These color changes could not be easily detected when the addition was stepwise, maybe because the concentration of the colored species was not enough to be detected visually. The addition of more reagents did not allow time to generate the colored species. Manganese has different color depending on its oxidation state, Mn(II) solutions are colorless, MnO2(IV) is gray, and Mn2O3(III) is brown, mixed valence corresponds to darker color (Tian et al., 1997). According to reports, the oxidation state of manganese changes during the reaction from 2+ to 4+ and then goes back to 2+ after the epoxidation reaction is finished (Quiñonero et al., 2009). A sample of 1 ml of reaction mixture was analyzed by UV–Vis at different time intervals (Figure 6). The sample taken at 31 min of reaction had intense brown color, and a band at 530 nm, this band has been attributed to UV–Vis spectra of reaction mixture at different times.
Conclusion
CPE can be epoxidized using the hydrogen peroxide/sodium bicarbonate/manganese sulfate system. The maximum selectivity obtained under the used conditions was 56%. The reactivity of CPE makes it difficult to improve selectivity. DMF was better solvent for this reaction than t-BuOH, since all the reagents were soluble in DMF. The volume of DMF used in the reaction media affects the conversion of CPE and selectivity to CPO. By using a ratio CPE:DMF of 1.6:51 mmol the selectivity to CPO was improved. The solvent is important not only to keep all the reagents in solution but also to reduce interactions and avoid side reactions. One-step addition of the H2O2/NaHCO3 solution at low temperature reduces the effect of heat released by the epoxidation reaction on the ring opening. The slow addition of H2O2/NaHCO3 had a deleterious effect on selectivity suggesting that the epoxide produced at one moment was further oxidized with the next addition. Manganese (II) was required for the selective oxidation of CPE to CPO under the conditions used. Sodium acetate and salicylic acid did not improve conversion of CPE and selectivity to CPO. Changes in manganese species during the course of reaction were detected by visible spectroscopy.
Footnotes
Acknowledgement
BH and LJG wish to thank Juan Diego Restrepo for all his help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Universidad de Antioquia. SLS acknowledges the support of the Office of U.S. Department of Energy, Office of Basic Energy Sciences, Division of Chemical, Biological and Geological Sciences under Grant DE-FG02-86ER13622.A000.