Abstract
Macroporous copolymers of ethylene glycol dimethacrylate and methacrylic acid containing immobilized molecules of Alizarin red S and similar polymers imprinted by gadolinium ions were synthesized by free radical bulk polymerization method. Sorption properties of two pairs of the polymers with cross-link ratio of 85.7 and 92.6% were compared. The resulting materials were characterized by scanning electron microscopy and infrared spectroscopy. Sorption performance of the polymers synthesized toward gadolinium ions was investigated. It has been found that the maximum removal of gadolinium ions from solutions is observed at pH 6. The experimental data on gadolinium ion interaction with the sorbent surface were shown to fit Langmuir adsorption theory. Maximum gadolinium sorption capacity of nonimprinted polymers with cross-link ratio of 85.7 and 92.6% is 19.4 and 14.9 mg/g, respectively. Sorbent selectivity for rare earth ions was studied. According to the obtained results, sorbents under study reveal affinity to triple-charged ions. Tetrad effect observed in rare earth distribution patterns reflects chemical nature of sorption on such polymers.
Keywords
Introduction
Due to their unique properties, rare earth elements (REE) are required for many different applications such as magnet, alloy, phosphor, catalyst, ceramics industries, and the like (Jordens et al., 2013). In the view of this, the quantitation of REE in various materials is essential. However, this task is often complicated by complex matrices or large amounts of coexisting rare earths.
To address this issue, separation of REEs from the matrix is needed in most cases. The methods that are most widely used for extraction and preconcentration of REE include coprecipitation (Taicheng et al., 2002), solvent extraction (Shabani et al., 1990), solid phase extraction (Liang and Chen, 2005), ion exchange (Barrat et al., 1996), HPLC (Pedreira et al., 2002), and adsorption (Inagaki and Haraquchi, 2000). The latter is recognized as one of the most promising methods due to its simplicity, high efficiency, availability and variety of adsorbents, low cost, and wide-range applicability.
Among various sorption materials available, synthetic polymers should be highlighted. Considerable diversity of monomers bearing different functional groups together with wide selection of polymerization techniques enables one to control resulting sorbent properties during the synthesis or modify obtained polymers subsequently. Therefore, synthetic polymers are widely used as efficient sorbents for heavy metal ions (Kara et al., 2004; Lemos and Baliza, 2005; Prabhakaran and Subramanian, 2003; Uğuzdoğana et al., 2010). Ion exchange (Jia et al., 2008) and chelating (Inagaki and Haraquchi, 2000) resins, as well as ion-imprinted polymers (Biju et al., 2003), are commonly applied for rare earth ion extraction and preconcentration.
Ion and molecular imprinting is a powerful and dynamically developing technique which nowadays allows scientists to obtain such sophisticated materials as imprinted multilevel nanocomposite membranes (Wu et al., 2016, 2017) that can rapidly recognize and separate different compounds and large molecules like proteins (Wu et al., 2015). Ion-imprinted polymers as highly selective sorbents for a particular chemical form of a given element are prepared by copolymerization of functional and cross-linking monomers in the presence of a target metal ion (template) to form complexes with the functional monomer which are being trapped in the polymer matrix during the synthesis. Removal of these ions leaves cavities in the polymer which are arranged to match the charge, size, coordination number, and coordination geometry of the template. With these recognition sites, such materials can selectively bind the target species in the presence of various concomitant ions (Branger et al., 2013).
It has been shown that the most suitable matrices for imprinted materials are macroporous polymers (Cormack and Elorza, 2004). It should be highlighted that the term “macroporous” in this case just reflects the type of the polymers obtained by the certain synthetic technique and does not imply any information about pore size and configuration (Sherrington, 1998). These polymers are formed during separation of the polymer phase when a prepolymerization mixture contains a porogen and a large amount of cross-linker. Thus, after the synthesis, the materials consist of the cross-linked polymer continuous phase and the porogen discontinuous phase acting as a pore template. Porogen removal followed by drying allows obtaining permanently porous materials with pore size independent of the solvent presence. High degree of cross-linking provides rigidity for the polymer network (Sherrington, 1998). Structure peculiarities of the macroporous polymers allow realization of so-called trapping—ion-imprinting approach when a ligand is not covalently bound to the polymer matrix but is trapped during the synthesis and retained therein (Branger et al., 2013).
Synthesis of the blank polymers, which are obtained under similar conditions but in absence of the target ion, is a prerequisite. Such polymers have always been investigated as control materials and have never been considered as independent sorbents. Nevertheless, in our opinion, such polymers with a permanent pore structure containing entrapped ligand molecules are promising sorption materials for metal ion extraction and preconcentration.
Alizarin red S (ARS) dye, which is one of the most widely used analytical reagents, has been chosen to serve as a ligand in our study (Fain, 1999). This dye was proposed for quantitative REE spectrophotometric determination in the view of its ability to form colored complexes with a number of metals including lanthanides (Rinehart, 1954).
Gadolinium as a representative of REE has been chosen for the present research because it is on the border between light and heavy rare earths. It is widely employed in optical ceramics, luminescent materials, magnets, and in the nuclear and cryogenic technologies. As an important application, gadolinium compounds are vital components of contrast agents for magnetic resonance imaging (Elvers et al., 2011).
The present study describes the synthesis of the macroporous polymers containing ARS with different cross-link ratio and the investigation of sorption performance of the obtained materials. The efficiency of gadolinium imprinting in such polymers was discussed, as well as possibility to use these sorbents for selective REE extraction from various matrices was investigated.
Experimental
Reagents
Methacrylic acid (MAA) and ethylene glycol dimethacrylate (EGDMA) were purchased from Merck KGaA, Germany; azobisizobutyronitrile (AIBN) was purchased from OOO Ukrorgsintez Scientific and Production Enterprise, Ukraine; state standard reference metal solution samples of Al, Fe, Cu, Zn, Sr, Cs (1 g/l) were purchased from A.V. Bogatsky Physico-Chemical Institute of NAU, Ukraine; ARS, nitric acid, hydrochloric acid, NaOH, and Gd(NO3)3·5H2O were of laboratory reagent grade; dimethylsulphoxide (DMSO) was of analytical reagent grade; oxides of La, Ce, Eu, Gd, Lu used to prepare metal ion solutions were of special purity grade.
Preparation of polymers
Polymeric nonimprinted sorbents, NIP1 and NIP2, with different cross-link ratios were synthesized via free radical bulk polymerization. ARS, MAA (functional monomer), and EGDMA (cross-linker) were added to 4 ml of DMSO (porogen) under stirring. AIBN initiator (0.7 mol% of the total amount of polymerizable methacrylate units) was mixed with the obtained solution in a reaction vial. The vial was purged with argon gas under sonication for 30 min, sealed, and placed in a water bath at 65℃ for 24 h. After polymerization, the resulting polymer was ground using a pestle and a mortar and subsequently sieved (≤150 µm). The particles were exhaustively washed with 0.1 M HCl and then with deionized water and dried in a vacuum oven at 80℃ for 8 h prior to use.
Ion-imprinted polymers, IP1 and IP2, were synthesized using the same protocol but with addition of gadolinium nitrate to the reaction mixture as a template. The synthesis pathway is schematically represented in Figure 1. According to Fain et al. (2004), ARS in metal complexes predominantly exists as tautomeric 2,9 - or 1,2-antraquinoid form.
Schematic representation of the synthesis of the Gd(III)-imprinted polymers.
Compositions of the prepolymerization mixtures for the polymer synthesis.

Adsorption studies
Sorption performance of the polymers synthesized was studied using batch experiments. Twenty-milliliter aliquots of test solutions containing specified amount of the polymers (25 mg) were mechanically shaken at 250 r/min for 1 h. Bulk phase pH was adjusted to a desired value with NaOH or HCl solutions and monitored using STARTER 3100 pH-meter (OHAUS, USA). Thereafter, the solutions were filtered and the concentrations of unextracted Al(III), Fe(III), Cu(II), Zn(II), Sr(II), La(III), Ce(III), Eu(III), Gd(III), and Lu(III) were measured by inductively coupled plasma atomic emission spectrometry using Thermo Scientific iCAP 6300 Duo spectrometer (USA). The concentrations of Cs(I) ions were determined by flame atomic emission spectrometry using Thermo Scientific iCE3500 spectrometer (USA).
The effect of solution pH on the adsorption efficiency of polymers toward Gd(III) was studied over the pH range from 2 to 7. The initial Gd(III) concentration was 5 mg/l. The uptake rate (%) of gadolinium was calculated by the following equation

Experimental adsorption isotherms were obtained as follows: portions of the polymers were equilibrated with solutions of varying Gd(III) concentrations ranging from 5 to 200 mg/l at pH 6. Sorption capacity (the amount of gadolinium ions adsorbed per unit mass of the adsorbent) can be obtained by

The above-described experimental procedure was also used to study competitive sorption of gadolinium in the presence of coexisting ions. Lanthanides such as Ce, La, Lu, Eu at concentration of 3 mg/l of each metal (series 1) as well as Al, Cu, Fe, Sr, Zn, Cs at concentration of 10 mg/l of each metal (series 2) were chosen as concomitant ions.
Distribution coefficients for investigated ions were determined using the following equation

Selectivity coefficients were estimated as follows

To elucidate the imprinting effect, relative selectivity coefficients were also calculated using the following ratio

Results and discussion
Characterization
The IR spectra of the obtained polymers were recorded in KBr pellets using Spectrum One spectrometer (Perkin-Elmer, USA). For liquid samples (MAA and EGDMA), a liquid cell was used. Figure 2 represents IR spectra of the monomer precursors and the synthesized polymers. As expected, all obtained materials demonstrate practically identical spectral patterns. The intensive absorption band observed at ca. 1700 cm−1 for both monomers and polymers can be attributed to C=O stretching vibrations. Intense absorption bands corresponding to C=C stretch (1635 cm−1) and C–H out-of-plane bend at a double bond (948 cm−1) are present in IR spectra of MAA (Figure 2(a)). The similar peaks (1638 and 944 cm−1) are found in IR spectra of EGDMA (Figure 2(b)). The corresponding absorption bands at 1641 and 962 cm−1 were observed for the polymers but with very low intensity (Figure 2(c)). This indicates high monomer conversion and confirms successful polymerization.
IR spectra of MAA (a), EGDMA (b), and the polymers synthesized (c). EGDMA: ethylene glycol dimethacrylate; MAA: Methacrylic acid.
The particle morphology of the obtained polymers was studied using JSM-6390LV scanning electron microscope (JEOL, USA). The small amount of the polymer was suspended in water, then one drop of the suspension was deposited on the carbon substrate, and water was evaporated. The SEM images were obtained in secondary electrons mode. Analysis of the microphotographs (Figure 3) shows no significant structural difference between the four synthesized materials. A layered particle surface morphology is observed with no pores visible under magnification used.
SEM micrographs of IP1 (a), NIP1 (b), IP2 (c), and NIP2 (d).
Specific surface area of the polymers was determined via argon sorption using Brunauer–Emmett–and Teller method. The obtained values of specific surface area for IP1, NIP1, IP2, NIP2 materials are 330, 450, 410, and 500 m2/g, respectively.
Effect of pH
The curves of gadolinium uptake rate dependence on bulk phase pH for NIP1 and NIP2 sorbents are depicted in Figure 4.
Effect of pH on sorption of Gd(III) ions on polymeric particles: 1—NIP1, 2—NIP2.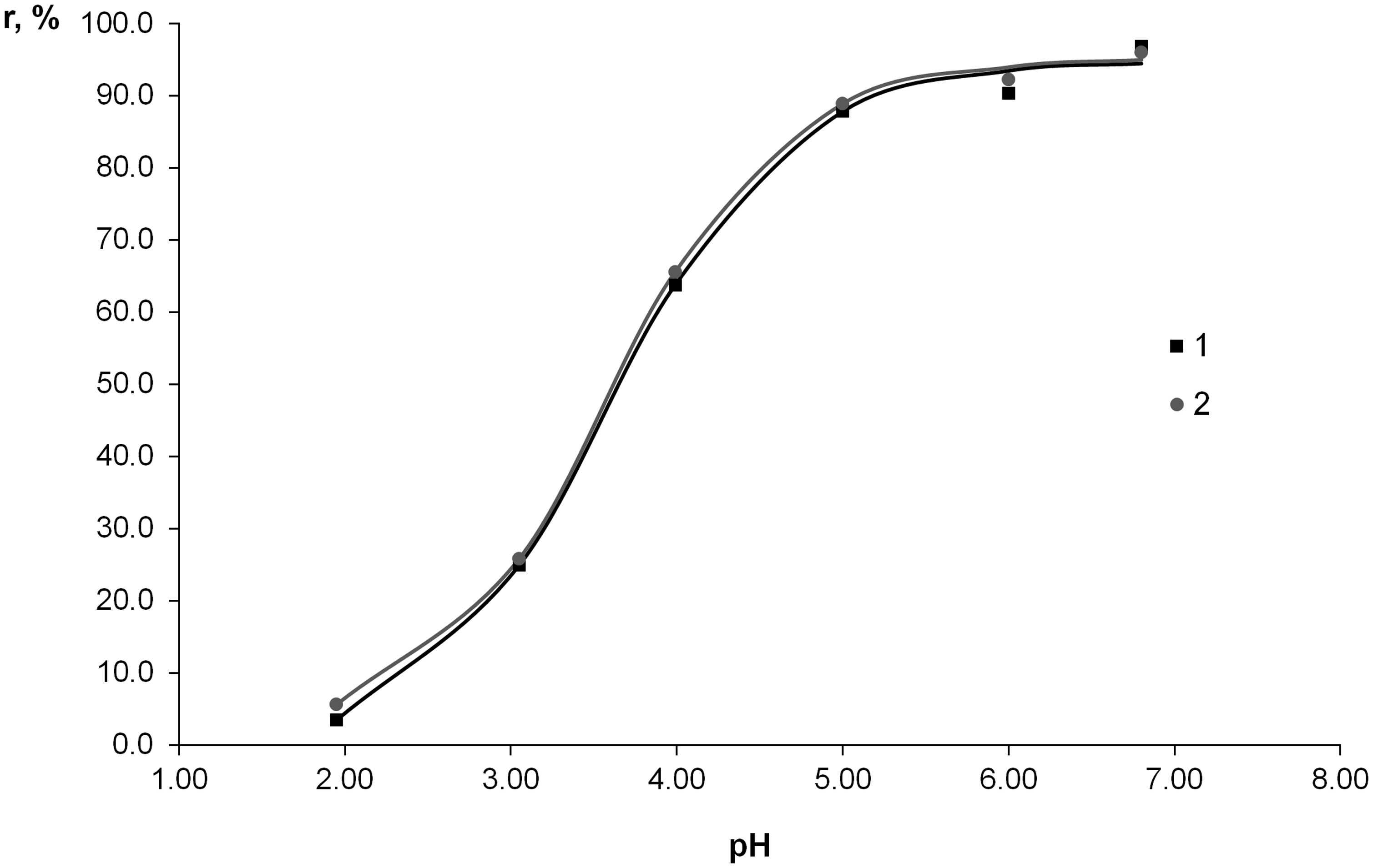
As can be seen from Figure 4, gadolinium sorption efficiency increases within pH range of 2–5 and then reaches a plateau. It can be explained by the fact that polymer functional groups are partially protonated at low pH. Increase of pH favors gadolinium ion binding by the sorbent surface. However, pH values above 7 were not studied in the view of gadolinium ion hydrolysis. Thus, pH value of 6 was chosen to be an optimum for further study of polymer sorption performance.
Kinetics of gadolinium ion sorption by the synthesized polymers was also investigated. According to the results obtained, the optimum equilibrium time to adsorb Gd(III) ions onto the polymers was attained in 10 min. Such rapid kinetics may result from high contact area between the sorbent particles and a solution, and high affinity of adsorbent to adsorbate. Also, occurrence of the cross-linker ether groups within the polymer network endows surface hydrophilicity, thus, facilitating interaction of the sorbent and dissolved species.
Sorption isotherms
To reveal sorption mechanism and sorption characteristics of the synthesized materials, adsorption isotherms for NIP1 and NIP2 were constructed (Figure 5). According to Giles classification (Parfitt and Rochester, 1986), the presented dependencies can be attributed to Adsorption isotherms for Gd(III) on blank polymers: 1—NIP1, 2—NIP2. Initial metal ion concentration: 5–200 mg/l, contact time: 1 h, temperature: 20℃, adsorbent dose: 1.25 g/l.
The obtained data were treated by Langmuir isotherm equation

where q is the equilibrium sorption capacity (mg/g), q∞ is the saturated monolayer adsorption capacity (mg/g), KL is the Langmuir equilibrium constant (l/mg), and ce is the ion concentration at equilibrium (mg/l). In linearized form it can be written as

Fitting parameters for Langmuir isotherm to experimental adsorption isotherm of the obtained polymers.

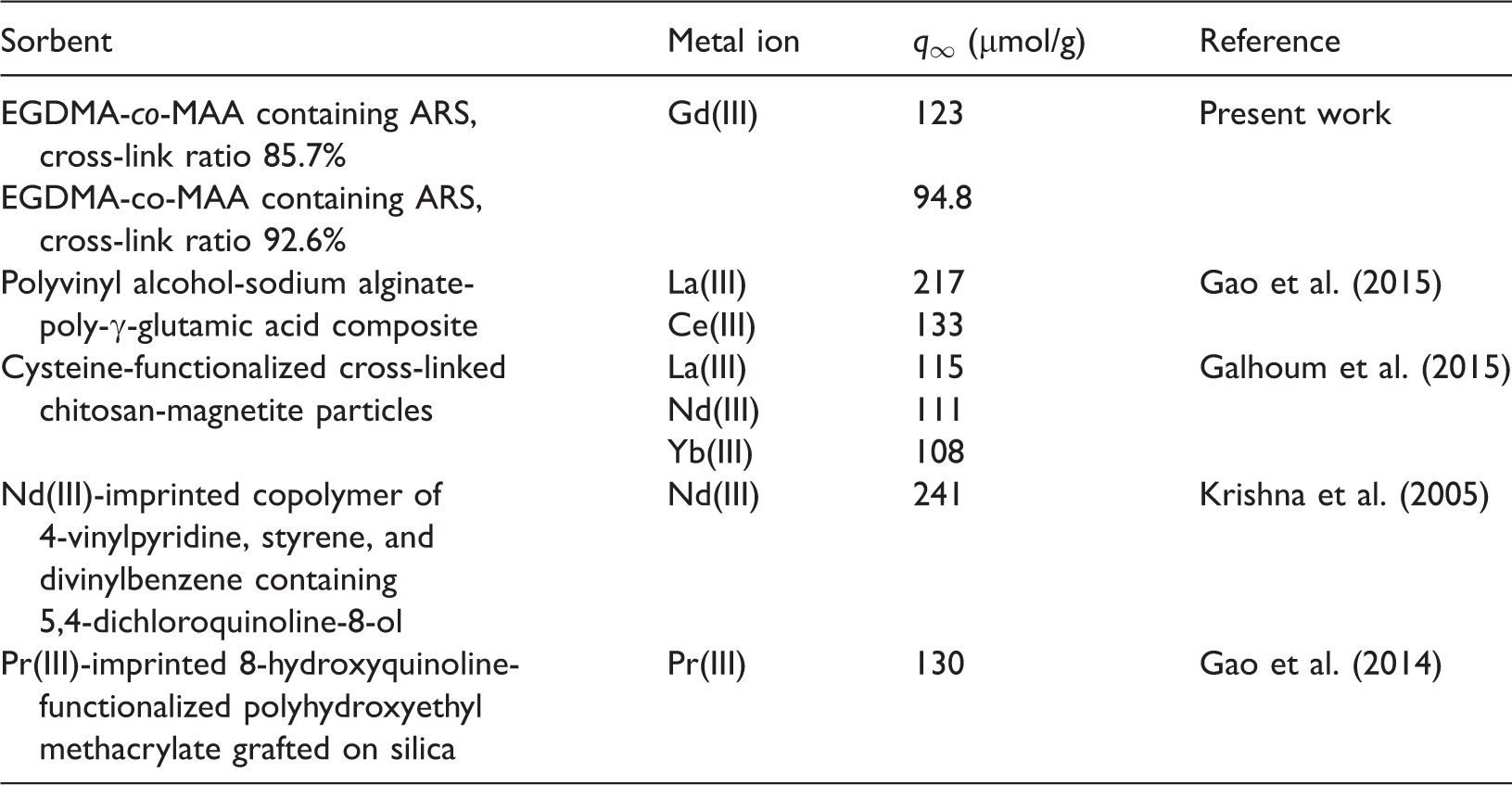
Comparison of sorption capacities.
Selectivity experiments
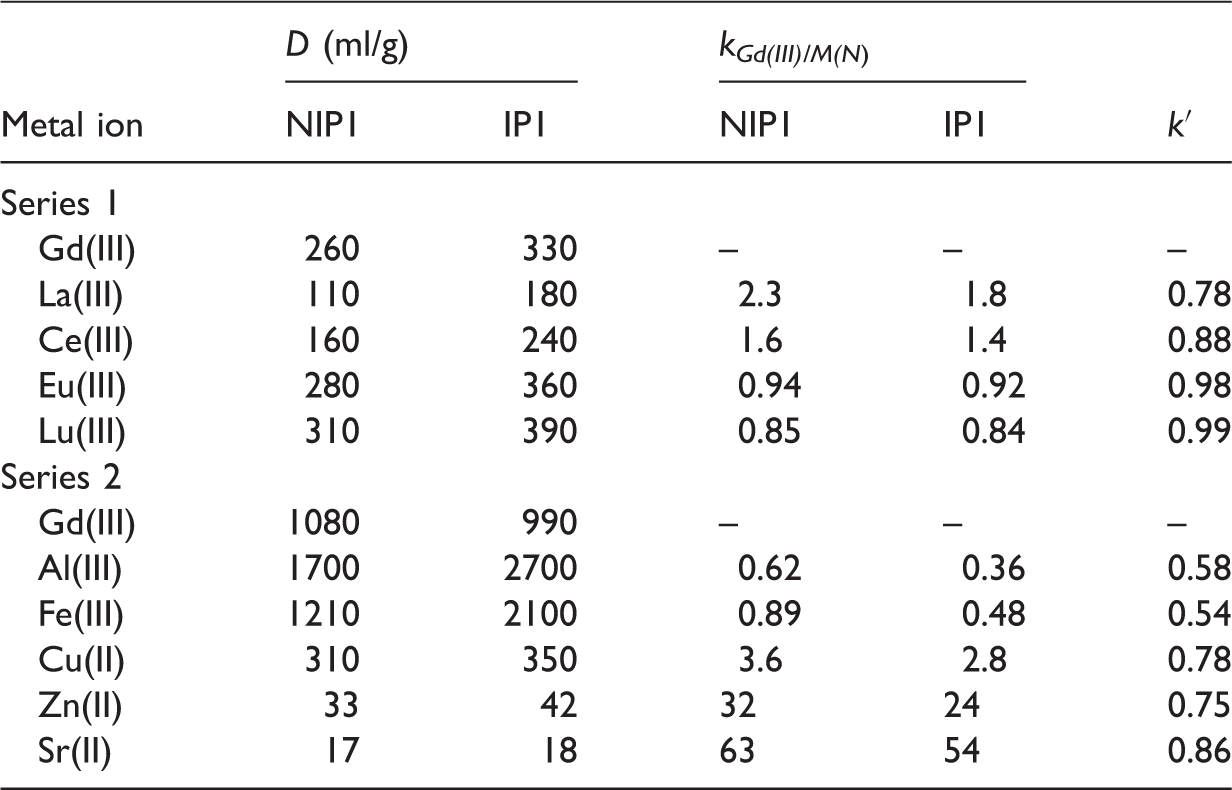
The binding parameters of NIP1 and IP1 toward metal ions.
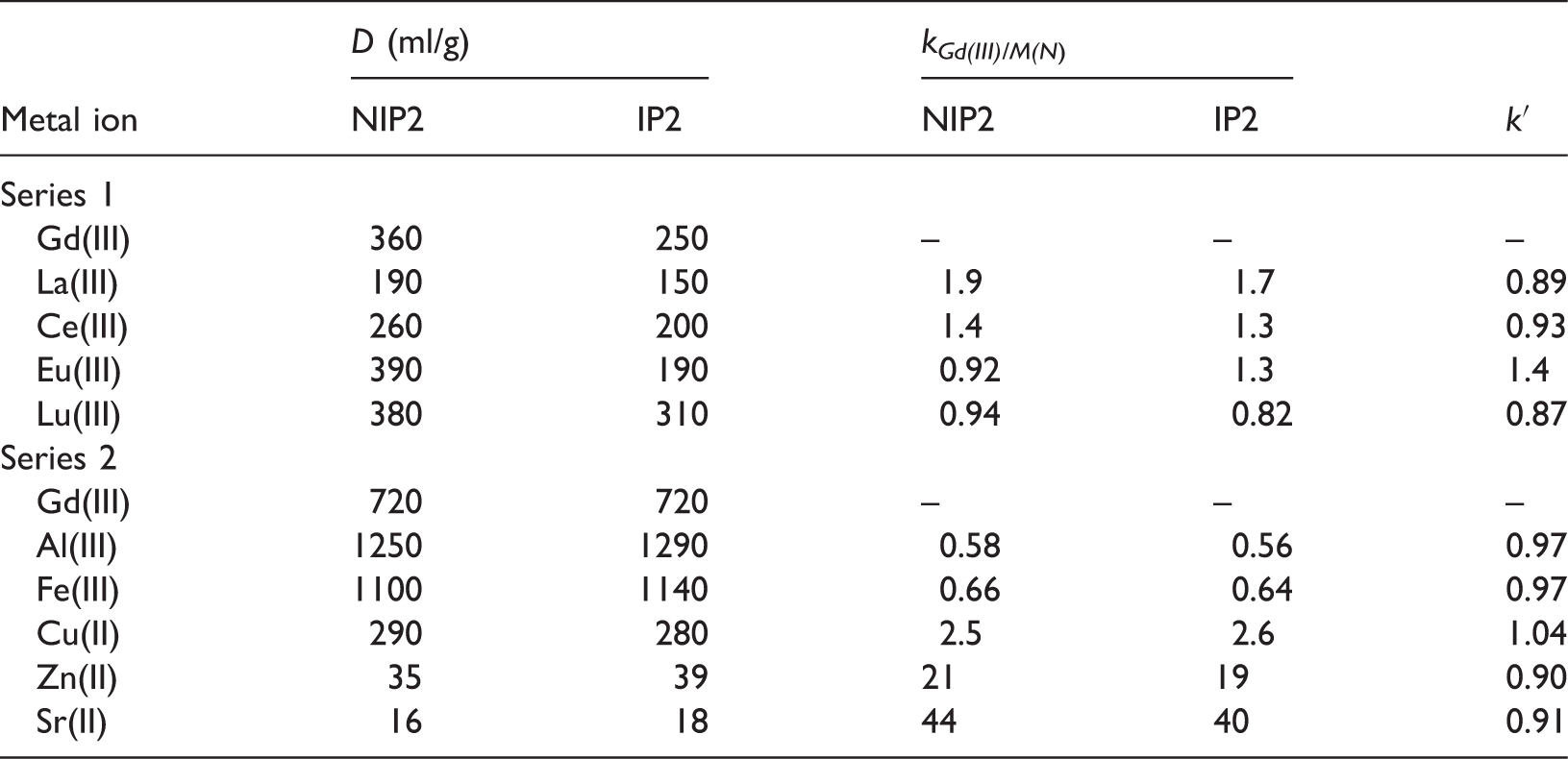
The binding parameters of NIP2 and IP2 toward metal ions.
Such values for Cs(I) ions were not estimated because they were not removed from solutions by the sorbents.
The discontinuity observed in the distribution coefficient dependence on atomic number of REE can be attributed to the so-called tetrad effect.
Tetrad effect consists in specific changes in physical and chemical properties of lanthanides depending on their atomic numbers which results in splitting them into four subgroups (tetrads): La–Nd, Pm–Gd, Gd–Ho, Er–Lu. Within each tetrad, properties change gradually, with surges occurring at the ends. These points can correspond with quarter-, half-, three-quarter or completely full 4f electronic shells. The effect is explained in terms of changes in the electron–electron repulsion parameters (so-called Racah parameters) while coordination bonds with lanthanide ions are being formed (Kawabe, 1992).
Tetrad effect explains a typical dependence of the REE distribution coefficients on their atomic numbers in extraction (Peppard et al., 1969), sorption (Coppin et al., 2002) and coprecipitation (Kawabe et al., 1999) experiments. Similarly, some scientists explain abnormal dependences of REE contents on their atomic numbers in some minerals, sedimentary rocks, and other geological samples (Monecke et al., 2002).
The display of tetrad effect indicates the chemical nature of lanthanide adsorption on the synthesized polymers at pH 6. It reflects the REE cation desolvation and formation of the inner sphere complex with surficial functional groups of the polymers. This conclusion is confirmed by other experimental studies suggesting that REEs are adsorbed on the sorbent surfaces in the form of inner sphere complexes (Takahashi et al., 2004, 2005; Tertre et al., 2008).
The highest values of the lanthanide distribution coefficients were obtained for NIP2 polymer.
As shown in Tables 4 and 5, the resulting polymers exhibit a greater affinity for trivalent elements. However, large values of the distribution coefficients for Al(III) and Fe(III) ions could have been obtained due to the partial hydrolysis of these ions at pH 6.
The relative selectivity coefficient values close to 1 indicate the absence of imprinting effect in the synthesized polymers. It can be explained by the fact that ARS is not a selective ligand for gadolinium ion in the presence of other rare earth ions, so binding sites created are not effective. Also, the nonselective adsorption by polymer matrix containing large amount of ester functional groups may play an important role.
Conclusions
Novel sorption materials with a high degree of cross-linking containing ARS as a complexing agent were synthesized by free radical bulk polymerization technique. The formation of the high-molecular compounds was confirmed by IR spectroscopy. The obtained polymer particles have a layered morphology and high specific surface area (from 410 to 500 m2/g). Gadolinium sorption efficiency of investigated sorbents increases within pH range of 2–5 due to gradual deprotonation of polymer functional groups with decreasing solution acidity and reaches a plateau after pH 5. Thus, pH 6 is optimal for effective removal of REEs from aqueous solutions by the synthesized sorbents. Binding of rare earth ions is described by Langmuir adsorption model. High sorption kinetics allows using such sorbents for extraction of REEs from aqueous solutions in a dynamic mode. Among the tested sorbents, NIP2 exhibits the best sorption performance toward the mixture of REEs. This finding may be attributed to higher cross-link degree and thereafter higher rigidity of the binding sites.
The specific form of the REE distribution coefficient patterns implies tetrad effect and therefore indicates chemical nature of lanthanide adsorption on the synthesized sorbents.
Applying of ion-imprinting technique was unable to yield materials selective for gadolinium ions because of nonselectivity of ARS ligand and nonselective ion sorption by polymer matrix. Therefore, the size and configuration of the sites do not significantly influence on the selectivity of the sorbents in this case.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to PV Mateichenko and DS Sofronov for their help and support.
Author's Note
The present study was first presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
