Abstract
Zeolite Na–X and chitosan-modified Na–X–CS were used as adsorbents for the removal of Cu(II), Fe(III), Mn(II) and Zn(II) from aqueous solutions. They were prepared based on fly ash by hydrothermal reaction using the NaOH solution under controlled conditions. Characterization of the Na–X and Na–X–CS sorbents was made using Fourier transform infrared spectroscopy with the attenuated total reflection mode, X-ray fluorescence and X-ray diffraction, nitrogen adsorption and desorption, acidity measurements and scanning electron microscopy techniques. The effects of adsorption time, the amount of sorbent and the concentration of metal ions on the removal percentage and sorption capacities were studied. The sorption percentage increased with the increasing phase contact time and zeolite amount. The adsorption mechanism was also discussed. The results revealed that Na–X–CS adsorbs Cu(II), Fe(III), Mn(II) and Zn(II) ions more effectively than zeolite Na–X and proved that modification by CS leads to an increase in the adsorption efficiency of the zeolites.
Introduction
The problem of waters and waste waters pollution with heavy metals has been known and studied by scientists around the world for a long time. Although the effects of heavy metals on organisms are well known and focused worldwide attention, this exposure is constantly growing because of their wide use in many fields (Järup, 2003). Therefore, new methods are developed to remove these compounds from the environment, especially from waters and wastewaters. Adsorption is a well-known method, which is considered to be both efficient and inexpensive and often with the possibility of adsorbent reuse (Huang et al., 2016). Studies on the sorption of heavy metals are carried out on zeolites. A variety of adsorbents includes active carbons, ion exchangers and zeolites as well as a large group of low-cost sorbents such as fly ashes (FAs), biochars obtained from feedstock materials, etc.
FA produced from coal combustion belongs to wastes and its proper management is very important not to cause environmental problems. It is used in many industries, including cement industry, but its large part is stored in landfills (Zhao et al., 2010). Negative impact of FA on the environment cannot be completely eliminated. Therefore, the use of this waste for production of zeolites applied for wastewaters treatment is the solution of these two problems at the same time. It is well known that both FA (Jovanovic et al., 2012; Petrus and Warchoł, 2005; Sprynskyy et al., 2006) and synthetic zeolites (Visa, 2016) can be used to purify waters and wastewaters from heavy metals (Cho et al., 2005; Mohan and Gandhimathi, 2009; Saakshy et al., 2016), dyes (Kumar et al., 2005; Vlková et al., 2004), phenols (Zhang et al., 2015). Synthetic zeolites produced based on FA proved to be an effective option in the treatment of water and wastewater from this type of pollution.
Zeolites can be classified on the basis of their framework structure. The International Zeolite Association recommends the use of the three letter code for zeolites with various structures. In our studies zeolite NaX was chosen.
Na–X is a synthetic form of naturally occurring mineral faujasite. This is a microporous zeolite with a well-known three-dimensional structure (Wu et al., 2016). Among the various types of zeolites, Na–X is characterized by a large pore diameter (0.74 nm) (Zhang et al., 2013b). Because of its desirable properties it can be used as molecular sieves, dehumidifiers, ion exchanger and also as catalysts (shape-selective catalysts). Zeolites of this type are also used in the purification processes of soil, air, water, in an oil refinery as well as ingredients in laundry detergents (Erdem et al., 2004; Kazemian et al., 2009; Khowatimy et al., 2014; Zhang et al., 2013a).
Physiochemical characterization of FAs, zeolites and their modifications is essential for precise description of particles interactions with solutions especially with respect to removal of impurities from waters and wastewaters. In this study the sorption process of heavy metals Cu(II), Fe(III), Mn(II) and Zn(II) on synthetic zeolite Na–X produced by the hydrothermal method under controlled conditions and the chitosan (CS)-modified zeolite was investigated. There are many studies describing the adsorption abilities of FA or zeolites for the removal of heavy metal ions from water and wastewaters, but only few studies have reported their modification by CS. CS is a polysaccharide obtained by N-deacetylation of chitin, having good complexing capacity due to specific interactions of –NH2 groups of heavy metal ions (Sobahi et al., 2014). For the process of modification CS flakes were used. The molecular weight of the prepared CS was found to affect the viscosity. Suitable CS modifiers determine the preferential adsorption of specific components from the purified solutions (selective sorption) and thus affect selectivity of the obtained sorbents. Therefore, the innovative element of the presented research is the use of FA activated in the milling process by means of magnetic core and the utilization of post-reaction solution of sodium hydroxide to modify the obtained zeolites with biodegradable and environmentally friendly CS. The fractions obtained by magnetic core, e.g. 2006 and 3004 were denoted as FA. Characterization of initial FA, Na–X and Na–X–CS sorbents was made using the Fourier transform infrared spectroscopy (FTIR) with the attenuated total reflection (ATR) mode, elemental analysis (CHN), X-ray diffraction (XRD), nitrogen adsorption and desorption isotherms (ASAP), acidity measurements and scanning electron microscopy (SEM) techniques for different CS modifiers. The effects of phase contact adsorption time, amount of sorbent, concentration of metal ions and pH on the removal percentage and sorption capacities were studied. Another important element is to make sorbent capable of operating under cyclic mode: sorption–desorption. High capacity of the sorbents affects the reduction of their amount especially in large-scale processes which is important from the economical point of view.
Materials and methods
FA, zeolites and CS
The raw FAs from fractions 2006 and 3004 (FA) used in this study were obtained from the heat and power generating plant in Kozienice, Poland. The synthetic zeolite Na–X was produced in the hydrothermal conversion reaction of FA with NaOH described by Bandura et al. (2015) and Wdowin et al. (2014) on a laboratory scale. Additionally, zeolite Na–X was treated with 1 M NaCl and 1 M CaCl2 solutions (Na–X(Na) and Na–X(Ca)) followed by washing with DI and drying at 353 K before use.
Chemical modification of Na–X was made by dissolving CS flakes with the deacetylation degree > 75% (Sigma Aldrich) in 1% glycolic acid (POCh, Poland). After 24 h of stirring using a magnetic stirrer at 1000 r/min at room temperature, the appropriate amount of Na–X zeolite was added (to obtain different FA and CS ratio). The mixture was stirred for another 6 h using a magnetic stirrer at 1000 r/min. Next a suitable amount of 1 M NaOH was added to the solution to achieve pH 9. Then the mixture was filtered and washed with distilled water to obtain a natural pH of filtrate. The resulting material (Na–X–CS) was dried and ground in the mill. All steps were carried out under controlled conditions using the analytical and measuring set (Metrohm).
Heavy metal ion solutions
The stock solutions of Cu(II), Fe(III), Mn(II), Zn(II) (1000 mg/l) were prepared by dissolving appropriate amounts of CuCl2·2H2O, FeCl3·6H2O, MnCl2·4H2O and ZnCl2 in DI. In order to obtain solutions with the appropriate concentrations stock solutions were diluted. 0.1 M HCl or 0.1 M NaOH solution was used to adjust pH to 5.0 and 6.0. The other solutions NaCl, CaCl2, etc. were of analytical grade and obtained from Avantor Performance Materials (Poland).
Physicochemical sorbents analysis
The solids were characterized by the X-ray diffraction (XRD), FTIR with the ATR mode, X-ray fluorescence (XRF), nitrogen adsorption and desorption isotherms (ASAP), acidity measurements by ammonia desorption and SEM.
The mineral composition of the samples was determined using the XRD method on the Philips APD X'pert diffractometer (PANalytical) with a goniometer of high angular resolution as well as Cu lamp and graphite monochromator. The analyses were performed in the 2θ angular range 5–65°. The phase structures were identified with the use of JCPDS-ICDD database.
The FTIR-ATR analyses were carried out to identify the functional groups of Na–X and especially Na–X–CS, e.g. to characterize newly synthesized zeolites and confirm that the final products comply with the defined specifications. Spectra were obtained using the FTIR-ATR spectrometer with the monolithic diamond crystal, Cary 630 (Agilent Technologies). Assays were made in the range 4000–400 cm−1 with the scan speed 2 scans/s (4 cm−1) and corrected using the MicroLab FTIR software.
The chemical composition of FA and Na–X was performed by the XRF method. Analysis was carried out using the Epsilon 3x spectrometer with an X-ray tube equipped with Rh anode (PANalytical). The maximum power used as the excitation source was 50 kV.
The measurements of average pore diameter, total pore volume and specific surface area were made by means of N2 adsorption/desorption isotherms at 77 K using a porosimeter ASAP 2420 (Micromeritics, Inc.).
SEM investigations of FA, Na–X and Na–X–CS samples were carried out by means of Quanta 3D FEG (FEI) microscope equipped with the EDX system for chemical analysis.
The point of zero charge (pHpzc) was determined by the drift method using 0.01 M NaCl solutions at pH 2–12. On this basis, the difference between the final and initial pH values was found. The average of three measurements for each sample was used to estimate the point of zero charge. The modification process and pHpzc determination were controlled using the analytical set equipped with Titrando, Dossino, pH-meter units with appropriate electrods and sensors (Metrohm).
Metal ions concentrations were analysed by the inductively coupled plasma optical emission spectrometer ICP-OES 720 ES (Varian). The sample introduction system consists of a OneNeb® nebulizer and a cyclonic spray chamber.

The adsorption experiments on the FA and Na–X, Na–X–CS zeolites were conducted at temperature 293 K. The adsorbent (0.1 g) was weighed into the Erlenmeyer flask and mixed with 20 ml of a solution of the appropriate concentration. The samples were stirred using the mechanical shaker at the amplitude 7 and 180 r/min, at room temperature during the period of time 1–120 min. For zeolites Na–X obtained from 2006 and 3004 FAs the effect of sample dose was also investigated. Sorbents in the range from 0.1 to 0.5 g were weighed to the Erlenmeyer flask and 20 ml of solutions was added. The mixtures were then stirred 120 min under analogous conditions. In order to investigate the effects, the additional amount of interfering Cl−,
Results and discussion
FA, Na–X and Na–X–CS characteristics
The XRD patterns of 3004Na–X zeolite are presented in Figure 1. It can be seen that the material is rich in zeolite Na–X. The zeolite phase also includes quartz, mullite and Na–P1 phase forming unconverted FA residues. The content of Na–X zeolite is about 60% and was determined based on the characteristic distances dhkl = 14.47; 3.81; 5,73; 8.85; 4.42; 7.54; 4.81; 3.94.
Mineralogical composition of zeolite Na–X.
As follows from the FTIR analysis of the 3004Na–X and 3004Na–X–CS zeolites it was found that the broad bond at 3650 cm−1 is connected with the adsorbed water (Figure 2). The band at 1170 cm−1 is attributed to the antisymmetric Si–O–Si stretching vibration of SiO4 tetrahedron while the bands at 1104 cm−1 assignable to the antisymmetric stretching vibration of Si–O–Al. The bands in the 900–400 cm−1 range are connected with stretching vibration of Al–O–Al. The band 464 cm−1 can be assigned to bending vibration of O–Si–O in SiO4. As for the CS analysis also the strong broad band was found at 3393 cm−1 and associated with the –OH group of CS. The next band 1640 cm−1 was assigned the presence of the CO group. The modified zeolite has also an evident band connected with the methyl group at 1376 cm−1. The sharpest band at 959 cm−1 is associated with Al–O and Si–O (Dogan and Durmaz Hilmioglu, 2010; Malek and Malek, 2012; Patil and Aminabhavi, 2008).
FTIR analysis of 3004Na–X and 3004Na–X–CS zeolites. FTIR: Fourier transform infrared spectroscopy.
Comparison of the contents of 3004FA obtained by the XRF method.

XRF: X-ray fluorescence.
It is well known that the shape of adsorption/desorption N2 isotherms for zeolites corresponds to the II type, both for natural (such as clinoptylolite) and synthetic (such as Na–P1) zeolites. With the increase of the relative pressure (p/p0) = 1, an increase of isotherm is observed due to the presence of mesopores. The effect of the hysteresis is also observed near p/p0 equal to 1 and its loops match type H3. The adsorption is low in a wide p/p0 range.
Zeolite 3004Na–X is characterized by a good homogeneity. As follows from the particle size distribution both are characterized by bimodal shape (Figure 3).
Particle size distribution of (a) 2006Na–X and (b) 3004Na–X.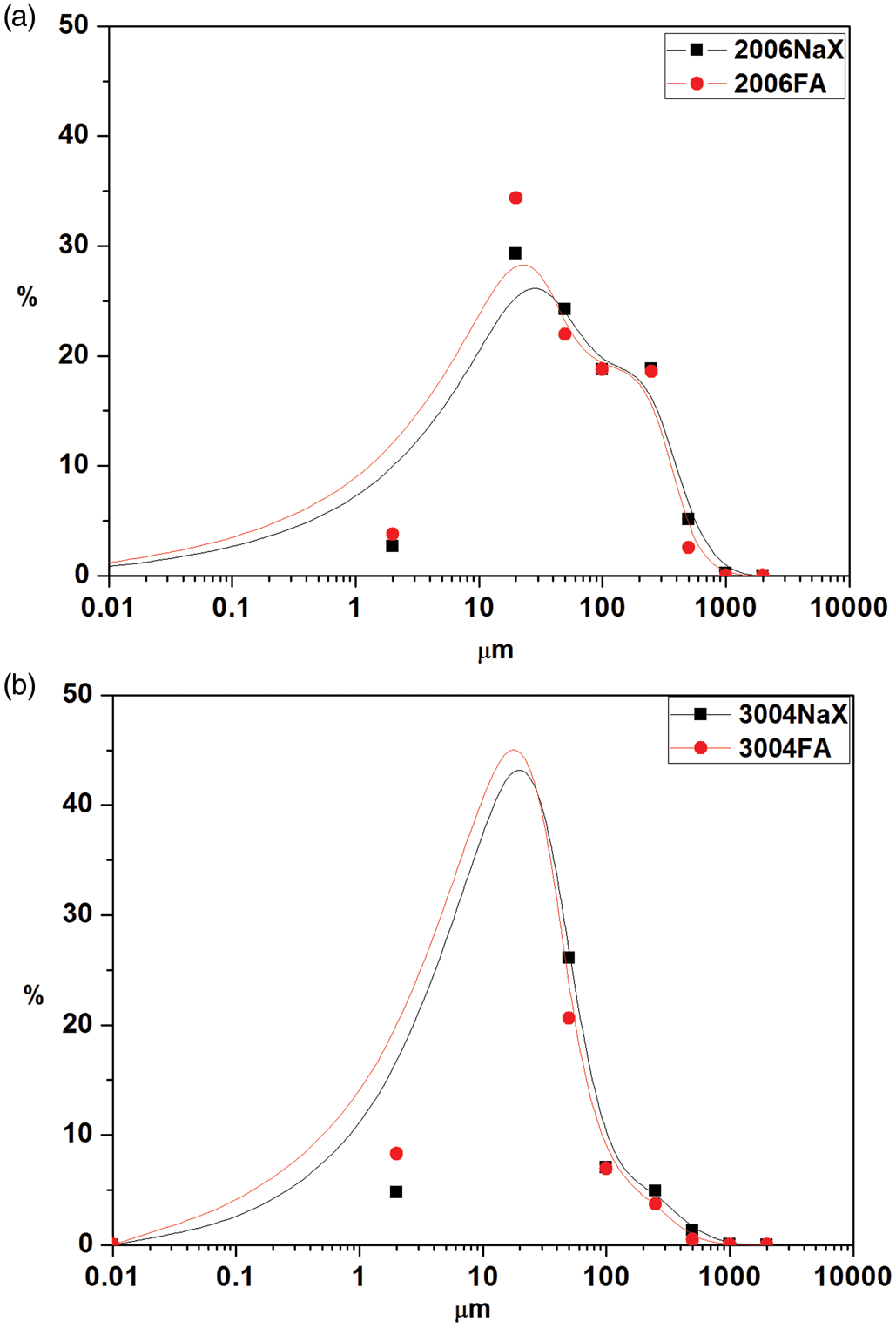
The scanning electronic micrographs (Figure 4) show the morphology of FA and obtained Na–X zeolite. FA is a fine dust whose particles are spherical in shape. As for Na–X zeolite, single crystals with irregular conformation and size 3–6 mm are shown. The edges of the particles are often sharp and jagged.
SEM image of (a) 3004 FA and (b) 3004Na–X. SEM: scanning electron microscopy.
The point of zero charge (pHpzc) values for 2006Na–X–CS and 3004Na–X–CS are shown in Figure 5. pHpzc values for them are equal to 7.20 and 7.57. This indicates that at pH value smaller than that at pHpzc the surface of the zeolites is positively charged, while at pH value greater than that the pHpzc the surface is predominated by negative charges.
Point of zero charge of 2006Na–X–CS and 3004Na–X–CS zeolites.
As shown in Figure 6 it is evident that the hydrothermal conversion reaction of FA with NaOH to Na–X leads to the improvement of CEC values of the adsorbent. This process resulted in an increase of the CEC values for 3004FA from 10.85 mval/100 g to 125.52 mval/100 g for 3004Na–X. For 2006FA and 2006Na–X these values are even higher.
Comparison of the cation exchange capacity (CEC) for 3004FA, 3004Na–X and 2006Na–X.
Sorption experiments
The sorption capacity qt (mg/g) of adsorbent at appropriate pH was calculated using equation (1)

The sorption percentage was calculated based on equation (2)

The removal of Cu(II) on FA, Na–X and Na–X–CS at different pH values in the range from 2.0 to 6.0 was studied using the Cu(II) solutions at the initial concentration of 100 mg/l. As was found in Adamczuk and Kołodyńska (2015) the adsorption of Cu(II) on FA is strongly affected by pH. The maximum removal was obtained at pH value below 4.0. With the increasing pH from 1.0 to 5.0, the residual concentration of Cu(II) ions decreases from 100 to 64 mg/l which corresponds to an increase of %S from 20 to 50%. Therefore, pH 5.0 was chosen as the more adequate for further experiments. Similar results were obtained for Na–X and Na–X–CS. For example, the most favourable pH values for both Na–X and Na–X–CS were 5.0 and 6.0, respectively. Analogous results were described in Liu et al. (2016) and they are connected with sorption on low-cost sorbents modified by CS. It should be noted that also for the mixture of Cu(II), Fe(III), Mn(II) and Zn(II) ions it was found that at pH 2.0 only 20% of Cu(II), 8.67% of Fe(III), 6.12% of Mn(II) and 3.32% of Zn(II) were sorbed. Under these conditions active sites are protonated and the competition between H+ and metal ions occurs. At high pH values %S increases and all metal ions exhibit the same trend. The pH dependence of Cu(II) adsorption on FA, Na–X and Na–X–CS can be related to both the type of functional groups (FA), cave and canals (in zeolite structure) and amine groups of CS on the surface of Na–X–CS zeolite as well as the aqueous chemistry of Cu(II) ions. According to the special diagrams produced using the MINEQL studies up to the pH 5.8 value, Cu(II), Mn(II) and Zn(II) ions were predominant as positively charged species of Cu2+ and Zn2+ ions. As for Fe(III) it exists in the Fe2O3 form (Figure 7). The same results were obtained by Lee et al. (2012). As for the mechanism of sorption at pH above 7.0, where the sorbent surface tends to be negatively charged, not only cation exchange but also metal ion precipitation should be taken into account. Typically when the concentration of hydroxyl ions increases they form hydroxides with metal ions.
Comparison of the species distribution for Cu(II), Fe(III), Mn(II) and Zn(II) solutions at initial concentration 10 µM.
The studies of the phase contact time on adsorption of Cu(II), Fe(III), Mn(II) and Zn(II) were carried out for different concentrations of metal ions and at different time intervals. However, in the first stage the sorbent dose was investigated. It was found that dosage of the used sorbent decreased with the increasing FA amount (Figure 8).
Effects of adsorbent dosage on the sorption effectiveness of Cu(II), Fe(III), Mn(II) and Zn(II) ions on FA.
As for effect of the phase contact time on the sorption effectiveness it can be noticed that the rate of removal of studied ions increases with the increasing phase contact time to a certain extent. Further increase in contact time does not increase the uptake due to deposition of metal ions on the available adsorption sites. It was found that the process of sorption of metal ions Cu(II), Fe(III), Mn(II) and Zn(II) has two main stages (Figure 9). During the first stage, fast and intensive sorption is observed. This stage lasts about 10 min. This can be explained by large availability of surface active sites. The second stage is significantly slower which indicates that the process reaches a state of equilibrium. Over the second stage sorption is on the same level because most of the active sites become adsorbed. Also other processes can occur during the second stage: ionic exchange, interactions between the metal ions and CS functional groups as well as metal diffusion into the zeolite structure. Additionally, it was found that the maximum sorption capacities for both Na–X and Na–X–CS were higher than those of FA-CS (both 2006FA-CS and 3004FA-CS).
Comparison of %S of (a) Cu(II), (b) Zn(II), (c) Mn(II) and (d) Fe(III) ions in the four-component solutions on 3004Na–X–CS.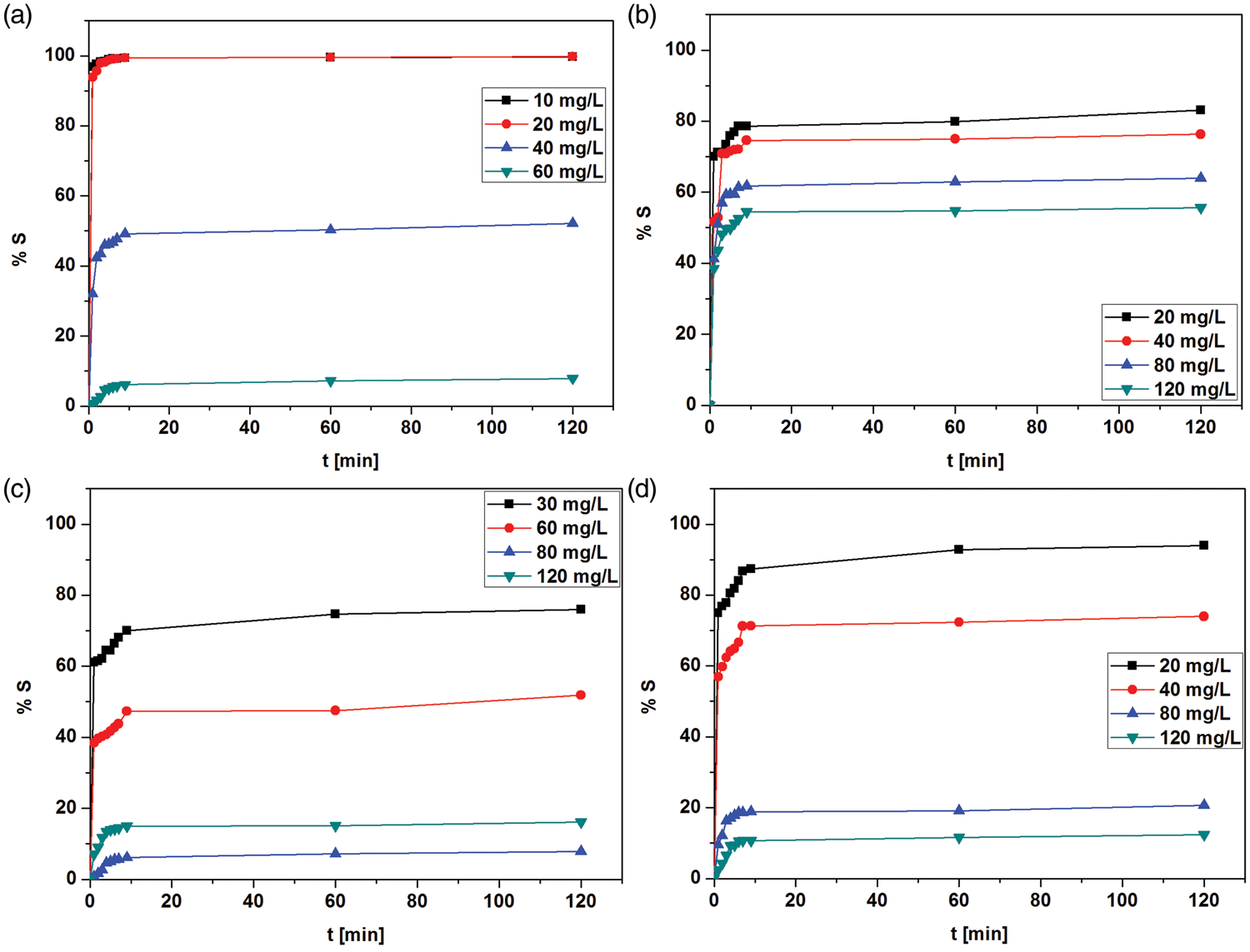
The kinetic parameters for sorption of Cu(II), Fe(III), Mn(II) and Zn(II) ions (mix3) on 3004Na–X–CS.
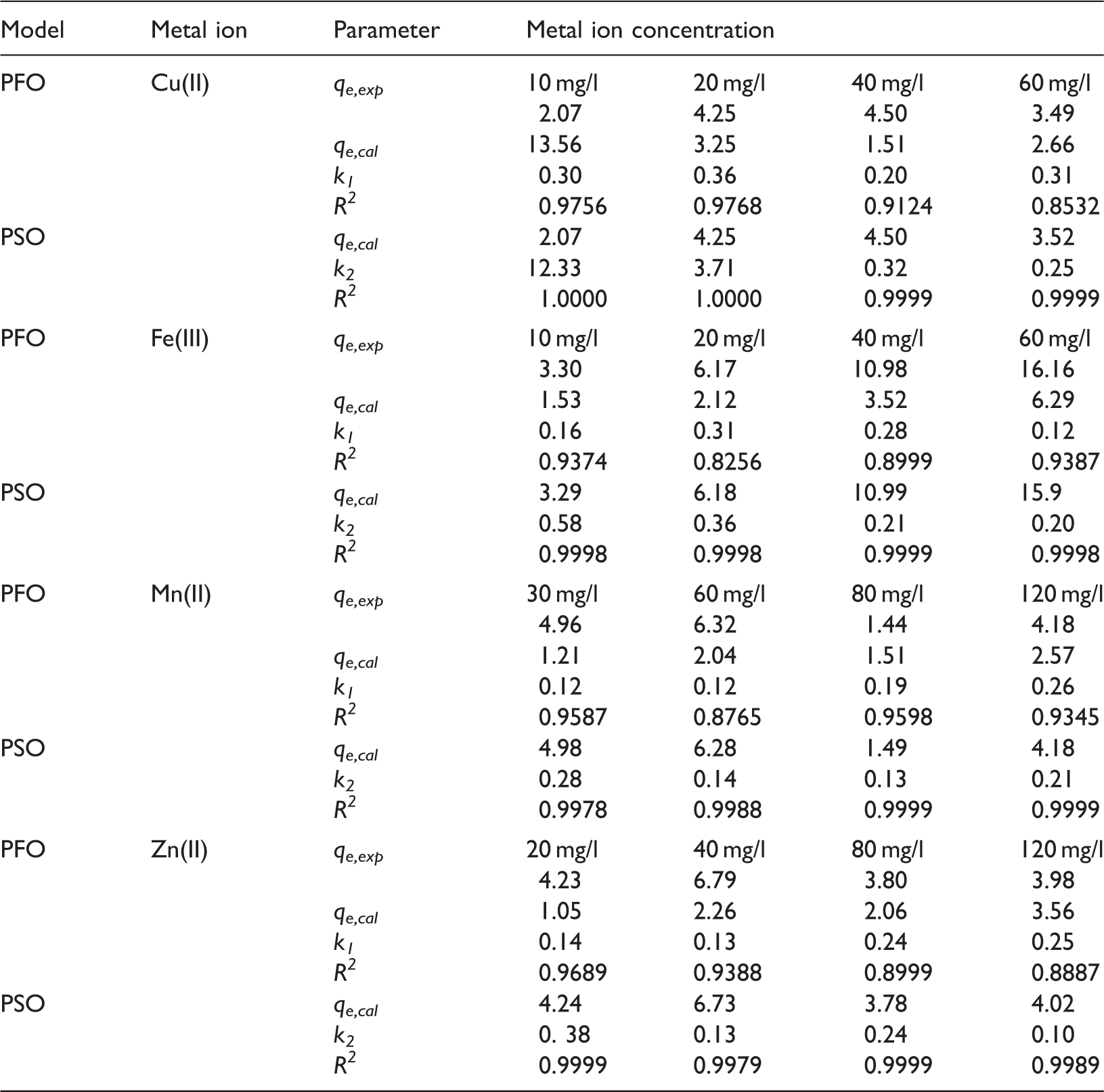
PFO: pseudo-first-order model; PSO: pseudo-second-order model.

Effect of foreign ions on sorption effectiveness of (a) Cu(II), (b) Fe(III), (c) Mn(II) and (d) Zn(II) on 3004NaX–CS.
Conclusions
The presented method allows to obtain a new kind of products with wide potential applications in the removal of pollutants from the environment. The conversion of FA under the controlled conditions was performed on a pilot scale for 24 h at 353 K. XRD, FTIR, XRF, ASAP and SEM indicated that CS was successfully attached to both FA and Na–X zeolite lattice. The XRD patterns showed that the zeolite structure has remained unchanged after the CS immobilization. Additionally, in the case of the zeolites modification by CS the results showed that the adsorption behaviour is not affected by physical properties but by chemical interactions. The solution pH plays a very important role where the highest adsorption capacity occurred at pH 5.0. It was found that the optimum sorbents dosage 20 g/l, the phase contact time 120 min and temperature 293 K can be used in sorption process. The kinetic study showed that the pseudo-second-order model was suitable for describing the removal of Cu(II), Fe(III), Mn(II) and Zn(II) ions on FA, Na–X and Na–X–CS sorbents.
Footnotes
Acknowledgements
The study was first presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the financial support from NCBiR within Project GEKON 2/O2/266818/1/2015.
