Abstract
The present article reports the easy to use approach on the application of tetraethoxysilane (TEOS) for the enhancement of drug release from mesoporous acrylic resin, in which a water insoluble drug is embedded. The investigated system combines the swelling of polymer with gelation of silica precursor within the polymer matrix. The nonsteroidal anti-inflammatory naproxen was used as a model drug and Amberlite XAD7HP was used as a drug carrier. Tetraethoxysilane was introduced into the loaded polymer up to the total pore filling. The transformation of tetraethoxysilane in aqueous solution at different pH produces condensed silica and ethanol which is a good solvent of naproxen. Appearance of ethanol in the sample facilitates naproxen release into an external solution at differentiated pH. The rate of release strongly depends on the pH of release solution due to differences in the hydrolysis and gelation rates of tetraethoxysilane. The addition of tetraethoxysilane to the polymer-naproxen composite appears beneficial for drug transport into the external media.
Introduction
The prevailing number of papers concerning the controlled release systems of drugs are focused on slow or long-time release formulations (Tarcha, 1990; Uchegbu and Schatzlein, 2006). In fact, the release of a drug is desorption of an organic component from a carrier, which is affected by the chemical character and the size of the specific surface area (sorption capacity) of the solid as well as diffusion of a release medium through the pores. Mesoporous solids are attractive hosts due to their high surface area, well-defined surface properties and tunable pore sizes (Contessotto et al., 2009; Slowing et al., 2008; Vallet-Regi et al., 2007). The chemical modification of the carrier surface with organic functional groups is a universal method of efficient drug loading and controlling of release process (Datt et al., 2012; Szegedi et al., 2011; Wang, 2009; Zou et al., 2015). Most frequently, the efforts are directed to dose the same portion of the drug in equal time periods. The constancy of dosage is of fundamental meaning for an effective long-term therapy. However, in a number of cases, mainly when the elimination of pain is required, a burst release of the drug is desirable. The fast release poses a challenge in the case of insoluble drugs in water, buffers or simulated body fluids. The rate of the release of an insoluble drug is, especially at the beginning of the process, negligibly small and without any therapeutic meaning. According to Biopharmaceutics Classification System (BCS), 30% of all oral pharmaceuticals belong to Class II model drugs, which have low solubility and high permeability (Murillo-Cremaes et al., 2014). For these reasons, further studies on increasing the solubility, and, hence, the bioavailability of this class of drugs are necessary.
Among the variety of drug release systems, those sensitive to the pH of body fluids seem to be especially promising (Chen and Zhu, 2012; Zhu and Shi, 2007). The controlled release may be initiated by a variety of external stimuli, such as for example pH, temperature or applied magnetic field (Chen et al., 2015; Deng et al., 2011; Liu et al., 2013; Ma et al., 2008; Sasidharan et al., 2012).
The aim of this paper is to demonstrate the pH effect of the release fluids on the rate of hydrolysis and condensation of tetraethoxysilane (TEOS) introduced into the polymer–drug composite, which, consequently, regulates the drug release. The drug chosen for this study is naproxen ((S)-6-methoxy-a-methyl-2-naphthalenacetic acid), which is insoluble in water, especially at a lower pH. In this study, a mesoporous non-ionic acrylic polymer, Amberlite XAD7HP, was used as a carrier for naproxen. TEOS, was used as an agent enhancing the solubility of the drug. In this place, it should be emphasized that many studies prove that live tissues demonstrate high biotolerance for silica produced via sol–gel process in liquid media (Cho et al., 2008; Radin et al., 2002). Thus, we believe that the present study is a promising new approach to developing a targeted fast drug release system.
Experimental
Materials
Amberlite XAD7HP (Rohm & Haas), naproxen (NXA, 98 %) and TEOS (98%) were purchased from Sigma Aldrich. NH4OH (25%), HCl (33–35%), and ethyl alcohol (96%) were purchased from POCH (Poland). Di-sodium hydrogen phosphate and sodium dihydrogen phosphate (98%) were received from Chempur (Poland). Prior to the sample preparation, Amberlite XAD7HP was washed with deionized water and dried at 80℃ under vacuum.
Samples’ preparation
Naproxen was loaded into XAD7HP polymer beads by wetting them with ethanol dissolved naproxen (27 mg NXA/1 cm3 of ethanol), drop by drop, until all polymer beads became swollen and glassy (1.76 g EtOH + NXA/1 g of XAD7HP). Afterwards, the sample was left under vacuum for 2 h at room temperature. Next the sample was dried for 6 h at 60℃. This procedure was repeated three times. The dried sample was designated as PNH. Naproxen content determined from the mass differences before and after drug loading exceeded slightly by 15 wt.%. Next, a part of PNH sample was loaded with the silica precursor by wetting the beads with TEOS (1.46 g TEOS/ 1 g of PNH), until all beads were swollen and glassy but did not stick to each other. TEOS is a very effective agent causing the swelling of polymers (Halasz et al., 2011; Kierys et al., 2010; Krasucka et al., 2015; Zaleski et al., 2012). Therefore, it is important for the described preparation procedure to add only so much TEOS, which can be totally entrapped within the pores of the polymer beads. The final, visually dry, products containing TEOS was labelled as PNHT.
Naproxen release
The drug release measurements were performed by adding 240 mg of PNH or 590 mg PNHT samples to a vessel containing 50 cm3 of a controlled pH release-solution and placed in a shaking incubator. These masses were chosen so that the amount of drug in samples with and without TEOS was the same. Naproxen release solutions were the following: aqueous solution of HCl at pH 1.2, 4, sodium phosphate buffer at pH 7.4 and aqueous solution of NH4OH at pH 12.5. The pH range includes physiological values between 1.2, 4 for the stomach, and 7.4 for further sections of digestive system/blood buffer. The pH 12 basic solution served as a reference system. The temperature was fixed to 37℃ and the stirring rate was 250 r/min. At predetermined time intervals, 5 cm3 of release fluid was taken out for analysis of the drug concentration with UV-Vis spectroscopy (Varian Carry 100 Bio) at a wavelength of 330 nm for naproxen. The taken aliquot was replenished with a fresh release medium.
The kinetic of naproxen release from all samples was characterized by finding the best fit of the release data (drug-released fraction versus time) to various kinetic models
First-order model describes the rate of drug release versus its initial concentration. Higuchi model is used in case of drug release from solid insoluble matrix filled with excess of drug and is based on Fickian diffusion. Korsmeyer and Peppas model describes the release of drug from polymer matrix taking into account the structure and geometry of polymers network/tablet. The Fickian diffusion mechanism is observed while n ≤ 0.5, and depends on pore geometry. Non-Fickian diffusion is observed if n value is above 0.5. In swelling-controlled release the n = 1.0, 0.89 and the 0.85 values belong to the slab, cylinder and the sphere, respectively.
Characterisation
The textural properties of the investigated materials were characterized by nitrogen adsorption experiment conducted at −196℃. The adsorption/desorption isotherms of nitrogen were measured by volumetric method using an automated sorption analyser, ASAP 2045, Micromeritics Inc. The pore size analysis was based on the Barrett–Joyner–Halenda procedure (Barrett et al., 1951). The specific surface area, SBET, was calculated using Brunauer–Emmett–Teller method (Brunauer et al., 1938) for adsorption data in a relative pressure p/p0 range from 0.01 to 0.25. The total pore volume, Vp, was estimated from a single point adsorption at p/p0 = 0.989.
Optical images of dry polymer XAD7HP beads and the beads swollen in TEOS were taken with an inverted material microscope, MA200 M, Nikon Co.
Results and discussion
Swelling experiment
The Amberlite XAD7HP resin swells very easily both in the alcoholic solution of naproxen and in TEOS. The swelling ratio S% was calculated as: S% = (VS − VI)/VI × 100%, where VS is the volume of swollen polymer/polymer-naproxen beads, VI is the initial volume of polymer/polymer-naproxen beads. The addition of naproxen solution and TEOS causes more than twofold increase of the polymer particle volume S% = 170% and 168%, respectively. The swelling ratio determined by the volume change before and after adding TEOS for the polymer with naproxen (PNH) equals S% = 80% and differs significantly from those obtained for the pure polymer. The much lower swelling ratio for the composites is the result of the presence of the drug in the sample and the stiffening of the polymer skeleton (Halasz et al., 2011; Kierys et al., 2010; Zaleski et al., 2012).
Textural characterization
Drug loading changes also the structural parameters of polymer sample. Figure 1(a) shows the nitrogen adsorption–desorption isotherms for the initial polymer and the sample loaded with naproxen, while Figure 1(b) depicts the BJH PSD curves. The pure polymer is characterized by a bimodal pore size distribution, demonstrating a narrow peak at smaller pores and a wide PSD for the second group of larger pores. The specific surface areas, the total pore volumes, and the mesopore diameters at the peaks of PSD for the investigated samples are given in Table 1.
(a) Nitrogen adsorption (solid points) and desorption (open points) isotherms at 77 K and (b) the pore size distribution of XAD7HP polymer – black triangles, PNH – blue squares. The parameters characterizing the porosity of the investigated samples determined by the nitrogen adsorption–desorption isotherms. Dn: pore diameters at the peak of pore size distributions (PSDs) of two main groups of pores.

As seen in Table 1, the total pore volumes strongly differ for the parent polymer and the samples with embedded naproxen. The pore volume is strongly elevated in the case of PNH. The increase of Vp after the drug loading seems to be somewhat surprising and may be explained by the irreversibility of the swelling of the initial polymer in the ethanol–naproxen solution. The solid component (drug particles), embedded via impregnation, might cause the stiffening of the polymer matrix and the stabilization of polymer particles in the swollen state. The presence of naproxen results in a partial pore blocking in the parent polymer and, simultaneously, the creation of new pores, representing the space between the nanoparticles/nanocrystals of naproxen. The total pore volume of PNH is moderately changed in comparison to the initial polymer. The formation of new interparticle pores is reflected by the appearance of a wide peak of PSD centred at D2 = 33 nm (Figure 1(b)).
The addition of the liquid silica precursor (TEOS) in the last stage of the sample preparation procedure leads to the total pore filling. Thus, the parameters characterizing the porosity of PNHT sample are negligibly small.
Drug release
The curves of the naproxen release are presented in Figure 2(a). As it follows from the kinetic curves, the pH of the release medium affects the process of naproxen desorption from both the polymer–drug composite and the sample containing TEOS. The release curves for pH 1.2, characterized by very low drug release (0–2%), are ommitted for the clarity of the presentation.
Naproxen release curves (a) at pH 4 – red circles, pH 7.4 – green squares and pH 12.5 – black diamonds, full points: samples without TEOS, open points: samples with TEOS and (b) amount of released naproxen after 30 minutes at pH 1.2, 4, 7.4 and 12 for PNH red (full squares) and PNHT (open squares).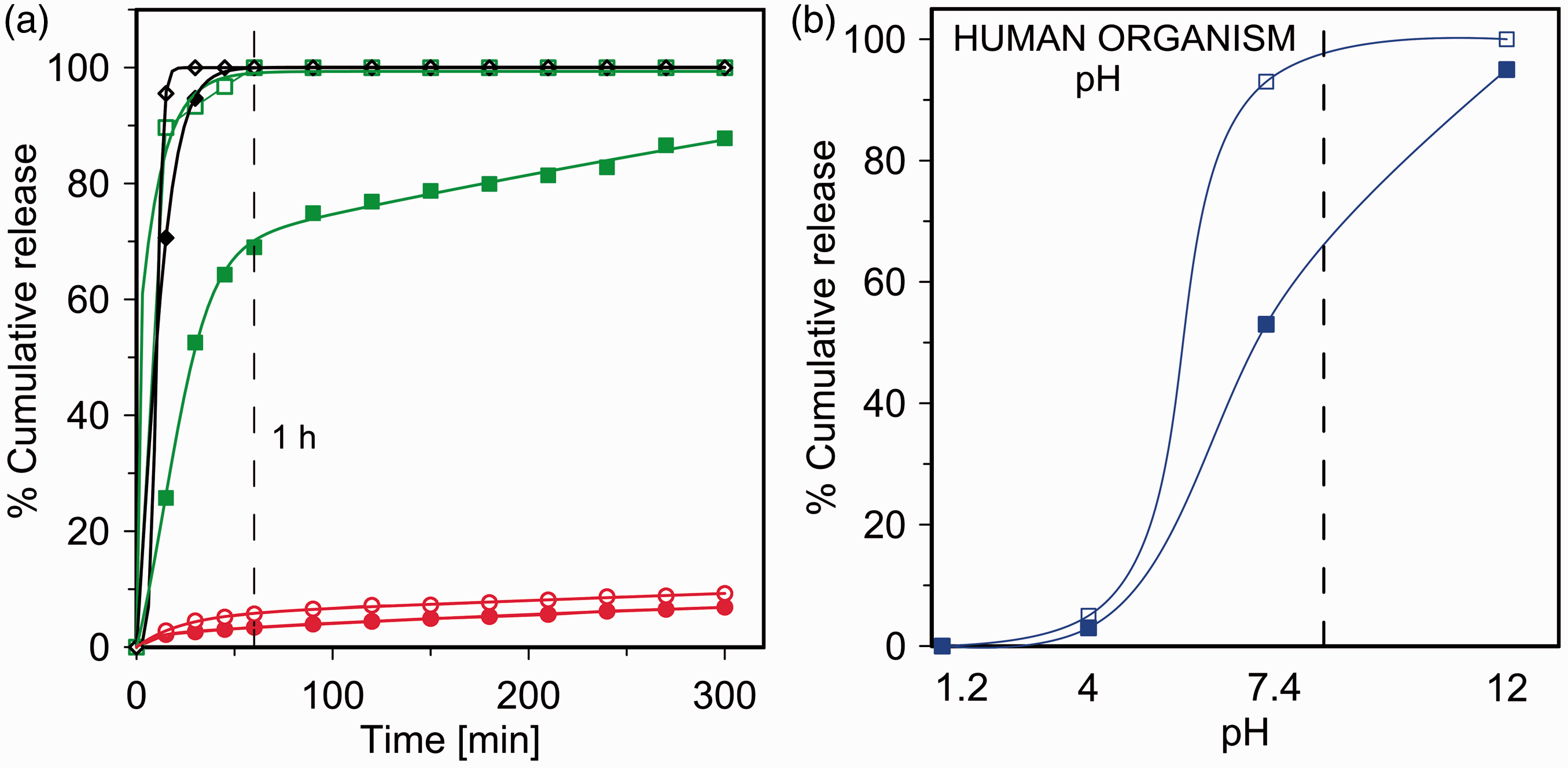
The release of the drug depends not only on the solubility of naproxen in the release solution, but also on the presence of TEOS in the composites. The amount of the released drug increases together with the increasing pH value, which is the result of an enhanced solubility of naproxen in the basic environment. The sample PNH demonstrates 100% release in the solutions at higher pH. In general, the composite sample show a lower release rate of naproxen for all used pH than the sample saturated with TEOS. TEOS enhances the release for both pH 4 and 7.4, which are the physiological pH values.
For samples immersed in the release solution of pH 7.4 and 12.5, the burst effect is observed with more than 80% of naproxen being removed within the first hour. Sample PNHT exhibits 90% burst release after 15 minutes at pH 7.4 and about 100% after 1 hour. In this case, the presence of the TEOS causes the highest increase in the solubility of naproxen, which is highly beneficial because it is the physiological pH. At pH 4, there is a negligible release of naproxen. At this pH, naproxen dissolves to a small extent, because it is an acid (pKa = 4.15). However, the polymer with TEOS exhibits more than 5% release within 45 minutes. The delay of the release rate shows that the hydrolysis of TEOS in an acidic medium is slow and the production of ethanol, in which naproxen is soluble, is also a lengthy process, resulting in only 10% after 5 h in both samples. For illustrative purposes and the convenience of the observation of the TEOS effect, Figure 2(b) shows the rate of release for a series of samples with and without TEOS at different pH. The broken line indicates the limit of physiological range of pH. The release is improved as the pH increases from typical stomach pH values 1.2–4 to 7.4, characteristic for fluids in further segments of gastrointestinal tract and human blood. The observed tendency in drug release is beneficial because of the relatively low release in the stomach and strongly elevated release at higher pH. In a basic solution, the release is always high whether or not the sample contains TEOS. Higher rates of naproxen release for the samples with TEOS may be due to the fact that, in the process of TEOS hydrolysis, ethanol is produced (Buckley and Greenblatt, 1994), which is a good solvent for naproxen (Roberts, 2007; Roberts et al., 2007).
Correlation coefficient (R2) and rate constant values (K) for release kinetics of naproxen.
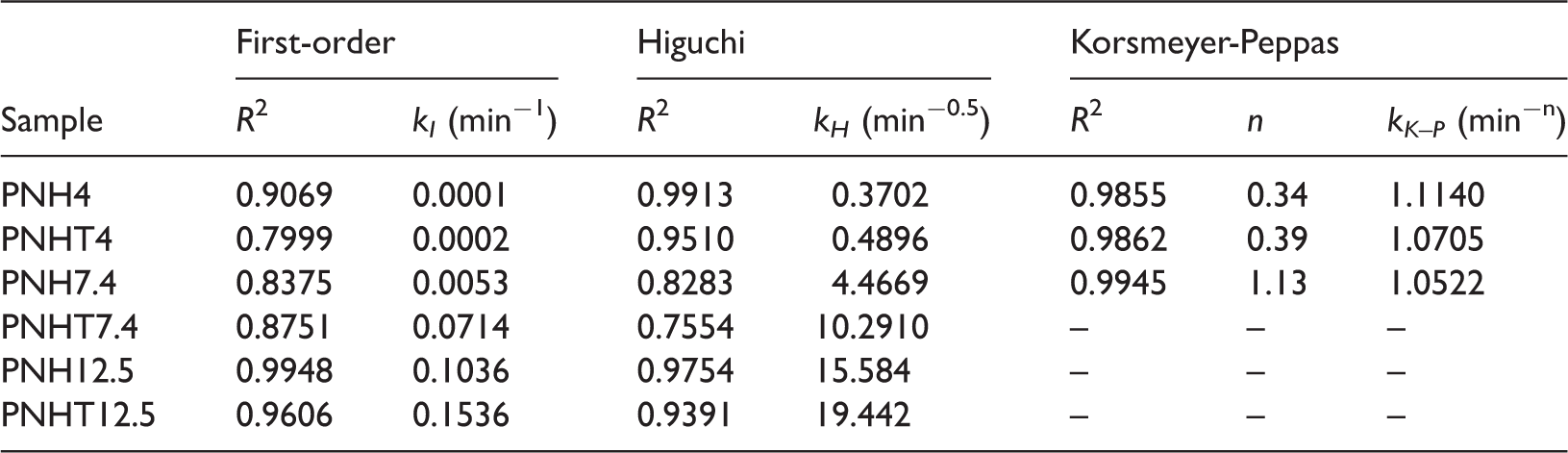
The parameters collected in Table 2 (Dash et al., 2010; Lao et al., 2011; Siepmann and Peppas, 2001) correlate reasonably well with the shape (the slope of the initial segment) of the release curves. In the case of the PNH sample the best match at pH 4 is Higuchi model (R2 = 0.9913) and Korsmeyer–Peppas model for the sample with TEOS (R2 = 0.9862; n = 0.39) characteristic for classical Fickian diffusion release mechanism for n ≤ 0.43. At pH 7.4, Higuchi model seems to be appropriate for the sample with TEOS (R2 = 0.9395) but Korsmeyer–Peppas fits best polymer–drug composite in the absence of TEOS (R2 = 0.9945). With a strong basic solution (pH = 12.5), naproxen is released with the first-order kinetic mechanism for the sample with and without TEOS. This mechanism corresponds likely to the fast dissolution and diffusion of drug from porous materials (Dash et al., 2010).
Conclusions
Naproxen was successfully loaded into the mesoporous acrylic resin Amberlite XAD7HP by the impregnation method. A new type of drug polymer carrier was prepared through the modification of the internal pore system of polymer–drug composite with TEOS as a release supporting component. It has been shown that the swelling of the polymer, hydrolysis of TEOS with the simultaneous production of ethanol and the gelation of silica species all affect the drug release rate. The results of this study prove that hydrolysis of TEOS introduced into the pores enhances the solubility of naproxen. The TEOS effect is most pronounced in solutions characterized by an intermediate pH level. During the hydrolysis of TEOS ethanol is produced, which is a very good solvent for naproxen. The silica species produced in the same process condense to silica gel. As the formation of alcohol takes place in situ, the polymer–drug–TEOS particles behave like a microreactor, in which the initiation of dissolution takes place when the system is exposed to basic body fluids. The level of drug release is the function of the actual pH of these fluids.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


