Abstract
MWCNTs produced by using Chemical Vapor Deposition (CVD) technique were functionalized with nitric and sulfuric acid to ensure a good dispersion in an aqueous solution. The FTIR analysis further confirmed the presence of carboxyl and hydroxyl groups on the surface of MWCNTs after acid treatment. The two types of MWCNTs composites were prepared using pristine and functionalized MWCNTs (p-MWCNTs and f-MWCNTs) in alumina matrix by sol-gel processing and to the best of our knowledge it is a new approach in the field of CNT based ammonia gas sensors. The Raman analysis showed that both the pristine and functionalized MWCNTs retain its pristine characteristics in alumina matrix even after the sol-gel processing. A precisional electronic circuit was developed to measure the sensing response of these two composite samples to detect ammonia gas and a detailed comparative analysis was done. While comparing the sensing performance, the functionalized MWCNTs composite sample was found to be more sensitive to ammonia gas with short response time but did not recover at room temperature because of the strong bonding between ammonia molecule and oxygen containing groups on the surface of MWCNTs.
1. Introduction
Since the discovery by Iijima in 1991 carbon nanotubes (CNTs) [1] have been drawing the prime attention of researchers because of its exceptional chemical, mechanical and electrical properties. It is well known that CNTs can be considered as appropriate candidates to design high feature gas sensors due to their good conductivity, excellent mechanical properties, and large specific surface[2] hence capable of detecting a small concentration of molecules, such as CO, NO2, NH3 H2 etc. [3,4]. Although tremendous progresses have been achieved in this area, the underlying sensing mechanism still remains unclear.
In recent years, incorporating CNTs into various matrix materials such as polymer and ceramic matrix, has attracted much attention since it offers further scope for improving their sensing characteristics, such as enhancing sensitivity and selectivity, lowering the detection limit, extending detection capacities to an ever-increasing number of gases, sensing at room temperature, etc. [5–6]. As ceramic materials are known to have excellent mechanical properties and relatively low density, hence reinforcement of CNTs into a ceramic matrix might be expected to impart stiffness, strength, and toughness to CNTs/ceramic composites[7].
However in such application as reinforcement of fillers in ceramic matrix, CNTs must demonstrate good dispersion, high interfacial stress transfer and better alignment of CNTs in the matrix before they can be used as a gas sensor. To enhance the solubility of CNTs several methods of chemical functionalization have been developed. The most widely used approach, is wet chemical oxidation process which is initiated by mixing CNTs with strong acids such nitric acid, sulfuric acid or a mixture of the two[8].
In this work two types of CNT based ammonia gas sensors have been developed by depositing pristine and functionalized MWCNTs in Alumina matrix by sol-gel method that is reported for the first time. A detailed comparative study was carried out for the various sensor parameters such as sensitivity, response-recovery time and possible underlying sensing mechanism. We have been successful in our attempt to explain the possible ammonia gas sensing mechanism with experimental evidences, for both pristine and functionalized MWCNTs and they are in agreement with the theoretical predictions reported by many authors [9,10].
2. Experimental Section
2.1. Preparation of alumina solution using sol gel process
Alumina solution was prepared from 20 gm of Al-Sec.-Butoxide (Al (OCH [CH3] C2H5)3) (procured from Alfa Aesar)used as a precursor, dissolving in 150 cc hot water and keeping the solution at 90°C under constant stirring for 45 minutes using magnetic stirrer. Peptization was done by adding 0.6 cc of concentrated HCL (procured from Merk) and the solution was kept overnight for refluxing under constant stirring. After that the calculated amount of Poly Vinyl Alcohol (PVA) (obtained from Sigma-Aldrich) used as a binder, was added to the solution and a transparent sol solution was obtained [11, 12].
2.2. Purification and Functionalization of MWCNTs
MWCNTs produced by CVD (with a diameter of 10–30 nm and a length of 6–15 μm) were used as a starting material. As produced MWCNTs were first purified by heating at 400°C for 5 hours in air to remove carbonaceous impurities. Then purified MWCNTs were ultrasonicated for one hour using Ultrasonicator(model Elmasonic P) at 37kHz frequency(50Watt), in a mixture of sulfuric acid and nitric acid (3:1 H2SO4:HNO3). Then the mixture was refluxed at 70 °C for four hours with constant magnetic stirring, followed by filtration of the acid mixture using cellulose Nitrate Membrane filter paper with pore size of the order 0.2μm. The residue was re-suspended in deionized water obtained from a Millipore system. This process was repeated several times until the pH of the solution reached up to neutral value. The acid treatment removed all the metallic impurities and greatly enhanced the solubility of MWCNTs in polar solvents by introducing carboxylic (-COOH) functional groups to the sidewalls and ends of MWCNTs[8], that is further confirmed by FTIR characterization as shown in Fig.2.

Picture of the sensor fabricated on alumina substrate patterned on PCB
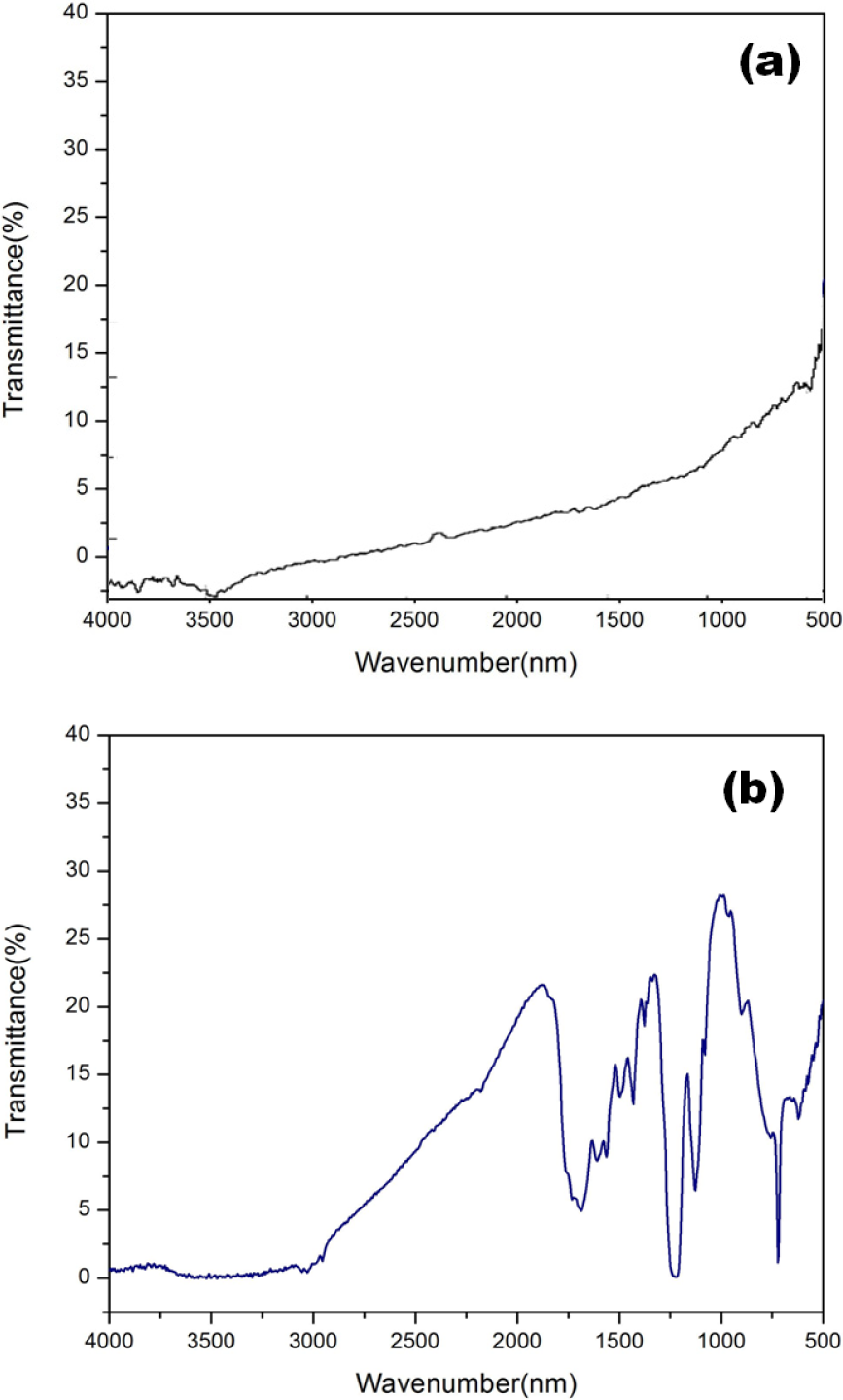
FTIR spectra of
2.3. Preparation of MWCNT /Alumina composite films
The two types of composite were prepared by mechanical mixing of MWCNTs (60mg p-(pristine) or f-(functionalized)) in 5ml Alumina sol solution for 24 hours using a magnetic stirrer. The MWCNTs/Alumina composite was obtained in the form of a paste and it was coated on Alumina substrate of dimension 2cm ×2cm by screen printing technique to get a thin film.
2.4. Gas sensing device fabrication and measurements
MWCNTs/ Alumina composite (paste) coated Alumina substrates were cured at 450°C in air to get a fine rigid film. Then Silver electrodes were made at the two end terminals of the cured samples and fired again at 200°Finally these samples were patterned on PCB (printed circuit board) to obtain a thin film gas sensor as shown in Fig. 1.
The gas sensor was placed in a small and sealed sensing chamber made up of steel. Nitrogen gas (which acted as a carrier gas) was made to flow into the chamber continuously before the test gas was introduced. The resistance of the composite film was recorded when the system reached to steady state. The device was evaluated for its sensor response by measuring the change in voltage across the two terminals of the dummy resistance of 1KΩ when exposed to alternate cycles of ammonia (Test Gas) and nitrogen(carrier gas) using a precisional electronic circuit(applied voltage: 30 V) at room temperature as shown in section 3.2. On exposure of the ammonia gas the resistance across the sensor terminals changes that cause the change in current in the circuit. This leads to the change in voltage across the dummy resistance (1KΩ) that we measured as electrical response of the sensor.
In order to examine effects of the functionalization of MWCNTs on its ammonia sensing properties, two samples were fabricated and tested for 100 ppm of ammonia at room temperature, one with p- MWCNTs /Alumina composite and other with f- MWCNTs /Alumina composite.
3. Results and Discussions
3.1. Material characterization
3.1.1. FTIR analysis
Fig. 2 shows comparative FTIR spectra for p-MWCNTs and f-MWCNTs, which confirms the presence of –COOH and –OH functional groups on the surface of MWCNTs after acid treatment [13–14].
3.1.2. Raman analysis of CNT composite film
The Raman measurements of the samples were carried out by micro-Raman spectrophotometer (LabRAMHR800, JY) fitted with peltier cooled CCD detector and an Olympus BX-41confocal microscope. The excitation of the samples was performed with an air-cooled Ar+- laser (Spectra Physics) tuned at 488 nm. The spot size was 1.19 μm at sample surface under optimal conditions. Measurements were carried out in the back scattering geometry using a 50X LWD microscope objective. The laser power was kept low on the sample surface to avoid excessive heating.
Fig. 3 A shows the Raman spectra for both pristine and functionalized MWCNTs. It is clear that acid treatment does not destroy the structure of MWCNTs as both the characteristic peaks of D (at 1330 cm-1) and G (at 1580 cm-1) bands can still be recognized in Raman spectra of f-MWCNTs and p-MWCNTs [15]. Also it does not produce any significant amount of defects as there is negligible change in intensity ratio (Id/Ig) of D to G band.

(A) Raman spectra of
The Raman analysis of composite films as shown in the Fig. 3 C implies that pristine and f-MWCNTs retain its characteristics and remained in alumina matrix even after the sol gel processing, where the characteristic (D and G) bands of MWCNTs are clearly visible while those are absent in the Raman spectra of pure Aluminum oxide (Fig. 3 B). Another noticeable fact observed is that the Raman signal of both p-and f-MWCNTs/Alumina composite is due to its MWCNTs origin not overlapped either by the signal of Alumina in composite nor from the substrate. It may be due to the right stoichiometry of CNT as well as its good dispersion achieved during composite formation and this has been confirmed by taking the series of Raman spectra on the surface of both the samples.
3.1.3. Scanning Electron Microscope (SEM) analysis of composite film
Scanning Electron Microscope images of both p- and f-MWCNTs composite films are shown in Fig. 4. SEM images were taken using Scanning electron microscope (JSM 6380 JEOL). The SEM observations clearly indicate that both f-MWCNTs and p-MWCNTs are homogeneously dispersed in Alumina matrix that is in good agreement with Raman spectroscopy results.

Scanning electron microscope (SEM) images of
3.2. Sensor response
The precisional electronic circuit, which was developed to measure electrical response of the sensor upon ammonia gas exposure, is shown in Fig. 5.
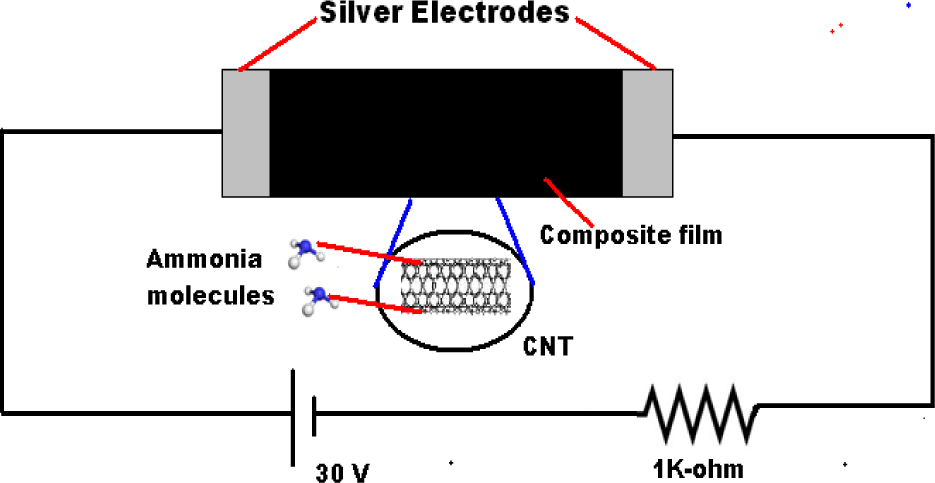
Precisional electronic circuit that was developed to measure electrical response of the sensor upon ammonia gas exposure
Fig.6 shows the electrical response of two types of sensors to approximately 100ppm of ammonia gas when examined consecutively for five days. The increase in electrical resistance upon ammonia exposure on sample was observed that leads to decrease in voltage across dummy resistance. It is observed that the complete recovery for f-MWCNTs composite sample was not achieved at room temperature even after nitrogen flow (for 24 hours), while p- MWCNTs composite samples recovered completely at room temperature within 24 hours.

Voltage variation across dummy resistance
Our observations for change in sensitivity with time are shown in Fig. 7. On day one the sensitivity for f- MWCNTs sample was found to be extremely high in comparison to the first day sensitivity of p-MWCNTs sample. But because of the fact that f-MWCNTs sample did not recover to its initial value at room temperature, its sensitivity became almost half the initial value on day two but no appreciable fall in sensitivity was noticed further as shown in Fig. 7 (b). While in case of p- MWCNTs sample the sensitivity was kept on falling day by day with almost same order of magnitude in spite of having complete recovery as shown in Fig. 7a. These experimental results can be explained as during the acid treatment of CNTs it is possible that oxygen chemisorbed at defect sites, where dangling bonds were present. So no longer it remains pure carbon and there are oxygen containing groups present on the acid treated CNTs surface. And it is the presence of those groups that enhances the interaction between the CNTs and ammonia gas molecules at any temperature.

Sensitivity of
Sensor parameters analysis at room temperature.

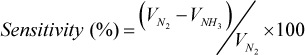
Where VNH3 the voltage is across dummy resistance in presence of ammonia gas and VN2 is the voltage in nitrogen gas. It can clearly be observed that p-MWCNT composite sample recovered completely to its initial position at room temperature in 24hours but f-MWCNT composite sample does not even after 96 hours. These results support the fact that ammonia interaction with p- MWCNTs is dominated by physisorption which can easily be overcome at room temperature by nitrogen flow [9]. While ammonia interaction with f-CNTs where oxygen containing functional groups are present on CNTs surface chemisorptions is dominated and due to the strong chemical bonding chemisorbed molecules are hardly removed from CNTs upon air exposure [8–9], resulting in slow recovery of functionalized CNTs based ammonia sensors [16–17]. Hence it is clear that some external energy is needed to break the strong chemical bonds between ammonia molecule and oxygen containing groups of f-MWCNTs to achieve complete recovery.
5. Conclusions
In this work the effect of functionalization was studied on the ammonia sensing properties of MWCNTs/ Alumina composite film. A rigid and sophisticated electronic system was developed using these films as sensing element, which could successfully detect 100ppm of ammonia gas at room temperature. It was observed that the sensitivity of the MWCNTs/Alumina composite sensor was enhanced with very short response time when f- MWCNTs were used to prepare sensing film in place of p-MWCNTs. It happened because of the presence of oxygen containing functional groups on MWCNTs surface which have very high affinity for ammonia molecules as compared to p-MWCNTs at room temperature. Hence according to this work f- MWCNTs are the best candidate for making an appreciable device that senses ammonia exceptionally well. But at the same time the recovery of this sensor to its initial position at room temperature is a big problem and presently our research group is working on this issue and upto certain extent we have been successful in our objective and to be reported shortly.
Footnotes
6. Acknowledgements
The authors are thankful to Ministry of Information and Technology (MIT) for providing the financial support
