Abstract
The objective of the present study was to investigate the adsorption behavior of manganese oxide nanowires as an adsorbent for the removal of dissolved toluene from underground water. The nanowire membrane, composed of three-dimensional porous nanostructures plus superhydrophobic character, is very suitable for removal of the hydrophobic molecules. The effects of adsorbent dose, contact time, initial solution concentration, pH, salinity, and recyclability on the uptake of toluene by the adsorbent in batch mode are examined. The adsorption data compared to the adsorption of two kinds of activated carbons show that manganese oxide nanowire is competitive to activated carbons. Furthermore, the equilibrium data are fitted to different types of adsorption isotherms. Freundlich isotherm model illustrated the best fit to the data. The results of this study suggest that nanowires of manganese oxides can be used as a low cost, highly efficient adsorbent for the removal of dissolved hydrocarbon from aqueous solution.
Introduction
Accidental or intentional leakage from oil reservoirs, gas stations, and pipelines are the most common cause of groundwater oil pollution. Consumption of contaminated groundwater can have serious environmental and health effects. Among the various petroleum hydrocarbons, benzene, toluene, ethylbenzene, xylene (known as BTEX) and their derivatives are the most common soil and groundwater contaminants (Picone, 2012). High water solubility of BTEX compounds relative to other petroleum components results in higher mobility in the subsurface water (Farhadian et al., 2008). Problems such as offensive odor, taste, and color appearance in underground water were clearly observed at a few kilometers far from the local refineries by our research group. The presence of such contaminants has a detrimental impact on the quality of life, drinking, and agricultural water in the area. Observations showed that the polluted water around local refineries contains compounds such as benzene, hexamethylbenzene, methylbenzene, toluene, ethylbenzene, etc. (Ansari and Habibagahi, 2006; Azimi and Habibagahi, 2003; Roghanian, 2008; Sadeghi, 2011; Talebi and Habibagahi, 2011). In this research, toluene was selected as a representative, because it has good solubility and less toxicity.
A short period of exposure time to the toluene at levels above the maximum contaminant level (for toluene concentration above 1 mg/l) may cause health effects such as kidney, liver, and nervous system problems. For the long-time exposure, more pronounced nervous impairments, disorders in speech, vision, memories, and liver and kidney damages are expected (EPA, 2009).
For soils and groundwater contaminated with oil, different treatment procedures have been proposed which are based on physical-chemical or biological processes. Among physical–chemical methods, air stripping, chemical oxidation, and adsorption are the most recommended methods (US Army, 1988). Techniques that incorporate methods such as flotation, gravity, and coalescing, which are traditionally employed to remove dispersed hydrocarbons, can only have a limited effect on the removal of dispersed hydrocarbons. Thus, other fundamental mechanisms like absorption, adsorption, extraction, membrane filtration, or oxidation have to be used in order to remove hydrocarbons (Meijer and Madin, 2010).
Adsorption is a process that occurs when a gas or liquid solute accumulates on the surface of a solid or a liquid (adsorbent), forming a molecular or atomic film (the adsorbate). Granular activated carbon (GAC) has been used successfully and recommended for the advanced (tertiary) treatment of municipal and industrial wastewater. However, it has some disadvantages (EPA, 2000), namely construction, maintenance and regeneration costs, and unsuitability for harsh conditions (e.g. temperature, pH, etc.) (Crittenden et al., 2012).
Many other kinds of adsorbing materials have been introduced. Sorbents for removing oil from water can be classified as inorganic mineral, synthetic organic, and natural organic materials. However, high cost, low sorption capacity, and environmental problems after usage are the main drawbacks of these techniques (Cojocaru et al., 2011; Korhonen et al., 2011; Payne et al., 2012; Radetic et al., 2008; Teas et al., 2001; Wang et al., 2012, 2010; Yuan and Chung, 2012; Zhu et al., 2011).
Meanwhile, several attempts have been made to invent high efficient materials (which means superporous and superhydrophobic); superabsorbent polymer materials (Kabiri et al., 2011; Kabiri and Zohuriaan-Mehr, 2003, 2004; Zohuriaan-Mehr and Kabiri, 2008), nanoporous materials like hydrogel composites (Nguyen et al., 2014), carbon nanotube (Hashim et al., 2012), nano manganese oxide nanowires (Yuan et al., 2008), and other materials (see Adebajo et al., 2003).
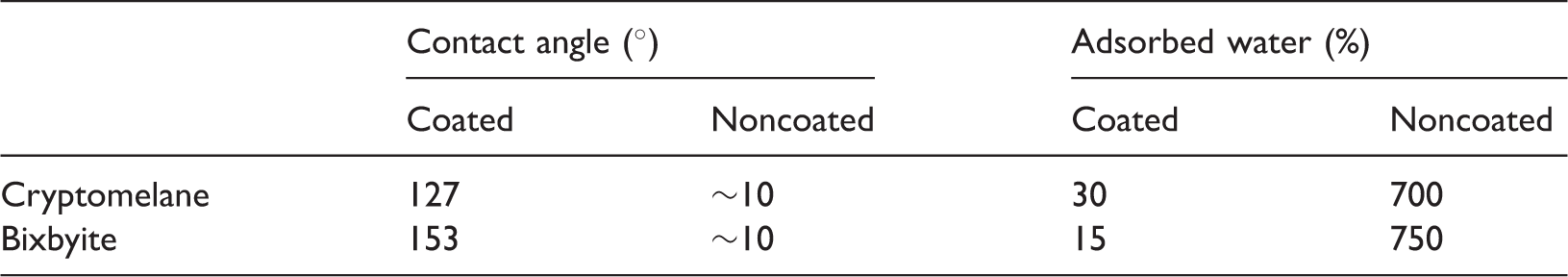
All of these attempts were made to structure a porous hydrophobic material. Hydrophobic and hydrophilic are frequently used descriptors of surfaces. A surface is hydrophobic if it tends not to adsorb water or be wetted by water and a surface is hydrophilic if it tends to adsorb water or be wetted by water. A simple, quantitative method for defining the relative degree of interaction of a liquid with a solid surface is the contact angle of a liquid droplet on a solid substrate. If the contact angle of water is less than 10°, the surface is designated superhydrophilic. For the contact angle less than 30°, more than 90°, and more than 150°, the surface is considered as hydrophilic, hydrophobic, and superhydrophobic, respectively (Arkles, 2011).
Nanowires of manganese oxides were first made and examined for decontamination of oily water by Yuan et al. (2008). Despite many synthesis methods to form one-dimensional manganese oxides (nanorods and nanowires) (Händel et al., 2013; Haris et al., 2014; Luo et al., 2000), their synthesis method conditions successfully fabricated elongated nanowires, forms an intertwined, self-assemble, and free-standing membrane. The final product was composed of superhydrophilic fibers that form a net of open superwetting capillaries coated with a superhydrophobic coating. They tested this membrane for a collection of organic solvents and dispersed oil, just to show the potential of the uptake capacity of the membrane. However, in their research, detailed study on the removal of dissolved hydrocarbons was not conducted to consider the effects of different parameters.
In this study, removal of dissolved toluene in water with nanowires of manganese oxide in different environmental conditions was investigated. Also, the synthesis process was optimized for the coating time. Effect of concentration, pH, salinity, and recyclability of this material was evaluated and the equilibrium data were fitted to different types of adsorption isotherms. For the sake of comparison, adsorption of toluene by two kinds of activated carbon (AC) was also evaluated. The results showed that this adsorbing material can be used in the purification systems such as permeable reactive barriers to decontaminate underground water from dissolved oil pollutions.
Materials and methods
Toluene (99%), manganese sulfate monohydrate MnSO4·H2O (MW 169.02), potassium sulfate K2SO4 (MW 174.27), and potassium persulfate K2S2O8 (MW 270.35) were all purchased from Merck (Germany). Two kinds of GAC were obtained from Merck: charcoal activated catalog no. 1.02514.1000 and 1.02518.1000.
The surface morphology of the synthesized nanowires was analyzed using FE-SEM (HITACHI (S-4160)) for powder samples. X-ray diffractometry (XRD) spectra were obtained using a D8ADVANCED diffractometer operating at 2θ step size of 0.01° and speed 1°/min. Total organic carbon (TOC) was used to screen samples for toluene. TOC of samples was measured by Shimadzu 500 TOC analyzer. The Shimadzu TOC analyzer adopts the 680℃ combustion catalytic oxidation method. For each specimen, five samples were analyzed, by injecting 15 µl sample into the TOC analyzer. The results include total carbon (TC) and inorganic carbon (IC) and TOC (=TC−IC).
Synthesis
Nanowires were synthesized via a hydrothermal reaction described by Yuan et al. (2008). Reactants for this process consisted of 19.1 mmol of K2SO4, K2S2O8, and MnSO4·H2O in a ratio of either 1:2:1, respectively, in 35 ml of deionized water. The solution was transferred to a Teflon vessel held in a stainless steel vessel. The sealed vessel was placed in an oven and heated at 250℃ for four days.
The product was suspended in 500 ml of deionized water and stirred for a day. The suspension was filtered and washed five times until all soluble impurities were removed from the solid. Moisture from the solid was removed by dispersing the solid on a Teflon substrate in an oven at 85℃ for 24 h.
The cryptomelane type of manganese oxide (K2Mn8O16) was produced through the above-mentioned method. Hydrophilicity before coating and hydrophobicity after coating were evaluated as described later.
To transfer this material to a higher degree of hydrophilicity (which means a higher degree of hydrophobicity after coating), the following procedure was carried out as described by Yuan et al. (2008). The cryptomelane type of manganese oxide nanowires was first placed in a solution of NH4Cl (27 g/l), resulting in ion exchange of K+ within the cryptomelane structure with NH4+ cations. Heating of the nanowires which were doped with NH4+ at 600℃ leads to the oxidation NH4+ and subsequent conversion to N2 gas. This product is bixbyite type of manganese oxide. For this type of manganese oxide, hydrophilicity before coating and hydrophobicity after coating were also measured. The morphology and crystallinity of the produced nanowires were analyzed via scanning electron microscopy (SEM) analysis and XRD.
Coating
The nanowire of manganese oxide is a superhydrophilic material. To use this material as an oil adsorbent, it needs to be coated with a hydrophobic material. Here, a simple vapor deposition method (VDM) was employed (Yuan and Dong, 2014; Yuan et al., 2008). A polydimethylsiloxane (PDMS) stamp and the nanowire membrane were placed in a sealed box heated at 250℃ for a specific time.
Batch tests
Solution preparation
The toluene solution was prepared by adding 0.52 g of toluene to 1000 ml of distilled water. Maximum solubility of toluene is 0.52 g/l (20℃) reported by Merck product information. The solution was stirred vigorously and then gently for 30 min at 25 ± 1℃. The solution was then poured in an aspirator bottle with stopcock and sat for 10 min to let the dispersed toluene float on the water surface. By the stopcock beneath the aspirator bottle, water–toluene solution was acquired.
Adsorbent preparation
A coated membrane made of nanowires of bixbyite type of manganese oxide was crushed, powdered, and sieved. The residual between sieve no. 10 to sieve no. 30 was taken and used in batch tests.
Tests
At the beginning of each batch test, the nanowires were placed into the autoclave glass bottles and the contaminant solution was then poured into the bottle and was immediately sealed. Adsorption tests were performed in 80 ml autoclave glass bottles, kept under continuous agitation (250 r/min) on a multiposition orbital stirrer. The temperature was maintained at 25 ± 1℃. Each batch test was conducted in duplicate to reduce the measurement errors.
Results and discussions
Synthesis
The morphology and crystallinity of the hydrothermally synthesized cryptomelane-type manganese oxide and bixbyite-type manganese oxide were analyzed via SEM analysis and XRD.
Figure 1 shows the X-ray powder diffraction pattern of the cryptomelane and bixbyite type of manganese oxide membranes. Almost all peaks can be assigned to diffraction from cryptomelane and bixbyite-type crystals (ICDD Cryptomelane-Q Reference code: 00-004-0778 and ICDD Bixbyite Reference code: 01-075-1573).
X-ray powder diffraction pattern of the cryptomelane and bixbyite type of manganese oxide membranes.
A SEM image (Figure 2(a) and (b)) shows a view of the nanowire membrane. A large portion of the nanowires assemble in bundles over a length scale longer than several tens of micrometers, forming a net of open capillaries. From SEM results, the observed morphology of coated and noncoated nanowires was almost similar. However, it seems that coating the nanowires may change the distribution to a further porosity and the woven nanowires were unknitted. This result was observed over many SEM pictures gained from synthesized-coated nanowires.
Characterization of the synthesized nanowire membrane. (a) SEM image of the noncoated nanowires, (b) SEM image of the silicone-coated nanowires, (c) FTIR spectrum of the silicone-coated manganese oxide nanowires, and (d) optical image of the cryptomelane membrane.
FTIR spectrum of the silicone-coated nanowires of manganese oxide is illustrated in Figure 2(c). The spectra interpretation was according to Tian et al. (2007) in which silicone-coated potassium bromide pellet with a similar procedure to the nanowire coating was conducted. The result shows the presence of short chains of PDMS on the nanowires.
Characterizations of coated and noncoated cryptomelane and bixbyite.
Effect of coating time and adsorbent doses
VDM was used to coat the nanowires to convert them from hydrophilic to hydrophobic material. As described, a PDMS stamp and nanowires were placed in a sealed container at 250℃. The optimum time in which the best coating was formed was obtained. Coating was carried out in 30, 60, and 120 min. Then, at different doses of adsorbents, the adsorption was measured. The results are illustrated in Figure 3. Obviously, increasing the adsorbent doses will increase the adsorption of toluene. However, increasing the coating time to 60 and 120 min does not show significant improvement in adsorption. So, the optimum time for coating was captured as 60 min for this type of coating for the rest of the experiments.
Effect of coating time and adsorbent doses (time of stirring: 60 min).
Effect of contact time (Kinetic adsorption test)
In order to establish the equilibrium time of adsorption–desorption for maximum uptake of toluene from toluene–water solution, the amounts of toluene adsorbed on the adsorbents were studied as a function of stirring time, which varied from 10 to 360 min, using initial toluene concentration of 101.5 mg/l.
The relationship between the amount of toluene adsorbed as a function of the time is shown in Figure 4. It is clear that the amount of toluene adsorbed increased with increasing contact time. These data indicated that the reasonable time for adsorption equilibrium was 60 min. Therefore, 60 min was considered as an adequate time for the adsorption of toluene from toluene–water solution under the used operating conditions.
Effect of contact time (kinetic adsorption test) (coating time: 60 min).
Effect of pH
The pH of underground water is affected by several factors. One of the most important factors is the soil composition and bedrock through which the water moves. Also, dumping the chemicals into the groundwater by industries is another factor which affects the pH. The effect of pH on toluene removal was carried out by adjusting pH of the toluene–water solution from 2 to 12 using 1 M HCl or 1 M NaOH prior to the addition of adsorbent. As it is shown in Figure 5, variations of pH from 2 to 7 (acidic solution) may have no effect on the adsorption. On the other hand, variations of pH from 7 to 12 will decrease the adsorption. This may have two explanations. One is that, alkaline environment may harm the coating. Subsequent tests showed no harmed coating. The combination of NaOH, H2O, and toluene will make a polar molecule (IUPAC name: sodium; toluene; hydroxide; hydrate) and the superhydrophobic adsorbent is incapable of adsorbing the polar molecule (US National Library, Pubchem CID: 86738162). Therefore, the adsorption of toluene will decrease in NaOH alkaline environment.
Effect of pH (time of stirring: 60 min, time of coating: 60 min).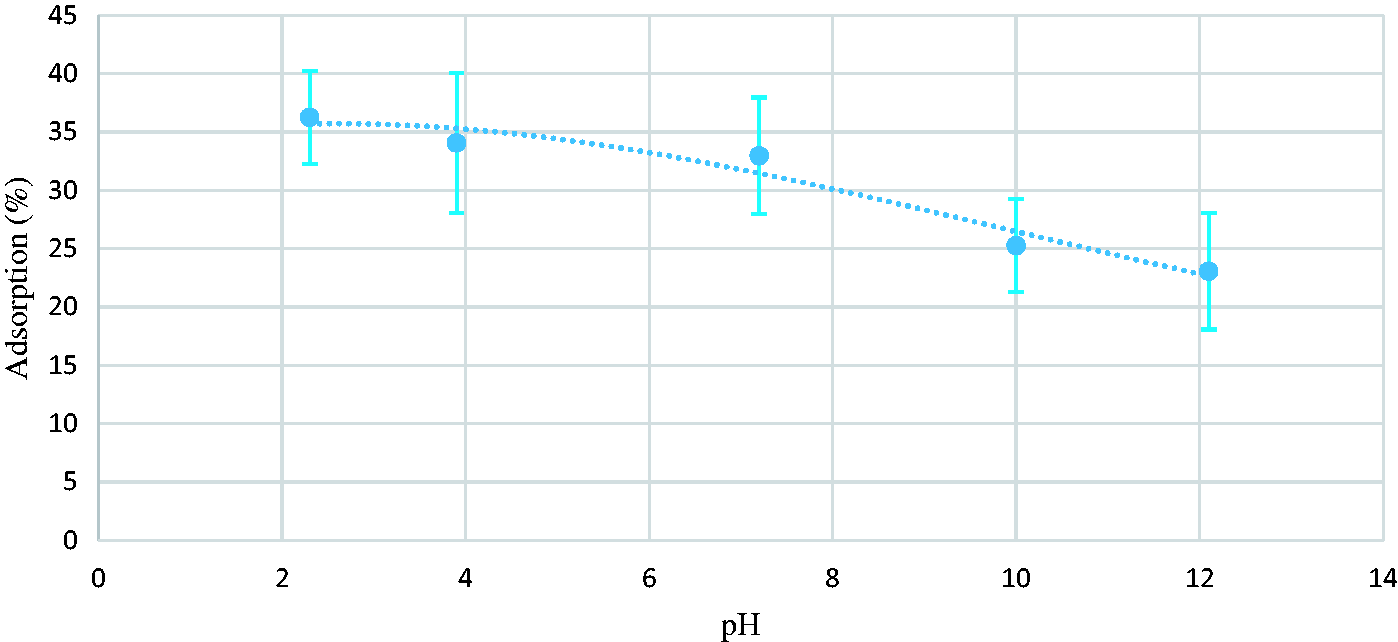
Effect of saline water
Many underground water flows had been contaminated by saline water from the disposal of oil-field brine. The capacity of coated bixbyite type of manganese oxide to adsorb toluene from water–toluene solution at different salinities was investigated. Saline water was first prepared at different salinities (0, 100, 200, 250, 300 mg/l). Then, toluene was added to the solution. The results are shown in Figure 6. The adsorption decreased by increasing salinity. This is due to the reduction of solubility of toluene in saline water (Figure 6) (also see Keeley et al., 1988). Therefore, decreasing the solubility of toluene in water would decrease the amount of dissolved toluene in the container and adsorption rate of toluene will decrease accordingly due to lower initial toluene concentration in liquid phase.
Effect of salinity on adsorption and solubility of toluene in water.
AC adsorption
The most recommended material to adsorb dissolved hydrocarbons is ACs. It has been studied, recommended, and used for many years (Adebajo et al., 2003; EPA, 2000; Okiel et al., 2011; Wu, 2004) but still has some disadvantages. Maintenance and regeneration of ACs are costly and have low stability in harsh conditions. Unlike ACs, nanowires of manganese oxide have good stability (Wang et al., 2012). Here, the adsorption of two kinds of ACs (Merck; Charcoal activated catalog no. 1.02514.1000 and 1.02518.1000 named: AC514 and AC518) is compared with manganese oxide nanowires. As it is shown in Figure 7, adsorption of manganese oxide nanowires is higher than that of ACs at the same adsorbent weight. The weight-base comparison is needed for economical evaluations.
Comparison of adsorption of manganese oxide nanowire and activated carbons.
Effect of regeneration method
One of the most important economic issues when using adsorbent materials is their recyclability and regeneration. Two methods were used to regenerate the nanowires. In the first method, used nanowires were placed in an oven for 1 h at 100℃ and reused. In the second method, nanowires were placed at the room temperature at 25 ± 5℃ for one week and then reused. The results are depicted in Figure 8. From the results, it seems that heating the nanowires may harm the coating and reduce adsorption after each regeneration. Although, all the coating will be totally removed from nanowire surface at 360℃ (Yuan et al., 2008), heating up to 100℃ will impair the coating and the adsorption will be decreased. However, regeneration of the nanowires at the room temperature seems to save the workability of nanowires and the coating will be safe. The second method could be used just for hydrocarbons with high vapor pressure like BTE, which would evaporate at low (room) temperature. Also, a system could be designed and set to collect the evaporated hydrocarbons.
Effect of regeneration method.
Adsorption isotherms
Parameters of the Langmuir and Freundlich isotherm for the adsorption of toluene on to the coated bixbyite-type manganese oxide nanowires.

To obtain the parameters of Langmuir isotherm (KL and b), equilibrium experimental data for the nanowires of manganese oxide were analyzed by plotting Ce/qe against Ce, in which the linearized form of the Langmuir equation is commonly written as

Applying the Langmuir isotherm model (the graph of Ce/qe versus Ce), it was observed that the linear trend line fitted to the adsorption data is not coinciding appropriately with the data and does not exhibit linear behavior and therefore the value of R2 was not satisfactory (R2 = 0.12). For the best fitted line, KL is equal to 1.089, b (l/mg), and the Langmuir constant is equal to 0.0024.
On the other hand, the shape of the Langmuir adsorption isotherm is indicated by the separation factor or equilibrium parameter r. For r = 1, the adsorption is known as linear, 0<r<1, favorable, r>1, unfavorable, and for r = 0 irreversible. The r value calculated by equation (2) is 0.82 which falls between 0 and 1 confirming that the isotherm is favorable. The separation factor (r) is expressed in terms of a dimensionless constant as follows

where C0 is the initial concentration of adsorbate (mg/l).
To obtain the parameters of Langmuir isotherm (Kf and n), the equilibrium experimental data for Freundlich isotherm were analyzed by plotting log qe against log Ce, where the linear form of Freundlich isotherm model is expressed as

where Kf is the Freundlich equilibrium constant which indicates the adsorptive capacity and n is the Freundlich constant indicative of the affinity of the adsorbate for the surface of adsorbent.
Applying the Freundlich model, the linear trend line fitted to the adsorption data had a better agreement than Langmuir isotherm with R2 = 0.86. The Kf value is 1.41 and n value is 1.11. Therefore, based on the correlation coefficients, Freundlich isotherm was best to describe the sorption of toluene from toluene–water solution by the bixbyite type of manganese oxide nanowires. On the other hand, the high values of n (especially values between 1 and 10) indicate favorable adsorption (e.g. Errais et al., 2011; Girardello et al., 2016; Xiong and Mahmood, 2010). The n value found to be equal to 1.11 in our study indicating Freundlich adsorption is favorable.
The Langmuir and Freundlich isotherm relationships fitted to the experimental data are shown in Figure 9. Also, the isotherm parameters are listed in Table 2.
Langmuir and Freundlich adsorption isotherm of toluene on nanowires of manganese oxide.
Conclusions
In this study, we investigated the possibility of using manganese oxide to separate the dissolved toluene from toluene–water solution. Hydrophilicity of two types of manganese oxide was evaluated. The results showed that noncoated (coated) bixbyite type of manganese oxide had higher hydrophilicity (hydrophobicity) than cryptomelane type. Several batch tests were conducted to evaluate the effects of adsorbent dose, contact time, initial solution concentration, pH, salinity, and recyclability on the uptake of toluene by the adsorbent. Bixbyite type of manganese oxide was found to be effective in the removal of toluene from aqueous solutions. Comparison between the results of adsorption of nanowires with two kinds of ACs indicated that the nanowires had higher adsorption at the same weight and nanowires are competitive to ACs. The isotherm analysis showed that Freundlich isotherm best describes the adsorption of toluene from toluene–water solution by nanowires of manganese oxide.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
