Abstract
Porous structure, surface chemistry and catalytic properties of carbon catalysts with various acid groups in ethyl tert-butyl ether synthesis were studied. Carbon catalysts were obtained by phosphoric acid activation followed by sulphonation of lignocellulosic feedstocks of different origin. Porous structure of carbon catalysts was characterized by nitrogen adsorption. Surface chemistry was investigated by potentiometric titration. Carbon catalysts obtained from lignosulphonate and sucrose showed the highest catalytic activity in ethyl tert-butyl ether synthesis (reaction rate at 120℃ is 4.3–5.2 × 10−6 mol·g−1·s−1), is comparable to activity of Amberlyst-15 (5.0 ×10−6 mol·g−1·s−1). Calculated turnover frequency (TOF) of sulphonic groups (pKa = −2.5) equals 0.00116 s−1, polyphosphate groups (pKa = 1.9) 0.01019 s−1, strong carboxylic (pKa = 3.6) 0.00202 s−1 and 0 s−1 for weak carboxylic (pKa = 6) groups. The activity of acid groups with pKa lower than 3.6 is higher than the activity of sulphonic groups of Amberlyst-15 (TOF 0.00115 s−1).
Introduction
The worldwide production and consumption of most popular fuel additive, methyl tert–butyl ether (MTBE), has sharply decreased for environmental reasons, whereas production of oxygenates from bio-resources, particularly, ethyl tert–butyl ether (ETBE), is growing (Yee et al., 2013). ETBE exhibits higher octane rating, higher boiling point, lower flash point, lower blending Reid vapour pressure and reasonably high oxygen contents (Thiel et al., 1997; Westphal et al., 2010; Yang et al., 2000). Moreover, the attraction of the ETBE industrial application is defined by the possibility to use bioethanol, and to obtain gasoline with reduced volatility. Thus, production of ETBE as gasoline fuel oxygenate additive conforms the principles of Green Chemistry (Clark et al., 2012; Sheldon et al., 2007) since it not only increases octane number, but also decreases pollution by reducing exhaust CO and unburned hydrocarbons. Commercially, ETBE is produced by exothermic reversible reaction between iso-butylene (IB) and ethanol (EtOH) over sulphonic ion exchange resins (Badia et al., 2013; Umar et al., 2009; Yee et al., 2013).
However, sulphonic ion exchange resins possess low mass transfer and low thermal stability, which decrease the catalytic activity and cause environmental problems. Furthermore, ion exchange resins tend to swell in polar reaction medium that causes technological difficulties in practical applications.
Sulphonated carbons are considered as promising catalysts in acid–base reactions due to rigid morphology, thermal stability and the lack of swelling in polar reaction medium, They can be inexpensively produced by partial carbonization of sulphopolycyclic aromatic hydrocarbons or sulphonation of partially carbonized organic compounds (Okamura et al., 2006; Toda et al., 2005; Zhao et al., 2010). Carbonaceous catalysts could be prepared from renewable resources as biomass (White, 2015).
Recently, the significance of weak acid surface groups in some catalytic reactions occurring in soft conditions was reported (Bucko et al., 2004; Gianotti et al., 2014; Kim et al., 2003; Vlasenko et al., 2006). The purpose of the present study is revealing the role of surface groups with different acid strength in ETBE synthesis.
Experimental
Carbon catalysts
Carbon catalysts were obtained by phosphoric acid activation (impregnation ratio 0.85, activation temperature 400℃) of lignocellulosic feedstocks of different origin followed by sulphonating with sulphuric acid at 180℃ for 5 h. Carbon catalysts were abbreviated according to the feedstock origin as: AP – apricot stones (Puziy et al., 2005), DW – dogwood stones, CG – coffee grounds, LS – lignosulphonate (Borregaard LignoTech, Sarpsborg, Norway) (Myglovets et al., 2014), SH – sunflower husks.
For comparison purpose, carbon catalyst was obtained by template method using silica gel as hard template and sugar as carbon source (Puziy et al., 2009, 2010a, 2011). Silica gel (for column chromatography, Acros Organics™) was impregnated by sucrose (carbon source) aqueous solution containing phosphoric acid (impregnation ratio 0.25), followed by drying, pre-carbonizing at 230℃ and carbonizing up to 400℃ in flow of argon (0.5 L min−1) with temperature ramp 10° min−1. After carbonization silica template was removed by dissolution in 40% HF. Obtained carbon was sulphonated using the same procedure as for lignocellulosic-derived carbons.
Characterization
Porous structure
Nitrogen adsorption isotherms were measured at −196℃ using Autosorb-6 adsorption analyser (Quantachrome, USA). Pore size distributions were calculated by Autosorb-1 software (Quantachrome, USA) using QSDFT method and slit pore model (Gor et al., 2012; Landers et al., 2013; Neimark et al., 2009). Specific surface area, ABET, was calculated by BET method using nitrogen adsorption data in the relative pressure range chosen by recently proposed procedure (Rouquerol et al., 2007). The total pore volume, Vtot, was calculated by converting the amount of nitrogen adsorbed at a maximum relative pressure to the volume of liquid adsorbate. The micropore, Vmi, and mesopore, Vme, volumes were calculated from the cumulative pore size distribution as the volume of pores with sizes less than 2 nm and between 2 and 50 nm, respectively.
Surface groups
Surface groups of carbon catalysts were investigated by potentiometric titration method (Lützenkirchen et al., 2012) performed in thermostatic vessel at 25℃ using a 672 Titroprocessor combined with 655 Dosimat (Metrohm, Herisau, Switzerland). The proton concentration was monitored using an LL pH glass electrode (Metrohm). The amount of protons adsorbed at each titration point was calculated using the following equation

Catalytic activity
Catalytic activity of carbon catalysts was investigated in flow reactor with fixed bed (Puziy et al., 2010b; Vlasenko et al., 2006) loaded with 1.0 cm3 of carbon catalyst. Molar ethanol/isobutene ratio was 1.5, liquid hour space velocity – 1 h−1, carrier gas helium. Commercially available ethanol (azeotropic mixture of C2H5OH; with 4.43% H2O), isobutene (99%) (Sigma–Aldrich) and helium were used. The pressure was 1.0 MPa, temperature range 80–160℃.
Reaction products were analysed using gas chromatograph Agat (Mashpriborkomplekt, Russia) equipped with Chromaton N-AW column (3 mm id, 2 m length) with 10% Carbowax 600 and a thermal conductivity detector.
Results and discussion
Porous structure
Nitrogen adsorption isotherms for all carbons (Figure 1(a)) belong to a combination of types I and II of IUPAC classification (Thommes et al., 2015). A steep nitrogen uptake at low relative pressures corresponds to the adsorption in micropores while gradual increasing the adsorption at intermediate and high relative pressures is characteristic of formation of multilayer adsorption.
Nitrogen adsorption isotherms (a) and pore size distributions (b) for lignocellulosic-derived carbons AP, DW, CG, LS, SH and templated carbon TC.
Parameters of porous structure of lignocellulosic-derived carbons AP, DW, CG, LS, SH and templated carbon TC.

Main porosity of all carbons lies in the region of pore sizes smaller than 3–4 nm (Figure 1(b)). Templated carbon TC and apricot stone-based carbon AP show bimodal PSD while other carbons exhibit unimodal distribution. The widest micropores are observed for templated carbon TC and lignosulphonate-derived carbon LS, while the narrowest micropores are in apricot stone-based carbon AP (Table 1).
Surface groups
Proton-binding isotherms by all investigated carbons lies in negative region indicating that only proton dissociation occurs in investigated pH range (Figure S1 online). Proton-binding isotherms are relatively flat without steep inflection points that imply highly heterogeneous character of surface groups.
Site concentrations (in mmol g−1) for lignocellulosic-derived carbons AP, DW, CG, LS, SH and templated carbon TC.
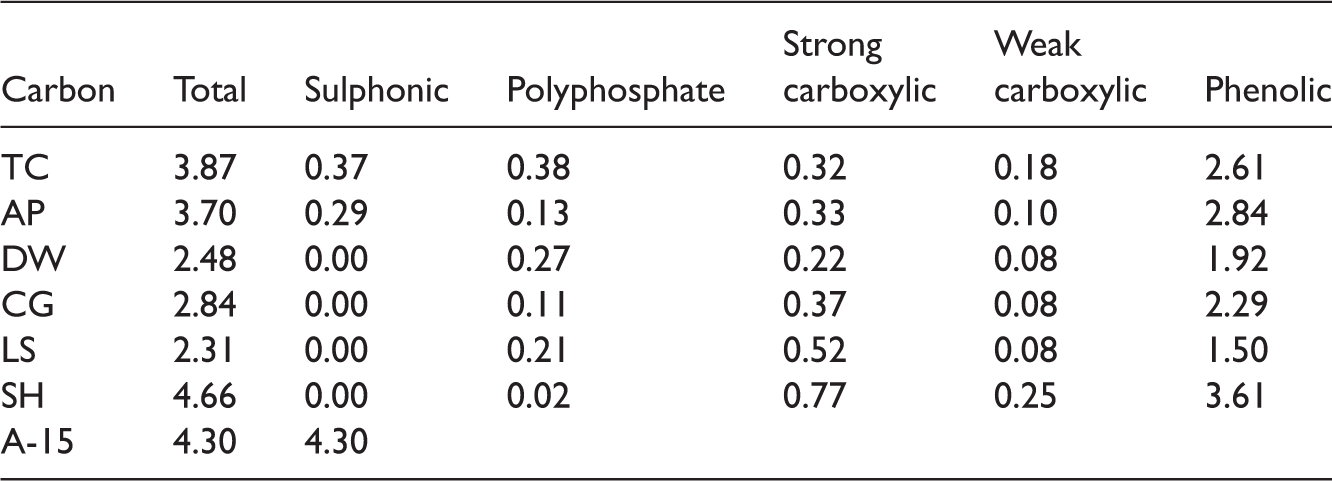
All investigated carbons show high total amount of surface groups (Table 2). Only templated and apricot stone-derived carbons feature sulphonic groups despite the fact that all carbons were sulphonated using the same experimental procedure. This fact implies that sugar-based (templated) and apricot stone-based carbons offer more reactive carbon material than the rest of the carbons.
Catalytic activity
Investigated carbons show high catalytic activity in ETBE synthesis from ethanol and isobutene (Figure 2). With increasing reaction temperature, the activity of the catalyst increases due to enhancing diffusion of reactants and products. At higher temperatures the reaction rate decreases due to thermodynamic limitations since the formation of ETBE is exothermic (Oudshoorn et al., 1999; Vila et al., 1993; Vlasenko et al., 2011).
ETBE formation rate over lignocellulosic-derived carbons AP, DW, CG, LS, SH and templated carbon TC at different temperatures.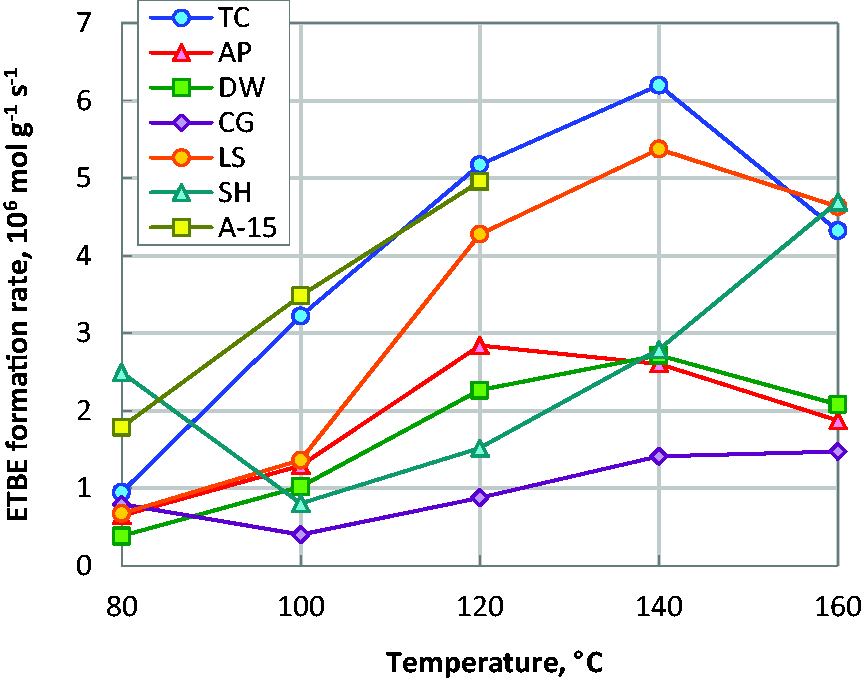
The activity of carbon catalysts is comparable to the activity of ion exchange resin Amberlyst-15 (A-15), which is widely used as catalyst for commercial production of ETBE (Yee et al., 2013). ETBE selectivity for investigated carbons is in the range 60–90% with a tendency to improve with increasing temperature. Side product of the reaction is tert-butyl alcohol with selectivity 10–30% shows a tendency to decrease with increasing temperature.
To reveal the role of different factors in ETBE synthesis, a correlation between reaction rate at 120℃ and porous structure parameters or surface chemistry was considered. There are no satisfactory correlation between reaction rate and porous structure parameters.
To gain a better insight on impact of surface chemistry on catalytic properties, the total activity of carbon catalyst in ETBE synthesis was modelled by the partial activity of each type of surface groups, turnover frequency (TOF), expressed by the equation

Fitting the equation (2) to experimental data allows calculating TOF of surface groups. A good correlation between experimentally determined and predicted ETBE formation rate at 120℃ (r@120) was obtained (Figure 3). The coefficient of determination, R2, is about 0.8. The following TOF values were obtained: for sulphonic groups (pKa = −2.5) 0.00116 s−1, for polyphosphate groups (pKa = 1.9) 0.01019 s−1, for strong carboxylic groups (pKa = 3.6) 0.00202 s−1 and 0 s−1 for weak carboxylic (pKa = 6). This fact implies that only surface groups having pKa lower than 3.6 are active in ETBE synthesis. Weaker acid surface groups are not involved in ETBE synthesis.
Correlation between experimental reaction rate at 120℃ (r@120) and predicted by equation (2) in the ETBE formation over lignocellulosic-derived carbons AP, DW, CG, LS, SH and templated carbon TC.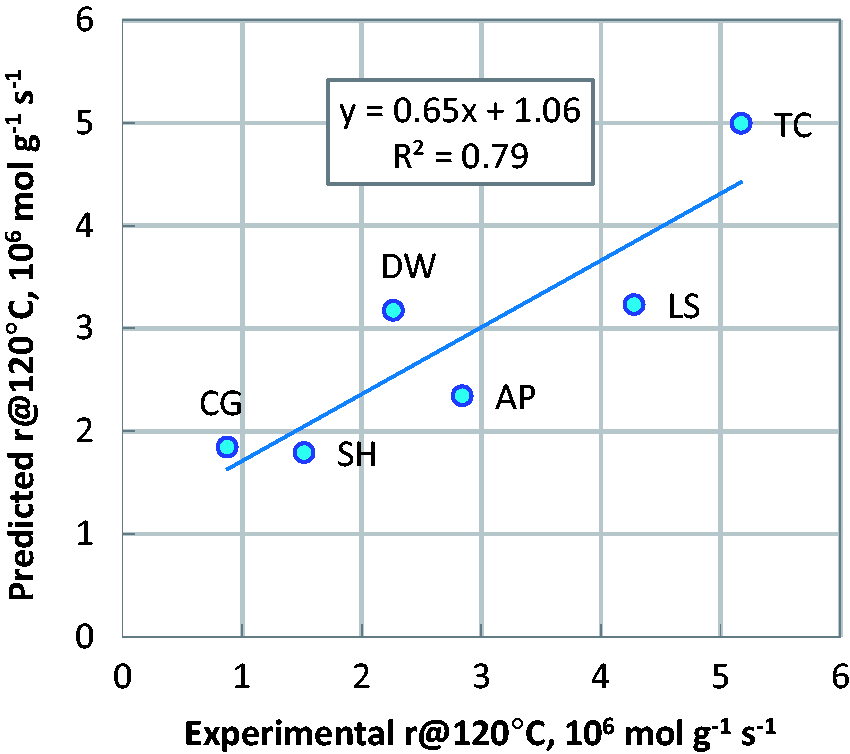
Among the catalytically active groups, polyphosphate groups with moderately strong acidity are the most active in ETBE synthesis. High catalytic activity in ETBE synthesis of polymer-based carbons containing polyphosphate groups was also reported (Puziy et al., 2010b). This fact substantiates our previous conclusion on the crucial role of moderately weak acid groups in ETBE synthesis on zeolite catalysts (Vlasenko et al., 2006).
Conclusions
Carbon catalysts have been obtained by phosphoric acid activation followed by sulphonation of lignocellulosic feedstocks of different origin. Carbon catalysts obtained from LS and sucrose (TC) showed the highest catalytic activity in ETBE synthesis (reaction rate at 120℃ is 4.3–5.2 × 10−6 mol·g−1·s−1), is comparable to activity of Amberlyst-15 (5.0 ×10−6 mol·g−1·s−1). Estimated TOF showed inactivity of surface groups with pKa greater than 3.6. The highest activity showed polyphosphate groups with moderate acidity. Calculated TOF of sulphonic groups (pKa = − 2.5) equals 0.00116 s−1, polyphosphate groups (pKa = 1.9) 0.01019 s−1, strong carboxylic (pKa = 3.6) 0.00202 s−1 and 0 s−1 for weak carboxylic (pKa = 6) groups. The activity of acid groups with pKa lower than 3.6 is higher than the activity of sulphonic groups of Amberlyst-15 (TOF = 0.00115 s−1).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material is available for this article online.
