Abstract
Sorption of naphthalene on modified red clay and halloysite was carried out successfully. It was established that the process was of pseudo-second-order mechanism. The maximum sorption capacities were 9.23 mg/g and 112.79 mg/g for red clay and halloysite, respectively.
Introduction
Polycyclic aromatic hydrocarbons (PAHs), such as naphthalene, anthracene, benzopyrene and phenanthrene are spread widely in the natural environment due to different industrial activities, e.g. coal and petroleum oil burning. Paper mills also release these compounds to the environment. PAHs are hydrophobic, have high melting, boiling points and electrochemical stability; therefore, they can stay for a long time in the environment (Chang et al., 2004) and can be dangerous owing to their toxic and carcinogenic properties. For these reasons, World Health Organization introduced the limit of PAHs in drinking water. There are different methods for the elimination of PAHs from the aqueous phase, such as bioremediation (Meulenberg et al., 1997), ozonation (Rivas et al., 2000), photodegradation, coagulation, flotation and adsorption (Walters and Luthy, 1984). The last method is effective in the removal of persistent organic pollutants. Adsorbents based on activated carbons are widely used, but their main disadvantages are the high cost and difficulty in regeneration (Ania et al., 2007; Gong et al., 2007; Luna et al., 2008; Valderrama et al., 2009). The clays modified with surfactants are a good alternative for activated carbons. Surfactants are known to hinder the migration of PAHs in water. The modification of minerals, especially soil, with surfactant increases their hydrophobicity and therefore enhances their adsorption capacity toward the aromatic hydrocarbons (Jaynes and Vance, 1999; Koh and Dixon, 2001).
The aim of our work is to compare the modified red clay and halloysite in the sorption of naphthalene. We have intentionally selected these minerals because of the fact of their occurrence in Poland in big resources. The mechanism of naphthalene sorption on modified clays (Lee and Kim, 2002; Osagie and Owabor, 2015) is not fully resolved, and hence we apply the quaternary ammonium salts which differ remarkably in their structure, i.e. we used the organic cation with naphthyl radical in its structure and two ammonium salts with benzyl and phenyl rings. We considered the ideas of many researchers, that the structure of surfactants affects the extent of aromatics partition (Li and Bowman, 1998; Klapyta et al., 2001; Sheng et al., 1997).
Materials and methods
Adsorbents
Percentage chemical composition of halloysite from Dunino mine and red clay from Pałęgi mine.

Preparation of organosorbents
The raw clay mineral samples (5 g) were contacted with 100 cm3 sodium chloride solution (1 mol/dm3) (Sigma-Aldrich, 99.5%) to obtain Na-clay. The red clay or halloysite was treated with surfactants for the purpose of sorption enhancement. Two commercial surfactants, namely phenyltrimethylammonium bromide (PTMA–Br Sigma-Aldrich 99% purity) and benzyltrimethylammonium chloride (BTMA-Cl, Sigma-Aldrich 99% purity) were used in the study. The third surfactant (surf_3) N,N-dimethyl(naphthalen-1-yl) methanaminium chloride used to modify the clays was a new non-commercial surfactant having the chemical formula: C10H7CH2NH(CH3)2Cl. The preparation of organoclays, namely PTMA-, BTMA- and surf_3- sorbents was done by the following procedure: 100 cm3 of 10 mmol/dm3 of 0.01 M BTMA-Cl, PTMA-Br or surfactant_3 solution was shaken with 1 g of Na-clays for 4 h at 60℃. Then, the obtained samples were decanted, washed with distilled water and dried in air. The prepared absorbents, namely BTMA-halloysite (BTMA-hal), PTMA-halloysite (PTMA-hal), surfactant_3-halloysite (surf_3-hal) and BTMA-red clay (BTMA-red), PTMA-red clay (PTMA-red), surfactant_3-red clay (surf_3-red) were used for the adsorption of naphthalene.
Adsorption experiments
The adsorption experiments were carried out using the batch method at 22℃. The reaction mixture was composed of 0.025 g of adsorbent and 50 cm3 of naphthalene solution. The initial concentration of naphthalene in the solution varied in the range of 0.05 to 0.2 mmol/dm3 for the study of adsorption isotherms and was 0.2 mmol/dm3 for kinetic studies. In the next step, the mixtures were shaken for 3 h, then filtered using paper filter (Filtrak 390) and centrifuged at 10,000 r/min for 10 min. The initial and equilibrium concentrations of naphthalene were determined spectrophotometrically at 275 nm. The amount of naphthalene adsorbed on sorbents was calculated from the equation

The percentage adsorption of naphthalene from aqueous solution was computed as follows

All the experimental data were the averages of triplicate determinations. The relative errors of the data were about 5%.
Instrumental techniques
The FTIR spectra of the clay samples were recorded in the transmission mode at room temperature using the Nicolet 8700A FTIR spectrometer (Thermo Scientific, USA, 2009), equipped with the TGS detector with the resolution of 2 cm−1. The frequency accuracy measurement of 0.1 cm−1 was maintained. Nitrogen adsorption measurements at 77K were performed using the adsorption analyzer ASAP 2420 (Micromeritics Inc., USA, 2012). Prior to the adsorption measurements, each sample was degassed at 100℃. Room temperature UV-Vis absorption/reflectance spectra were registered using a V-660 spectrophotometer (JASCO). Stationary emission spectra were obtained in room temperature with a Photon Technology International Inc. spectrofluorimeter capable of solid sample measurements, equipped with a continuous wave Xe-arc lamp as a light source. Spectral resolution was maintained at 1 nm. Powder samples were filled into a front face reflectance cell. The excitation wavelength was fixed at 276 nm.
The samples for FTIR, N2 adsorption and fluorescence measurements have been prepared by shaking 50 ml of naphthalene solution and 2.5 g of organosorbent for 3 h. After this time, the solid phase was separated on a filter and dried at room temperature. The initial concentration of naphthalene solutions was 0.01 mol/dm3.
Results and discussion
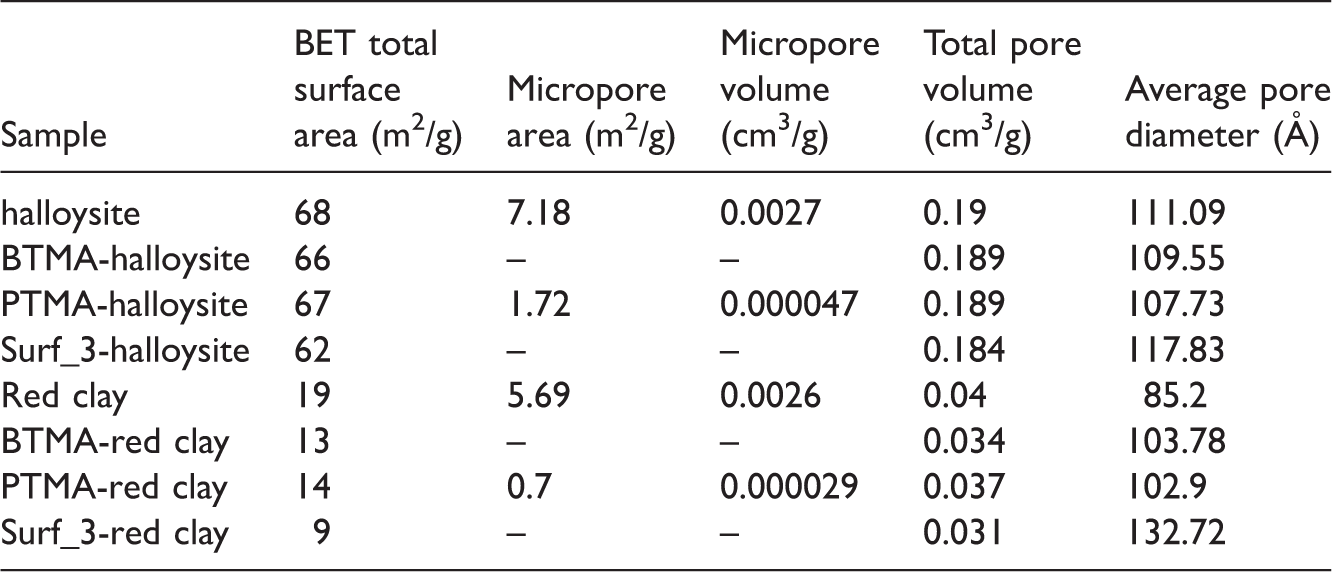
Textural analysis of sorbents
Textural analysis of halloysite and red clay.
Adsorption study
The adsorption isotherms of naphthalene on modified red clay and organohalloysite are shown in Figure 1(a) and (b). The Langmuir-Freundlich (equation (3)) and Dubinin–Radushkevich (equation (4)) isotherms were used to describe the adsorption of naphthalene on the sorbents. The models are given by the following equations
The adsorption isotherms (a, b) and the adsorption kinetics (c) of naphthalene. Parameters of adsorption isotherms and kinetic model for adsorption of naphthalene onto modified halloysite and red clay.




The mean adsorption energy, according to Dubinin-Radushkevich model, was found to be in the range of 5.3–5.87 kJ/mol, which confirms the existence of the physical adsorption reactions. Thus, the adsorption process of naphthalene takes place as a result of the intermolecular van der Waals forces between the molecules of adsorbate and the surface of the adsorbent. A comparison of the regression coefficient (R2) for the two isotherms is shown in Table 3. For the Langmuir-Freundlich isotherm, we have a range of 0.978 to 0.999 and for the Dubinin-Radushkevich isotherm, the range of R2 value is 0.949 to 0.982. We can therefore conclude from the average values of R2 that the Langmiur-Freundlich isotherm gives a very good fit to the adsorption process. The maximum sorption capacities were 9.23 mg/g for red clay and 112.79 mg/g for halloysite. The results obtained for halloysite were really good, compared to literature data. The amount of naphthalene adsorbed in the clay sample was 39.84 mg/g, in the sandy soil was 19.92 mg/g (Osagie and Owabor, 2015) and 58.36 mg/g for activated carbon (Cabal et al., 2009).
The adsorption of naphthalene on modified halloysite and red clay increased rapidly at the initial contact time (Figure 1(c)). After 3 hours from the beginning of the adsorption process, the maximum amount of adsorbate was achieved. The experimental data of naphthalene adsorption on modified forms of halloysite and red clay were analyzed by the well-known in physical chemistry pseudo-second-order model. The pseudo-first-order model was also applied to the calculation but the correlation coefficients R2 values were in the range of 70–75%. The pseudo-second order model proved to be a better one in the evaluation of the experimental data than the pseudo-first order as in the case of adsorption of naphthalene on boehmite (Abu-Elella et al., 2013) and zeolite (Chang et al., 2004).
FT-IR spectra of modified sorbents
The different regions of FT-IR spectra of modified sorbents are given in Figure 2(a) and (b). The band characteristic of Si–O stretching vibration of red clay is shifted from 979 cm−1 to 985, 1032 and 1031 cm−1 for Surf_3-red, BTMA-red and PTMA-red, respectively. This is the consequence of the strong ion-dipole interaction of organic cations with H2O, which is concurrent with the complexation of siloxane oxygen by H2O. The sorption of naphthalene on clay, which proceeds probably via π–π bond formation between benzyl and phenyl rings of organic cations and naphthalene, manifests as return of the bands from the positions: 985, 1032, 1031 to 981, 984, 980 cm−1. The strength of interaction of naphthalene with organic cations follows the order: Surf_3≪BTMA=PTMA. There is probably an obstacle in the formation of π–π bond between naphthalene and spaciously extended Surf_3 cation. For halloysite, the sequence of the bands shift is very similar, i.e. the bands shifts of BTMA-, PTMA-hal differ remarkably in comparison with those one for Surf_3-hal.
FTIR spectra of modified red clay (a) and halloysite (b).
The bands responsible for Si-O-Si bending vibrations are given in Figure 2(a) and (b). One can notice that the band 521 or 520 cm-1 shifts to 534 or 546 cm−1, which is the evidence for the strong interaction of organic cations: BTMA and PTMA with siloxane oxygens of clay structure, cancelled via interaction of naphthalene with organic cations, which manifests in the return of the bands into the lower wavelengths: 521, 520 cm−1 for BTMA and PTMA, respectively. For halloysite, the perturbation of Si–O–Si bending vibrations under the influence of organic cations is very similar to the red clay case with Surf_3 cation, showing the lack of the influence on these deformation vibrations.
The successful intercalation of Surf_3, BTMA and PTMA organic cations into the red clay structure is confirmed by the bands: 1513, 1452–1487, 1461–1487 characteristic for naphthalene and –C–C–C– vibrations in aromatic rings. For halloysite (Figure 2), the disappearance of the bands 1458–1491, 1463–1491 cm−1, originating from –C–C–C– stretching vibrations in BTMA-hal-naph, PTMA-hal-naph, provides evidence regarding the strong π–π interaction of naphthalene molecules with the aromatic rings of organic cations. In turn, the bands 1518, 1516, 1513 cm−1 confirm the presence of adsorbed naphthalene.
Luminescence study
The presence of naphthalene sorbed on the surface of surfactant modified clays was confirmed by the steady-state luminescence measurements, as a quick and non-destructive method. The band in the region of 320–335 nm characteristic for naphthalene was noticed. The maximum luminescence was found for PTMA-hal, which confirms the results from the sorption experiments.
Conclusions
The introductory results of naphthalene sorption on natural clays show evidently better sorption on halloysite compared to red clay, which probably results from the adsorbate interactions with the mineral both on the surface and in the interior. The adsorption energy calculated on the basis of the Dubinin-Radushkevich isotherm model indicates the physical character of the reaction. Kinetic evaluation of the equilibrium data showed that the adsorption of naphthalene on the studied sorbents follows well the pseudo-second-order kinetic model.
