Abstract
For the design of a fixed-bed reactor, intraparticle diffusivity of adsorbents is one of the most important parameters. However, determining this diffusivity is difficult as measuring the effects of fluid film resistance of adsorbents. Shallow-bed (differential reactor) technique is commonly used to determine intraparticle diffusivity. The conventional shallow-bed technique is based on the assumption that fluid film resistance is negligible because of high fluid velocity; hence, the fluid film mass transfer is not calculated. For an activated carbon–phenolic compound system, both intraparticle diffusivity and fluid film mass transfer coefficient were determined using a shallow-bed reactor. However, no one has confirmed the accuracy of the conventional assumption for a synthetic resin adsorbent–phenolic compound system. In general, synthetic resin adsorbents have a larger fluid film resistance than activated carbon. Therefore, this study focused on the effect of fluid film resistance based on the conventional assumption. The conventional analysis method (intraparticle diffusion controlling model) and the new analysis method (both intraparticle diffusion and fluid film mass transfer controlling model) were compared, and the results indicated that the conventional assumption, which neglects the elimination of fluid film resistance, had no effect on intraparticle diffusivity. Therefore, the conventional analysis method is useful for determining intraparticle diffusivity for a resin adsorbent–phenolic compound system.
Keywords
Introduction
Resin adsorbents are widely used in a number of applications, such as the extraction of active components from plants (Huang et al., 2008; Kühn et al., 2014; Monsanto et al., 2015; Sevillano et al., 2014; Weisz et al., 2010), removal of pesticides (Naushad et al., 2014; Otero et al., 2014; Schummer et al., 2014; Zhang et al., 2013), purification of antibiotics (Chao et al., 2014; Jia et al., 2014; Zou et al., 2015), and water treatment (Bilgili, 2006; Li et al., 2002; Lin and Juang, 2009). To design a fixed-bed reactor, intraparticle diffusivity of adsorbents is one of the most important parameters. For activated carbon systems, shallow-bed reactor (Fujiki et al., 2011; Fujiki et al., 2014; Satoh et al., 2008; Sonetaka et al., 2009a, 2009b) and completely mixed batch reactor (CMBR) (Fujiki et al., 2010; Furusawa and Smith, 1973; Suzuki and Kawazoe, 1975) techniques are commonly used to determine intraparticle diffusivity. The shallow-bed (differential reactor) technique is generally based on the assumption that fluid film resistance is negligible with an extremely high fluid velocity; hence, the fluid film mass transfer coefficient cannot be determined. As a result, Sonetaka et al. (2009a, 2009b) proposed a new analysis method to determine both intraparticle diffusivity and fluid film mass transfer coefficient of activated carbon using a shallow-bed reactor (Fujiki et al., 2011; Sonetaka et al., 2009a, 2009b). Their analytical method, using the Biot number (Bi), can be applied to the CMBR technique for activated carbon systems (Fujiki et al., 2010).
In contrast, resin adsorbents have a larger fluid film resistance; hence, the CMBR technique cannot be applied for such resins as the rotational speed of CMBR cannot sufficiently reduce the fluid film resistance. Therefore, resin adsorbent systems require the determination of intraparticle diffusivity using the shallow-bed technique. However, the assumption that a high fluid velocity eliminates the larger fluid film resistance of resin adsorbents has not been experimentally certified.
This study focuses on the effect of fluid film resistance of resin adsorbents using a shallow-bed reactor and validates the accuracy of the conventional assumption described above. To determine intraparticle diffusivity, two different simulation models, i.e. the intraparticle diffusion controlled model and both intraparticle diffusion and fluid film mass transfer controlled model, are employed.
Materials and methods
Materials
Characteristics of XAD-2000.

Actual value.
Provided by Organo Corporation, Japan.
Experimental
The shallow-bed technique was applied to determine intraparticle diffusivity, and the procedure is as follows. The resin adsorbent was packed in a glass column (8 mm diameter and 1 cm height) and known concentrations of solutions (PCP including 20 wt% alcohol aqueous solutions, 100–3000 mg/L) at known temperatures (288.2, 298.2, 308.2, and 318.2 K) were fed upward through the column with a flow rate of 0.14 m/s. After a predetermined time interval, the solution in the column was removed with a small volume of distilled water and extracted with 0.1 M NaOH aqueous solutions to determine the amount adsorbed. The resin adsorbent seemed to absorb the molecules present in the resin microparticles; therefore, the amount adsorbed between microparticles was determined by measuring the amount desorbed. Details of the adsorption equilibrium and experimental procedure have been reported in previous studies (Aguwa et al., 1984; Furuya et al., 1989).
Fundamental equations
The shallow-bed reactor has a small bed length and high fluid velocity. Therefore, for a shallow-bed reactor, the effluent at the interface between the fluid and solid is considered as the same, and fluid film resistance can be considered negligible. A similar intraparticle-diffusivity determination procedure for activated carbon–phenol systems by combining an experimental adsorption uptake curve (EAUC) with a theoretical uptake curve (TUC) has been reported (Fujiki et al., 2011; Sonetaka et al., 2009a, 2009b). Generally, intraparticle diffusion of resin adsorbents is measured using the pore diffusion model (Fujiki et al., 2014; Furuya et al., 1989). Therefore, in this study, fundamental equations are established using the following assumptions: (a) constant temperature, (b) constant influent flow rate, (c) equal influent and effluent concentrations; hence, single-particle adsorption concept is applied, (d) Freundlich-type isotherm, (e) local equilibrium established on the pore surface of adsorbent, (f) pore diffusion controlling, (g) constant effective intraparticle diffusivity and fluid film mass transfer coefficient, and (h) two-step mass transfer concept applied (liquid film mass transfer and intraparticle diffusion). Dimensional fundamental equations are listed below.
Dimensional Fundamental Equations
Intraparticle diffusion

Fluid-to-solid film transport

Interface transport

Average amount adsorbed

Equilibrium relationship

These dimensional equations were solved for the following dimensionless variables, as shown below, to reduce rigorous numerical calculations. As a result, dimensional fundamental equations were transformed into dimensionless fundamental equations. Please note that the dimensionless fundamental equations include a kinetic parameter Bi.

Results and discussion
Determination procedure of DP and kF
The procedure for determining DP and kF is similar to that reported previously (Fujiki et al., 2011; Sonetaka et al., 2009a, 2009b). The procedure is described with reference to the following example of Run 4.
Determination of Bi
As mentioned above, the dimensionless kinetic parameter, Bi, affects the shape of uptake curves; therefore, its value needs to be determined. Each Bi is calculated by use of Freundlich constant n. Time ratios at Qt = 0.7 and Qt = 0.3 were calculated using dimensionless equation A. Figure 1 shows the relation between Bi and time ratio T0.7/T0.3. The values of the dimensional and dimensionless time ratios, t0.7/t0.3 and T0.7/T0.3, respectively, are equal (equation 18). Figure 1 shows the relation between Bi and t0.7/t0.3 = T0.7/T0.3.
Plot of Bi vs t0.7/t0.3 (Run 4).

Determination of t0.7/t 0.3 = T0.7/T 0.3 from EAUC
The value of t0.7/t0.3 = T0.7/T0.3 can be estimated from EAUC (Figure 2). In the following example (Run 4), t0.7 = 11.40 and t0.3 = 1.69; hence, t0.7/t0.3 = 6.77. The value of Bi = 84 at t0.7/t0.3 = 6.77 was determined as shown in Figure 1.
Determination of t0.7/t0.3 from EAUC (Run 4).
Determination of Dp
Dimensionless fundamental equation A was employed to determine DP. EAUC (Qt vs t) was overlaid with the AUC (Qt vs T) of the calculated Bi, and the ratio of dimensionless and dimensional time, T/t (min−1), was determined (Figure 3). In the following example, T/t = 0.005 (min−1) when EAUC and AUC is overlaid. After obtaining a successful fit of the curves, DP can be determined from equation (19), obtained by rearranging equation (6).
Curve fitting to determine T/t (Run 4).

In Figure 3, DP is calculated in the following manner:

Determination of kF
Dimensionless fundamental equation B was employed to determine kF. EAUC (Qt vs t) can be overlapped with TUC (Qt vs T′) (Figure 4). After obtaining a successful curve-fit, T′/t can be determined and kF can be calculated from equation (20), obtained by rearranging equation (12).
Curve fitting for determining T′/t (Run 4).

In Figure 4, kF is calculated in the following manner:
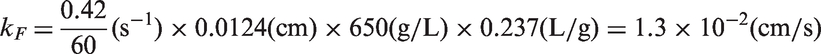
Finally, Bi is calculated to confirm the accuracy of the calculated values of DP and kF, obtained using equation (6).
Comparison of conventional and new analysis methods
Experimental results.

DP for conventional analysis method is provided in Furuya et al. (1989).

Comparison of conventional analytical DP and new analytical DP .
Conclusion
In this study, Dp of XAD-2000 was estimated by two types of analytical methods. The first method is the conventional analytical method, which assumes that adsorption is controlled by intraparticle diffusion only and eliminates the effect of fluid film mass transfer, whereas the second method is the new analytical method, which considers both intraparticle diffusion and fluid film mass transfer. Both methods show similar DP values, thus indicating that the conventional assumption of negligible fluid film mass transfer in a shallow-bed reactor is correct. Therefore, the shallow-bed technique can sufficiently reduce the fluid film resistance and can be used for determining the intraparticle diffusivity of resin adsorbents with low calculation cost.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
