Abstract
A novel polypropylene hollow fiber membrane with a new function of selective adsorption of mercury ions in aqueous solutions was successfully prepared. The surface of the polypropylene hollow fiber membrane was initially modified with polydopamine by surface polymerization, and subsequently grafted with polyacrylamide (PAM) polymer brush via the surface initiated atom transfer radical polymerization (SI-ATRP) technique (thereafter named as PP-PAM). This study investigated the adsorption performance of Hg(II) ions by PP-PAM and the effect of various influencing factors on Hg(II) ion adsorption. The experiment indicated that the Hg(II) adsorption capacity of the PP-PAM increased with the increase of the pH, and the Hg(II) adsorption kinetics was consistent with the pseudo-second-order kinetic model. The adsorption isotherm followed the Langmuir model, with the maximum adsorption capacity calculated to be 0.854 mmol/g for Hg(II) ions. The adsorption study in multi-component system indicated that PP-PAM preferentially adsorbs Hg(II) over Pb(II) ions, with significant adsorption capacity difference of the two heavy metal ions. This study provided an efficient method for the preparation of the adsorptive polypropylene hollow fiber membrane, which expands its application for the selective removal of heavy metal ions.
Introduction
The rapid development of industrialization brought unprecedented tension to the natural environment. Especially, heavy metal pollution of the water body has become an increasingly serious problem, which affects the ecological environment as well as the human health (Alloway and Jackson, 1991; Al-Yousuf et al., 2000; Jarup, 2003; Jeng and Swanson, 2006). Mercury (Hg), as a heavy metal extensively used in industrial production, is one of the most hazardous elements to human beings. Therefore, it has been listed as a priority pollutant by the Environmental Defense Fund of the United States (Cameron, 1992). The excessive intake of Hg mainly harms the central nervous system, digestive system and kidney of the human body. It also has great negative influence on respiratory system, skin, blood and eyes (Clarksonand and Magos, 2006; Zahir et al., 2005).
Currently, conventional methods for treating heavy metal wastewater mainly include chemical precipitation, electrochemical process, ion exchange technique, etc. (Fu and Wang, 2011; Kongsricharoern and Polprasert, 1996; Lai and Lin, 2003). These methods are generally applied with problems of high energy consumption, secondary pollution and low treatment efficiency. Under most circumstances, the heavy metal wastewater in industrial production is presented with multiple heavy metal ions instead of a single component. The conventional methods often lack the selectivity for heavy metal ions, which makes it difficult to recycle the specific or the target heavy metal ion. For instance, industrial Hg wastewater often contains other recyclable heavy metal ions, such as copper and lead, but the existence of Hg ions makes it very difficult to effectively recycle these heavy metal ions. The selective separation and efficient elimination of the heavy metal ions with economic means remain a challenge.
With the development of economy and the enhancement of the environmental protection awareness of the people, research attention has been focused on new wastewater treatment methods that are inexpensive, highly efficient and free from secondary pollution. Among them, adsorption with new adsorptive materials has been regarded as one of the most efficient methods, as the adsorptive materials are given more features by means of chemical modification such as coating and grafting, while keeping the original physical properties of the material. Polypropylene hollow fiber membrane (PPHFM) is a membrane material extensively applied in the field of water treatment, which features good impact, wear and corrosion resistance, as well as a large specific area and a high separation efficiency. Traditional PPHFM has been widely used in treating wastewater containing pollutants such as micro-particles and colloids, due to its porous structure which acts as a filtration sieve to keep off those pollutants. However, in a more complexed wastewater system containing particles, colloids as well as heavy metal ions, the function of physical filtration only may limit its application in removing heavy metal ions. It is desirable that the adsorptive functional groups be immobilized on PPHFM’s surface, so that the adsorption of heavy metal ions would take place during water filtration. However, as PPHFM almost possesses no active groups on the surface, the immobilization of the adsorptive functional groups seems difficult. Therefore, it would be favorable that the heavy metal ion adsorption property be given to PPHFM, while the original property remains, so as to expand the application of the PPHFM in the field of wastewater treatment.
It was discovered in recent years that dopamine has oxypolymerization – cross-linking reaction in water solutions and is able to form a composite layer of extreme adhesion on the surface of almost all the solid materials (Lee et al., 2006, 2007; Waite, 2008). Rich in catechol group, polydopamine provides the active sites for the introduction of various functional polymer brushes into the polydopamine layer and consequently realizes further functionality of the material surface. The introduction of the polymer brushes (Milner, 1991) is one of the focuses of the surface modification technologies in recent years, which can effectively change the chemical and physical properties of the material surface. Through various kinds of surface-initiated polymerization, the variety of polymer brush structures can be introduced, and thus, its usage in practice can be expanded.
In this study, the PPHFM was consecutively surface-polymerized with polydopamine and grafted with polyacrylamide (PAM) polymer brush via the controllable SI-ATRP technique for the selective adsorption of mercury (Hg(II)) ions. While retaining the good properties of the original material, the PPHFM has gained new functionality through the polymer brush grafting as the adsorptive hollow fibers. To the best of our knowledge, there is no publication so far regarding the PAM-grafted PPHFM and its investigation on the selective adsorption of Hg(II) ions. The modified PPHFM prepared in this study possesses multi-functions of both physical filtration of the micro-particles and the selective adsorption of heavy metal ions. The modified PPHFM was characterized and the selective adsorption of Hg(II) was explored in Hg(II)/Pb(II) mixed solutions. This study would provide insight into the surface modification of the widely used PPHFM and expand its application to remove a variety of pollutants in waters and wastewaters.
Experiment
Experiment material
PPHFM was provided by Yuanxiang Technology Co., Ltd. 2-bromide isobutyl bromide (BIBB, AR, 98%), pyridine (AR, 99%), 2,2-bipyridine (AR, 99%), cuprous bromide (CuBr, AR, 99%), copper bromide (CuBr2, AR, 99%), dopamine hydrochloride (AR, 98%), 3-(hydroxymethyl) aminomethane (Tris HCl, AR, 99.8%), acrylamide (AR, 99.9%), N,N-dimethyl formamide (DMF, AR, 99.5%), disodium ethylene diamine tetraacetate (EDTA, AR) and lead nitrate (AR) were all purchased from Shanghai Aladdin Co. Ltd. (2-(dimethylamino) ethyl) amine (Me6TREN, AR, 99%) was purchased from Beijing J&K Scientific Ltd. HgCl2 was purchased from Global Reagent Co., Ltd. All other reagents were of analytical grade.
Polydopamine coating on PPHFM’s surface
The pristine membrane PPHFM (0.1 g) was placed in a plasma cleaner (Harrick, PDC-002, USA) and surface modified with plasma in air atmosphere for 5 min. Then, the PPHFM was immersed into the dopamine solution (2 g/L in 200 mM Tris buffer solution at pH 8.5), and the polymerization of dopamine was performed on the surface of PPHFM at 60℃ for 2 h. After that, the fiber membrane was washed with deionized water, dried in an oven and named as PP-PDA (PDA for polydopamine).
Bromination
First, 0.1 g PP-PDA and 15 mL dichloromethane (DCM) were added into a 100 mL test tube, which was then placed into an ice–water bath. Afterwards, 3 mL BIBB was slowly pipetted into the tube, followed by the addition of 1 mL pyridine. The reaction mixture in the test tube was stirred at the speed of 300 r/min on a magnetic stirrer for 24 h. After the reaction, the fiber membrane was taken out and washed with acetone for 20 min, with deionized water for 10 min, and dried in a vacuum oven. The brominated PP-PDA was named as PP-PDA-Br.
PAM grafting
For the graft polymerization of acrylamide on the surface of PP-PDA-Br, the surface initiated atom transfer radical polymerization (SI-ATRP) was carried out in a CuBr/CuBr2/Me6TREN catalytic system at 30℃ (Liu and Bai, 2014). The schematic layout of the SI-ATRP mechanism was shown in Figure S1 in the Supplementary material. 0.15 g copper bromide (CuBr), 0.024 g cuprous bromide (CuBr2), 7.56 g acrylamide (AM) and 0.1 g PP-PDA-Br was put into a 100-mL test tube. Then, 2 mL DMF was added into the test tube, and the reaction mixture in the test tube was bubbled with argon for 10 min to remove the oxygen. After the monomer was completely dissolved in the solution, 0.24 mL Me6TREN was injected into the test tube. The reaction mixture in the test tube was stirred at the speed of 150 r/min on the magnetic stirrer for 48 h at 30℃. After the reaction, the fiber membrane was taken out and sequentially washed with DMF, 0.1 M EDTA and ethanol. The thus-prepared PPHFM was named as PP-PAM. The schematic diagram of PPHFM surface functionality was shown in Figure 1.
Schematic diagram of PPHFM surface functionality.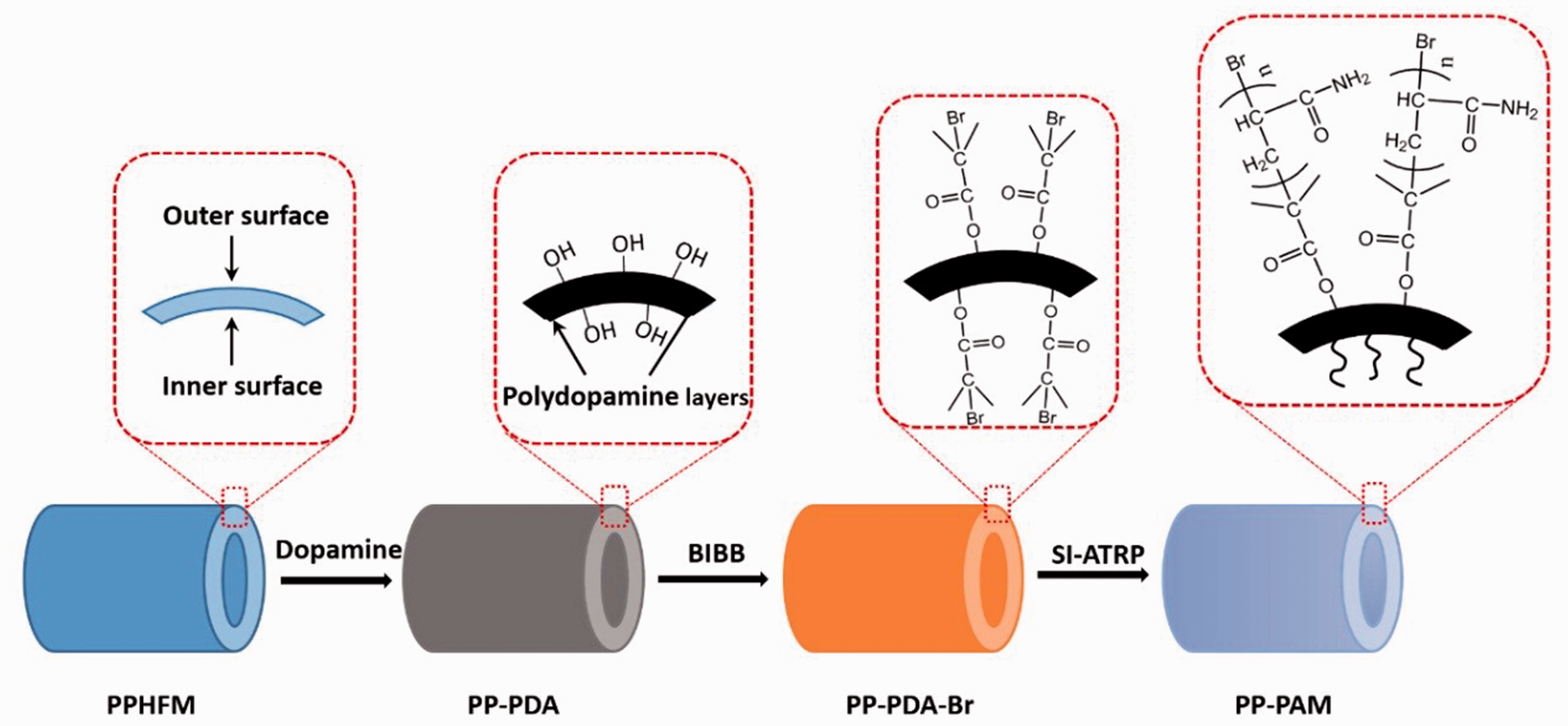
Material characterization
The surface chemical functional groups of the PPHFM-related materials were analyzed with Vertex 70 ATR FT-IR spectrometer (Bruker Corporation, Germany). The surface morphology of the materials was observed with JSM-7800F field emission scanning electron microscope (FESEM, JEOL, Japan). The surface elements of the material were identified and the corresponding atom percentage was tested with the ESCALAB 250Xi X-ray photoelectron spectrometer (XPS, Thermo, USA). The surface water contact angle of the material was tested with the SD-S1 water contact angle tester (Shengding Precision Instrument Co., Ltd., China).
Adsorption performance test
The pH effect, the adsorption kinetics and the adsorption isotherms were investigated in both the single species system (only Hg(II) ion was present) and the binary species system (both Hg(II) and Pb(II) were present). The schematic layout of the adsorption test was shown in Figure S2 in the Supplementary material. The concentrations of Hg(II) and Pb(II) before and after the adsorption were determined with inductively coupled plasma optical emission spectrometry (ICP-OES, Optima 2100DV, PerkinElmer). The adsorption capacity of Hg(II) and Pb(II) was calculated from equation (1).

In order to study the pH effect in single species system, each of 30 mg PP-PAM samples was placed in 30 mL Hg(II) solutions (pH value being 1–5, respectively; Hg(II) concentration: 4 mM). The adsorption was carried out for 24 h at 25℃. For the adsorption in binary species system, the adsorption conditions was similar with those in single species system, except that the Hg(II)/Pb(II) binary species solutions were used, each heavy metal ion being 4 mM in the solution. The pH range of 1–5 was chosen according to the MINEQL+ software (Schecher and McAvoy, 2003) as no metal ion precipitate would be formed for both Hg(II) and Pb(II) ions.
For the investigation of the adsorption kinetics, 0.3 g PP-PAM was placed in 300 mL heavy metal ion solution (4 mM, pH = 5) for the adsorption and the solution was stirred at the speed of 150 r/min and 25℃. The samples were taken at different time intervals for the analyses with ICP-OES. The adsorption kinetic experiments were carried out in both single and binary species systems.
For the investigation of the adsorption isotherm, each of the 30 mg PP-PAM samples was placed into a series of 30 mL heavy metal ion solutions with the concentrations ranging from 0.2 to 4.0 mM at pH 5. The adsorption was allowed for 24 h at the rotation speed of 150 r/min at 25℃. After the adsorption equilibrium was achieved, the concentrations of the samples were determined by ICP-OES. The adsorption isotherm experiments were carried out in both single and binary species systems.
Results and discussion
Characterizations of the PPHFM-related materials
ATR-FTIR spectra were used for the characterization of the chemical components of PPHFM before and after surface modification. As shown in Figure 2, a new characteristic peak appeared at 1540 cm−1 for PP-PDA, which was an overlapping peak of C=C stretching vibration and N–H bending vibration on the aromatic ring of the polydopamine structure. After the PAM polymer brush was grafted, N–H asymmetric and symmetric stretching vibration peaks appeared at 3361 cm−1 and 3200−1 cm, respectively. The characteristic peaks at 1620 cm−1 (C=O stretching vibration peak) (Nalwa, 1997) and 1400 cm−1 (C–N stretching vibration) were attributed to the amide groups, indicating that PAM was successfully grafted on the surface of PPHFM.
Infrared Spectroscopy of PPHFM, PP-PDA and PP-PAM.
Water contact angle was used to evaluate the surface hydrophilic performance of the materials. As shown in Figure 3, the water contact angle of the original PPHFM was about 88° and can be considered as hydrophobic. After the layer of polydopamine was coated on the surface, the hydrophilic property was increased as the water contact angle was changed to 51°. The water contact angle of PP-PDA-Br was 72°, which was due to the ester generated from the reaction of BIBB with the polydopamine composite layer (Huang et al., 2013). After the PAM polymer brush was grafted, the water contact angle of the PP-PAM was reduced to 0°, showing a fully hydrophilic surface.
Water contact angles of PPHFM, PP-PDA, PP-PDA-Br and PP-PAM.
The FESEM images of PPHFM before and after surface modification were shown in Figure 4, the original PPHFM was relatively porous, regular and with even aperture. The aperture decreased on the porous membrane surface after the membrane was coated with polydopamine. There was obvious protrusion and wrinkles on membrane surface after the membrane surface was grafted with PAM.
FESEM images of the PPHFM, PP-PDA and PP-PAM.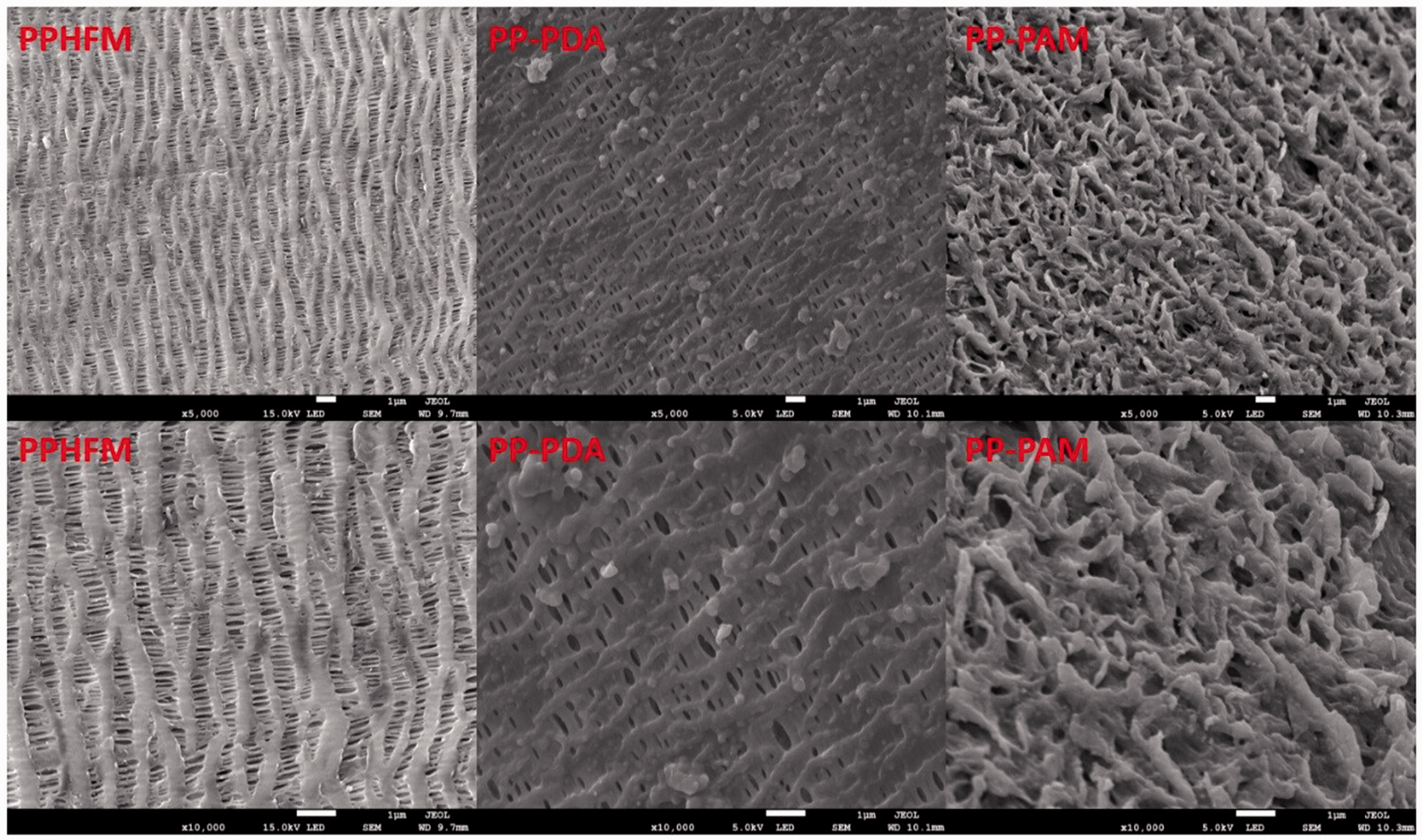
Figure 5 showed the XPS wide-scan spectra of the material in different preparation stages and the PP-PAM adsorbed with Hg(II). The original PPHFM consisted of elements carbon (C) and hydrogen (H). After polydopamine coating and bromination reaction, the characteristic peaks of N 1s, Br 3d and Br 3p (located at the binding energy 399, 69 and 182 eV, respectively) can be obviously seen from the PP-PDA-Br wide-scan spectrum, indicating the successful introduction of nitrogen and bromine elements on the membrane surface. The bromination step was important in order to produce the macro-initiator for the subsequent grafting of the PAM polymer brush via the SI-ATRP technique (Liu et al., 2017; Zheng et al., 2011). After the PAM polymer brush was grafted on membrane surface, the characteristic peak of N 1s obviously increased compared with that of PP-PDA-Br, and the [N]/[C] atom content increased to 11.8% compared with 6.18% of the PP-PDA-Br, proving that PAM polymer brush was successfully grafted onto the PPHFM surface. After the adsorption of Hg(II) ions with PP-PAM, Hg 4f characteristic peak appeared at the binding energy of 101 eV, indicating that PP-PAM can be used for Hg(II) ion adsorption.
XPS wide-scan spectra of PPHFM, PP-PDA, PP-PAM and PP-PAM adsorbed with Hg(II).
Effect of pH
Figure 6(a) showed the pH effect on Hg(II) ion adsorption with PP-PAM. The adsorption capacity of Hg(II) ion with PP-PAM increased as the pH value increased, and the adsorption was obviously inhibited when the pH value was relatively low. This is because the amide group on PAM is likely to be protonated at low pH values. As the Hg(II) adsorption occurred due to the reaction of the amide groups of the PAM with Hg(II) to form the covalent bond (Sonmez et al., 2002), the protonated amide could hardly provide reaction sites for the covalent bonding with Hg(II) ions, resulting in the low adsorption capacity. As the solution pH increased, the protonation of the amide groups was reduced, providing more sites for the covalent bonding with Hg(II) ions. When the pH value reached 5, the adsorption capacity with PP-PAM reached the maximum.
(a) Adsorption of Hg(II) ions with PP-PAM with different pH values; (b) adsorption of Hg(II) ions in binary species system (Hg(II) and Pb(II) ions) with PP-PAM with different pH values.
Figure 6(b) showed the selective adsorption behavior of Hg(II) ions in binary species system at different pH values. The Hg(II) ion adsorption performance with PP-PAM gradually increased as pH increased and reached the maximum value when the pH was 5. On the other hand, the Pb(II) ion adsorption performance was less influenced by the pH value and the adsorption capacity kept at a lower amount. The PAM grafting on the modified PP membrane played an important role on the selective adsorption of Hg(II). This is because the PAM grafted contains the amide group, which can react with HgCl2 to form the covalent bond in the form of amide-Hg (Li et al., 2005), while other heavy metal ions (e.g., Pb ions) cannot form the covalent bond with amide groups. Although the –NH2 group owns the ability to form coordination with other heavy metals, the carbonyl (C=O) group in –CONH2 attracts the electron cloud of the nitrogen atoms of –NH2 and reduces the possibility of forming coordination with Pb(II) ions by contributing the lone pair electrons to form the hybrid orbitals with Pb(II) ions (Nugent et al., 2015). Therefore, the coordination ability between PP-PAM and Pb(II) ions was greatly eliminated. The selectivity of Hg(II) ions over Pb(II) ions was mainly due to the different adsorption mechanisms of these two heavy metal ions with PP-PAM.
Adsorption kinetics
Figure 7(a) showed the adsorption kinetics of Hg(II) ions with PP-PAM. At the initial stage, the adsorption rate for Hg(II) ions with PP-PAM was higher, and it became slower after 60 min because the available adsorption sites for Hg(II) ions gradually decreased as more Hg(II) ions were adsorbed on PP-PAM surface. When the adsorption time reached 300 min, the adsorption reached equilibrium, with the equilibrium adsorption capacity of 0.584 mmol g−1. The Hg(II) ion adsorption kinetics of the PP-PAM was fitted with the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetics equations (Ho and McKay, 1999; Zheng et al., 2010), with the relevant equations and parameters shown in Table 1. Based on the calculation result in Table 1, the adsorption behavior of Hg(II) was suitable for the fitting with PSO kinetics equation, with the (a) Adsorption kinetics curve of Hg (II) with PP-PAM; (b) adsorption kinetics curve of Hg (II) – Pb (II) mixed solution with PP-PAM. Adsorption kinetics model fitting parameters of Hg (II) adsorption with PP-PAM.

The selective adsorption kinetics for Hg(II) ions in binary species system with PP-PAM was shown in Figure 7(b). At the initial adsorption stage, both heavy metal ions were adsorbed due to the large amount of the available sites. As the adsorption time continues, the amide groups were gradually occupied by Hg(II) ions forming the covalent bonds, and the adsorption of Pb(II) ions were greatly inhibited, with Pb(II) ions initially adsorbed even replaced by Hg(II) ions.
Adsorption isotherm
The Hg(II) adsorption isotherm with PP-PAM was shown in Figure 8(a). As Hg(II) equilibrium concentration increased, the Hg(II) adsorption capacity with PP-PAM showed a steady increase. When the Hg(II) equilibrium concentration was higher than 3 mmol L−1, the adsorption capacity of Hg(II) reached an equilibrium. The Hg(II) adsorption isotherms were fitted with Langmuir and Freundlich adsorption models, and relevant equations and parameters were shown in Table 2. The Langmuir isotherm model fitted the isotherm better than Freundlich isotherm model, indicating that the adsorption process was in the form of single layer adsorption and the maximum adsorption quantity was about 0.854 mmol/g as determined from the Langmuir model parameters, which was comparable to chitosan-based adsorbent (0.324 mmol/g) (Kyzas and Kostoglou, 2015), and EDTA functionalized magnetic graphene oxide (1.012 mmol/g) (Cui et al., 2015). Figure 8(b) showed the selective adsorption isotherm for Hg(II) ions in the binary species system. As the Hg(II) and Pb(II) equilibrium concentrations increased, the adsorption of Hg(II) ions showed a steady increase, but the adsorption of Pb(II) ions was greatly inhibited. The equilibrium adsorption capacity for Hg(II) and Pb(II) ions were 0.622 mmol/g and 0.089 mmol/g, respectively, with significant adsorption capacity difference between the two heavy metal ions (Luo et al., 2014a, 2014b).
(a) Hg(II) adsorption isotherms with PP-PAM fitted with Langmuir and Freundlich models; (b) adsorption isotherms of Hg(II) – Pb(II) mixed solution with PP-PAM. Adsorption isotherm model fitting parameters of Hg (II) adsorption with PP-PAM. For Langmuir model, 

Conclusions
In this study, an adsorptive PPHFM was successfully prepared via the surface polymerization of dopamine, followed by the SI-ATRP of PAM polymer brush. The prepared PP-PAM showed efficient and selective adsorption of Hg(II) ions in aqueous solutions. The experimental results indicated that the adsorption of Hg(II) ions was best obtained at the pH value of 5. The adsorption kinetics was fast in the first 1 h, reached equilibrium within 5 h, and could be fitted with the pseudo-second-order kinetics model. The adsorption isotherm was best fitted with the Langmuir isotherm model, and the maximum theoretical adsorption capacity was derived to be 0.854 mmol/g. The PP-PAM also showed obvious selectivity towards Hg(II), with significant adsorption capacity difference between Hg(II) and Pb(II) ions. This study successfully prepared a multi-functional PPHFM and greatly expanded its application in the field of water or wastewater treatment. Furthermore, this study also provided an efficient method for the functionalization of the polypropylene material with the grafted polymer brush, which would expand its application in various fields as a widely used material.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Research Foundations of Shenzhen (KQCX20140519103908550, JCYJ20160308105200725, JCYJ20140418095735550, ZDSYS20160606153007978), and the National Natural Science Foundation of China (21307083).
