Abstract
The pollution of natural waters with aromatic amines is of serious environmental concern. The aim of this work was to enhance the ability of chitosan for selective and effective removal of aromatic amines from natural waters. The chitosan was modified with the inclusion of nano-Al2O3 and then the impregnation with Cu2+ solution. The study on the chemical structure of the modified adsorbent by Fourier transform infrared spectroscopy showed the formation of a complex between amine groups of the chitosan and Cu(II) on the adsorbent surface. It was found that the modified adsorbent, Cu–chitosan/nano-Al2O3, has more porous surface morphology and so more specific surface area available for adsorption. The adsorption properties of the modified adsorbent toward aniline and dimethyl aniline as typical aromatic amines were investigated using kinetics and isotherm studies at room temperature. The adsorption equilibrium data were fitted well by the Freundlich isotherm model. The kinetic data exhibited a non-linear fit to pseudo-second-order model for both aniline and dimethyl aniline. The adsorption capacity of Cu–chitosan/nano-Al2O3 was much higher than that of neat chitosan and chitosan/nano-Al2O3. The adsorbent had high selectivity for adsorption of aniline and dimethyl aniline in the presence of chloride, nitrate, sulfate, phosphate, and carbonate as natural waters common anions. The modified adsorbent exhibited good regeneration ability. The results showed that the introduced adsorbent can be employed as an effective chitosan-based adsorbent for selective removal of aromatic amines from natural waters.
Introduction
The pollution of natural waters by synthetic organic materials is a serious environmental problem (Schwarzenbach et al., 2010; Vörösmarty et al., 2010). Aromatic amines, a class of organic pollutants, can enter bodies of water from many industries associated with pharmaceuticals, pesticides, dyestuffs, petrochemicals, cosmetics, textiles, paper, plastics, and agrochemical production (Pinheiro et al., 2004). The occurrence of these materials in natural waters even at low concentrations has marked negative impact on living organisms and human health (Liu et al., 2012). Furthermore, these compounds are hard to decompose by natural chemical and biological processes. Accordingly, aromatic amines are classified as refractory organic materials by U.S. Environmental Protection Agency (USEPA) (Sun et al., 2009; Zhao et al., 2002) and Europe’s Economic Community (EEC) (Li and Jin, 2009). Therefore, removal of aromatic amines from water is of great importance.
Various treatment methods such as oxidation by ozone (Qi et al., 2002), membrane processes (Ho et al., 1982; Sarode et al., 2001), dialysis (Klein et al., 1972), reverse osmosis (Gómez et al., 2009; Hidalgo et al., 2011), photocatalytic degradation (Kamble et al., 2003), biodegradation (Gheewala and Annachhatre, 1997), and adsorption (Datta et al., 2003) are commonly applied to remove aromatic amines from water. Among these methods, adsorption is considered as a more simple and cost-effective technique for removal of contaminants from water. Various adsorbents such as activated carbon powders and fibers (Chai et al., 2007; Han et al., 2006), carbon nanotubes (Yang et al., 2008), iron oxide magnetite nanoparticles (Bettini et al., 2015b; Kakavandi et al., 2013), montmorillonite (Essington, 1994), Cr-bentonite (Zheng et al., 2009), MCM-41 mesoporous material (Eimer et al., 2003), tetraborate-modified Kaolinite (Unuabonah et al., 2008), sulfonic-functionalized styrene-divinyle benzene co-polymer (Jianguo et al., 2005), and methacrylic acid-modified silica gel (An et al., 2009) have been developed to remove aromatic amines from water.
Biomaterials based on renewable resources such as chitosan and cellulose are of great interest as potential adsorbents for removal of various contaminants from water (Annadurai et al., 2002; Bettini et al., 2014; Crini and Badot, 2008; Gerente et al., 2007). Chitosan, a natural biopolymer, is used extensively as an adsorbent for removal of heavy metals and dyes from contaminated waters (Ngah et al., 2011). The presence of a large number of amino and hydroxyl functional groups as active adsorption sites in the chitosan structure, and its non-toxicity, biodegradability, abundance and low cost, make it a promising adsorbent for aquatic systems (Kumar, 2000). Chitosan-based adsorbents have been also used for removal of organic contaminants from water (Ali et al., 2012; Crisafully et al., 2008). To the best of our knowledge, there is no published study about the removal of aromatic amines from water using chitosan-based adsorbent. Considering chemical structure of chitosan and aromatic amines, a weak interaction, and thus a low adsorption capacity is expected due to the presence of electron-rich functional groups in both chitosan and aromatic amines structures. Therefore, we decided to develop a chitosan-based adsorbent for effective and selective removal of aromatic amines from water.
In the present study, a chitosan-based adsorbent was prepared and employed as adsorbent for the removal of aromatic amines. The porosity of the chitosan beads increased with the inclusion of nano-Al2O3 to obtain chitosan/Al2O3 nanocomposite. The nanocomposite was treated with Cu2+ solution to form Cu–chitosan/Al2O3 nanocomposite. It should be noted that metallic ions such as Cu2+ can be chelated by amine functional groups of chitosan. The main reason for this treatment is that amine containing compounds such as aromatic amines have great tendency to Cu(II) on the adsorbent surface (Le Cocq et al., 2012). Indeed, the condition for chemical adsorption of aromatic amines is provided by applying this modification. The adsorption behavior of aniline (An) and N,N-dimethy aniline (DMA) as predominant aromatic amines in contaminated waters was investigated. Chitosan and chitosan/Al2O3 nanocomposite were also prepared and their properties and behaviors were compared with those of Cu–chitosan/Al2O3 nanocomposite.
Experimental
Materials
Medium molecular weight chitosan with deacetylation degree of 85–95% was purchased from Sigma-Aldrich Chemicals. The nano-filler, γ-Al2O3 with average particle size of 20–30 nm supplied by TECNAN Nanoproducts was employed for adsorbents preparation. All other chemicals including aniline, N,N-dimethyl aniline, oxalic acid, hydrochloric acid, and sodium hydroxide were of analytical grade and purchased from Sigma-Aldrich Chemicals. Distilled water was used for preparation of all solutions.
Preparation of adsorbents
In order to prepare chitosan/nano-Al2O3 beads, γ-Al2O3 nanoparticles were dried in an oven for 6 h at 100℃. The dried nano-filler was mixed with oxalic acid solution for 5 h at room temperature. The acid-treated nano-filler was then filtered, washed with water, and dried in an oven at 80℃ for 3 h. About 10 g of medium molecular weight chitosan was slowly mixed with 200 mL of 10 wt.% oxalic acid solution to form a viscous mixture. The mixture, then, was diluted with 200 mL distilled water and heated to 50℃ to facilitate mixing. About 0.4 g of acid-treated nano-Al2O3 was added to the mixture and stirred for 24 h. Excess oxalic acid in the mixture was neutralized with NaOH solution to settle chitosan/nano-Al2O3 beads. The chitosan/nano-Al2O3 beads were filtered, washed with water completely, and dried in an oven at 60℃ for three days.
In order to prepare Cu-bonded chitosan/Al2O3 nanocomposite, the chitosan/nano-Al2O3 beads were mixed with CuSO4 solution for 2 h. The concentration of CuSO4 and chitosan/nano-Al2O3 beads in the mixture was selected based on maximum adsorption capacity of the prepared adsorbent. In the optimum condition, the molar ratio of Cu2+ to glucosamine groups of the adsorbent was 1.3. Then, the modified adsorbent was filtered, washed with water and dried.
All chemicals employed for preparation of the adsorbent are abundant and commercially available that enables the preparation of the adsorbents with reasonable cost.
Characterization of adsorbents
Surface morphology of adsorbents was studied using scanning electron microscopy (SEM) and atomic force microscopy (AFM). A Vega Tescan SEM (Czech Republic) and a Dual scope C-21 AFM from DME Instruments (Denmark) were employed for morphological studies. Fourier transform infrared (FTIR) spectra of adsorbents were recorded on Bruker Tensor 27 spectrometer (Germany). X-ray diffraction (XRD) patterns of specimens were obtained using a Siemens D-500 X-ray diffractometer (Germany) with wavelength, λ = 1.54 Å (Cu–Kα), at a tube voltage of 25 kV, and a tube current of 30 mA.
The surface area of all adsorbents was determined on the basis of Brunauer–Emmett–Teller (BET) adsorption/desorption isotherm (Lowell and Shields, 2013) using a Quantachrome ChemBET-3000 instrument by means of adsorption/desorption of pure nitrogen gas at 77 k.
To determine pH at the point of zero charge (pHpzc), batch equilibration method was applied (Lataye et al., 2006). A certain amount of each adsorbent (0.2 g) was introduced into a known volume (100 mL) of 0.1 mol/L KNO3 solution. The ionic strength of solutions in all experiments was kept constant by using of KNO3 as inert background electrolyte. The pH values of KNO3 solutions were adjusted from 4 to 10 by addition of 0.1 M HCl or NaOH solutions. The initial pH (pHi) of the solutions was determined exactly and recorded. Suspensions were allowed to equilibrate for 48 h in a shaker at room temperature. Then, the suspensions were filtered and the pH values of the filtrates (pHf) were measured. The pHpzc for the adsorbents was determined by plotting ΔpH (pHi–pHf) versus pHi. The point of intersection with pHi-axis is considered as pHpzc.
Batch adsorption experiments
All batch adsorption studies were performed with aqueous solutions of aniline (An) and dimethylaniline (DMA) with certain concentrations (40, 80, 120, 160, and 200 mg/L). The pH of solutions was adjusted in desired amount using 0.1 M HCl or NaOH solutions. Equilibrium isotherm measurements were carried out by constant solution volume of 50 mL and adsorbent amount of 0.05 g, and varying amounts of An or DMA concentrations at room temperature. The solutions of aromatic amines in the presence of adsorbents were allowed to attain equilibrium by stirring at 100 r/min in a water bath for 4 h. After equilibration, adsorbents were filtered from solutions and filtrates were analyzed for An or DMA.
The amount of An and DMA adsorbed (mg) per unit mass of the adsorbent (g), qe, was obtained by using the following equation
Kinetic studies were carried out by measurement of An or DMA concentration at certain time intervals to determine the rate of aromatic amines removal by adsorbents with the initial An and DMA concentration of 40 mg/L for all adsorbents.
Analytical method
The concentration of the aromatic amines in the aqueous solutions was analyzed using UV spectrophotometry (Łabudzińska and Gorczyńska, 1994; Pinheiro et al., 2004) at the wavelength of maximum absorbance (280–300 nm). Measurements were carried out by using a Shimadzu UV-1800 spectrophotometer.
Interfering effect
In order to determine the interfering effect of common natural water anions including chloride, nitrate, sulfate and phosphate on the aromatic amines adsorption, the experiments were performed with 40 mg/L An or DMA solutions containing 400 mg/L of the interfering anion in the form of its sodium salt.
Desorption and regeneration
To investigate the regeneration ability of Cu–chitosan/nano-Al2O3 adsorbent, it was reused in repeated adsorption–desorption cycles and its capacity was determined after each cycle. The adsorption experiment was carried out using 0.05 g of the adsorbent in 50 mL solutions with An and DMA concentration of 40 mg/L. For desorption experiment, the loaded adsorbent was filtered and washed with distilled water. Then, it was placed in 50 mL of Cu2+ solution (10 g/L) and stirred at 100 r/min for 2 h at room temperature. Afterward, the adsorbent was filtered and washed with distilled water completely. The regenerated adsorbent was dried in an oven at 60℃ for reuse in the next cycle.
Results and discussion
Morphological properties of adsorbents
The surface morphology of the adsorbents was characterized by using SEM. Micrographs of chitosan, chitosan/nano-Al2O3, and Cu–chitosan/nano-Al2O3 beads are shown in Figure 1. It is obvious that the inclusion of nano-Al2O3 in the chitosan matrix leads to a more porous surface morphology for chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3 adsorbents compared to neat chitosan beads. Increased roughness of chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3 adsorbents may provide more surface area for aromatic amines adsorption.
SEM micrographs of (a) neat chitosan, (b) chitosan/nano-Al2O3 and (c) Cu–chitosan/nano-Al2O3.
To further verify the results of SEM studies, the specific surface area was determined for all adsorbents using BET isotherm for nitrogen adsorption/desorption as shown in Figure 2. The specific surface area for chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3 was 6.1, 41, and 31 m2/g, respectively. The surface area for nano-Al2O3 containing adsorbents was much higher than that of pristine chitosan. The presence of nano-Al2O3 may induce the formation of more porous surface morphology as confirmed by SEM imaging. The modified adsorbent, Cu-chitosan/nano-Al2O3, exhibited lower surface area compared to chitosan/nano-Al2O3 adsorbent. The bonding of Cu2+ to chitosan on the adsorbent surface may reduce the surface area somewhat.
N2 adsorption/desorption isotherms at 77 k for neat chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3.
Figure 3 illustrates AFM phase image of Cu–chitosan/nano-Al2O3 adsorbent. An advantage of AFM imaging is the relatively high contrast between soft and hard phases. There is a difference between stiffness of chitosan biopolymer and crystalline alumina regions of Cu–chitosan/Al2O3 nanocomposites, and it is sufficient for contrast development. When the AFM tip scans across the alumina phase, it works in repulsive regime with the bright phase contrast, whereas the dark contrast represents the soft chitosan phase, where the tip works in attractive regime. The bright spherical particles in the figure correspond to nano-Al2O3 phases dispersed in the dark polymeric matrix. The images reveal a relatively uniform dispersion of alumina nanoparticles in the chitosan matrix with little agglomeration which confirms the formation of chitosan-matrix nanocomposites.
AFM phase images of Cu–chitosan/nano-Al2O3; scan sizes: (a) 1 µm and (b) 500 nm.
XRD studies of adsorbents
Figure 4 presents XRD patterns for neat chitosan, nano-Al2O3, chitosan/nano-Al2O3, and Cu–chitosan/nano-Al2O3. For neat chitosan, two peaks around 2θ = 7.8° and 10.9° relate to the hydrated crystalline structure of chitosan and the characteristic peak at 2θ = 19.9° indicates the amorphous structure of chitosan. For the nano-Al2O3 sample, all peaks in the XRD pattern match with standard value of γ-Al2O3 crystalline phase according to the Joint Committee on Powder Diffraction Standards (JCPDS) file No. 29–63. Furthermore, the broadening of XRD peaks indicates the nanocrystallinity of the γ-Al2O3 sample. The most prominent peaks in the γ-Al2O3 diffractogram appeared at 2θ = 45.6° (400 reflection) and 66.9° (440 reflection) which correspond with the values mentioned in the literature (Ogawa et al., 1984; Wang et al., 2005). By analyzing the XRD pattern of chitosan/nano-Al2O3 sample, the main crystalline phases were identified as chitosan, nano-Al2O3, and Na2C2O4. The diffraction peaks for chitosan were the same as those of neat chitosan with little shifts in peaks positions. The reflections for sodium oxalate appeared at 2θ = 31.63°, 34.43°, 38.54°, and 30.82°. The presence of oxalate phase in chitosan/nano-Al2O3 structure is expected as it has been reported that carboxylate group of oxalate has an electrostatic interaction with amine groups of chitosan (Boddu et al., 2003). Due to the nano-size nature and low concentration of nano-Al2O3 in chitosan/nano-Al2O3 composition, the characteristic peaks of nano-Al2O3 are of low intensities which are covered by reflection peaks of other phases. In the Cu–chitosan/nano-Al2O3 sample, copper ammine sulfate, Cu(NH3)2SO4, and copper oxalate, Cu(C2O4) were also identified from its diffractogram in addition to chitosan and alumina phases. The diffraction peak at 2θ = 23° is associated with (NH3)2CuSO4 phase. The reflection peaks at 2θ = 23°, 35.5°, and 39° correspond to Cu(C2O4) phase. The peak appeared at 2θ = 30° relates to sodium carbonate or bicarbonate phases. The presence of Cu(NH3)2SO4 phase in the sample confirms the formation of a bond between chitosan and Cu(II) on the adsorbent surface.
XRD patterns of neat chitosan, nano-Al2O3, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3.
FTIR studies
FTIR spectrometry is a useful technique to identify the presence of certain functional groups on a material surface as a specific chemical bond often has a unique energy adsorption band (Bettini et al., 2014, 2015a). Therefore, chemical properties of three types of adsorbents were evaluated by FTIR spectrometry as shown in Figure 5. The most important characteristic bands for pristine chitosan can be assigned as follows: 3400–3200 cm−1 (a broad band corresponds to –OH and –NH stretching vibration), 2920 and 2865 cm−1 (stretching vibration of aliphatic C–H bonds), 1659 cm−1 (bending vibration of –NH in primary amines), 1382 cm−1 (deformation vibration of –NH in primary amines) and 1081 cm−1 (stretching vibration of –CO in –COH). Compared to the spectrum of neat chitosan, two new bands at 775 and 515 cm−1 appeared in the spectrum of chitosan/nano-Al2O3 which correspond to stretching and bending vibration of Al–O bond. The presence of characteristics bands of chitosan in chitosan/nano-Al2O3 spectrum without significant changes indicates that the incorporation of nano-Al2O3 into chitosan bead has no significant effect on chitosan structure. In the spectrum of Cu–chitosan/nano-Al2O3, the noticeable point is the absence of a broad band between 3400 and 3200 cm−1 observed for chitosan and chitosan/nano-Al2O3. This indicates the formation of complexes between amine and hydroxyl groups on the chitosan surface and copper ions.
FTIR spectra of (a) chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3 before adsorption and (b) Cu–chitosan/nano-Al2O3 after adsorption.
To understand the nature of aromatic amines adsorption on Cu–chitosan/nano-Al2O3, FTIR spectra of the modified adsorbent after adsorption of An and DMA were also obtained as presented in Figure 5. It is noticeable that the two spectra are so similar. After adsorption of An and DMA on the modified adsorbent, two major changes can be seen in the related spectra. One is a significant increase of the intensity of bands around 1650–1400 cm−1 and 3400–3200 cm−1 assigned to bending vibration of –NH groups in primary amines and stretching vibration of –OH and –NH groups of chitosan in the adsorbent composition, respectively. It may be due to the formation of a complex between aromatic amines and Cu(II) on the adsorbent surface and weakening of N–Cu and O–Cu bonds and so strengthening of N–H and O–H groups. Another is the appearance of new peaks at 1620 and 1363 cm−1 corresponding to stretching vibration of C–C bond of benzene ring, and at 1319 cm−1 corresponding to stretching vibration of C–N bond in aromatic amines. The emergence of these new peaks in the FTIR spectra can be attributed to the presence of aromatic amines adsorbed on the modified adsorbent. Briefly, the FTIR results suggest that the nature of aromatic amines adsorption on Cu–chitosan/nano-Al2O3 is based on the chemical bonding between aromatic amines and Cu(II) on the adsorbent surface.
Adsorption isotherms
Adsorption isotherms describe the equilibrium distribution of an adsorbate between a solid phase as adsorbent and a liquid phase at constant temperature and provide a quantitative measure of the adsorption efficiency and the nature of adsorption. The most prevalent isotherm models, namely Langmuir and Freundlich isotherms (Foo and Hameed, 2010) were examined in the present study. Langmuir isotherm can be expressed as:
The Freundlich isotherm is defined by the following equation
The experiments for studying the adsorption isotherms were conducted using aqueous solution of the aromatic amines with initial concentrations varying from 40 to 200 mg/L at neutral pH range (pH = 6.5–7.0), adsorption time of 4 h at room temperature. The data for adsorption of An and DMA by three types of the adsorbents were fitted to non-linear model of Langmuir and Freundlich isotherms as shown in Figures 6 and 7, respectively. The fitting parameters along with the isotherm constants for both models are given in Table 1. The fitting parameters, correlation coefficient (R2), sum of square errors (SSE) and root mean square error (RMSE), indicate that the adsorption data are in relatively good agreement with both isotherm models. For the modified adsorbent, Freundlich model with higher R2 and lower SSE and RMSE values seems to be better model to describe the adsorption data. According to the fitting data obtained from Langmuir model, the magnitude of maximum adsorption capacity (qm) for three types of adsorbents toward both aromatic amines is in the order chitosan < chitosan/nano-Al2O3 < Cu–chitosan/nano-Al2O3. It implies that the incorporation of the particulate nano-filler into chitosan bead and the surface modification with Cu2+ have considerable effect on chitosan efficiency for the adsorption of aromatic amines. Based on the results of morphological studies, the surface area of the chitosan bead increases with the inclusion of a nano-filler such as nano-Al2O3 and it may provide more adsorption sites for aromatic amines adsorption. For Cu2+-treated chitosan/nano-Al2O3 adsorbent, more favorable conditions may be provided through chelating of aromatic amines by Cu(II) on the adsorbents surface as indicated by XRD and FTIR studies.
Fitting of adsorption data to Langmuir model for adsorption of (a) aniline and (b) dimethyl aniline on chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3. Fitting of adsorption data to Freundlich model for adsorption of (a) aniline and (b) dimethyl aniline on chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3. Langmuir and Freundlich isotherm constants and related fitting parameters for adsorption of aniline and dimethyl aniline on chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3. SSE: sum square errors; RMSE: root mean square errors.


Adsorption kinetics
The prediction of the rate of adsorption for removal of a pollutant from aqueous solution is so important for scale-up and design purposes. The kinetic data for adsorption of the aromatic amines on the adsorbents were fitted to pseudo-first-order and pseudo-second-order kinetic models as shown by equations (4) and (5), respectively.


The non-linear forms of pseudo-first-order and pseudo-second-order kinetic models are expressed, respectively, by equations (6) and (7) obtained by integrating the differential equations with the boundary conditions of qt = 0 at t = 0 and qt = qt at t = t.


The non-linear fitting method was employed to obtain the correlation between the experimental kinetic data and the kinetic models. Table 2 presents the value of the rate constants, k1 and k2, and the correlation parameters of both models for adsorption of An and DMA on three types of adsorbents. Although the fitting parameters are rather satisfactory for both kinetic models, the values of equilibrium uptakes predicted by pseudo-second-order kinetic model present considerable differences with experimental data. In other words, pseudo-first-order kinetic model has good compliance with kinetic data in the case of both fitting parameters and the predicted values of equilibrium uptake. Figure 8 shows experimental kinetic data and the pseudo-first-order non-linear model for adsorption of An and DMA for three types of adsorbents. It can be seen that the adsorption of An and DMA on three types of adsorbents is rather rapid and reaches to equilibrium state within the first 30 min of contact. The initial adsorption rate (V0) for three types of adsorbents was calculated using pseudo-first-order kinetic by placing qt = 0 at t = 0 in the kinetic model as follows
Pseudo-first-order kinetic model for adsorption of (a) aniline and (b) dimethyl aniline on chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3. Parameters of pseudo-first-order and pseudo-second-order kinetic models fitting of aniline and dimethyl aniline adsorption data on chitosan, chitosan/nano-Al2O3, and Cu–chitosan/nano-Al2O3 adsorbents.
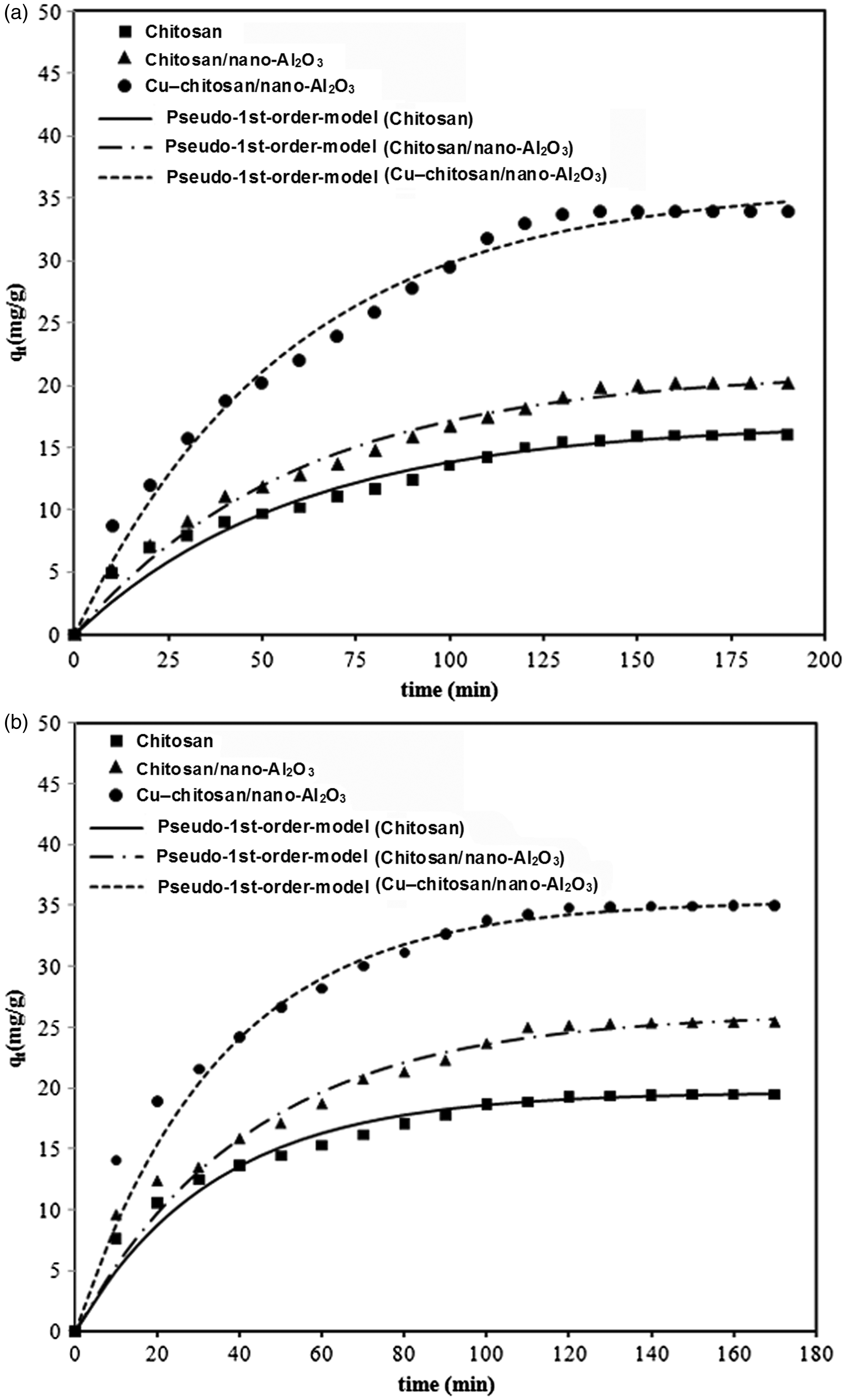
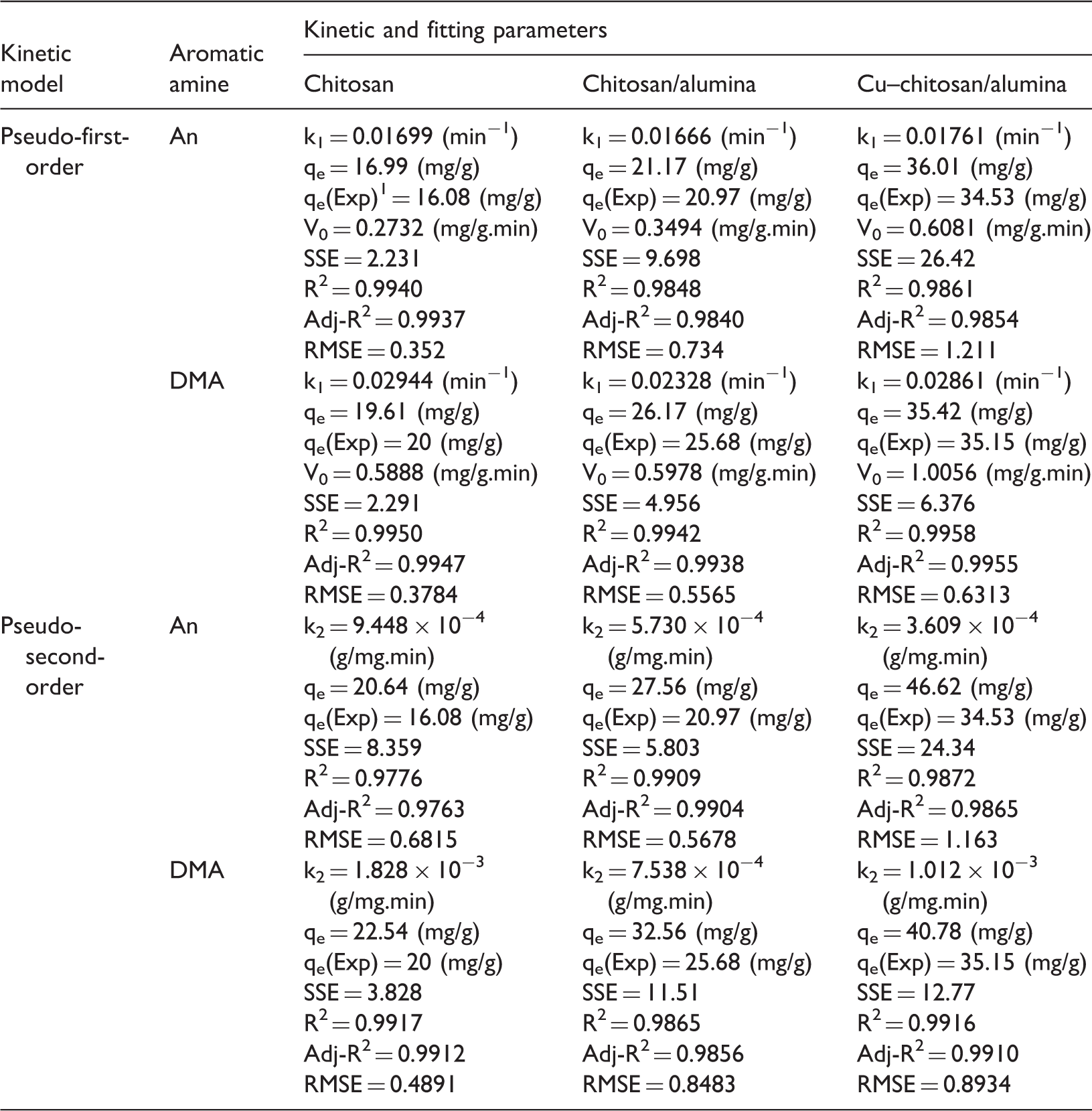
The comparison of k1 values shows no significant difference between adsorbents, while the magnitude of initial adsorption rates and maximum adsorption capacities for Cu–chitosan/nano-Al2O3 are much higher than those of neat chitosan and chitosan/nano-Al2O3 adsorbents. It implies that Cu–chitosan/nano-Al2O3 can more quickly absorb much higher amounts of aromatic amines during short time of exposure than neat chitosan and chitosan/nano-Al2O3 adsorbents. After initial adsorption, the adsorption rate of Cu–chitosan/nano-Al2O3 is almost the same as that of chitosan and chitosan/nano-Al2O3 adsorbents. Considering the results of kinetics studies, Cu–chitosan/nano-Al2O3 presents much better kinetic characteristics for effective removal of An and DMA as typical aromatic amines.
Effect of initial pH
The adsorption capacities of three types of adsorbents toward An and DMA were studied in the pH range of 4.0–11.0 at 25℃. Initial concentration of the aromatic amines was selected to be 40 mg/L. Figure 9 presents the adsorption capacities of three types of adsorbents versus the initial pH of the aromatic amine containing solution. A similar trend is observed for adsorption of An and DMA by each adsorbent. For neat chitosan bead, the adsorption capacity increases steeply with the increase of pH value from 4.0 to ∼7.2 and then decreases in the pH range 7.2–11.0. In the case of chitosan/nano-Al2O3 adsorbent, a gradual increase in the adsorption capacity is seen over pH range 4.0–8.0 and a decrease is observed at higher pH values. For Cu–chitosan/nano-Al2O3, the adsorption capacity increases with increase of pH from 4.0 to ∼7.2 and decreases at higher pH values. To explain the behaviors observed, the pH at the point of zero charge (pHpzc) was determined for the adsorbents. The pHpzc for chitosan, chitosan/nano-Al2O3 Na and Cu–chitosan/nano-Al2O3 was determined to be, respectively, 7.6, 8.15, and 6.3 using the results of batch equilibrium experiments as shown in Figure 10. At pH values lower than pHpzc of an adsorbent, the surface is positively charged, while at pH values higher than the pHpzc, the surface is negatively charged. The relation between pH value and the aromatic amines adsorption on chitosan and chitosan/nano-Al2O3 can be explained by the pHpzc of the adsorbents. As pH increases up to the pHpzc, the positively charged surface sites decrease, on the one hand, and the concentration of protonated aromatic amines as the positively charged species decreases on the other hand, causing an increase in the adsorption. Thus, the adsorption of the amines on chitosan and chitosan/nano-Al2O3 adsorbents can be addressed to the electrostatic interactions between the aromatic amines species and the surface of adsorbents. For Cu–chitosan/nano-Al2O3, the mechanism based on electrostatic interactions cannot describe the variation of aromatic amines adsorption against pH value duo to its different behavior. The adsorption of aromatic amines increases with increase of pH value up to ∼7.2 higher than the pHpzc (6.3). In other word, a considerable increase occurs after the pHpzc and it cannot be explained by electrostatic interactions on the surface. Furthermore, the adsorption capacity for Cu–chitosan/nano-Al2O3 is much higher than that of others. It suggests that the adsorption on the modified adsorbent may be occurred by complex formation rather electrostatic interactions as evidenced by FTIR and XRD studies. Aromatic amines such as aniline can react with Cu(II) at room temperature to form Cu(II)–aromatic amines complexes (Le Cocq et al., 2012). At pH > 7.2, the adsorption capacity decreases gradually but is still higher than that of neat chitosan and chitosan/nano-Al2O3 adsorbents. It may be due to the formation of Cu(II) hydrate in competition with the formation of the complex between Cu(II) and aromatic amines. Formation of copper oxide at pH values higher than 10 was clearly visible as the adsorbent color changes from blue to dark brown.
Effect of initial pH on adsorption of (a) aniline and (b) dimethyl aniline by neat chitosan, chitosan/nano-Al2O3 and Cu–chitosan/nano-Al2O3. Determination of pHpzc for neat chitosan, chitosan/nano-Al2O3, and Cu–chitosan/nano-Al2O3 adsorbents.

Reusability of adsorbent
Considering economic aspects of an adsorption process for commercial applications, it is important that an adsorbent can be regenerated and reused in a cyclic manner. The reusability of the modified adsorbent was evaluated by determining the adsorption capacity after several regeneration cycles. Figure 11 shows the variation of the adsorption capacity after each adsorption–desorption cycle. A slight reduction in the adsorption capacity was observed for An and DMA after five cycles of the adsorption–desorption process. It indicates that the studied adsorbent can be regenerated and reused repeatedly and so has a high durability.
Adsorption efficiency of Cu–chitosan/nano-Al2O3 toward aniline and dimethyl aniline versus regeneration cycle.
The modified adsorbent, Cu–chitosan/nano-Al2O3 showed desirable stability of its capacity when stored in air at room temperature so that there was no measureable change in its capacity after four weeks of preparation.
Interfering effects
The interfering effect of natural waters common anions (chloride, nitrate, phosphate, carbonate, and sulfate) on An and DMA adsorption by Cu–chitosan/nano-Al2O3 was studied. The concentration of interfering anions was selected to be 10 times higher than that of An and DMA. As shown in Figure 12, the presence of nitrate and chloride anions had no considerable effect on the adsorption capacities. In the presence of phosphate, carbonate and sulfate anions, the adsorption capacities were decreased slightly indicating low interfering effect of these anions.
Adsorption capacity of aniline and dimethyl aniline in the presence of interfering anions (400 mg/L). The experiments conditions: aniline and dimethyl aniline concentration of 40 mg/L and the adsorbent amount of 1 g/L.
Conclusions
FTIR and XRD studies of the modified adsorbent indicated the presence of Cu(II) bonded to amine groups of chitosan on the surface. Cu–chitosan/Al2O3 nanocomposite provided more surface area for adsorption compared to neat chitosan as confirmed by SEM and BET studies. Experimental adsorption data could be well explained by both Freundlich and Langmuir isotherm models. Maximum adsorption capacities for adsorption of An and DMA by the modified adsorbent were, respectively, 84 and 61 mg/g, much higher than those for neat chitosan The adsorption of aromatic amines on the modified adsorbent was to be based on chemical bonding to Cu(II) on the adsorbent surface as confirmed by FTIR studies of the modified adsorbent after adsorption. The modified adsorbent showed a reasonable selectivity for An and DMA in the presence of natural waters common anions. The results showed that the Cu–chitosan/Al2O3 nanocomposite can be employed as a chitosan-based adsorbent for effective and selective removal of aromatic amines from natural water.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the University of Maragheh.



