Abstract
This paper is aiming at analyzing the desorption performance of the four polyamine-functionalized adsorbents after copper ion adsorption, with nitric acid as the desorption agent. The four adsorbents (namely, P-EDA, P-DETA, P-TETA and P-TEPA) studied were immobilized with ethylenediamine (EDA), diethylenetriamine (DETA), triethylenetetramine (TETA) and tetraethylenepentamine (TEPA), respectively. Nitric acid concentration, ionic strength, and solid-to-liquid (S/L) ratio were taken into consideration as the influencing factors on the desorption performance. The findings showed that higher desorption efficiency had no necessary connection with the higher concentration of nitric acid, and the highest desorption efficiency was realized for P-EDA and P-DETA at 0.10 mmol/L, and for P-TETA and P-TEPA at 0.25 mmol/L, in the nitric acid concentration range of 0.01–2.00 mmol/L studied. In addition, Ionic strength showed obvious influence on the desorption efficiency, and higher ionic strength resulted in significant lower desorption efficiency. Different S/L ratios studied showed almost no differences on the desorption efficiency. The copper ion desorption kinetics, best described by the Elovich model, was discovered to be very fast that more than 85% of the desorption efficiency was attained within 2 min. A slight decrease in Cu ion adsorption capacity was found through ten cycles of adsorption-desorption-regeneration for all the four adsorbents.
Introduction
Heavy metal pollution poses serious threat to the environment and public health, due to the toxic and non-biodegradable nature of the heavy metal ions (Naushad et al., 2015a, 2015b; Wang and Chen, 2008). It is a common practice in industry that adsorption technology has been widely applied for heavy metal ion removal. Adsorbents immobilized with sulfonic, carboxylic or amine groups often play a vital role in the adsorption efficiency and selectivity of the heavy metal ions (Anirudhan et al., 2015; Volesky and Holan, 1995; Wan Ngah and Hanafiah, 2008; Wang and Chen, 2015). To increase the reusability of the adsorbents, as well as to decrease the industrial cost, most of the adsorbents after adsorption have adopted the desorption and regeneration process for the purpose of recovery and reuse (Alshehri et al., 2014). Therefore, in terms of significance, the desorption and regeneration process nearly equals the adsorption process.
The heavy metal ion desorption has been investigated with the soil materials as adsorbents, including minerals, clay, sand, etc. In addition, detailed metal ion desorption investigations were also conducted on biosorbents which mainly involved biopolymers such as alginic acid and cellulose, and biomass such as alga and activated sludge (Huang et al., 2013; Kumar et al., 2014; Rabiul Awual et al., 2015; Wang and Peng, 2010). Hydroxyl, carboxylate or metal oxyhydroxide functional groups were generally considered as basic components of the soil materials and biosorbents for metal ion desorption investigation. However, few reports were issued on the systematic and comprehensive desorption study of heavy metal ions with the amine-based adsorbents, which have often been regarded as one of the most effective adsorbents for heavy metal ion adsorption (Wu et al., 2013). Most papers which reported amine-based adsorbents emphasized the heavy metal ion adsorption performances; only a limited portion of research work was focused on the desorption of heavy metal ions.
Another important reason for us to study the desorption performance of the amine-based adsorbent is from the economical point of view. By investigating the influencing factors such as desorption agent concentration and solid-to-liquid (S/L) ratio, the best desorption efficiency can be achieved with the least amount of desorption agents (e.g. nitric acid). As a result, a sustainable use of the adsorbents for cycles of adsorption-desorption of heavy metal ions would be favored with higher desorption efficiency and much lower processing cost in the industry (Abdolali et al., 2015; Zhang et al., 2015). Moreover, the desorption kinetics of amine-based adsorbents, as well as the surface properties of such adsorbents in heavy metal ion adsorption–desorption–regeneration process have not been thoroughly studied, but these investigations are also important in better understanding of the desorption and regeneration mechanisms (Liu et al., 2002; Naushad et al., 2015c; Yu et al., 2011).
In this study, investigations were conducted on copper ion desorption performance with the polyamine-based adsorbents after copper ion adsorption. The adsorbents used were P-ethylenediamine (P-EDA), P-diethylenetriamine (P-DETA), P-triethylenetetramine (P-TETA), and P-tetraethylenepentamine (P-TEPA), prepared by amination of poly (glycidyl methacrylate) (PGMA) crosslinked polymers with EDA, DETA, TETA and TEPA, respectively, as reported elsewhere (Liu et al., 2008). Nitric acid (HNO3) solution was used as the desorption agent throughout the experiments. Influencing factors such as HNO3 concentration and S/L ratio were studied for the copper ion desorption performance. Furthermore, investigations were also carried out on surface chemistry property, the optimum desorption conditions, desorption kinetics, and the copper ion adsorption–desorption–regeneration cycle.
Materials and methods
Materials
PGMA polymer was prepared via the suspension polymerization of the glycidyl methacrylate (GMA, AR, Aladdin) monomer and the crosslinker monomer to form the polymer beads (Liu et al., 2008). Chemical agents including EDA (AR, Aldrich), DETA (AR, Aladdin), TETA (AR, Fluka), and TEPA (AR, Aladdin) were used for the amination of PGMA beads to produce P-Polyamines (PGMA polymers aminated with polyamines). Briefly, 1 g PGMA beads and 7 mL 1,4-dioxane were added in a 20-mL Pyrex test tube and degassed with argon for 20 min. Then, one kind of the polyamines (EDA, DETA, TETA, or TEPA) was added for the amination reaction. The amination reaction conditions were: in the form of P-Polyamines (polyamine concentration; reaction temperature; reaction time), P-EDA (2.76 mol/L; 70℃; 6 h), P-DETA (2.76 mol/L; 70℃; 6 h), P-TETA (2.76 mol/L; 70℃; 10 h), and P-TEPA (2.76 mol/L; 70℃; 10 h) (Liu et al., 2010). The prepared P-Polyamines were then vacuum-filtered using a Buchner funnel, and washed several times with water until the pH of the washing liquids became neutral. In the adsorption experiments, the copper ion solutions were prepared from copper (II) nitrate (Cu(NO3)2·3H2O, from Aladdin). Nitric acid solutions were prepared from the concentrated nitric acid (AR, Aladdin). Copper ion solutions for inductively coupled plasma (ICP) calibration in the analysis were prepared from 1000 µg/mL of the copper (II) nitrate standard solution from Aladdin. Deionized (DI) water was used for the preparation of all solutions.
Batch desorption studies
The four P-Polyamines adsorbents (P-EDA, P-DETA, P-TETA and P-TEPA) used in copper ion desorption performance investigation were first adsorbed with copper ions. Briefly, 500 mg of each of the P-Polyamines was put into 1000 mL beaker containing 500 mL of Cu(NO3)2 solution (4 mmol/L, pH 5). The beakers were shaken at 150 r/min in a shaker for 24 h (period far exceeding Cu ion adsorption equilibrium). Afterwards, the Cu ion-adsorbed P-Polyamines were filtered and vacuum-dried before use. The Cu ion-loaded adsorbents were named as P-EDA-Cu, P-DETA-Cu, P-TETA-Cu and P-TEPA-Cu, respectively (similarly, named as P-Polyamines-Cu). The four P-Polyamines-Cu were used in the subsequent Cu ion desorption experiments. All the desorption experiments were repeated twice and error bars as determined from the standard deviations of the two parallel experiments’ results were added in all the figures.
Detailed desorption studies were carried out with nitric acid solution as the desorption agent. The influencing factors including nitric acid concentration, ionic strength, and S/L ratio were examined during the desorption experiments. In addition, the desorption kinetics and repeated use of the P-Polyamines were also investigated. The concentration of copper ion desorption solutions were analyzed with inductively coupled plasma optical emission spectrometry (ICP-OES, Optima2100DV).
For the influence of nitric acid concentration on desorption, 20 mL of HNO3 solution with ranging concentrations from 0.01 to 2.00 mmol/L were used to desorb the copper ion from P
For the influence of ionic strength on desorption, nitric acid was added with NaNO3 to adjust the ionic strengths. Nitric acid solutions with five different ionic strengths (0.10, 0.25, 0.50, 1.00 and 1.50 mol/L in the form of NaNO3 concentration) were investigated for the ionic strength influence on the copper ion desorption from the four P-Polyamines-Cu. A set of 20 mL of the above-mentioned nitric acid solutions with the best desorption concentration for each P-Polyamine-Cu were used to desorb the copper ion from each of the four P-Polyamines-Cu, which were obtained from Cu ion adsorption with 50 mg P-Polyamines.
For the effect of the S/L ratio on desorption, a set of nitric acid solutions with the best desorption concentration for each P-Polyamines-Cu, were investigated with the four P-Polyamines-Cu, which were obtained from Cu ion adsorption with 60 mg P-Polyamines. The S/L ratio is defined as the ratio of the weight of the P-Polyamine to the volume of the nitric acid. The S/L ratios were 7.50, 5.00, 3.75, and 3.00 for the nitric acid solutions with the volume of 8, 12, 16, and 20 mL, respectively.
The desorption kinetics were investigated for the four P-Polyamines-Cu. 160 mL of nitric acid solutions with the best desorption concentration for each P-Polyamines-Cu were applied for each of the four P-Polyamines-Cu, which were obtained from Cu ion adsorption with 1.200 g P-Polyamines. The solutions were sampled at regular time intervals for the determination of the desorbed copper ion concentrations with ICP-OES in the desorption kinetics.
The repeated use of the P-Polyamines was conducted in the following consecutive steps: namely, Cu ion adsorption, Cu ion desorption, P-Polyamines regeneration, P-Polyamines rinsing, and Cu ion adsorption. The Cu ion desorption experiments followed the same procedure with that of the above-mentioned “Cu ion desorption kinetics” experiments. For the regeneration and rinsing step, P-Polyamines with Cu ion desorbed were put into 160 mL of 0.5 mol/L NaOH solution and shaken in a shaker for 2 h. After that, P-Polyamines were subsequently washed with 160 mL DI water in a beaker and shaken for 2 h. After the above mentioned procedures, the P-Polyamines were vacuum dried for the next round of Cu ion adsorption experiments.
Characterization
X-ray photoelectron spectroscopy (XPS, Thermo ESCALAB 250Xi, USA) was used to characterize the oxidation states of the amine groups (represented by the nitrogen element) on the surface of the P-Polyamines during the adsorption–desorption–regeneration process. An Al Kα X-ray source (1486.6 eV of photons) was used for the XPS analysis with a high vacuum of 10−7 Pa maintained in the XPS chamber. The XPSpeak 4.1 software was utilized to deconvolute the XPS spectra of the N element into subcomponents. The full width at half-maximum was maintained constant for all the subcomponents in a specific spectrum.
Results and discussion
Cu ion adsorption with P-Polyamines
Comparison of the Cu ion uptake of the P-Polyamines in this work with other amine-based adsorbents.

Cu ion desorption with different nitric acid (HNO3) concentrations
It was shown in Figure 1 that the desorption efficiency did not show a monotonous increase as the increase of the nitric acid concentrations, ranging from 0.01 to 2.00 mol/L. Instead, the best desorption efficiency was achieved at 0.10 mol/L for P-EDA-Cu and P-DETA-Cu, and 0.25 mol/L for P-TETA-Cu and P-TEPA-Cu. Other nitric acid concentrations, higher or lower, would lead to a lower desorption efficiency. As is often reported in the literature, desorption efficiency showed a monotonic increase with the increase of the desorption agent concentration (Chouyyok et al., 2011; Hammaini et al., 2007; Liu and Bai, 2006). This finding therefore showed a different phenomenon, indicating the importance to investigate the nitric acid concentration effects on copper ion desorption efficiency. From the XPS study (“Cu ion adsorption-desorption-regeneration cycles with P-Polyamines” section, Figure 6, P-EDA as the representative of the four P-Polyamines for the XPS study), it can be seen that the amine groups of the P-Polyamines-Cu existed in three forms, namely, pristine amine, protonated amine, and Cu ion-complexed amine. During the desorption process, two main equilibrium equations took place which were shown in equations (1) and (2), where R-NH2 denotes the P-Polyamines, R-NH2 … Cu2+ denotes the P-Polyamines-Cu, and H+ denotes the nitric acid. The addition of nitric acid would drive both equations (1) and (2) to the right side, and Cu ions were desorbed according to equation (1). However, as more nitric acid was introduced, the protonated amines (R-NH3+) generated from both equations would accumulate and tend to shift equation (1) to the left side, favoring less Cu ion desorption.

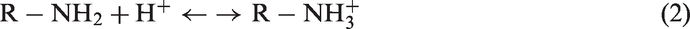
Cu ion desorption with different ionic concentrations
Ionic concentration can be another important factor during the metal ion desorption process from the adsorbents. Gao et al. (2003) showed an increase of the desorption ratio with the increase of the ionic strength in solution for the contaminated soil adsorbent. Yuan et al. (2007) reported the similar trend for the soil adsorbent studied. For amine-based adsorbent, however, few investigations were carried out for the ionic strength effect on metal ion desorption. Our study with P-Polyamines adsorbents showed an opposite trend as compared with the reported trend of the soil adsorbents. Figure 2 clearly showed that the desorption efficiency decreased as an increase of the ionic strength. One possible reason is that the increase of the ionic strength would cause the decrease of the activity coefficient of the ions in solution. Therefore, the quantity of the H+ ions participating in the desorption of copper ions from P-Polyamines-Cu would decrease, which would decrease the desorption efficiency. Another possible reason for the observed phenomena was related to electrical double layer of the P-Polyamines-Cu. The thickness of the double layer can be expressed as the reciprocal of the Debye-Hückel parameter κ (1/κ), which is shown in equation (3), where Nitric acid concentration effect on Cu ion desorption efficiency of P-Polyamines-Cu. Effect of ionic concentration on Cu ion desorption efficiency of P-Polyamines-Cu.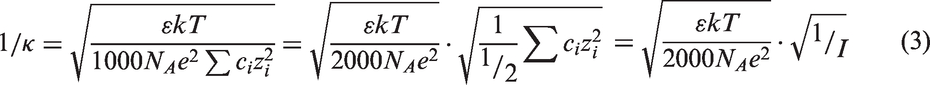


Cu ion desorption with different S/L ratios
The S/L ratio, which may affect the desorption efficiency as an important influencing factor, was also investigated in this paper. A higher S/L ratio would be favored since a relatively small volume of the desorption agent was able to achieve a high desorption efficiency of the adsorbents, saving cost for adsorbent regeneration. In this study, four S/L ratios were selected for Cu ion desorption of P-Polyamines-Cu, as shown in Figure 3. The best nitric acid concentrations (0.10 mol/L for P-EDA-Cu and P-DETA-Cu, and 0.25 mol/L for P-TETA-Cu and P-TEPA-Cu) were selected for the experiments. It was found that the desorption efficiencies remained almost the same for the four P-Polyamines-Cu, although a slight increase in the desorption efficiency was observed with the highest S/L ratio (7.50). The highest S/L ratio (7.50) in this study was more desirable for desorption, due to less nitric acid used to achieve the higher desorption efficiency. The observed phenomenon may result from the rich nitric acid content for Cu ion desorption, despite the change of the nitric acid solution volume.
Effect of solid-to-liquid (S/L) ratio on the Cu ion desorption efficiency of P-Polyamines-Cu.
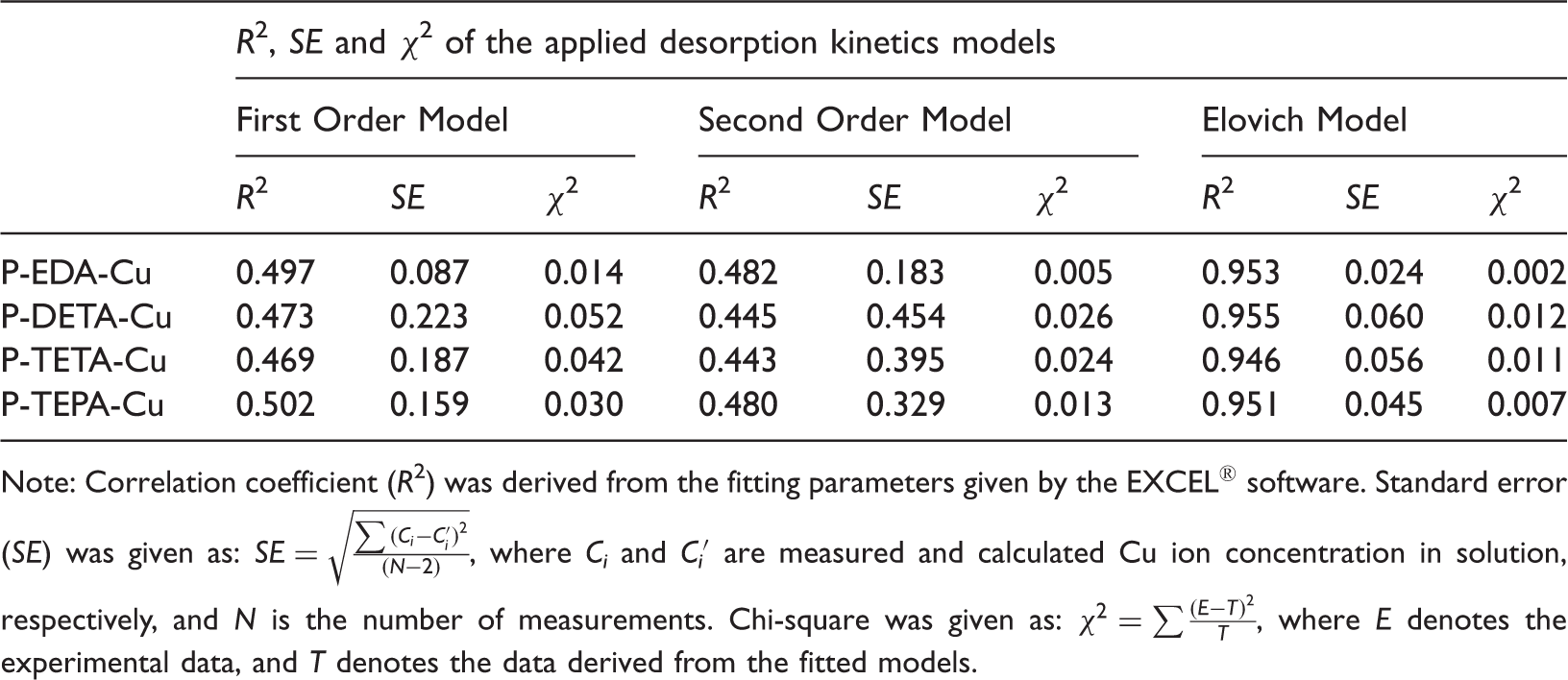
Cu ion desorption kinetics
Figure 4(a)–(d) presented the desorption kinetics performance of the four P-Polyamines-Cu. It was found that over 85% of the desorption efficiency was achieved within the first 2 min, indicating an extremely fast desorption kinetics. In addition, the desorption kinetics, in terms of Cu ion concentration released into solution as a function of desorption time, were also shown in Figure 4(a)–(d). A series of kinetic models, namely, First Order Model, Second Order Model, and Elovich Model, were applied to the Cu ion desorption kinetics from the four P-polyamine-Cu. The three different kinetic models and their model parameters were shown in Table 2. The correlation coefficient ( Desorption efficiency and desorption kinetics (with Elovich model fitting) of P-Polyamines-Cu in HNO3 solution. (Desorption conditions: for P-EDA-Cu and P-DETA-Cu, 1.0 g P-Polyamines-Cu, 160 mL of 0.10 M HNO3 solution; For P-TETA-Cu and P-TEPA-Cu, 1.0 g P-Polyamines-Cu, 160 mL of 0.25 M HNO3 solution.) DETA: diethylenetriamine; EDA: ethylenediamine; TEPA: tetraethylenepentamine; TETA: triethylenetetramine. Kinetic models and parameters for Cu ion kinetic desorption. Note: With the assumption of Correlation coefficient ( Note: Correlation coefficient ( Model parameters of the Elovich Model applied to the Cu ion desorption kinetics of different P-Polyamines-Cu. Note: 


Cu ion adsorption–desorption–regeneration cycles with P-Polyamines
The evaluation of adsorbent’s performance depends tremendously on metal ion adsorption–desorption–regeneration cycle performance tests. All the four P-Polyamines were examined for ten cycles of Cu ion adsorption, Cu ion desorption, and adsorbents regeneration in this study, as shown in Figure 5. For all the four P-Polyamines, in general, only a slight decrease in the Cu ion uptake was observed. The results hence indicated a stable reusability of the polyamine-based adsorbents and a great potential of these adsorbents to remove heavy metal ions for industrial application. To further examine in detail the oxidation states of the amine functional groups on the surface of P-Polyamines during one round of adsorption–desorption–regeneration process, N 1 s XPS study was carried out and the XPS spectra was shown in Figure 6(a)–(d). As have been reported earlier (Liu et al., 2008), the N 1 s XPS sub-peaks of N1, N2, N3, and N4 were representative of neutral amine (– Cu uptakes by (a) P-EDA, (b) P-DETA, (c) P-TETA, and (d) P-TEPA in ten adsorption–desorption-regeneration cycles. N 1s XPS spectra of (a) P-EDA, (b) P-EDA-Cu, (c) P-EDA-Cu-D (P-EDA-Cu after desorption in 0.1 M HNO3 solution), and (d) P-EDA-Cu-D-R (P-EDA-Cu-D after regeneration in 1 M NaOH solution followed by DI water rinse).

Conclusions
Metal ion desorption properties of the polyamine-based adsorbents were the main focus of this paper. Four polyamine-based adsorbents functionalized by EDA, DETA, TETA, and TEPA were used for Cu ion desorption and regeneration studies (P-Polyamines for all the four adsorbents), with nitric acid as the desorption agent. Several influencing factors that were surveyed included nitric acid concentrations, ionic strengths, and S/L ratios. The best nitric acid concentrations were determined as 0.1 mol/L for P-EDA and P-DETA, and 0.25 mol/L for P-TETA and P-TEPA, respectively. Higher nitric acid concentrations may not result in a higher Cu ion desorption efficiency. In terms of ionic strength effects, results showed a decrease in desorption efficiency as an increase of the ionic strength. No apparent changes were observed for the desorption efficiency with the four S/L ratios studied, and hence the highest S/L ratio was applied to save the cost. In the desorption kinetics test, more than 85% of desorption efficiency was achieved within the first 2 min. The fast desorption kinetics were best fitted with the Elovich kinetics model and the surface oxidation states of the amine groups on P-Polyamine surface were examined by XPS study during the Cu ion adsorption–desorption–regeneration process.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Natural Science Foundation of China (21307083), the Research Foundations of Shenzhen (JCYJ20160308105200725, JCYJ20140418095735550, JCYJ20140418091413506, KQCX20140519103908550, KQCX20130625164044956), and the Foundation for Distinguished Young Talents in Higher Education of Guangdong (No. 2013LYM_0079).
