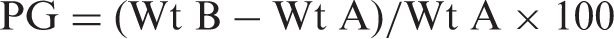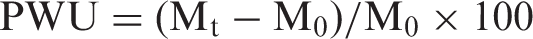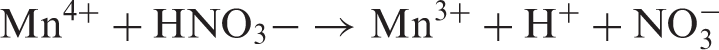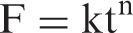Abstract
In this work, poly(acrylic acid) has been grafted onto cotton fibers through the free radical initiated polymerization and the resulting fibers have been characterized by Fourier transform infrared, thermogravimetric analysis, and scanning electron microscopy analysis. The grafted fibers have been loaded with copper nanoparticles using in situ approach. The Transmission Electron Microscopy (TEM) analysis of Cu nanoparticles revealed that almost 45% of the particles had a diameter range of 60–80 nm. The copper nanoparticles loaded fibers show slow release of Cu(II) ions, extended over a period of around 50 h. The release of Cu(II) ions followed a second-order kinetic model successfully. The fibers also exhibited an excellent antibacterial action against model bacteria Escherichia coli as tested by zone of inhibition method.
Introduction
Cotton fibers based gauze have been used as wound dressing material for hundreds of years because it is naturally soft, pliable, and has a strong water absorbing capacity. Cotton fibers consist mainly of cellulose, which is linear chain of several hundreds to over 9000 β (1→4) linked
There are several reports which describe different type of strategies to load metal nanoparticles into the cotton cellulose fiber networks for antibacterial applications. For example, Klemencic et al. [18] have proposed a novel two-step procedure utilizing the pad-dry-cure method to apply an inorganic–organic hybrid sol–gel precursor (reactive binder, RB) followed by the in situ synthesis of AgCl particles on the RB-treated fibers. The antimicrobial activity against the bacteria Escherichia coli and Staphylococcus aureus was estimated according to the ISO 20645:2004 (E) and AATCC 100-1999 methods. Similarly, polyvinyl alcohol/regenerated silk fibroin/AgNO3 composite nanofibers were prepared by electro spinning [19]. Most recently [20], calcium alginate impregnated cotton fabric has been loaded with copper nanoparticles to impart antimicrobial properties. The fabric, so prepared, was characterized by Transmission Electron Microscopy (TEM) and Fourier transform infrared (FTIR) analysis. The TEM is used to measure the size of the nanoparticles. No adverse effect was found on the mechanical properties of fabric due to alginate impregnation. The release of Cu(II) ions was studied in the physiological fluid at 37℃ under different experimental conditions, such as varying concentrations of sodium alginate and the cross-linker calcium chloride. The fabrics showed an appreciable release of Cu(II) ions, extended over a period of 50 h. The amount of Cu(II) ions released showed a negative dependence on the amount of alginate present within the fabric network and the concentration of cross-linker calcium chloride used. The release data were fitted on the Higuchi diffusion-controlled release model successfully. Finally, the antibacterial activity of fabric was tested by zone inhibition method against E. coli as model bacteria.
In order to use cotton fibers as an effective material for wound healing applications, we hereby propose a strategy, which involves attachment of a poly(AAc) chain to the cellulosic backbone via graft co-polymerization followed by entrapment of Cu(II) ions into the synthesized polymer network and their subsequent reduction to copper nanoparticles. The poly(acrylic acid) graft chains not only act as templates for incoming Cu(II) ions but the three-dimensional cross-linked grafted network, so formed, also provides them enough space for accommodation.
Experimental
Materials
Cotton fibers were purchased from a local textile mill (Indore, India) and were used as received without chemical treatment. The monomer acrylic acid (AAc), cross-linker N, N-methylene-bisacrylamide (MB), and initiator potassium permanganate were purchased from Hi Media Chemicals, Mumbai, India and were analytical grade. The copper salt, namely copper chloride, and other chemicals were also purchased from Hi Media Chemicals, Mumbai, India and were analytical grade. The double distilled water was used throughout the investigations.
Method
Preparation of poly(AAc)-g-fibers
The graft co-polymerization of acrylic acid onto cellulose cotton fibers was carried out using KMnO4–HNO3 system [21]. In a typical experiment, 5 g of cotton fibers was immersed in 100 ml KMnO4 solution (1% w/v) in a conical flask for 30 min at 50℃ with a continuous shaking to ensure the homogeneous soaking of MnO4 over the cotton fibers. After this treatment, the fibers were washed with distilled water several times to remove the excess of KMnO4 solution. They were then squeezed between two filter papers before being introduced into the polymerization solution. The treated fibers were placed in a round bottom flask which contained 130 ml of 0.1 N HNO3 solution, 15 g of monomer acrylic acid, and 0.5 g of cross-linker MB. The fibers were refluxed for 45 min at 60℃ and then taken out, washed extensively with de-ionized water to remove unreacted salts, and finally washed with 50% (v/v) aqueous solution of methanol to remove the homo-polymer formed. The fibers were dried in electric oven (Tempstar, India) till they attained constant weight. The percent grafting (PG) was calculated using the following formula
Preparation of Cu nanoparticles loaded poly(AAc)-g-fibers
The predetermined quantity of grafted fibers was placed in aqueous solution of Cu(II) ions of known concentration for a period of 4 h which was found to be a sufficient time to attain equilibrium loading. The Cu(II)-loaded fibers were then placed in aqueous solution of NaBH4 (1%) for a period of 6 h. Finally, the fibers were taken out and dried in an electric oven at 45℃ till they attained constant weight. The black color of resulting fibers indicated the formation of copper nanoparticles into the grafted polymer network.
Characterization of fibers
The FTIR spectra of plain and poly(AAc)-g-fibers were recorded with an FTIR spectrophotometer (Shimadzu, 8400 S Columbia, USA) using KBr. For this, fibers were cut into a number of small pieces and mixed with KBr. The scans recorded were the average of 100 scans and the spectral range was 400–4000 cm−1.
Thermogravimetric analysis (TGA) of plain and grafted fibers was performed using a thermogravimetric analyzer (Mettler, Teledo TGA/SDTA 851, Switzerland). A definite quantity of fibers was placed in a ceramic crucible and analyzed over the temperature range of 30–600℃ at the heating rate of 10℃/min, under the flow of N2 gas at the rate of 30 ml/min. The initial weight of plain and grafted fibers was 15.12 and 40.20 mg, respectively. In order to investigate the surface morphology of plain and poly(AAc)-g-fibers, scanning electron microscopy (SEM) images were recorded with a Hitachi S-4700 (New Jersey, USA) operating at an acceleration voltage of 15 kV. All samples were dried in vacuum at room temperature and coated with gold before scanning. Surface morphologies were imaged at different magnifications. The size and distribution of copper nanoparticles within the fibers was determined using a JEOL 1010 Transmission Electron Micrograph and were taken as a 200 nm thick microtome section, cut parallel to the surface of the fabric.
Dynamic swelling and diffusion measurements
The swelling behavior of grafted fibers was studied gravimetrically [22]. The preweighed fiber sample was placed in 500 ml of distilled water at 37℃ and it was taken out at different time intervals, wiped superficially with tissue paper to remove the extra surface water, weighed accurately in an electronic balance (Denber, Germany), and then placed back in water. The percent water uptake (PWU), obtained at different time intervals was determined using the following expression
Cu(II) release study
The release of Cu(II) from the grafted fibers was monitored by using method proposed by Singh et al. [23] with a little modification. The preweighed quantity of copper nanoparticles loaded poly acrylic acid grafted (CNLPAA)-g-) fibers were placed in 25 ml of release medium (i.e. physiological fluid) at 37℃. After definite time intervals, fibers were transferred into fresh release medium, and the amount of Cu(II) ions released was determined spectrophotometrically at 488 nm using the method reported elsewhere. The quantity of Cu(II) ions released from fibers was calculated using Lambert–Beer’s plot obtained for copper solutions of known concentrations. The ratio of volume of release medium to mass of fibers was maintained at a constant value of 50 ml/mg.
Antimicrobial studies
A total of 1 g of agar and 2.8 g of nutrient agar was mixed in 100 ml of water in a sterile conical flask and stirred for 15 min to ensure complete dissolution. The mouth of the flask was covered with cotton plug and foil, and the flask was autoclaved. The media was poured into sterilized Petri plate and allowed to get solidified which took almost 15 min. The bacterial culture was inoculated and speeded homogeneously over the media. Around 0.1 g of Cu loaded grafted fibers were put in the center of the Petri plate and the Petri plate was incubated for a period of 24 h in an incubation chamber. Finally the Petri plate was taken out and observed for zone of inhibition. The plate, supplemented with plain fibers, was used as control set.
Results and discussion
Preparation of grafted fiber
It is logical to assume that MnO particles are deposited uniformly all over the cotton cellulose and are located at certain sites, most probably at the hydroxyl groups of cotton cellulose. Once the accessible hydroxyl groups are occupied by MnO particles, extra deposition of MnO will be difficult and, if deposited, the particles will be easily removed during washing.
In the presence of an acid HNO3, primary radical species formation occurs as a result of the action of HNO3 on the MnO2 deposited as follows
Cotton cellulose macro radicals may also be formed by direct attack of Mn4+ or Mn3+ ions on the cellulose molecule via abstraction of the hydrogen atom
In the presence of a vinyl monomer, the cellulose is added to the double bond of the vinyl monomer, resulting in the formation of covalent bond between monomer and cellulose with further generation of free radical on the monomer, i.e. a chain is initiated. Subsequent addition of monomer molecules to the initiated chain propagates the grafting reaction onto cellulose fibers. Finally, the propagating chains are terminated through the recombination of two growing chains.
FTIR spectroscopy analysis of fibers
The FTIR spectra of cellulosic fibers and poly(acrylic acid)-g-cellulosic fibers are shown in Figure 1. The grafted sample shows abroad band at 3400–3600 cm−1 which may be attributed to sum of contributions from hydrogen bonded –OH stretching of cellulose as well as hydrogen bonded –OH from carboxylic groups of polyacrylic acid and –NH from amide groups of cross-linker MB. In addition, the grafted sample also contains typical signals of cellulose backbone (–CH 1431 cm−1, –COC 1059 cm−1, –β linkage 890 cm−1). The grafted sample exhibits a sharp peak at 1734 cm−1 attributable to the –C = O stretching vibrations from carboxylic groups of poly(acrylic acid) P(AAc). Moreover, there is also a sharp peak at 1030 cm−1 for –C–CHO stretching, thus confirming the formation of aldehyde group in cellulose network, the initiator during the grafting process. It is to be noted that the percent transmittance, observed in the spectra, is quite low. This could probably be due to the fact that greater quantity of polymer was taken in sample preparation for FTIR spectral analysis.
FTIR spectra of plain and poly(AAc)-grafted fibers.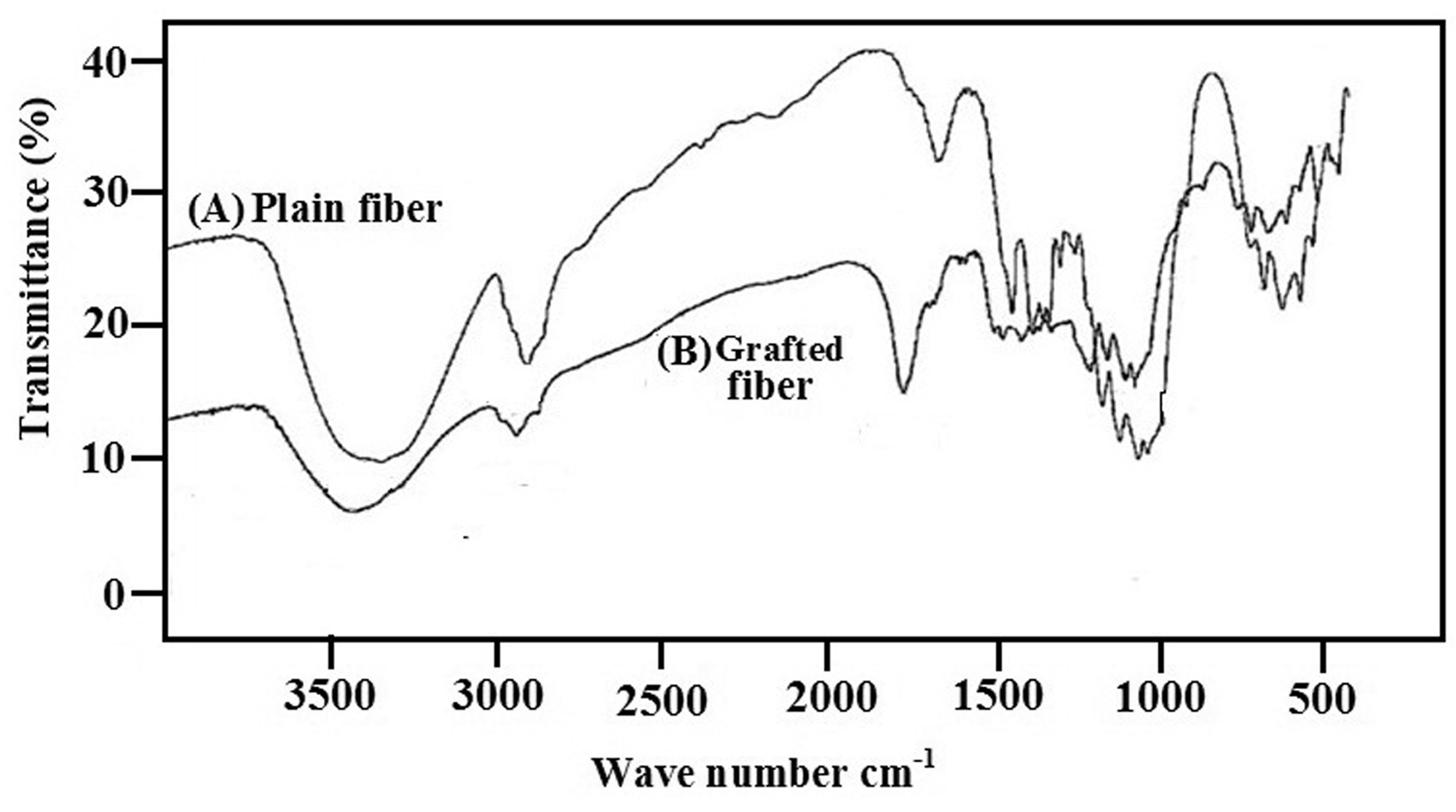
Preparation of CNLPAA-g-fibers
When poly(acrylic acid)-g-cotton fibers are placed in aqueous solution of Cu(II) ions, these ions enter into the grafted polymer network owing to the ion-exchange process between H+ ions of polymer network and Cu++ ions present in the external solution. These incoming copper ions enter into the polymer network and are bound to the highly electronegative O atoms present in the grafted poly(AAc) chains. In addition, the O atoms of –OH groups of cellulose also act as templates for these Cu(II) ions. When these copper ions loaded P(AAc)-grafted fabric is put in the sodium borohydride solution, these Cu(II) ions are reduced to Cu nanoparticles. The optical photograph of plain, P(AAc)-grafted and Cu nanoparticles loaded polymer grafted fibers are shown in Figure 2(a) to (c).
Optical photograph of (a) plain, (b) poly(AAc)-grafted, and (c) Cu nanoparticles loaded polymer-grafted fiber.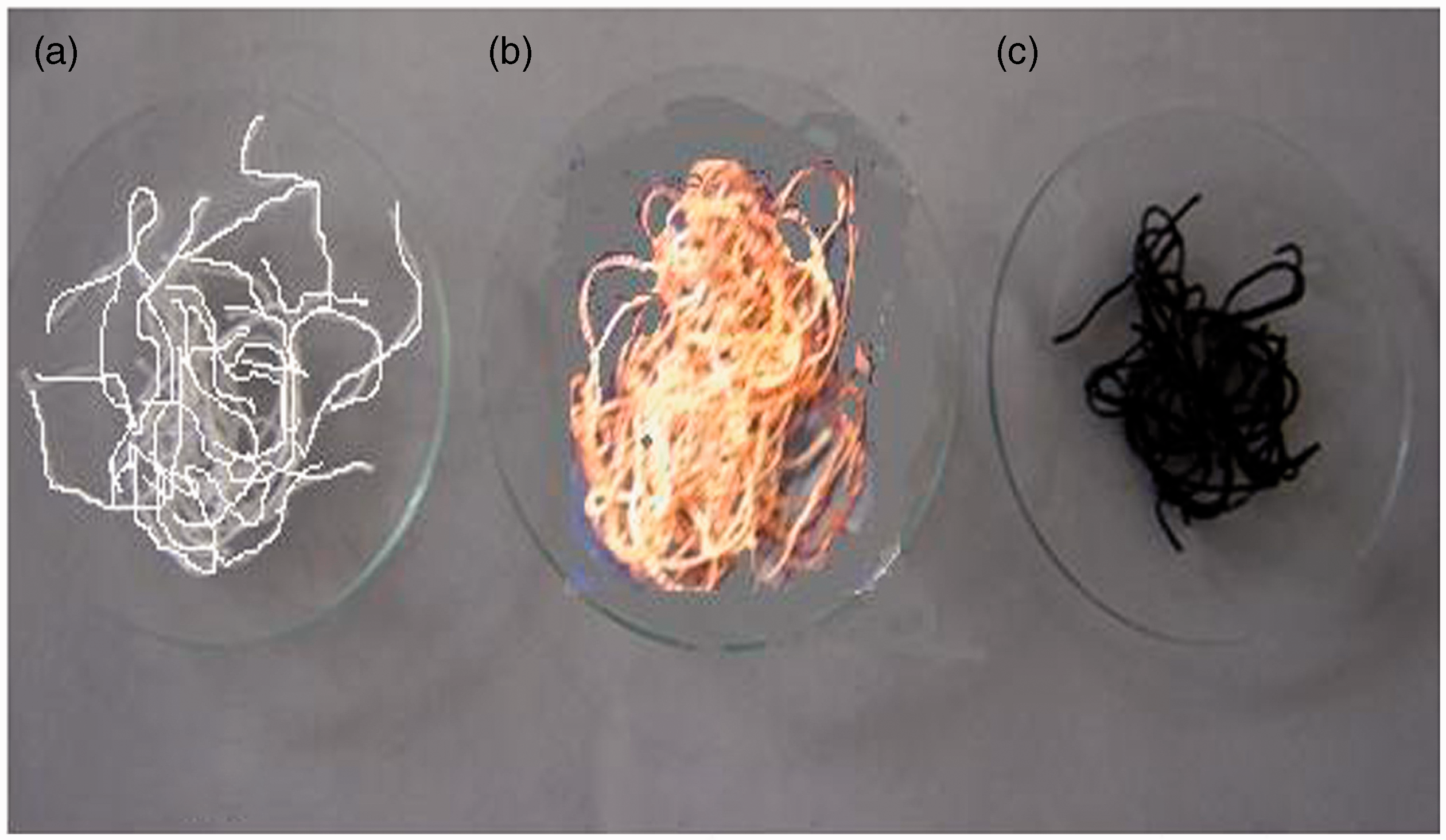
Characterization of poly(AAc)-g-cotton fibers
Thermo gravimetric analysis
The thermograms of plain and poly(AAc)-grafted cotton fibers are shown in Figure 3, respectively. The plain cotton fiber shows a fair thermal stability up to 250℃ and then loses its weight drastically up to 400℃ through the formation of levoglucosan and other volatile compounds [24], thereby showing a percent weight loss of nearly 90%. The total weight loss suffered by cotton fibers up to 600℃ is nearly 94%. The thermogram of poly(acrylic acid)-grafted cotton fibers shows almost different behavior. It shows three-step degradation, with initial decomposition in the range of 50–150℃ attributable to the loss of physically absorbed water. It may be mentioned here that presence of poly(AAc) macromolecular chains on the surface of fibers makes the surface more hydrophilic as polar-COOH groups of poly(AAc) chains have strong affinity for water. Hence, removal of the strongly absorbed water is responsible for the weight loss in the temperature range of 50–150℃. The second degradation starts from 150℃ to nearly 310℃. It has been reported [25] that the second degradation is associated with loss of water molecules through the formation of intra- and intermolecular anhydride linkages and also due to de-carboxylation of a fraction of –COOH groups. Finally, the third stage of degradation occurs in the temperature range of 310–550℃ which involves the decomposition of polymer with the elimination of CO and CO2 through abundant backbone scission. However, these three stages are not so enhanced due to presence of cotton cellulose in the main backbone of grafted poly(AAc) networks. The total weight loss of the grafted cotton fibers up to 650℃ is nearly 80%. The thermal behavior of Cu nanoparticles loaded grafted fibers is also shown in Figure 2(c). It can be seen that total loss suffered by these fibers up to 650℃ is almost 63% which is lowest of all three types of fibers, namely plain, poly(AAC) grafted and Cu nanoparticles loaded grafted fibers. This, in some way, confirms the presence of metallic copper in the fibers. It is also noteworthy that there is rapid weight loss in the thermogram of Cu nanoparticles loaded grafted fibers around 100℃. This is very unusual and it might be due to insufficient drying of fiber sample before running it for the Thermogravimetric Analysis (TG) analysis.
Thermograms of plain and poly(AAc)-grafted cotton fibers.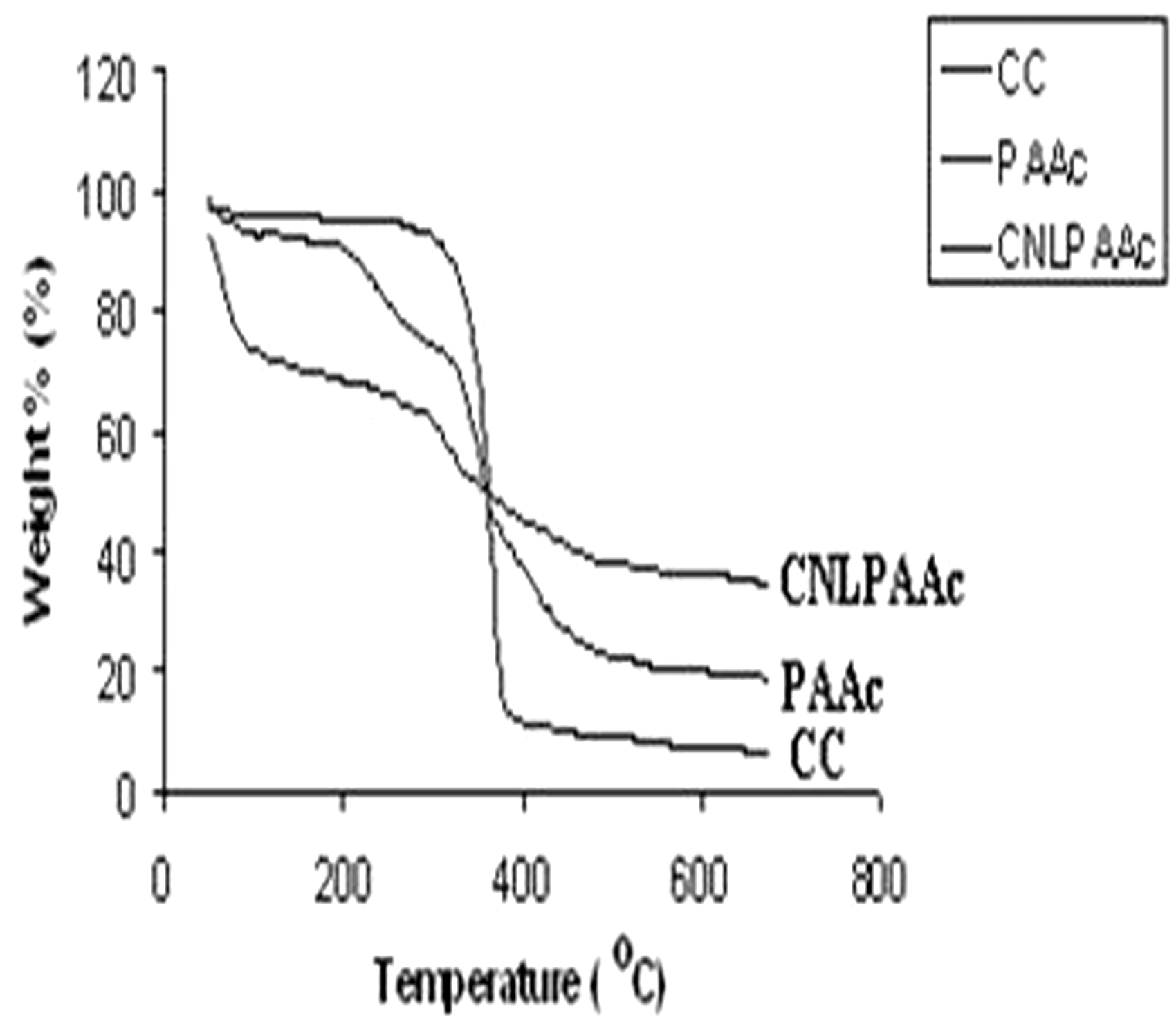
SEM
The SEM is a significant tool to analyze the surface texture and morphology of the surface of a substrate. In addition, it is also used to provide visual evidence in support of grafting onto a polymeric substrate. The SEM images of plain and poly(AAc)-g-fibers are shown in Figure 4(a) and (b), respectively. It can be seen clearly that there has been drastic change in the surface texture of grafted fibers as compared to the plain fibers. The plain cotton fibers have smooth surface while that of grafted fibers exhibits roughness throughout due to the formation of polymer skin layer. In addition, there is noticeable increase in the thickness of the fibers due to polymer grafting. It may probably be indicative of coating of cross-linked poly acrylic acid network onto the cotton fibers surface. Moreover, the grafting appears to be not so uniform.
SEM images of (a) plain and (b) poly(AAc)-g-fibers.
TEM image of copper nanoparticles
The results of the TEM analysis are shown in Figure 5. The image clearly reveals an almost uniform distribution of copper nanoparticles throughout the poly(AAc)-grafted cotton fibers. The particle size distribution curve (see inset) was obtained by measuring size of individual particles arbitrarily selected from different areas of image. On the basis of the distribution curve obtained, it was found that nearly 45% particles had an average diameter of 60–80 nm.
TEM image of copper nanoparticles loaded fibers (particle size distribution in inset).
Swelling behavior of poly(AAc)-g-fibers
The formation of graft chains of poly(AAc) along the cellulosic backbone renders pH-dependent water absorption properties to the resulting fibers. As a result, the polymer-grafted fiber is expected to exhibit a pH-dependent swelling behavior. To confirm this, the dynamic water uptake of the plain and poly(AAc)-g-fibers was investigated in the physiological buffers of pH 1.0 and 7.4 at 37℃ under sink conditions. The results are shown in Figure 6. It can be observed well that grafted fibers exhibit a percent swelling of nearly 236 and 887 in the media of pH 1.0 and 7.4, respectively. The observed low water uptake in the medium of pH 1.0 may be explained as follows: In acidic pH the –COOH groups of poly(AAc)-grafted chains do not undergo any ionization and remain in almost unionized state. This results in formation of H-bonding interactions between these –COOH groups thus serving as additional cross-links. Hence, the poly(AAc)-g-fiber network acquires a complex and compact structure, thus not permitting much water molecules to enter into the polymer network. This results in low water absorption by these grafted fibers. However, in the medium of pH 7.4, the fibers demonstrate appreciable water uptake of nearly 887%. This can be attributed to the fact that the –COOH groups, present along the macromolecular chains undergo ionization in the pH 7.4 to yield negatively charged –COO− groups along the macromolecular chains. These groups repel each other and cause the polymeric chains to relax or unfold, thus allowing more and more water to enter into the networks. This results in an enhancement in the percent swelling. On the other hand, plain fibers absorb water owing to the presence of abundant –OH groups and ultrafine pores within the fiber networks. As there is no any strong driving force, the water absorption is relatively low.
The dynamic uptake of water of the plain and poly(AAc)-g-fibers in the medium of pH 1.0 and physiological fluid of pH 7.4 at 37℃.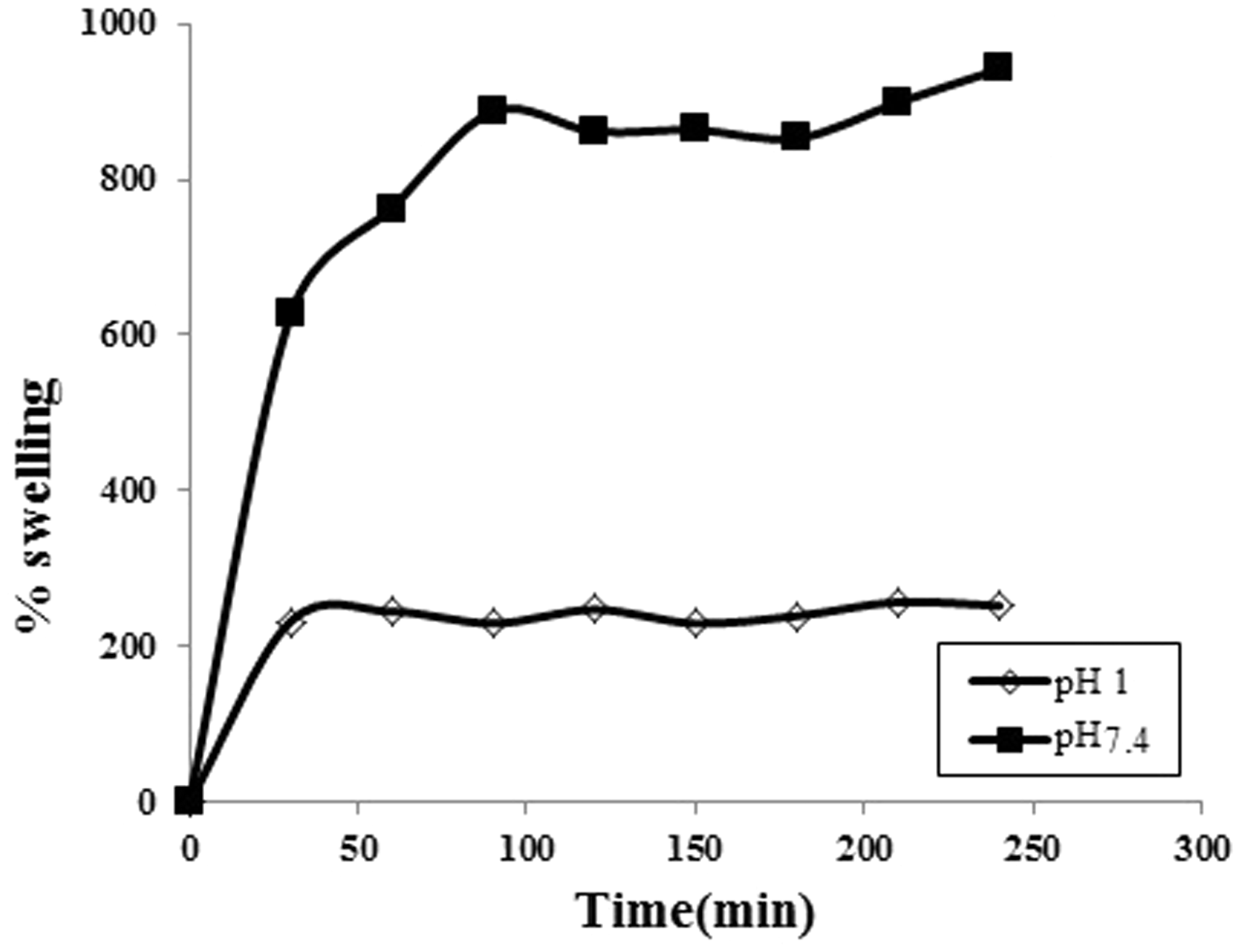
In order to see the effect of presence of Cu(II) in the poly(AAc)-g-fibers, the plain fibers, the grafted fibers, and the fibers equilibrated in 2% Cu(II) solution were allowed to equilibrate in phosphate buffer of pH 7.4 for a period of 24 h. The results are shown as bar diagram in Figure 7. It can be seen that the Cu(II)-loaded poly(AAc)-g-fibers show minimum percent swelling of 153 while plain and poly(AAc)-g-fibers demonstrate percent swellings of nearly 500 and 880, respectively. Here, it is worth mentioning that as a result of graft co-polymerization, the cross-linked poly(AAc) network is produced on the fibers surface only. However, the cross-linked polymer network makes an appreciable contribution toward the overall swelling of the fibers. The minimum swelling, shown by Cu(II) loaded grafted fibers is attributable to the ionic cross-linking caused by Cu(II) ions with the –COO− groups of poly(AAc) graft chains. The additional cross-links, offered by Cu(II) ions, render more compactness to the grafted polymer network, thus resulting in minimum water absorption. It can also be said that in the case of Cu(II)-loaded grafted polymers, the relaxation of poly(AAc) chains is not so prompt or it is overcome by the physical cross-links provided by Cu++ ions and negatively charged –COO− groups of polymer chains. Similar type of cross-linking of poly(acrylate) by Cu(II) ions has been reported [26].
The equilibrium water uptake of plain fibers, the grafted fibers, and the fibers equilibrated in 2% Cu(II) solution, in the physiological fluid of pH 7.4 at 37℃.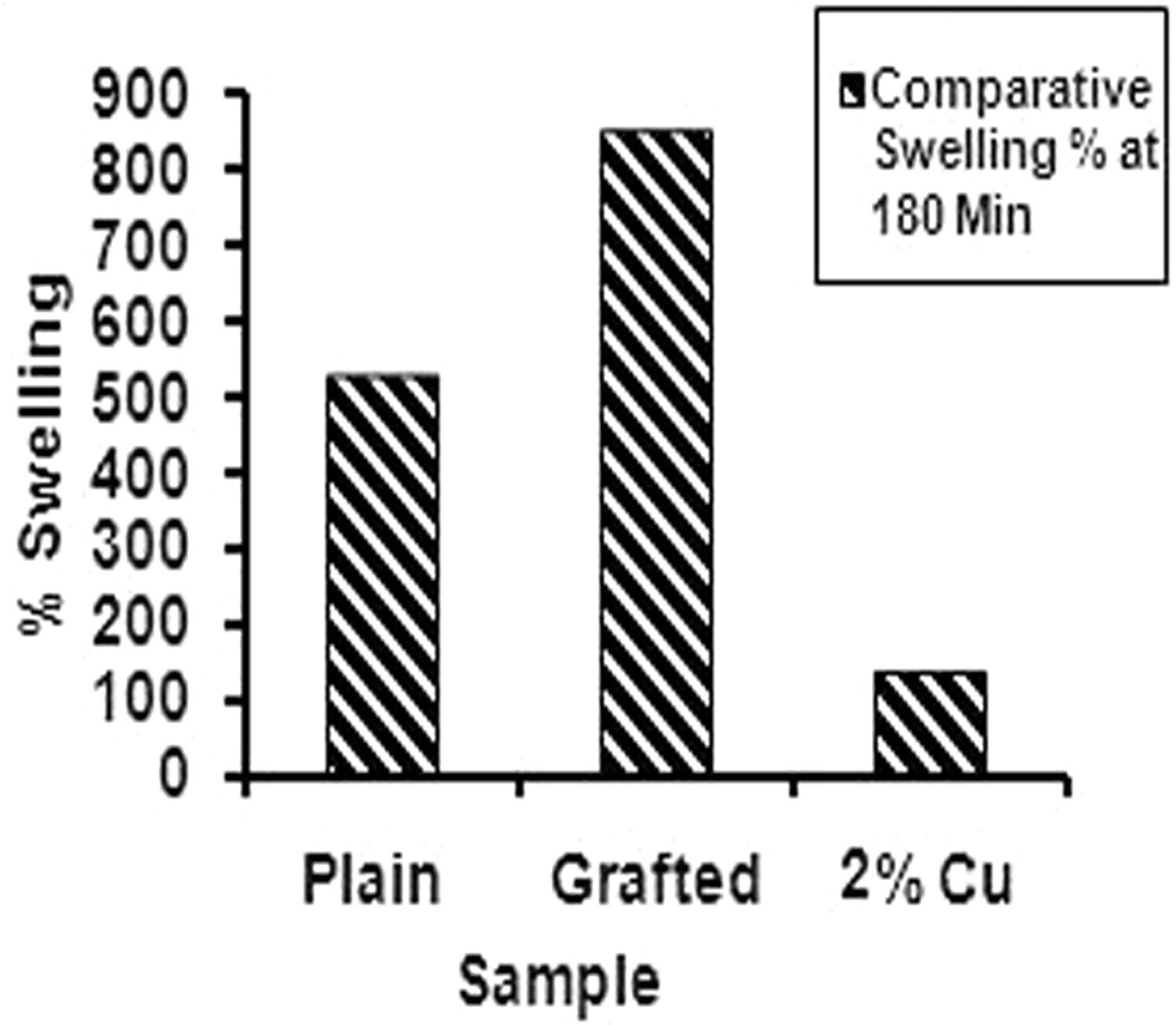
Cu(II) release studies
While investigating the release of an antibacterial drug from a matrix that is intended to be used as wound dressing, the nature of release medium is very important. The composition of wound fluid depends upon the nature of the wound. For example, Trengrove et al. [27] reported that wound fluid collected from leg ulcers contained 0.6–5.9 mmol/l glucose and 25–51 g/l protein. Similarly, Bonnema et al. [28] analyzed serum fluid formed after auxiliary dissection and reported that on the first operative day the drainage fluid contained blood and high concentration of creatine-phosphokinase while after day 1, it changed to lymph like fluid that contained different cells and more proteins. Therefore, looking to the variation in nature of wound fluid, we carried out in vitro release study in physiological fluid (PF) as suggested by British pharmacopeias. The PF contained 142 mmol/l of NaCl and 2.5 mmol/l of CaCl2.
In order to investigate the dynamic release of Cu(II) ions from the Cu(II)-loaded grafted fibers, we took 86.3% grafted poly(AAc)-g-fibers and equilibrated them in the Cu(II) aqueous solutions, having percent (w/v) concentrations of 0.5, 1.0, and 2.0. Finally, the Cu(II) loaded poly(AAc)-g-fibers were put in aqueous solution (1%) of sodium borohydride to reduce Cu(II) into Cu nanoparticles. These fibers, designated as CNLPAA-g-(0.5), CNLPAA-g-(1.0), and CNLPAA-g-(2.0), were put in a definite volume of PF to monitor the release of Cu(II) ions spectrophotometrically [29]. The kinetic run is shown in Figure 8. It is clear that as the time increases, amount of Cu(II) ions released also increases. In addition, for a given time the quantity of Cu(II) released increases with the concentration of loading solution. It was found that the release of copper ions continued for a total period of almost 50 h.
The dynamic release of Cu(II) from the fiber samples CNLPAA-g-(0.5), CNLPAA-g-(1.0), and CNLPAA-g-(2.0) physiological fluid of pH 7.4 at 37℃.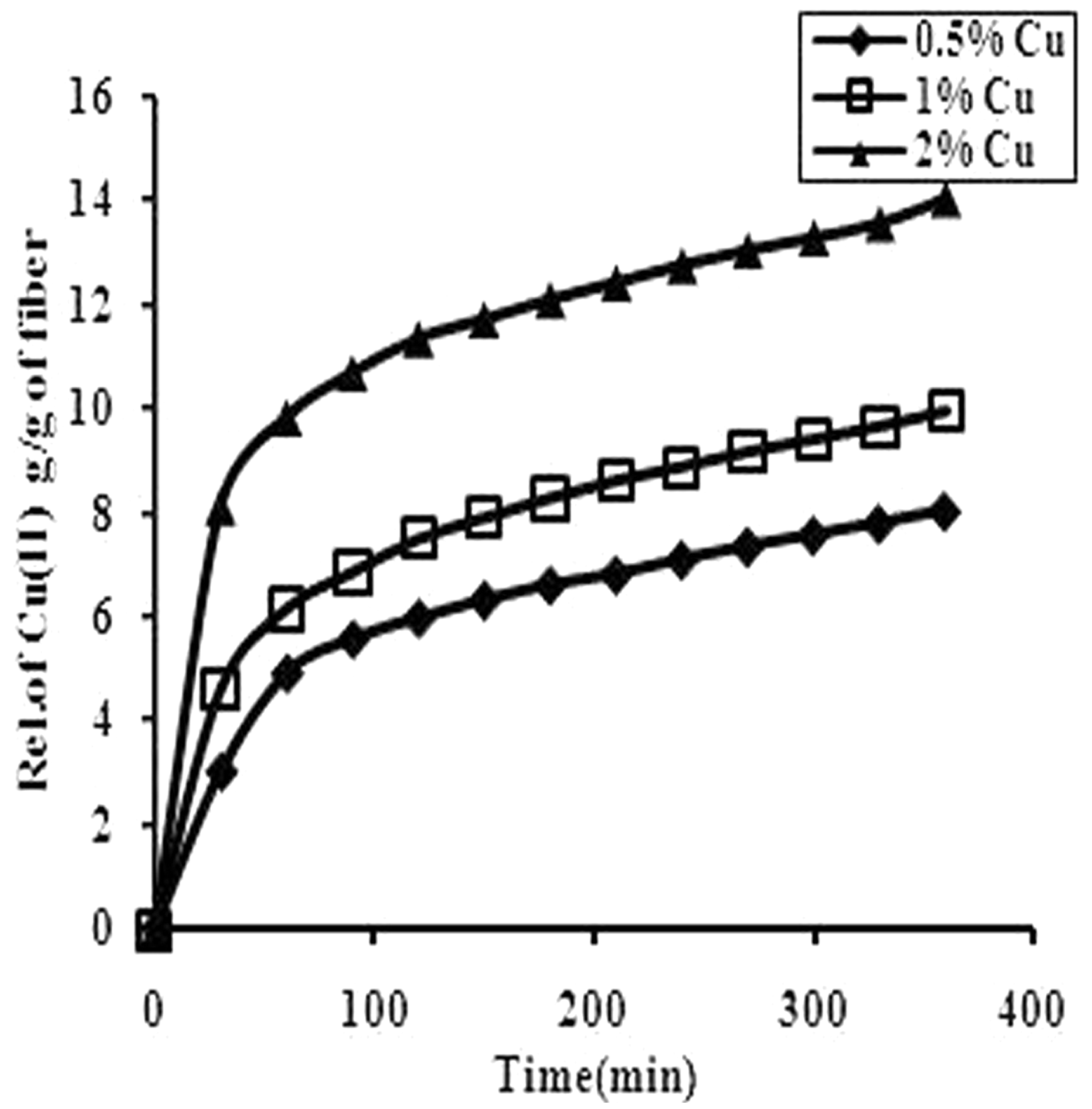
Analysis of Cu(II) release data
The release of Cu(II) from the polymer-grafted network is a swelling-dependent phenomenon. A critical analysis of the swelling process reveals that two underlying molecular processes act. First is the penetration of the solvent molecules into the void spaces in the network and the second one is the subsequent relaxation of the polymeric chains. The fundamental equation used to describe the water penetration mechanism is [30]
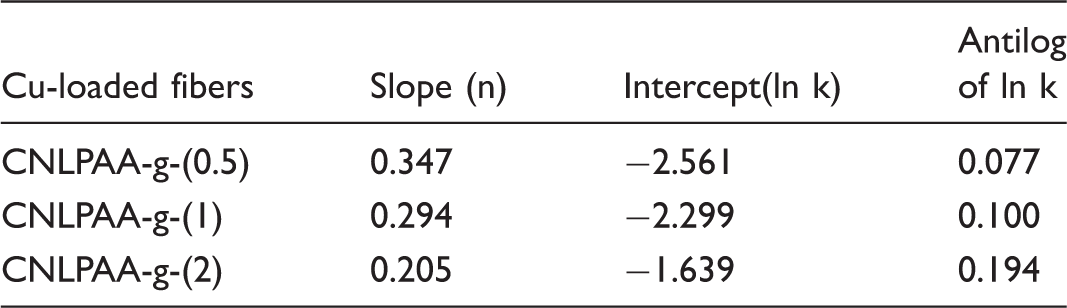
ln (1–Mt/M∞) versus t for the Cu(II) release data obtained from various copper loaded fibers. Release exponent n and gel characteristic constant k for Cu(II) release data obtained for different fibers.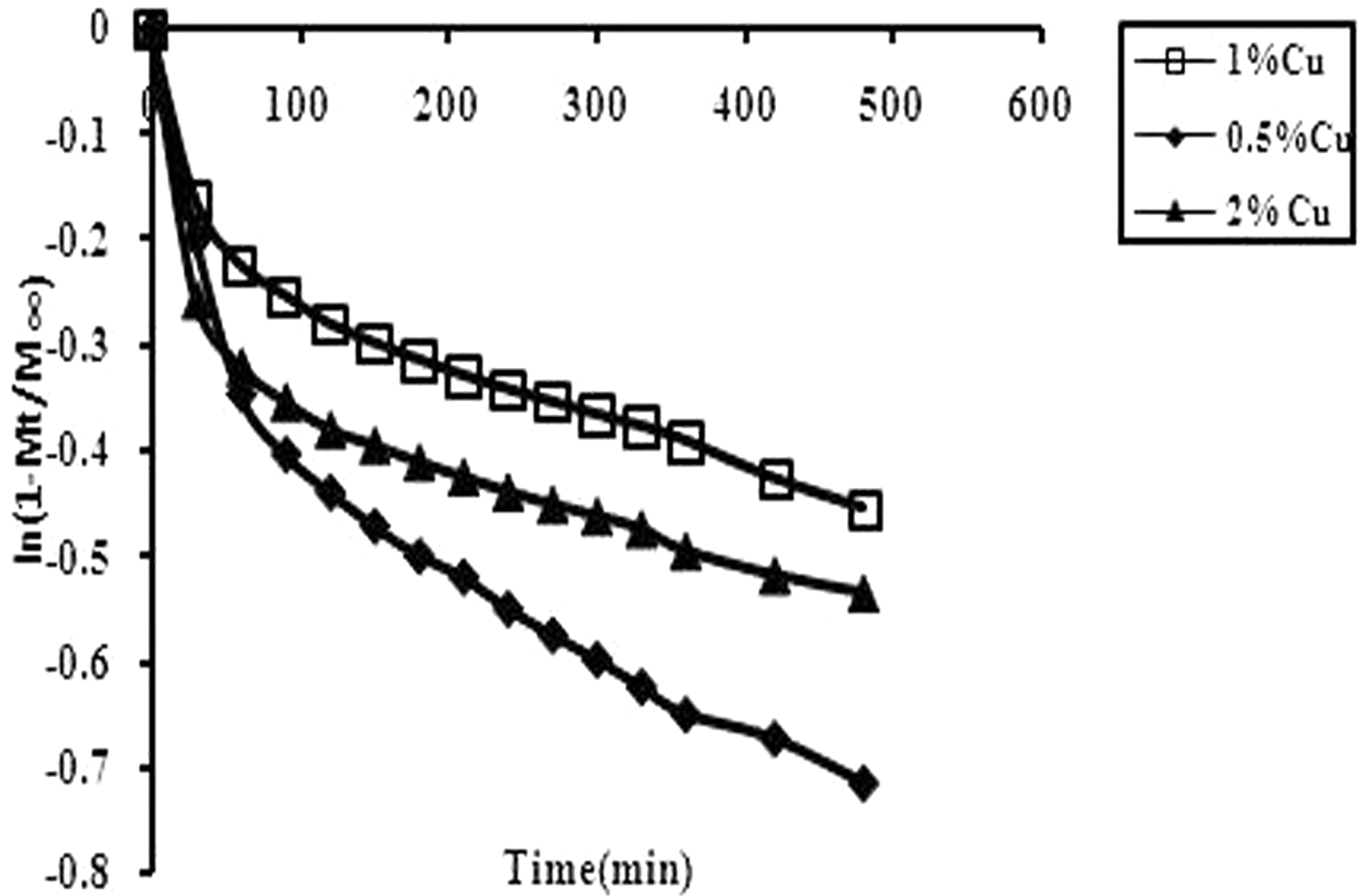
The kinetic Cu(II) release data were also applied on the first-order kinetic equation, developed by Beren and Hopfenberg [31], according to which the rate of drug releases at any time is proportional to the first power of the amount of drug to be released till equilibrium, i.e.
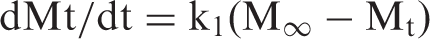
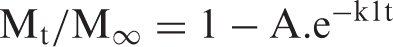
The logarithmic form of above equation is

A graph, plotted between ln (1–Mt/M∞) and t, is shown in Figure 10. A close look at the three different plots, obtained for release data from the Cu(II) loaded grafted fiber samples reveal that each plot exhibits biphasic linearity. The first initial portion passes through the origin while after a time interval of nearly 90 min there is a drastic change in the slopes of the linear plots. Smith and Fisher [32] have also reported such unusual behavior for water absorption by melamine–formaldehyde resins. It may be because initially the solvent approaches the Cu(II) loaded fibers from all sides thus allowing a faster swelling followed by Cu(II) release from the polymer-grafted network. However, as the solvent fronts meet, the fully swollen/hydrated grafted polymer network does not possess a sharp concentration gradient of dissolved Cu(II) species and therefore the release gets slower as indicated by the sudden change in the linear slopes for all fiber samples.
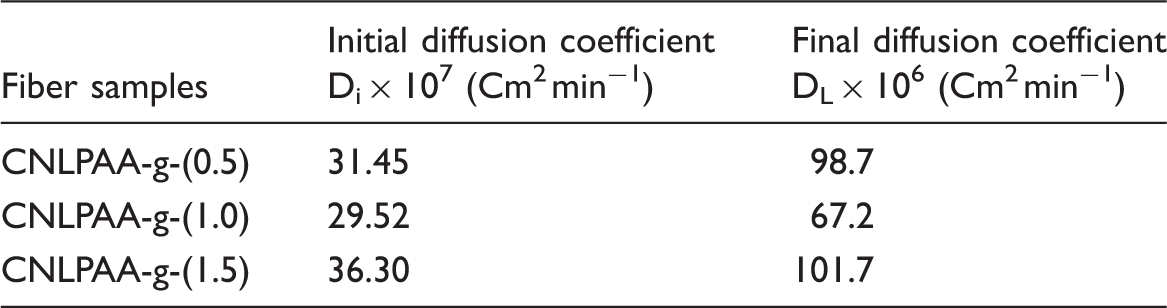
Evaluation of late time diffusion coefficient DL from the slopes of later portions of linear plots.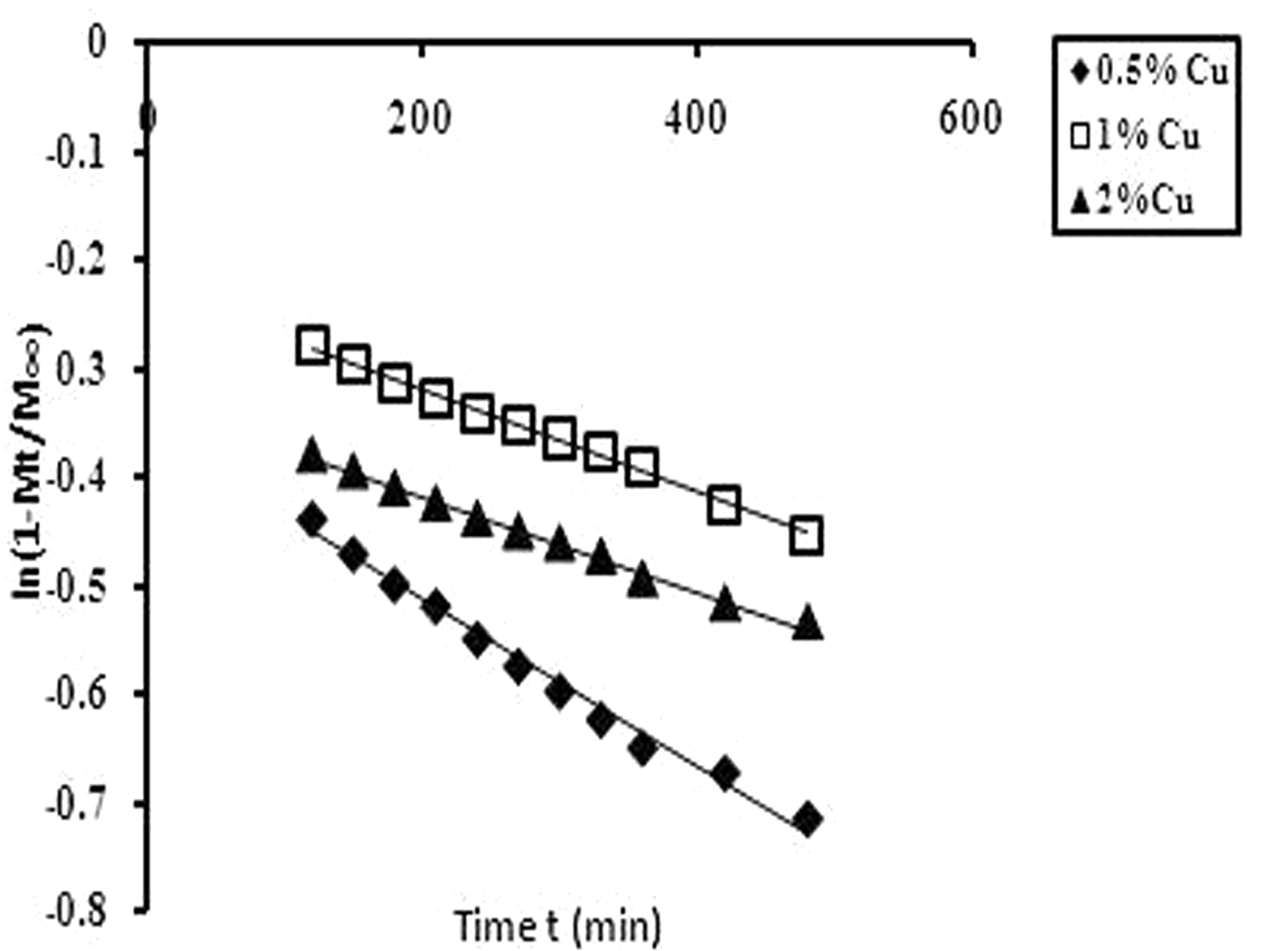
Finally, assuming that the Cu(II) release rate at any time is directly proportional to the quadratic of the quantity of the copper ions to be released before the attainment of equilibrium state, the second-order equation, proposed by Schott [33] was applied
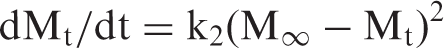
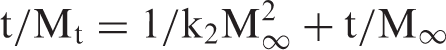
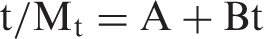
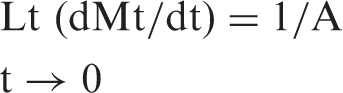
Therefore, the intercept A is reciprocal of initial release rate. In order to apply second-order model on the kinetic data obtained for all the three copper loaded fiber samples in the medium of pH 7.4, plots were obtained between t/Mt and t as shown in Figure 11. It can be seen that the plots obtained are linear with fair regression values. This suggests that second-order kinetic release model is quite successful in interpretation of the release data. All the parameters obtained with second-order model are given in Table 2.
t/Mt versus t plots for Cu(II) release from different CNLPAA-g fibers in the physiological fluid of pH 7.4 at 37℃. Parameters associated with second order kinetic model.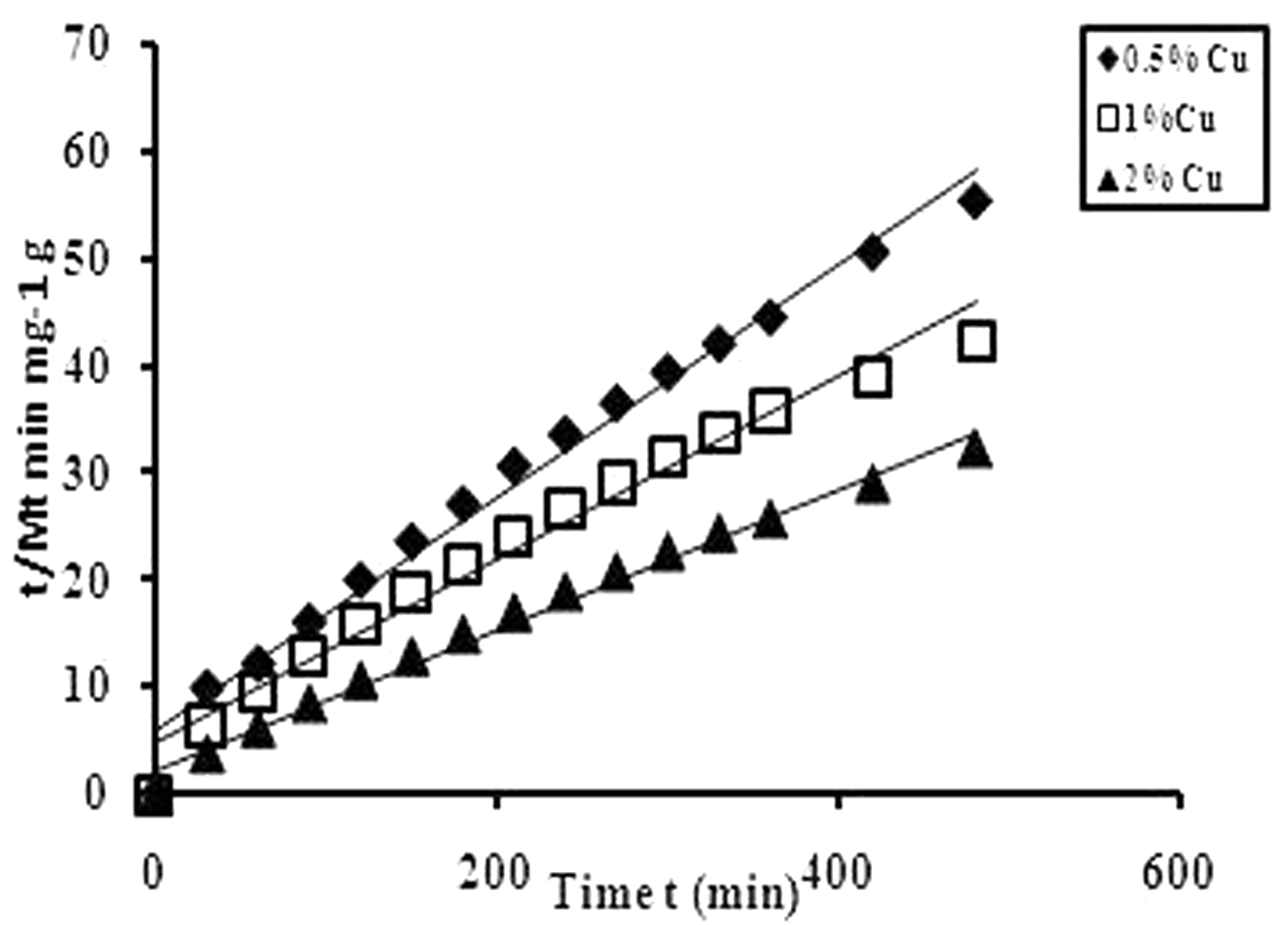
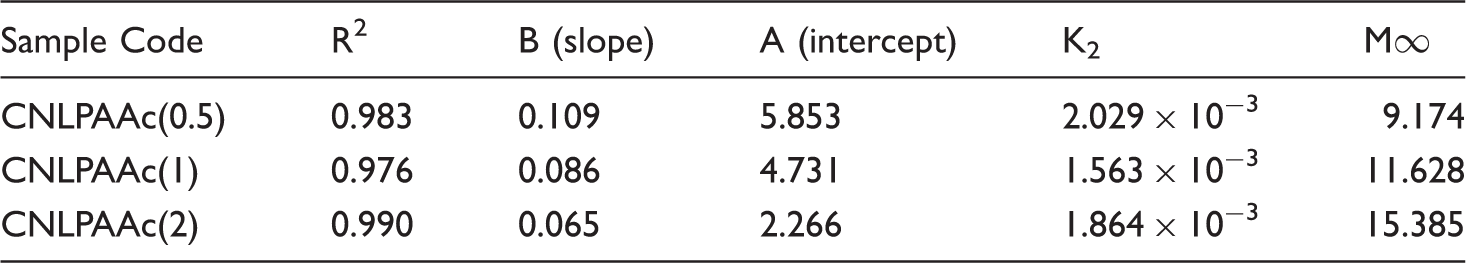
Most of the diffusion processes are best interpreted by the Ficks first and second law of diffusion. The initial diffusion coefficient DI gives an idea about the release of entrapped ingredient from the matrix in the initial stage. In order to determine DI, the following equation was employed [34]
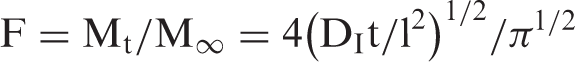
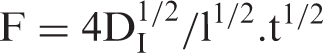
The slope of linear plot between F and t1/2 was used to calculate Di as

The initial 60% of the dynamic release data were used to draw linear plots between ln F and t1/2 (data not shown). The slopes of the linear plots were used to calculate initial diffusion coefficients Di.
In order to calculate the average diffusion coefficient Dave, F = 0.5 and t = t1/2 were substituted in equation (12), presuming that 50% of the total release, attained by fibers, can enable us to evaluate Dave. The above substitutions yielded following expression
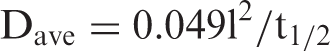
To calculate Dave, the dynamic release data were used to evaluate the time required for attainment of 50% of the total Cu(II) release from various fiber samples. When a polymeric fiber is placed in swelling medium, the solvent molecules penetrate from all sides into the outer circumference and cause the outer part to swell. There can be observed a sharp division between the relaxed, swollen region and interior dry region. In relation to this phenomenon, an interesting observation was made by Shankar [35] who claimed that a plot between the ln (1–Mt/M∞) and “t” gives a linear plot but with a distinct break in the slope. This is observed experimentally too for both Fickian and non-Fickian diffusion. The break in the slope corresponds to the time when the two advancing boundaries meet each other. Using the slope of the later part, it is possible to calculate DL. The late time diffusion coefficient DL was determined using later 60% of the total release data for various fiber samples. Using this data, the ln (1–Mt/M∞) values were plotted against t for various fiber samples. The plots obtained were bi-phasic linear plots. The slope, obtained for the later part of the swelling process, was used to calculate DL using the following expression [34]
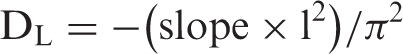
Diffusion coefficients for the dynamic release of Cu(II) from the copper nanoparticles loaded fibers.
Antibacterial studies
Copper is widely known as broad-spectrum biocide. Various forms of copper, such as zero-valent copper, copper oxide, ionic copper, and copper containing molecular complexes have all shown antimicrobial properties [36]. In contrast to low sensitivity of human tissues to copper, prokaryotes (i.e. bacteria) are extremely susceptible to copper [35]. The US Environmental Protection Agency have confirmed antimicrobial efficacy of copper against E. coli O157:H7, S. aureus, Enterobacter aerogenes, and Pseudomonas aeruginosa [37].
In this work, we prepared two copper nanoparticles loaded grafted fiber samples by equilibrating in Cu(II) solutions of concentrations 1 and 2% followed by borohydride reduction. These samples were designated as (CNLPAA)-g Antibacterial action against model bacteria E. coli.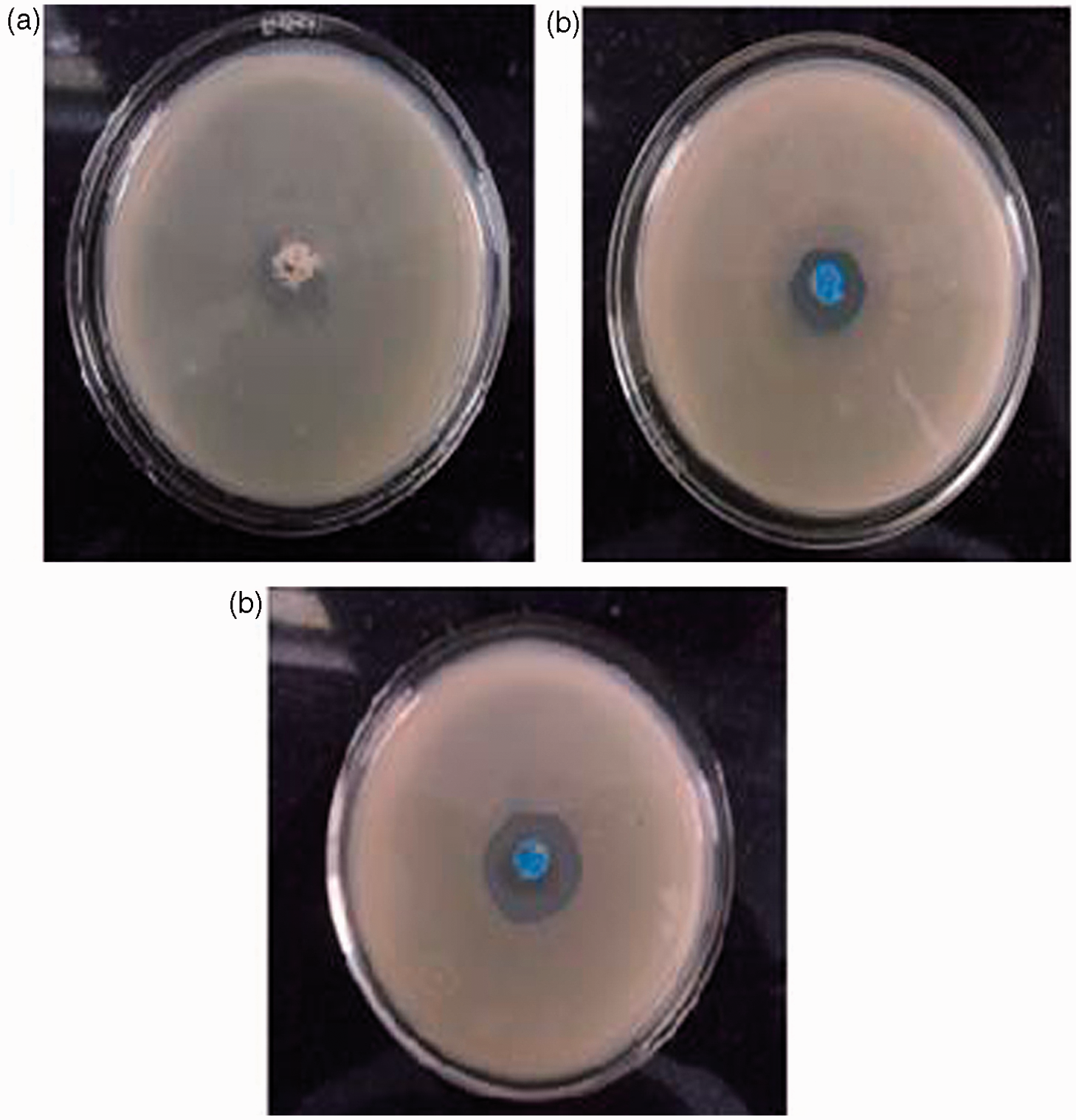
Conclusions
The above study concludes that Cu nanoparticles can be conveniently loaded into poly(AAc)-grafted cotton fibers at a desired level of concentrations. The ion-exchange process between Cu(II) ions from the loading solution and H+ ions from the grafted polymer network appears to be responsible for the entrapment of Cu(II) ions into the grafted fibers. The release of copper ions from the polymer network was observed for more than 50 h. The release data are best interpreted by second-order kinetic model. The fibers show fair antibacterial activity against model bacteria E. coli. The copper nanoparticles loaded poly(AAc)-g fibers seem to have great potential to be used for wound dressing applications. However, still it is required to carry out a detailed investigation of cytotoxic studies of these fibers to further ensure their suitability for wound management.
Footnotes
Acknowledgements
The authors are thankful to Dr O.P. Sharma, Head of the Department of Chemistry, Govt Model Science College Jabalpur, M.P., for his kind and unconditional support. We are also thankful to IIT Mumbai for the analysis of samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.