Abstract
Ethylene is recovered from ethylene/ethane mixture by adsorptive separation on CuCl2 loaded activated carbon. When 36.7% of CuCl2 were dispersed on activated carbon made from coconut shell (CSC) and calcined at 400℃ for some hours, a satisfactory adsorbent sample was prepared. The adsorption capacity of ethylene reached 23.55 mg/g and ethylene/ethane separation factor reached 7.6. Adsorbent characterization by various modern instruments demonstrated the reduction of CuCl2 to provide Cu+ cation on CSC, which is responsible to form π-complexation bonds for the adsorption of ethylene. Mechanism was discussed. Nature of related active centers was suggested.
Introduction
Ethylene is one of the most important raw materials for the production of many petrochemicals, including plastics, rubbers and textiles. Therefore, its demand is rapidly increasing in the latest decades. Ethylene is now produced mainly by steam cracking of naphtha from crude oil or decomposition of ethane from natural gas. In order to save these raw materials, recovery of ethylene from dry gas of fluid catalytic cracking (FCC) and delayed coking units in a refinery is also under consideration. In all of the above technologies, ethylene and ethane separation play an important role in the preparation of polymer-grade ethylene. Cryogenic distillation is known to be a conventional and reliable measure for this separation. However, it is also energy-intensive due to the close relative volatilities between ethylene and ethane. A number of potential alternatives have, therefore, been investigated, among which adsorptive separation is commonly considered to be one of the most potential approaches. Zeolite has specialized regular channel dimension and polarity, and can be also modified by ion exchange, therefore, it is often considered to be an ideal adsorbent for the separation of ethylene and ethane as reported in the literature (Meng et al., 2010; Mofarahi and Salehi, 2013; Narin et al., 2014; Triebe et al., 1996). Recently, a zeolite imidazolate framework, which is very close to the structure of zeolite, has been used as an adsorbent for this purpose, wherein a gate-opening mechanism is suggested (Ying et al., 2014). Additionally, a metal organic framework (MOF) has also been reported (Martins et al., 2015). However, the most promising alternative is the adsorptive separation based on π-complexation (Choudary et al., 2002; Yang and Kikkinides, 1995). It is reported that π-complexes are formed between the double bond of ethylene and some Cu+ or Ag+ cation containing transition metal salts. The π-complexation bonds are stronger than Van der Waals forces formed by physical adsorption, so π-complexation adsorption can achieve high selectivity and high adsorptive capacity for ethylene. On the other side, those bonds are still so weak, that ethylene can be desorbed by raising the temperature or decreasing the pressure. Related chemical mechanism and related conditions for this π-complexation reaction have been discussed in the literature. Cuprous chloride (CuCl), which contains Cu+ cation(Cu(I)), is recommended to be the first choice (Huang et al., 1999; Van Miltenburg et al., 2006) in this work, because silver is more expensive and Ag+ is often apt to be reduced to silver metal. This metal salt is always dispersed over a support with a large surface area to provide more adsorption sites (Aguado et al., 2012). Several kinds of supports have been examined (Padin and Yang, 2000; Rege et al., 1998; Yang et al., 2002), but zeolite materials are also often considered to have the excellent performance and have been widely applied to support cuprous chloride (CuCl) (Van Miltenburg, 2007; Xie et al., 1990) to prepare an adsorbent for the separation of ethylene.
However, cuprous chloride is not soluble in water and it must be dissolved in a concentrate hydrochloride solution, which requires a special tank to prepare CuCl solutions to solve corrosion problems. On the contrary, cupric chloride (CuCl2) is thoroughly soluble in water, while it is not able to produce π-complexation bonds with ethylene. Therefore, the adsorbent can be prepared by dispersing CuCl2 water solution on the zeolite support first and then converting it to CuCl by succeeding reduction of CuCl2 particles in hydrogen or carbon monoxide to provide Cu(I) (Xie et al., 1990).
Activated carbon (AC) is one of the conventional adsorbent and has been widely industrially applied. For example, it has been successfully applied to FCC dry gas separation to remove C3 and heavier compounds to prepare ethylene/ethane starting materials for this work. However AC itself is not able to produce high purity ethylene. Because of the porous structure and various fundamental groups on the AC surface, it has been applied as a support for the dispersion of transition metal compounds to prepare an adsorbent for ethylene and ethane separation via complexation. Initially CuCl is still used to prepare CuCl/AC for this purpose (Mei et al., 2002). If CuCl2 must be used instead of CuCl, a succeeding reduction step is considered to be necessary (Golden et al., 1992). More recently, it is observed that CuCl2 can be reduced to CuCl by AC itself in the preparation steps of this adsorbent as exemplified in a paper (Zhang et al., 2008). Therefore, the succeeding reduction step to convert CuCl2 into CuCl by hydrogen stream can be cancelled. As we have known, the reduction behaviors and mechanism for CuCl2 to CuCl by AC support have not yet been studied. This work aims to highlight the effect of important preparation parameters on the dispersion and reduction results of cupper species on CSC support. Reduction mechanism and related active centers will be also addressed. This information would pave the way to prepare more effective CuCl2/AC adsorbent for the recovery of ethylene from ethylene/ethane mixture.
Experimental
Preparation of the adsorbent
AC has varying pore dimensions. Adsorption is performed mainly in micropores, while the contribution by mesopores is rather small. Very few macropores is also formed in AC and they only play the role of passageway. All of these three pores link up each other. In addition, different oxygen-containing functional groups are exhibited on the surface of AC, including carboxylic group, lactonized carboxylic group, phenolic hydroxyl groups and carbonyl groups (Boehm, 2002; Lahaye, 1998). Modification of the above properties of AC by using hydrogen peroxide or nitric oxide has often been used in the literature (Gil et al., 1997).
In our previous work, it has been recognized that adsorption behaviors of CuCl2/AC adsorbent are firstly determined by the source of the starting materials to produce that AC. In addition, AC made from coconut shell (CSC) support provides substantial advantages over other AC that is in line with the reported results (Zhang et al., 2008). To clarify the effect of reduction extent and related mechanism, original CSC without any chemical treatment is selected to be a support for CuCl2 aqueous solution in this paper.
In this work, CSC was purchased from Shanghai activated company. It was pulverized and sieved to ϕ0.2–0.8 mm particles, then dried at 100℃ for 2 h before taken as CSC support. The length of the ϕ10 mm tube column is about 300 mm, therefore, it is possible to be a plug flow in the adsorption tube. CuCl2/AC was prepared by using incipient wetness technique, which is reported to permit effective dispersion of a monomolecular layer of metal compounds and avoid from their decomposition (Yang et al., 2002; Xie et al., 1998). A desired concentration aqueous solution of CuCl2 was prepared. A fixed volume of pre-dried CSC support was then impregnated in the solution at a fixed temperature for 8 h. After that, the solid product was dried at 110℃ for 6 h, and then it was purged with nitrogen stream at a flow rate of 60 mL/min in a tubular oven for 30 min under room temperature to thoroughly remove the oxygen therein. Afterwards, the tubular oven is temperature programmed heated at a ramp of 5℃/min to a fixed calcined temperature. After keeping for 4 h, the product was cooled down to room temperature.
Adsorption behaviors
To simulate the composition of the potential feed gas, an ethylene and ethane mixture in nitrogen (28:60:12 kPa) was used as a typical feedstock for the separation tests by adsorption in this work. Adsorbed amount and separation factor were measured in a conventional self-made fixed bed adsorption apparatus by using dynamic gas chromatograph method. It is composed of feed gas and adsorption system, which is connected to a thermal conductivity detector (TCD) of a gas chromatography (GC) system. A U-type adsorption tube (ϕ10 mm × 300 mm) is put in an electric thermostat to keep the temperature constant. Each of the adsorption or desorption step was preformed therein. Gas composition was analyzed by a GC9160 GC equipped with a Φ3 mm × 4 m GDX-502 packed column (copolymer of divinylbenzene and nitrogen containing heterocyclic monomers) and a TCD.
The adsorption capacity qk was calculated from the related breakthrough curves as follows:
Separation factor S for ethylene and ethane was also calculated from the related breakthrough curves as follows:
In our tests, three times of measurements were performed to obtain each amount of adsorbed ethylene and separation factor and their average results were used. The results have good reproducibility, and the relative standard deviation is less than 5%.
Characterization
Surface area and pore structure were measured on an ASAP 2020 surface area and porosity analyzer. Surface area was measured by using BET method, while the pore volume of the micropores was calculated by t-plot method (Lippens and De Boer, 1965). XRD patterns were recorded on an X-ray diffractometer D/max2550V. Temperature programmed reduction (H2-TPR) behaviors of the adsorbent was measured on a Micromeritics AutoChem II 2920 V4.02 set. H2-TPR profiles are used for characterization of changes in valence of metal cupper species. In this test, 70 mg of dried samples were loaded to a U-type tube and pretreated in a high purity nitrogen flow at 200℃ for 30 min to remove the adsorbed water, and then dropped to 50℃. After the base line is stable, the tube is switched to a 10/90 of H2/Ar gas line with a flow rate of 40 mL/min and was temperature programmed heated with a ramp of 10℃/min to 750℃. TCD was used to measure the signal for consumed hydrogen. Changes in functional groups were detected on a Nicolet 6700 FTIR spectrometer in the range of 4000–500 cm−1 with a resolution of 4 cm−1. The adsorbent sample was prepared as a KBr pellet, wherein AC:KBr was 1:400. Valence changes and surface element analysis of the cupper species on loaded AC were detected by X-ray photoemission spectra (XPS) (Padin and Yang, 2000), obtained on ESCAL-ab 220i-XL, produced by VG Scientific Inc. England. Measurement of the surface elements and their states on the absorbent sample were performed by using energy dispersion X-ray (EDX) analyzer from EDAX Inc., attached to S-3400N vacuum electric microscope, produced by Hitachi, Japan.
Results and discussion
Both CuCl2 content on CSC support and the calcinations temperature are the important parameters in the preparation of CuCl2/CSC adsorbent. At the same time, CuCl2 must be reduced to CuCl to provide Cu+ cations (Cu(I)) and well dispersed on CSC surface for the separation of ethylene via complexation. They will all be studied as follows:
Representative breakthrough curve
Adsorption behaviors are confirmed with adsorption breakthrough tests. A 17.44 g of a 36.7% CuCl2/CSC adsorbent sample is loaded, and the column porosity is 0.56. The feed gas is a mixture of ethylene/ethane/nitrogen (28:60:12) with a rate of 40 mL/min. The measurement is performed at 0.1 MPa and 313 K.
In Figure 1, it is seen that both ethylene and ethane are entirely adsorbed in the first few minutes, but ethane breaks through the tube column after 8 min. During the first 25 min, the adsorbent retains ethylene and the outlet stream is free from ethylene, while it breaks through afterwards. At last, the exit gas concentration returns back to that of feed gas after 50 min, demonstrating the saturation of that adsorbent sample. The amount adsorbed of ethylene is obtained by integration of the related breakthrough curve and the separation factor is calculated by the formula, as described in the above section (Aguado et al., 2012; Huang et al., 1999).
Breakthrough curve for a mixture of ethylene/ethane/nitrogen (28:60:12) on 36.7% CuCl2/CSC adsorbent.
Effect of loaded CuCl2 contents
Effect of loaded CuCl2 amount on C2H4/C2H6 adsorptive separation behaviors on CuCl2/CSC adsorbents is examined. It is shown in Figure 2 that the adsorbed amount of ethylene is lower than that of ethane on the pure CSC support, resulting from the inherent physical adsorption of CSC itself. With the increase of loaded CuCl2 content on CSC, the adsorbed amount of ethane sharply drops. On the other hand, the adsorbed amount of ethylene raises up initially, reaching the maximum at 36.7% and then gradually drops down. This decrease is probably because of their threshold limit and exceeding amount of CuCl2 would plug the pores on the surface. These results are in line with the increasing adsorption separation factors, as seen in Figure 2.
Effect of loaded CuCl2 amount on ethylene/ethane adsorptive separation behaviors (calcination temperature: 673 K, adsorption conditions: P = 0.1 MPa, feed flow = 40 mL/min, T = 313 K).
The separation behaviors of CuCl2/CSC adsorbents relate to their physico-chemical characteristics. Firstly, the relation of pore structures of CuCl2/CSC with varying CuCl2 content is listed in Table 1. It is shown that both specific surface area and pore volume rapidly decrease with increasing loaded CuCl2 amount. At CuCl2 content of 36.7%, the specific surface area and the pore volume of the micropores drop to 4.6% and 4.4% of that of the pure CSC, respectively. It must result in a significant decrease in the physical adsorption amount on these CuCl2/CSC adsorbents. Therefore, adsorptive ability for ethane rapidly drops in Figure 2. However, the adsorbed ethylene amount on the adsorbent increased instead, demonstrating the prominent effect of adsorption via complexation up to 36.7% CuCl2/CSC adsorbent. Secondly, the separation behaviors relate to reduction and dispersion results of the metal cations on the CSC support. XRD patterns of CuCl2 loaded CSC adsorbents are shown in Figure 3. Two wider peaks on pattern (a) for pure CSC support are observed near value of 2θ of 24° and 44°, respectively. They are attributed to specialized peaks of the graphite microcrystals, demonstrating the disordered layer structure of graphite on pure CSC support (JCPDS-ICDD, 1995). When CuCl2 is loaded on CSC and calcined at 400℃ in nitrogen stream, the peaks for graphite microcrystal significantly weaken with increasing amount of loaded CuCl2, that is in line with the change of the microstructure. At loaded CuCl2 content lower than 44%, no new specialized peak appears except that for graphite microcrystal of CSC itself, demonstrating good dispersion of the loaded metal salt. When the CuCl2 content in the adsorbent reaches 55%, new specialized peaks appeared at 2θ of 28.520°, 47.434°, 56.287°, attributed to peaks for CuCl crystal faces of 111, 220, and 311, respectively. These results reveal the reduction from Cu(II) into Cu(I), but dispersion of the latter sharply declines. Therefore, peaks attributed to CuCl clearly appear on the XRD pattern of 55% CuCl2/CSC, resulting in lower adsorptive ability as seen in Figure 2. Meanwhile, reduction extent of Cu(II) on 44% is not explained. It will be discussed in “Extent of reduction from Cu(II) to Cu(I) on 36.7%CuCl2/CSC adsorbent” section.
XRD patterns of CuCl2/CSC adsorbents with varying CuCl2 content (calcination temperature: 673 K, a: CSC, b: 15%CuCl2/CSC, c: 25%CuCl2/CSC, d: 3–36.7%CuCl2/CSC, e: 44%CuCl2/CSC, f: 55%CuCl2/CSC). Pore structural parameters of CuCl2/CSC adsorbents.

Effect of calcination temperature
Calcination is an important step in the preparation of modified AC adsorbent. In this step, changes in crystal particles and reduction extent of loaded cupper species have a strong influence on their dispersion and surface area.
In Figure 4, it is seen that at calcination temperature of 200℃, the adsorbed amount of ethylene on 36.7%CuCl2/CSC adsorbent is only 2.8 mg/g with a C2H4/C2H6 separation factor of 2.1. When the adsorbent is calcined at 400℃, the adsorptive capacity reaches an optimum of 23.6 mg/g with a separation factor of 7.6. On the other side, they dropped to 19 mg/g and 6.2, respectively, at a temperature of 500℃.
Effect of calcinations temperature in the preparation of 36.7%CuCl2/CSC on ethylene/ethane separation behaviors (adsorption conditions: P = 0.1 MPa, feed flow = 40 mL/min, T = 313 K).
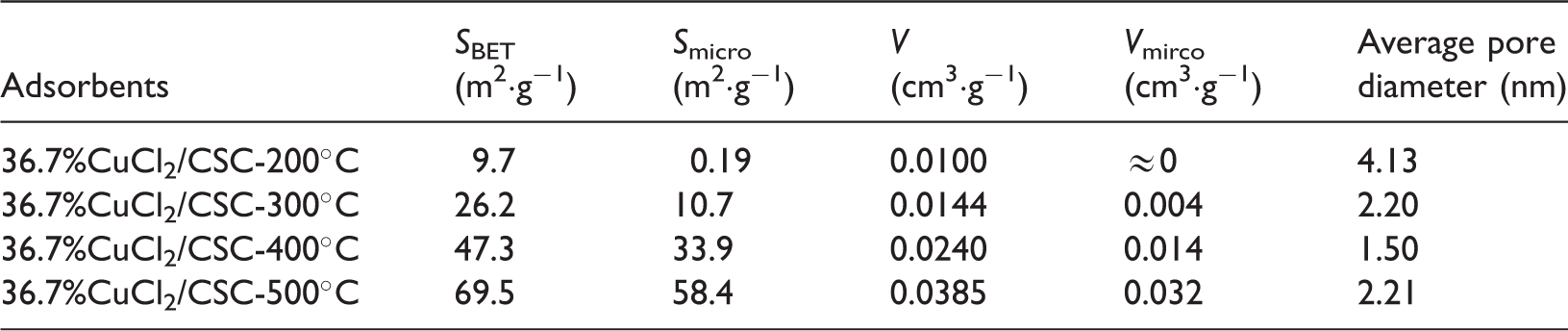
Pore structures of 36.7%CuCl2/CSC adsorbents, calcined at different temperatures.
Secondly, separation behaviors also relate to the changes in the extent of reduction and dispersion, which is drawn from characterization by XRD patterns. As shown in Figure 5, three peaks of uncalcined 36.7% CuCl2/CSC appear at 2θ of 15.4°, 25.9° and 30.7°, respectively, attributing to the diffraction peaks of CuCl2 crystals. For this adsorbent calcined at 200℃ under nitrogen stream, a weak peak at 2θ of 15.4° attributing to CuCl2 is still observed. It indicates that part of CuCl2 has not yet been reduced and has not well dispersed. On the other hand, no other peak except that for active carbon is observed for that sample, calcinated at 400℃ and 500℃. It is therefore concluded that active components can be well dispersed or no sintering of the metallic salt on CSC support is observed at calcination temperature of 400℃ and 500℃. In our work, calcination temperature is selected to be 400℃ to obtain the optimal separation behaviors.
XRD patterns of 36.7%CuCl2/CSC prepared at varying calcination temperatures.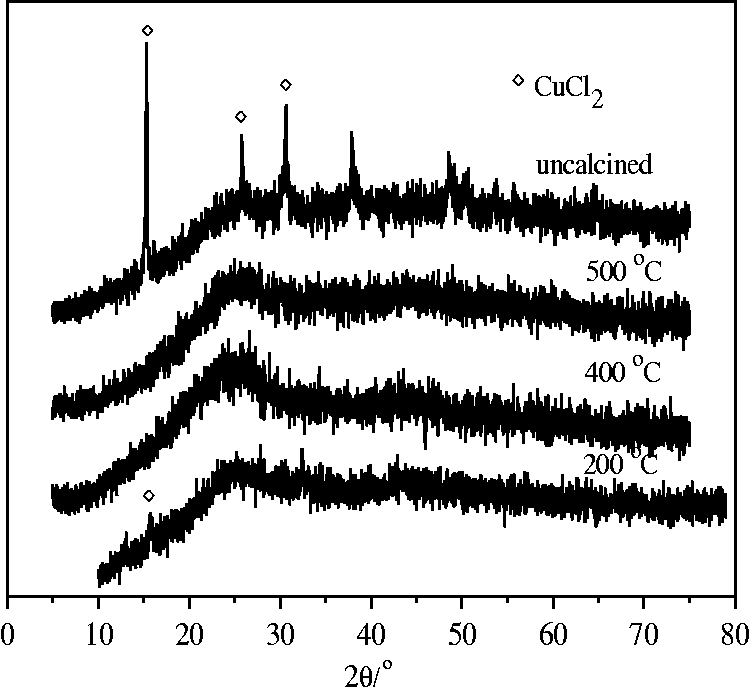
From the results in this section, it is concluded that the surface area and pore volume of adsorbent decrease with increasing CuCl2 content on CuCl2/CSC calcined at 400℃, while both of them increases with increasing calcination temperature. From XRD patterns in Figure 5, it is seen that calcination temperature must be above 400℃ to convert Cu(II) into Cu(I) for each 36.7%CuCl2/CSC. If CuCl2 content is higher than 36.7%, dispersion of Cu(I) will be deteriorated as shown in Figure 3 and that is in line with the decrease in adsorption ability as shown in Figure 2. A 36.7%CuCl2/CSC sample calcined at 400℃ has satisfactory dispersion, but the extent of reduction cannot be estimated from XRD patterns in Figure 3. It will be measured and discussed in the following section.
Extent of reduction from Cu(II) to Cu(I) on 36.7%CuCl2/CSC adsorbent
The main component to form π complex adsorption is only Cu(I) in CuCl. The selective adsorption behaviors of CuCl2/CSC adsorbent is related to the extent of the reduction of CuCl2 to CuCl.
Some surface element analysis of 36.7%CuCl2/CSC adsorbents calcined at different temperatures.

Main element analysis of CSC and 36.7%CuCl2/CSC by EDS. a

Calcination temperature: 673 K.
Mechanism of the reduction from Cu(II) to Cu(I)
TPR is one of the effective method to study the valence of cupper metal species. Changes in valence of cupper species in reduction step from Cu(II)→Cu(I)→Cu(0) were monitored by using the related peaks, appeared at varying temperatures on the TPR profiles with hydrogen stream. By comparison with the results in the literature, the peak appeared at 320℃ in Figure 6 is attributed to the reduction reaction of Cu(I)→Cu(0) on the oxygen-containing functional groups of CSC, while that appeared at 420℃ is attributed to the reduction reaction of Cu(I)→Cu(0) on the carbon on CSC. Moreover, one more reduction peak appears at 220℃ on the TPR profile for the uncalcined CuCl2/CSC sample. This peak is attributed to reduction of CuCl2. These results also indicate that CuCl2 has thoroughly converted to CuCl on the CuCl2/CSC adsorbent after calcination at 400℃, because that peak at 220℃ has thoroughly disappeared.
H2-TPR profiles of CuCl2/CSC adsorbents (calcination temperature: 673 K).
To further elucidate the mechanism for the reduction of Cu(II) to Cu(I) on CSC support. FTIR pattern measurement is performed for pure CSC and CuCl2 /CSC samples as shown in Figure 7. As reported in the literature (Biniak et al., 1997), the peak at 1000–1200 cm−1 is attributed to stretching vibration of phenolic hydroxyl groups, while the other one at 3000 cm−1 is attributed to that of C–H groups on the poly-aromatic rings. It is observed in Figure 6 that both of these two peaks drop with increasing CuCl2 content on CuCl2/CSC adsorbents. It reveals that these two groups play the role in the reduction of Cu(II) to Cu(I) on CSC support and demonstrates the existence of two reduction active centers. This result is in line with the measurement by TPR, where two active centers are observed. This reaction is postulated to partially proceed from chlorine substitution reactions between CuCl2 and para-position hydrogen atoms of C–H bonds on CSC poly-aromatic rings as follows:
FTIR spectra of CSC and CuCl2/CSC adsorbents (calcination temperature: 673 K).

This suggestion is proved by the fact that hydrochloride gas has been detected in the exit gas during calcination in our test.
Conclusions
CuCl2/CSC adsorbent is effective for ethylene and ethane separation via π-complexation, because Cu(II) has thoroughly been reduced to Cu(I) by CSC support in the preparation stages in this work. CuCl2 is soluble in water, while CuCl can only be dissolved in concentric hydrochloride solution. Therefore the corrosion problem resulting from the preparation of CuCl solution has been avoided.
Separation factor for the separation of ethylene and ethane on the CuCl2/CSC adsorbent enhances with increasing CuCl2 content thereon. Meanwhile, a maximum adsorbed amount of ethylene appears at CuCl2 content of 36.7%. It is clear that π-complexation plays the predominant role.
Calcination temperature is another one of important preparation parameters. When it is performed at 200℃, CuCl2 is not well dispersed on CSC support and the channels are seriously plugged that would give negative effect on the separation behaviors. At calcinations temperature of 400℃, reduction of Cu(II) to Cu(I) is proved to have been performed from our tests. They are well dispersed on CSC support and their separation ability is therefore apparently improved. However, it slowly drops at calcination temperature of 500℃.
It is postulated that there are two reduction active centers on CSC support. One is phenol-hydroxyl groups and the other is CH– groups connected to poly-aromatic rings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


