Abstract
Rapid urbanisation has significantly increased the impervious surface along with increase in pollutants such as nutrients (nitrogen, phosphorus), sediments, oil, and hydrocarbon in stormwater. Their removal is important as they adversely affect the aquatic ecosystem and the environment. Thus, this study evaluated the performance of various adsorbent materials (red gum mulch, pine mulch, flyash, oyster shell, sawdust, clay, sand, zeolite, dolomite, alum and lime sludges) for the removal of phosphorus from synthetic stormwater. Among them, alum and lime sludges appeared to be the most effective in removing phosphorus. Further analysis showed that alum and lime sludges remove 99 and 90% phosphorus within 60 minutes from the synthetic stormwater. Therefore, this study demonstrates that the lime and alum sludges could be highly useful as a sustainable and cheap filter media for stormwater treatment systems that are enriched in phosphorus.
Introduction
Rapid growth of urbanisation leads to a substitution of vegetated ground by large impervious surface such as roofing, pavements, roads and other civil structures. This results in reduction of infiltration and increase the volume of runoff. The stormwater washes various pollutants that come along its way such as sediments, heavy metals, hydrocarbon, bacteria and nutrients (phosphorus and nitrogenous species) (Burton and Pitt, 2001). Among them, phosphorus is one of the major elements that is considered to be a main nuisance as it is a nutrient for microbial growth. Lawn fertilizer, animal waste and detergent used in the daily lives heavily contribute phosphorus in stormwater and become rich in phosphorus (USEPA, 1998). Although phosphorus is essential nutrient for the plant growth, its excessive presence can endanger the quality of stormwater. This eventually deteriorates the receiving water bodies and also affects the aquatic life ecosystem and environment (Marsalek, 1991; Roesner et al., 2001; Walsh, 2000; Wong and Eadie, 2000). As it is hard to control the source of phosphorus, its removal from the stormwater prior to mixing in receiving water bodies is probably the best way to reduce its impact on the aquatic ecosystem.
Researchers have been trying to find economical and feasible technology to remove phosphorous from stormwater as it has substantial adverse effect on aquatic ecosystem. Many studies in the past attempted to remove phosphorus using various biological and physicochemical processes. Physicochemical process by using of such materials as zeolite (natural mineral or artificially produced alumina silicates), pelleted clay, opaka (a siliceous secondary rock), pumice (natural porous mineral), fly ash, blast furnace slag, alum, dolomite, calcite, sand, limestone, oyster shells were investigated and found to be effective as sorption media for phosphorus removal (Korkusuz et al., 2007). Similarly, DeBusk et al. (1997) used sand, fresh organic (peat) soil, crushed lime rock and wollastonite to remove phosphorus from stormwater and the wollastonite was found to be very effective in phosphorus removal (87.5%). Activated carbon, peat moss, compost and sand were also found to be effective in phosphorus removal from stormwater (Clark et al., 2001). Previous study, Hsieh and Davis (2005) reported that sand also has good phosphorus removal efficiency as it removes 40–48%. Although many studies have been conducted to explore various adsorbent media for phosphorous removal from aqueous solutions, waste products produced by many industries could be the potential adsorbents and they might be sustainable and cheap in removing phosphorous.
Moreover, in Australia, alum and lime sludges produced from groundwater treatment plants are dewatered and then disposed as a waste in landfill site since they contain high level of aluminium and calcium, respectively. Previous studies (Babatunde and Zhao, 2009; Li et al., 2013; Nazirul et al.,2014) have shown that alum can effectively remove phosphorus from the wastewater. Similarly, Nair and Ahammed (2015) reported that the 87% of phosphorus removal from the wastewater by using alum sludge. In addition, Craig et al. (2015) found that the alum sludge enhanced the sand filter and performed the highest phosphorus removal efficiency (80%). As the alum used in the treatment plant for the removal of natural organic matter in the water, the sludge obtained after water treatment could still possess a good ability to remove phosphorus. Further re-use of sludge including other waste product from industries is beneficial both environmentally and economically. Therefore, this study aims to investigate the effectiveness of lime and alum sludges in removing phosphorous from synthetic stormwater. Furthermore, their performance was compared against various materials such as industrial by-products (sawdust and fly ash), waste (oyster shells) from food processing industry, widely used mulch (pine and red gum), materials used in soil treatment (zeolite and dolomite) and readily available materials (sand and clay) for the removal of phosphorus.
Materials and methods
Adsorption materials collection and preparation
Eleven different materials (red gum mulch, pine mulch, fly ash, saw dust, zeolite, dolomite, oyster shell, sand, clay, alum sludge and lime sludge) were used as a phosphorus adsorbent media. Alum and lime sludges were obtained from Gnangara and Neerabup Ground Water Treatment plants, Perth, Australia while rest of the materials were obtained from the local home supply store. Sand mulch, oyster shell, fly ash, sawdust was washed before the test. Afterwards, alum sludge, lime sludge, sand, mulch, oyster shell, flyash, sawdust were oven dried at 105℃ for 24 hours and stored in room temperature while zeolite, dolomite and clay were directly used for adsorption experiment. Among them, sludges (alum and lime) and oyster shells were broken into small pieces as they were in bigger lump. Batch test was carried out to determine their adsorption capacity. Afterwards, two best adsorbents determined based on their phosphorous removal efficiencies were further analysed using jar and column tests to calculate the rate of adsorption and adsorption capacity, respectively.
Synthetic water preparation
Phosphorous stock solution of 10 g/L was prepared by dissolving an analytical grade of monopotassium phosphate in Reverse Osmosis treated water (Ibis IS0006, Ibis Technology, Australia). Stormwater contains various pollutant including phosphorus (<1 mg/L). However, only phosphorus was used in synthetic water to avoid the interference of other pollutant to determine the effectiveness of various adsorbents. The experiment was carried out at pH of 6–7and pH was adjusted in the solution using 1 M of sodium hydroxide and 1 M of sulphuric acid.
Analytical methods
The pH was measured using a portable pH meter (HACH 40d with PHC101 HAC electrode) and measurement error was ±0.1. Phosphorus in solution was determined by using an Aquakem 200 (Thermo Scientific, Finland), high precision wet chemistry analyser. The instrument had a detection limit up to0.002 mg-P/L and measurement error ±1.5% (95% confidence level). Each sample was filtered through 0.45 µm membrane filter (GE Water and Process Technologies) prior to phosphorous measurement.
Experimental design
Synthetic stormwater with phosphorus concentrations of 25 and 80 mg-P/L was employed depending on the experiment. Three different approaches were taken to study and evaluate the performance of different adsorbents as presented in Figure 1.
Experimental flow chart.
Batch test. To determine phosphorous removal efficiency of adsorbents, 5 g of each adsorption material was placed into 500 ml Erlenmeyer flask containing 250 ml of synthetic stormwater with phosphorus concentration of 25 mg-P/L and pH of 6.58. The top of each flask was covered by aluminium foil to prevent from any possible contamination and to avoid water losses through evaporation. The Erlenmeyer flasks were then placed in the shaking platform (Innova, 200, New Brunswick Scientific) and continuously shake at constant speed of 100 rpm at room temperature (18 ± 2℃) for 24 hours. Then, phosphorus concentration was measured in each sample. Phosphorus removal efficiency of each material was calculated using initial and final phosphorus concentration in solution.
Jar test. To determine the phosphorous adsorption rate, a jar test was carried by placing 5 g of each material (alum and lime sludges) and 500 ml of synthetic stormwater having phosphorus concentration of 25 mg-P/L and pH (6.0–7.0) in a 1 L beaker. The beaker content was then mixed at a constant speed of 100 rpm and water sample was collected every 10 minutes interval for 1 hour from the top of beaker to measure phosphorus concentration.
Column test. To determine the breakthrough time and adsorption capacity of alum and lime sludges, a column test was carried out by continuously feeding synthetic stormwater.Two columns (diameter: 3.9 cm and height: 30 cm) were used for this study and each column was filled with alum (10 g) and lime (10 g) sludges having particle size of 2 and 0.15 mm, respectively. A cotton pad was placed at the bottom and top of the alum and lime sludges column to prevent any loss of sludge. Further, to prevent the escaping of sludge from the column, glass beads were kept over the cotton at top of the bed. The synthetic stormwater having phosphorus concentration of 80 mg-P/L and pH 6.58 was then continually fed through the bottom of each column at flow rate of 5 ml/min using a peristaltic pump. In order to ensure the contact of water with the entire sludge bed, whole sludge bed was operated under fully submerged condition using upward flow in the column. Effluent samples were collected from the top of column and analysed for the residual phosphorus concentrations. The experiment was continued until the complete exhaustion of alum and lime sludges beds.
Characterisation of alum and lime sludges
The characterisation of alum and lime sludges was carried out using a scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (EDS). The samples were first air-dried and coated with approximately 3 nm of carbon using a Baltec MED 020 coater. Afterwards, they were viewed using focused ion beam scanning electron microscope (Zeiss Neon 40EsB) at 15 kV and a working distance of 8.5 mm and images were taken using the secondary electron. EDS was used to identify the element attached in both samples. For this, samples were kept under vacuum and excited to a higher energy state with an electron beam. As each element falls back down to its original energy state, it emits X-ray energy at different wavelengths for each element. Identification of the elements was done by matching the peak locations (on the x-axis) with known wavelengths for each element.
Metals leaching
To understand whether the alum and lime sludges release metals back into the water that were possibly adsorbed during the water treatment process, metal leaching test was carried out. It was examined by conducting a batch test (detailed as above) under four different pH (5, 6, 7 and 8), as stormwater pH normally remains within that range. After the test, water samples were collected and filtered through 0.45 µm filter paper (GE Water and Process Technologies). The soluble metals concentrations in each sample were then determined by an inductively coupled plasma spectrophotometer (ICP-OES, Perkin-Elmer Optima 4300 DW) at ChemCentrein Curtin University, Bentley, Australia.
Results and discussion
Phosphorus removal capacity of different materials
Figure 2 illustrates the effectiveness of 11 different materials in removing phosphorus from the aqueous solution. It clearly shows that fly ash, zeolite, sand, mulches, sawdust and clay are not very effective as they removed less than 20% phosphorus. The removal percentage was increased with dolomite and oyster shell but the efficiency remained only around 25% where as the lime and alum sludges out performed all these materials and offered phosphorus removal efficiency up to 76% and 94%, respectively. Results demonstrated that alum and lime sludges are effective phosphorous adsorbent materials comparing to other tested materials.
Phosphorus removal % of different materials.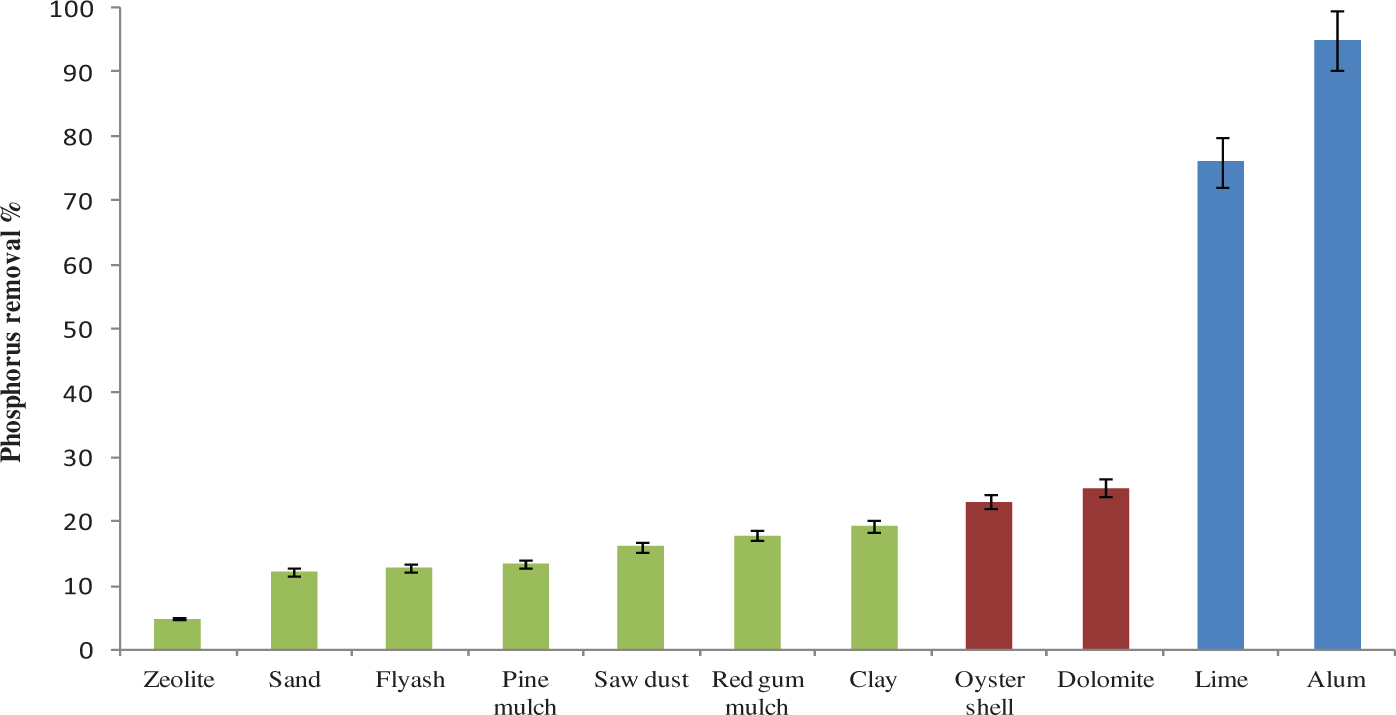
Effect of pH on phosphorous removal
Once the alum and lime sludges were identified as the best phosphorus removal materials, further investigation was carried out at four different initial pH (with the same initial phosphorous concentration of 25 mg-P/L) to understand the effect of pH on phosphorus removal capacity of alum and lime sludges. Figure 3 demonstrates the effectiveness of both alum and lime sludges in terms of phosphorus removal. Result clearly showed that removal of phosphorus decreases with increases the pH. Thus, higher removal of phosphorus is obtained at lower pH. The residual concentrations of phosphorus were significantly varied for lime sludge at different pH but no such difference for alum sludge. However, phosphorous removal by lime was not difference when compared the solutions having initial pH of 6 and 7. pH 5 was found to be the most effective for both sludges in removing phosphorus. This result is in agreement with the previous findings that higher removal efficiencies were obtained at lower pH (Yang et al., 2006).
Phosphorus residuals in the synthetic stormwater at different pH. Initial phosphorous concentration was 25 mg-P/L in the synthetic stormwater.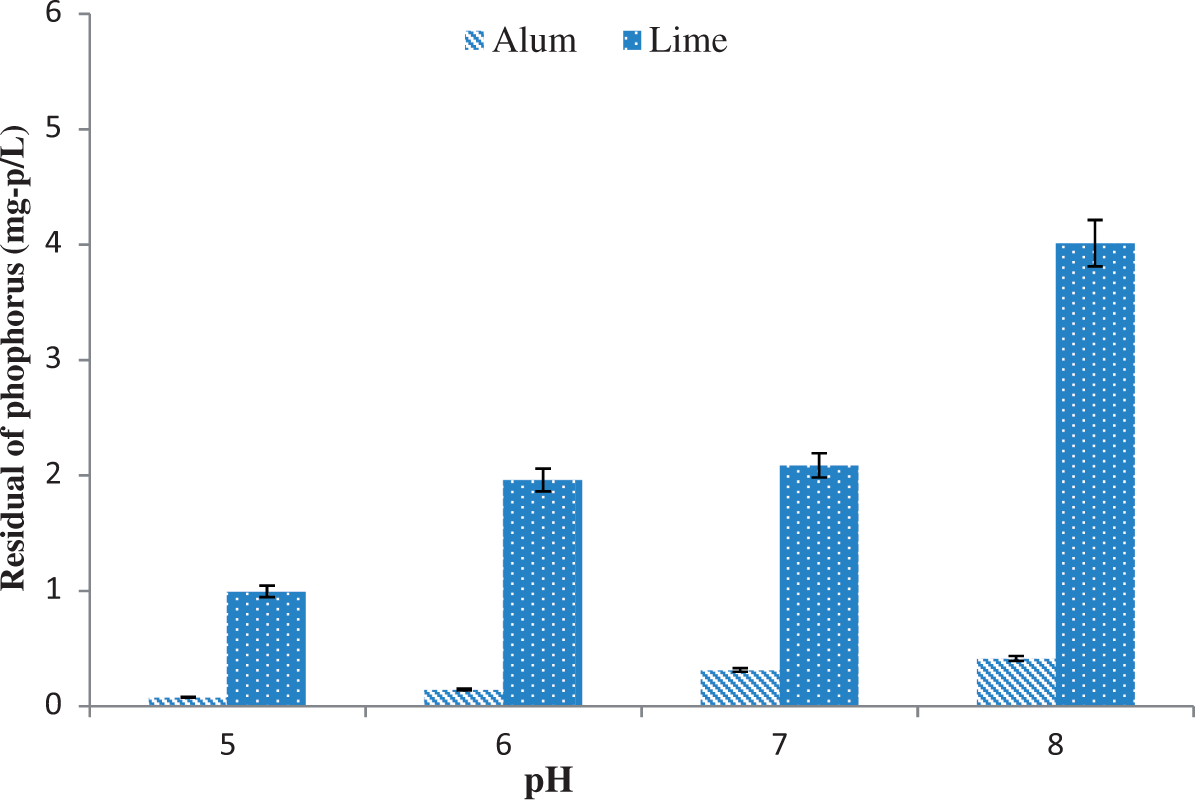
Determination of phosphorus uptake rate by alum and lime sludges
Similar to phosphorous removal efficiency, alum sludge was found to be better in up taking phosphorous from solution than the lime sludge. Alum reduced phosphorus concentration from 25 to 1.08 mg-P/L within 30 minutes while lime reduced to 5.17 mg-P/L (Figure 4). Result indicated that less contact time is enough for alum sludge to remove phosphorous from the solution than lime sludge as it removed 99% phosphorus within 60 minutes. This could be attributed to more favourable specific surface area, chemical reaction and precipitation in alum (Zhao et al., 2006). However, in lime sludge, presence of calcium could lead to the formation of tricalcium phosphate (Ca3 (PO4)2). This would eventually stabilise thermodynamically and becomes less soluble due to the formation of hydroxyapatite (Ca10 (PO4)6(OH) 2) (Roques et al., 1991). As the calcium phosphate compounds ages and increases in size, the adsorption area decreases resulting in slower phosphorus adsorption rates although, it needs further investigation (Brady, 1990).
Phosphorus removal efficiency of alum and lime sludges.
Determination of phosphorus uptake capacity of alum and lime sludges
The ratio of effluent (Ce) and influent (Co) was plotted against the column operation time with constant flow rate (5 ml/min) as presented in Figure 5. Breakthrough curve of phosphorus was used to quantify phosphorus adsorption under the continuous flow. Results showed a very slow increase from 48 hours for both sludges (alum and lime) while rapid increase of the ratio of effluent and influent of phosphorus was occurred after 144 and 168 hours in lime and alum sludges, respectively. Results indicated that the alum bed saturated after 168 hours of operation as there is no difference between the feed and effluent phosphorus concentrations. Similar to batch and jar test result, lime saturated earlier than alum (after 144 hours of operation). As the mechanism is physical adsorption for both materials, the removal was higher at the beginning followed by consistent loss of removal capacity resulting from exhausted adsorption sites. Further to understand the total removal capacity of these materials, the adsorbed mass of phosphorus was calculated as presented in Table 1. This clearly shows that alum has capacity to remove around 55 mg of phosphorus per unit g of alum sludge while lime has relatively less (around 44 mg-P/g of lime sludge) as expected.
Breakthrough curve of phosphorus during the column test. Phosphorus adsorption capacities of alum and lime sludges.

Characterisation of alum and lime sludges: SEM, EDS and metals leaching
Reusing industrial by-products such as alum and lime sludges is a promising alternative. However, these materials could release the unwanted contaminants like heavy metals back into the treated water. Thus, they were analysed for the possibility of metals leaching. Initially, their possible presence was identified using SEM and EDS. Figure 6(a) shows a clear peak for aluminium as alum (aluminium sulphate as a coagulant) was the main chemical used in the ground water treatment process. This analysis also showed the presence of calcium, iron and carbon attached on the sludge. Similarly, the presence of calcium, magnesium, iron and carbon was identified in lime sludge (Figure 6(b)).
(a) SEM (top) and SEM–EDS (bottom) of the dewatered alum sludge. (b) SEM (top) and SEM–EDS (bottom) of the dewatered lime sludge.
Result of metals leaching test at different pH from alum and lime sludges.

Significance of using alum and lime sludges
Coagulation is one of the widely used techniques in treating drinking water employed by the water utilities around the world. Various coagulants such as ferric chloride, aluminium sulphate, poly aluminium chloride, calcium hydroxide are used based on the source water characteristics in the treatment plants. Gnangara and Neerabup Ground Water Treatment plants, (Perth, Australia), respectively use aluminium sulphate and calcium hydroxide for the treatment of drinking water. This means both plants produce a significant amount of sludge which has to be disposed on a regular basis. Thus, reuse of these materials not only ease the sludge disposal process but also provides a sustainable phosphorus removal material in the potential stormwater treatment systems. This article shows the possibility of use of these by-products which could be used in the application of infiltration basin, bioretention systems and constructed wetland as a sustainable alternative filter media.
Conclusions
Eleven different materials were tested for their ability of removing phosphorus from synthetic stormwater. Result showed that dewatered lime and alum sludges, by-product of water treatment process have showed to be a potential phosphorus removing materials as they remove 76 and 94%, respectively. Similarly, the adsorption capacity of lime and alum were obtained 44.5 and 55.3 mg-P/g, respectively. This could be a promising alternative filter media for the sustainable use of by-product for the construction of stormwater treatment system.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
