Abstract
To understand methane adsorption characteristics on shale at high pressures, we conducted experiments of methane adsorption on dry shale up to 50 MPa and at 95.6 ℃, 75.6 ℃, and 40.6 ℃. Results show that the amount of excess adsorption on shale at high pressures is not the same as that at low pressures. The amount of adsorption increases with pressure until a peak value is reached and then declines with further pressure increases. Isosteric heat of adsorption is studied to explain the excess adsorption behavior. In addition, a new supercritical adsorption model was developed based on supercritical adsorption. This new model and Langmuir model were both used to fit the adsorption isotherms data. Langmuir model matched well with experiment data only at low pressures but failed at high pressures. The new model can fit the adsorption isotherms well, indicating that it reflects the characteristics of supercritical methane adsorption on shale at high pressure and temperature.
Introduction
Shale gas as a significant unconventional resource has recently attracted attention of many researchers. Shale gas can exist as adsorbed on shale rock and as free gas stored in micro-pores. The total amount of a shale gas reservoir is determined not only by the amount of free gas but also by that of the adsorbed gas. It is known that the amount of adsorbed methane is controlled by the composition, micro-structure of shale and temperature and pressure of the reservoir. As the density of adsorbed gas (adsorbed phase) is higher than that of free gas (bulk phase), more gas is stored in shale under this condition than that of free gas. Earlier studies show that the amount of adsorption takes up 20–85% in the total gas content (Jia et al., 2012; Li et al., 2011; Wang et al., 2013; Xiong et al., 2012), so understanding adsorption behavior is important for shale gas reserve evaluation and production.
Schettler and Parmoly (1990) and Schettler and Parmely (1991)) studied the adsorption characteristics of methane on shale by experiments, indicating that methane is adsorbed by kerogen and various clay minerals and adsorbents have different sorption capacities for their different specific surface areas. The adsorption capacity of some Devonian shale has positive correlation with total organic carbon (TOC), and is highly correlated with clay minerals (Schettler and Parmoly, 1990; Schettler and Parmely, 1991). Lu et al. (1995) found that illite's sorption plays an important role and that shale contains less adsorbed methane with less TOC and at higher temperature. Li et al. (2001) and Nuttall et al. (2005) expressed their belief that organic materials contain more adsorbed gas than clay minerals by conducting adsorption experiment with CH4 and CO2. Strąpoć et al. (2010) and Wang et al. (2009) reported that not only clay minerals and organic matter but also vitrinite reflectance (Ro) and micro-pore volume of shale have an impact on adsorption characteristics. Popular monolayer adsorption theory may not be suitable to calculate the total gas content of shale gas accurately and impact of organic and clay minerals should be considered in future research. It is also reported that kerogen in Posidonia shales plays an important role in methane adsorption while sorption of CO2 is mainly due to inorganic materials (Hartman et al., 2011; Rexer et al., 2014). Experiments of adsorption on shale were carried out under pressure from 1.0 MPa to 25 MPa at 46 ℃, and results showed that methane sorption capacity increased with TOC content and moisture content could reduce sorption capacity of shale (Tan et al., 2014).
Langmuir model is widely used to describe the isotherm of methane adsorption on shale and coal for pressures ranging from 0 MPa to 15 MPa. It is well known that the adsorption isotherms obtained by experiments with volumetric method is excess isotherms (Do and Do, 2003; Zhou and Bai, 2002; Zhou and Yang, 2000), and some studies showed that Langmuir model cannot describe these excess adsorption isotherms because this model does not reflect the physical interpretation of adsorption (Yan and Zhang, 1979; Zhou et al., 2000). Langmuir model matches excess adsorption isotherms well under very low pressures, because the excess isotherm approximately equals absolute isotherm at these conditions. Studies show that adsorbed gas increases with pressure until a peak value is reached, and then falls with further pressure increase. Hence, Langmuir model can fit the isotherm (Xing et al., 2002; Zhou et al., 1999, 2000). Lu et al. (1995) also proposed a Bi-Langmuir model for adsorption, but this model is in essence the same.
In China, the depth of shale gas is between 2500 and 4000 m with a pressure gradient of 1.3–2.0, and the pressure of gas in shale could be 30–80 MPa. The adsorption characteristics and law of adsorption may be different to those from elsewhere.
In this paper, we conducted a series of experiments to study the adsorption behavior of methane on shale. The experiment pressure is from 0 MPa to 50 MPa at three different temperatures of 40.6 ℃, 75.6 ℃, and 95.6 ℃. The experiment results were also analyzed to understand the characteristics of methane adsorption on shale. A new model was established to describe the isotherms obtained through these adsorption experiments.
Experiments of adsorption of methane on shale
Materials
The samples used in the experiments are Longmaxi Formation shale, from Southern Sichuan Basin, China. They were taken from a depth interval of 2300–2400 m below the surface. The reservoir temperature is about 95.6 ℃ and pressure of the reservoir is about 50 MPa. The Longmaxi shale belongs to the Silurian period and is the most commonly available stratum for exploration in the southern Sichuan Basin. Longmaxi shale has a thickness of 50–600 m, including black shale whose thickness is 20–260 m. Pure methane and helium used in the study were bought from a professional Gas Company (purity is 99.999%; the processing standard followed GB/T8980-1996). The sample was crushed into particle sizes of 0.15–0.25 mm (diameter), and then dried until their weight became constant (drying temperature 100 ℃ at normal atmospheric pressure) for adsorption isotherm test, mineral analysis, separation of kerogen experiment, and liquid nitrogen adsorption experiment.
We obtained the mineralogical composition of the sample by X-ray diffraction (XRD) with D8-DISCOVER apparatus and the SY/T 5163-2010 criteria were used to interpret the results of XRD for sample compositions. The experiment of separation of kerogen is done to obtain TOC percentage. The mineral composition and TOC percentage of the sample are shown in Table 1. The specific surface area of this sample is 19.36 m2/g based on result of liquid nitrogen adsorption on the shale. The corresponding pore size distribution (PSD) of this sample tested by N2 adsorption is presented in Figure 1. Based on the PSD of the sample we recognize that the shale sample mainly contains micro-pores and meso-pores, and hence the adsorption capacity of the sample is reasonable.
Pore size distribution of experimental sample tested by liquid nitrogen adsorption. Mineral weight composition of the sample.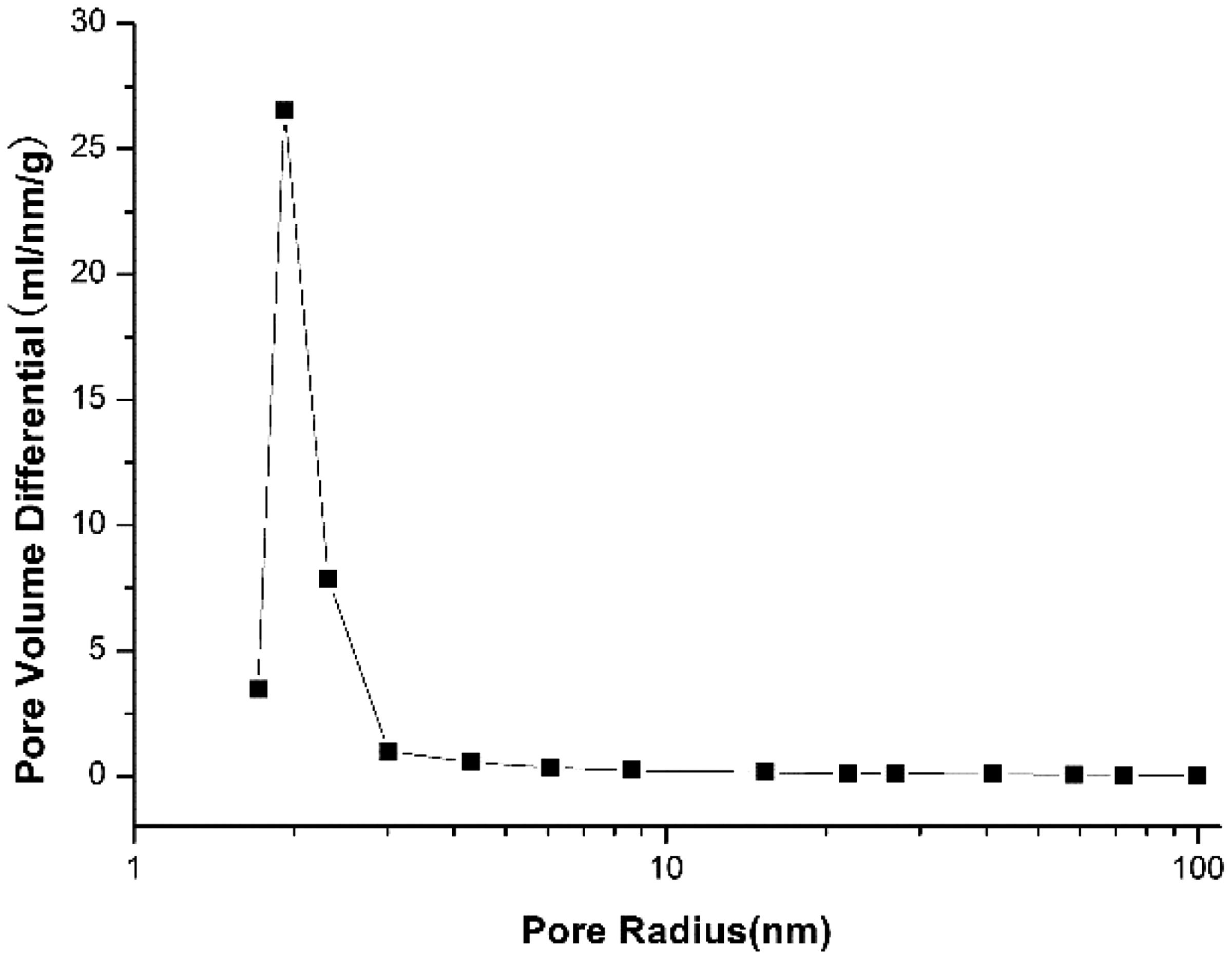

Experiment facilities
Volumetric method was used to measure the excess adsorption isotherms with GAI-100 instrument. The test pressure of the apparatus ranged from 0 MPa to 69 MPa with a precision of 0.01 MPa; the highest test temperature was 177 ℃ with a precision of 0.1 ℃. A Sketch of GAI-100 is shown in Figure 2. This apparatus consists of three units, so three samples can be tested simultaneously.
The schematic of GAI-100 adsorption apparatus.
Experiment procedures
Install sample cylinder
Clean the inner sample cylinder and the seal groove with acetone, then load the sample after the inner sample cylinder and the seal groove are aired. Wash hands and put sealing oil on the seal groove evenly after cleaning the seal groove with swab and acetone. Put three sieves with 325 meshes into the seal ring. Smear the seal ring with sealing oil evenly before putting the ring into the seal groove. Close the seal cover with torque spanner. Finally put the sample cylinder into the oil baths and connect the cylinder to the experiment system.
Check the sealing of the sample cylinder
For unit 1: 1) open A0, A1 and A3 valves, then wash the pipeline for 10–20 s by helium flow, then close A3 when the pressure sensor reads zero. 2) Open A1 to let the helium flow into the expansion cylinder and sample cylinder. 3) Pump helium into unit 1 until the pressure reaches the given value which is 500 lbf/in2 higher than the experiment pressure, then close A0 and A1. Data of pressure readings are recorded by software. If the pressure varies by 5 lbf/in2 within 24 hours, it is accepted that the seal works well. If leakage is detected, the sample cylinder should be unloaded and the above process repeated until the seal works. Follow the same processes for sealing tests for the other two units.
Calibrate void volume
Void volume has a vital effect on experiment accuracy, so it should be determined accurately. In this paper, AJP-100 calibration apparatus was used to measure the void volume by helium expansion. Four calibration tests were carried out for high accuracy. Void volume took the average of these values.
Experiment
Adsorption experiments were carried out at reservoir temperature 95.6 ℃, and the pressure varied from 0 MPa to 50 MPa. Prior to adsorption experiment, sample cylinder and expansion cylinder were vacuumed and the oil bath was heated until the oil temperature remained constant. Firstly, close valve 2 that connects the sample cylinder, and open the valve of expansion cylinder and pump methane into expansion cylinder to a pre-set pressure, then close the valve of expansion cylinder. Secondly, record the pressure data when the pressure of expansion cylinder stabilizes (varying within the range of 0–5 lbf/in2). Thirdly, open valve 2, let gas flow from the expansion cylinder to the sample cylinder. Record pressure data until the pressure variation was less than 5 lbf/in2. Finally, calculate the excess adsorption isotherms based on the temperature and pressure data.
High temperature and pressure adsorption model
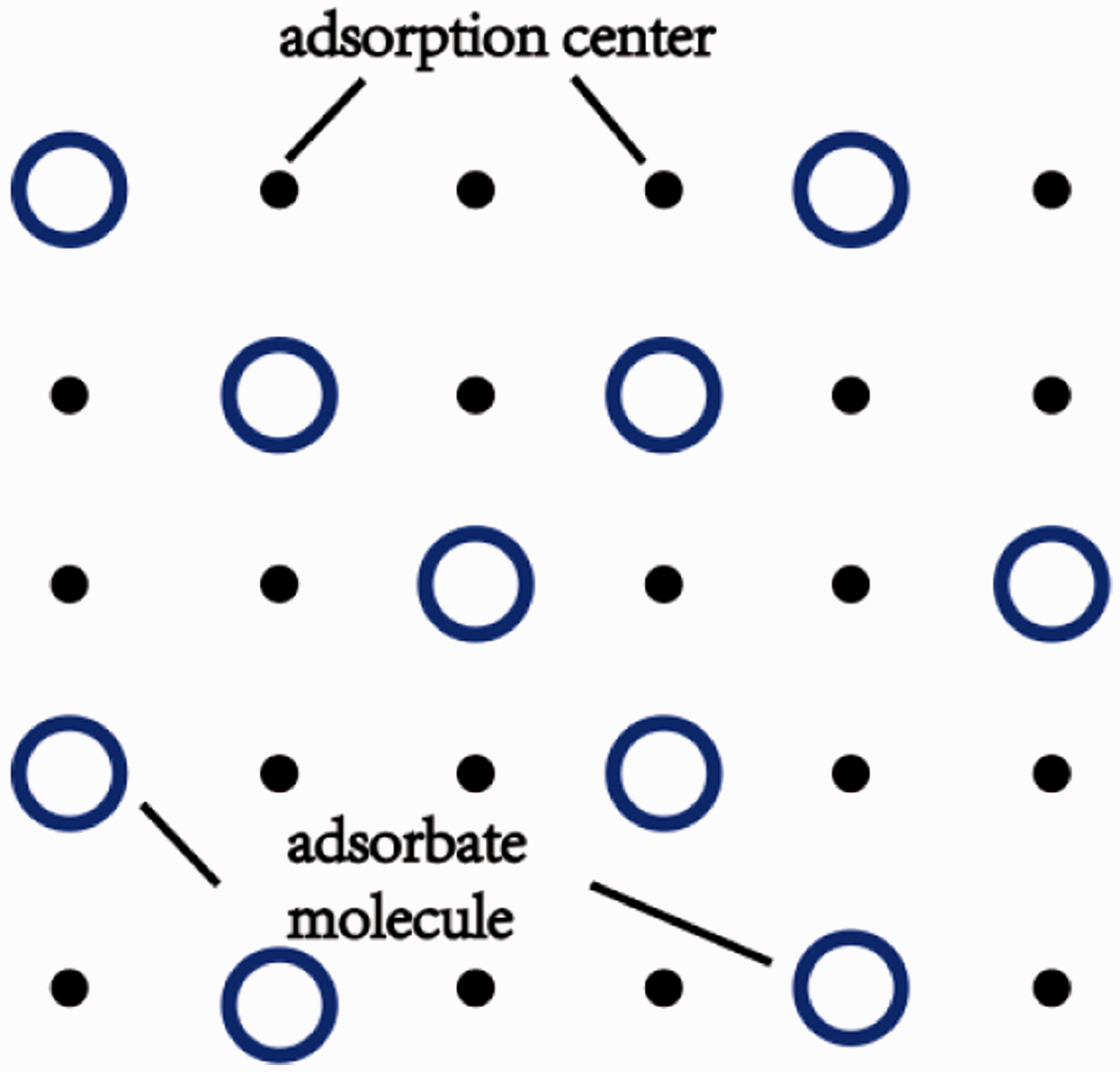
The temperature and pressure of shale gas reservoir are generally higher than the critical temperature and pressure of methane, the main component of shale gas, so adsorption occurred in reservoir is supercritical (Chareonsuppanimit et al., 2012; Zhou et al., 1999, 2000). Studies showed that the supercritical adsorption is almost monolayer adsorption (Zhou et al., 1999, 2000). The following assumptions are made: a) supercritical methane adsorption is monolayer adsorption; b) there are adsorption centers on the adsorbent surface and all the adsorption centers are equally distributed; c) adsorption and desorption of each center are independent and non-interactive; d) each adsorption center has the same thermodynamic property for adsorption and desorption. We consider that as the transition phase between adsorption phase and bulk phase.
We assumed that the non-adsorbed phase can be divided into non-adsorbed phase region I and non-adsorbed phase region II (Figure 3), and that the two regions have small differences in thermodynamic properties, but significant differences in thermodynamic properties exists between non-adsorbed phase (I region and II region) and adsorption phase.
Different thermodynamics regions in the pore.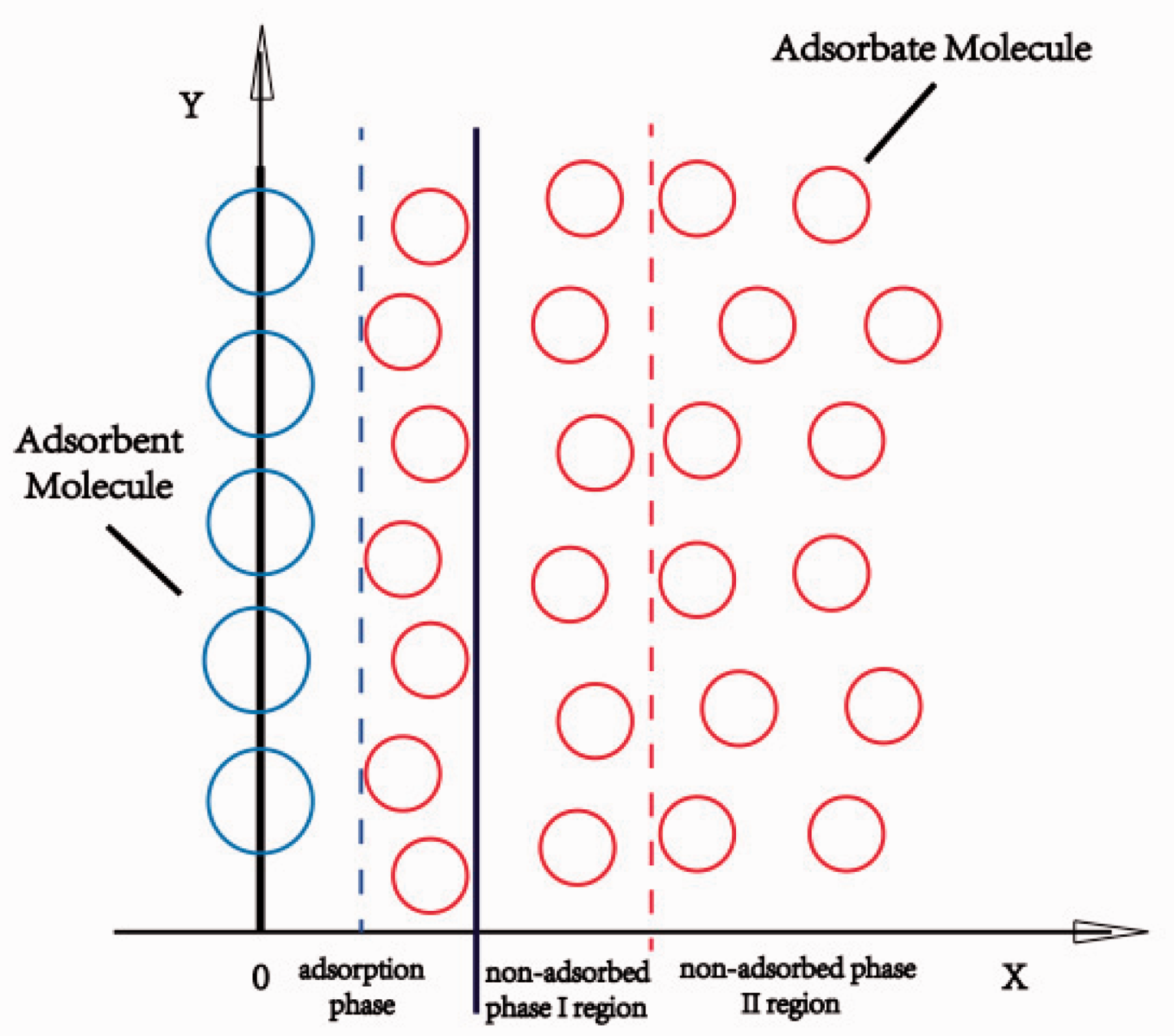
It is supposed that the number of adsorption centers in a unit area is The surface model of adsorbent.
Equation (1) can be transformed into the form below through Stirling's method (Prausnitz et al., 2006):

The chemical potential of adsorption phase is expressed by:

The partition function for any thermodynamic system can be described using quantum mechanics (Hirschfelder et al., 1954; Hocker et al., 1999):

It is assumed that the quantity of lattices which can be occupied by gas molecules is 
All the lattices occupied by gas molecules 
Usually by retaining the maximum term of equation (4) as the approximate value of equation (4) (Yan and Zhang, 1979), the partition function for each molecule of non-absorbed phase I region and II region is considered as 

The 

Therefore the partition function for all non-absorbed phase (I region and II region) can be obtained by combining equation (7) and equation (10):

Taking natural logarithm on both sides of equation (11):

Transform equation (12) with Stirling's method (Prausnitz et al., 2006):

Take the derivation of equation (13) with respect to 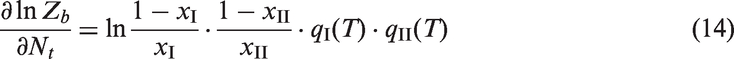
Because 
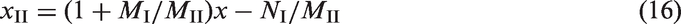
At a given temperature and pressure the values of 

Substituting equations (17) and (18) into equation (14), we have:

Hence the chemical potential of non-absorbed phase is:

The chemical potential of non-absorbed phase and that of the absorbed phase should be equal at the time of adsorption equilibrium. Combining equations (3) and (20), we can get:

From which θ can be derived:

The relationship between adsorption and θ is (Yan and Zhang, 1979):

The relationship between gas density and 
By substituting equations (23) and (24) into equation (22), a new high temperature and pressure supercritical adsorption model can be derived:

Results and discussions
The characteristics of supercritical high-pressure methane adsorption on shale
Adsorption isotherm analysis
Figure 5 shows experiment data at three temperatures: 40.6 ℃, 75.6 ℃, and 95.6 ℃. The amount of isothermal adsorption increases with increasing pressure until reaching a peak point and then decreases with further pressure increase, which indicates that these isotherms have the same characteristics as supercritical high-pressure adsorption isotherms (Chen et al., 1997; Xing et al., 2002; Zhou et al., 2000). The maximum amount of excess adsorption declines as the temperature increases. This phenomenon does not exist for methane adsorption on coal bed and some shale at shallow depth and low pressures. Results suggest that the amount of excess adsorption at 50 MPa is less than that at 10 MPa. The reason is that the amount of adsorption by experiment is amount of excess rather than amount of absolute adsorption (Do and Do, 2003). The amount of absolute (real) adsorption will increase to saturation as pressure increases. That is, the isotherm should be monotonically increasing based on adsorption theory (Do and Do, 2003; Zhou and Bai, 2002). Therefore, even if the experiment temperature and pressure are of reservoir conditions, the excess amount of adsorption obtained by the experiment cannot be considered as the real amount of adsorption of shale.
The adsorption isotherms of shale at different temperatures.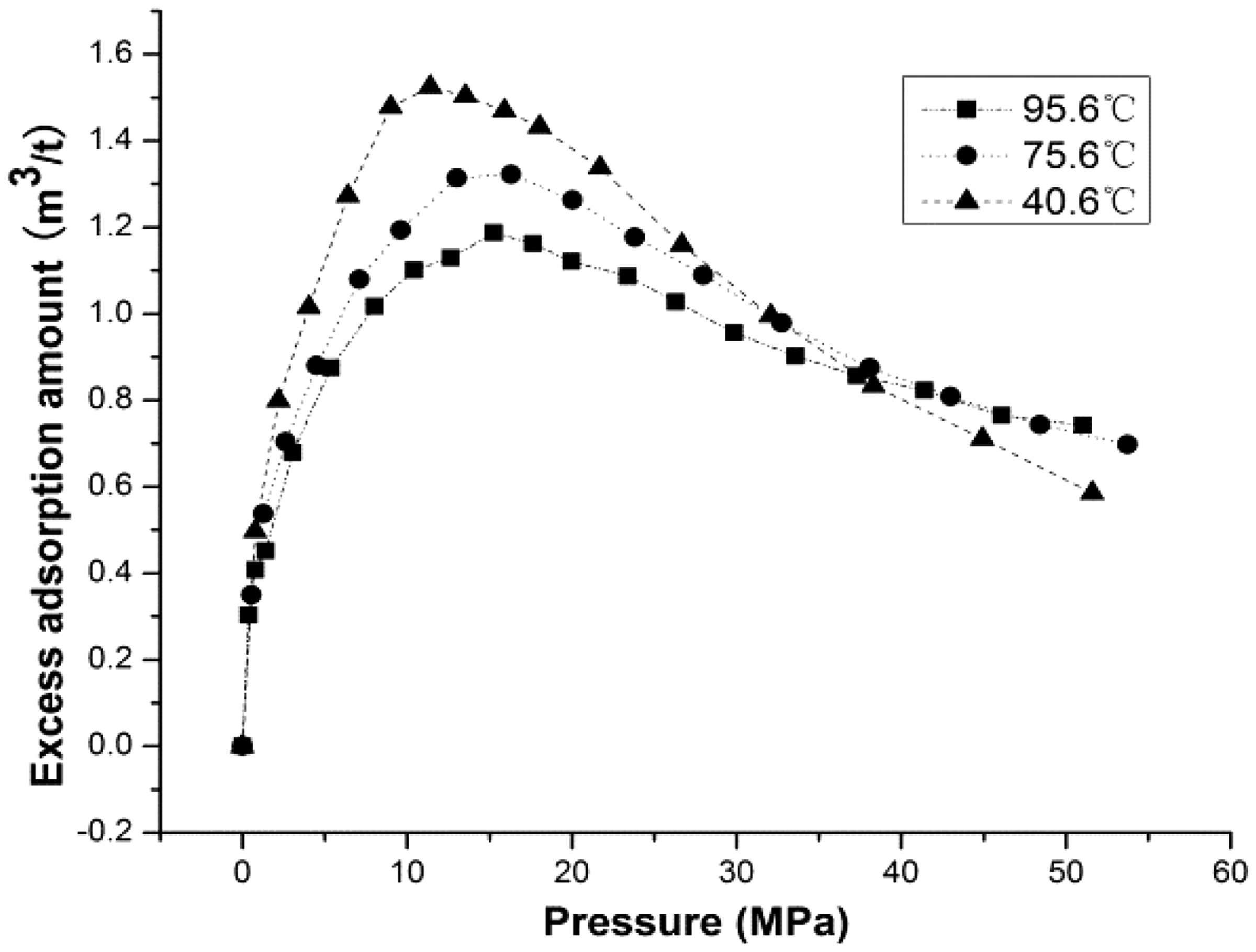
It is known that methane adsorption on shale is the physical adsorption, which is reversible. We therefore define the pressure corresponding to the maximum amount of excess adsorption to be critical desorption pressure. As the pressure increases the amount of excess adsorption also increases until it reaches the critical desorption pressure. Further increase in pressure beyond the critical desorption pressure results in decline in amount of excess adsorption.
The special phenomena indicates that during gas production free gas is firstly drained as the pore pressure is reduced. The amount of excess adsorption increases until the critical desorption pressure is reached. As the free gas is being produced, bulk phase density will be decreasing. At the same time the adsorption phase density hardly changes, and therefore density difference between adsorbed phase and bulk phase increases, leading to an increase in amount of excess adsorption (Do and Do, 2003). At the first stage of gas production, desorption does not occur. When the pressure is below the critical desorption pressure, gas will be desorbed from shale and produced together with free gas.
Isosteric heat of adsorption
To study the isosteric heat of adsorption, adsorption isotherm is divided into two parts: part A is upward-trending line below critical desorption pressure, and part B is downward-trending line above critical desorption pressure. Because these isotherms of adsorption consist of discrete points, we use the Clausius-Clapeyron equation to calculate the isosteric heat of adsorption (Ramirez-Pastor and Bulnes, 2000):

Integrating both sides of equation (26), we get:

In this paper, Wang's method (Wang, 2010) was applied to obtain the isosteric heat. Take part A as an example. Plot the experiment data of ln The relationship between ln The relationship between ln The value of ln The isosteric heat of different excess adsorption amounts.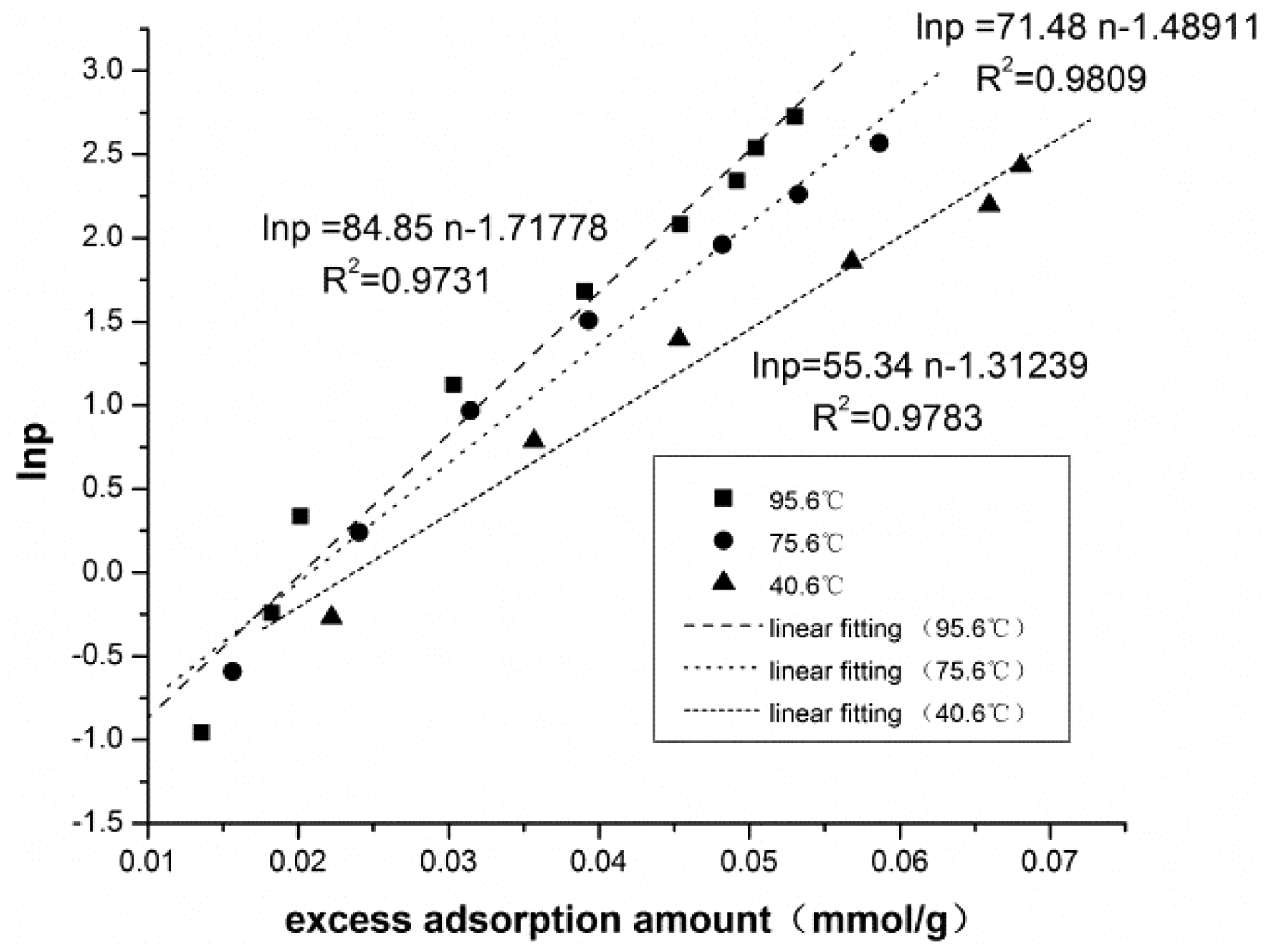



Table 3 shows that the isosteric heat ranges between 3.31 kJ/mol and 43.44 kJ/mol for part A and between −30.59 kJ/mol and −2.10 kJ/mol for part B. The isosteric heat of part A increases as the amount of adsorption increases, meaning that the adsorption of methane on shale becomes more and more difficult as the amount of adsorption increases. This is similar for part B. As the pressure increases the amount of adsorption declines and the isosteric heat increases. This indicates that methane adsorption on shale gradually becomes harder and harder as the pressure gets higher and higher. Besides, the isosteric heat goes from positive to negative separated by the critical desorption pressure. We believe that the reverse of isosteric heat is the reason why adsorption isotherm takes a convex form.
Models describing the isotherms of supercritical methane adsorption on shale
To validate the new adsorption model, we used the model to fit the adsorption isotherms data obtained by experiments; we also fitted these isotherms using Langmuir model and compared these two models. There are two ways to do this. One way is to fit all the data for each isotherm; the other way is to fit the data only at pressures lower than the critical desorption pressure and calculate the amount of adsorption at pressures higher than the critical desorption pressure with Langmuir parameters acquired. All the fitting parameters are presented in Tables 3 and 4. Apparently, the Langmuir model cannot fit all the isotherms data but our new model can fit all the data at a high accuracy (Figures 8–10, Tables 4 and 5). This new model is believed to represent the occurring physics and the process of methane adsorption on shale. It represents the characteristics of methane adsorption on shale at reservoir temperature and pressure.
New model and Langmuir model fitting the excess adsorption isotherm of shale at 95.6 ℃. The parameters obtained by Langmuir model fitting the excess adsorption isotherms. The parameters obtained by new model fitting the excess adsorption isotherms.


We also matched the excess adsorption isotherm of liquid nitrogen on activated carbon with our new adsorption model at low pressures, and achieved a fitting correlation coefficient of 0.9976 (Figure 11). The results confirmed that the new model can be applied to adsorption at low pressure range. In addition, we compared our model with some published adsorption models for high-pressure adsorption (Table 6). Comparisons confirmed that the new model has a better applicability and offers a better representation of the physical processes.
New model and Langmuir model fitting the excess adsorption isotherm of shale at 75.6 ℃. New model and Langmuir model fit the excess adsorption isotherm of shale at 40.6 ℃. New model fits the excess adsorption isotherm of N2 on activated carbon at −196 ℃. The comments of representative adsorption model published.


Conclusions
Experiments of isothermal adsorption on shale were carried out at pressures up to 50 MPa and at 95.6 ℃, 75.6 ℃, and 40.6 ℃. These isotherm data showed typical characteristics of supercritical adsorption. This adsorption, acquired by experiments at reservoir temperature and pressure, is excess adsorption, not the total amount of adsorption that occurred in the reservoir. A critical desorption pressure is defined based on the characteristics of isotherms data presented in this paper.
Because adsorption behaviors below and above the critical desorption pressure are different, the composition of produced gas will also be different. When the reservoir pressure is above the critical desorption pressure, the produced shale gas is mainly free gas. However, when the reservoir pressure has dropped below the critical desorption pressure, desorption will occur and the produced gas will consist of both free gas and desorbed gas. Isosteric heat of the adsorption increases with increasing pressure, which explains adsorption behavior on shale.
A new model for supercritical adsorption was developed. Results showed that the model can be used to match the experimental data well, indicating that it reflects the intrinsic characteristics of supercritical adsorption at high pressures. It also is verified that the model can be applied not only to adsorption of supercritical methane on shale but also to other supercritical gas adsorptions on porous material.
Footnotes
Associated Content
Isotherm data reported with this paper are included in the NIST/ARPA-E Database of Novel and Emerging Adsorbent Materials (NIST SRD-205), http://adsorbents.nist.gov, and may be accessed directly at ![]() .
.
Acknowledgements
We thank Institute of Porous Flow and Fluid Mechanics of Chinese Academy of Sciences for providing us the conditions for research and also appreciate the help and suggestion of its staff for this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
