Abstract
The isosteric heat of adsorption (IHA) is one of the key thermodynamic variables for evaluating the interaction between shale and methane, which is rarely studied especially under high pressure. In this work, we conducted methane adsorption experiments at pressures up to 30 MPa and different temperatures on shale samples collected from Longmaxi formation in Sichuan Basin, China. Based on the definition of IHA and Langmuir adsorption model, we proposed a new method to analyze the IHA of methane on shale under four conditions. The calculated results show that the commonly used Clausius–Clapeyron equation overestimates the true isosteric heat of shale, especially under high pressure. IHA under four conditions yield a fixed order as
Keywords
Introduction
Shale gas has received increasing attentions in recent years due to its remarkable success of commercial production in North America (Curtis, 2002; EIA, 2016; Montgomery et al., 2005), which has also promoted the exploration and development of shale gas in many other countries. The exploration of shale gas reservoirs in China, particularly in southern Sichuan Basin, has made significant progress in the past few years (Zou et al., 2015). Shale gas mainly consists of free gas and adsorbed gas, and exists in nanopores of organic matter and clay minerals (Ambrose et al., 2012; Chen et al., 2016; Zhou et al., 2016). Gas-in-place, the sum of free gas and adsorbed gas, is a key parameter for economic evaluation of a shale gas reservoir. Therefore, accurately determining the adsorbed gas capacity is critical for the assessment of shale gas reserves and the design of effective production strategies (Ambrose et al., 2012; Curtis, 2002).
Methane adsorption in gas shales has been extensively studied by isothermal adsorption experiments (Chareonsuppanimit et al., 2012; Clarkson and Haghshenas, 2013; Tian et al., 2016; Zhang et al., 2012; Zhou et al., 2018a ) and molecular dynamics simulations (Mosher et al., 2013; Xiong et al., 2017), mainly focusing on calculating adsorbed gas quantity and evaluating its effect on shale gas transport (Wu et al., 2016). However, its thermodynamic characteristics have been less reported (Chen et al., 2019; Rexer et al., 2013; Tang et al., 2017; Tian et al., 2016; Zhang et al., 2012). Previous studies have shown that shale gas adsorption belongs to physisorption, which is mainly controlled by London dispersion forces, the weakest type of van der Waals forces (Tang et al., 2017). Therefore, its adsorption heat is only 10–22 kJ/mol, which is significantly lower than that for chemical adsorption (Zhang et al., 2012). The isosteric heat of adsorption (IHA) is one of the key thermodynamic variables for evaluating the interaction between adsorbate and adsorbent, serving as an important descriptor of the shale gas physisorption system (Askalany and Saha, 2015; Chakraborty et al., 2006; Pan et al., 1998; Sircar et al., 1999).
In previous literatures, researchers commonly used the Clausius–Clapeyron (C-C) equation to calculate its IHA after conducting adsorption experiments under different temperatures (Gasparik et al., 2014; Ji et al., 2015; Tian et al., 2016; Wu et al., 2016; Zhang et al., 2012). The C-C equation is an oversimplified model that ignores the volume of adsorbed phase and uses the ideal gas law (Pan et al., 1998). However, neither of the two conditions can be satisfied for high pressure methane adsorption in shale because of the thermodynamic properties of methane (Askalany and Saha, 2015; Chakraborty et al., 2006). Methane in shale formations is typically at the supercritical state as the formation temperature and pressure are much higher than its critical temperature and pressure (190.65 K and 4.64 MPa), so the volume of adsorbed phase cannot be assumed to be negligible and the ideal gas law is no longer applicable (Do and Do, 2003; Zhou et al., 2000, 2018b). Therefore, the calculated results by the C-C equation may not reveal the true IHA of methane in shales and the influence of the adsorbed-phase volume and the real gas behavior on the IHA are also needed to be investigated.
Moreover, prior to calculating IHA, we need to transform the excess (observed) adsorption isotherms into the absolute adsorption isotherms for determining the volume of adsorbed phase by an appropriate adsorption model (Tang et al., 2017; Tian et al., 2016; Zhou et al., 2019). Many models have been proposed and applied to study high-pressure methane adsorption in shale, such as the extended Langmuir model, Dubinin-Radushkevich model, Dubinin-Astakhov model, simplified local density model, Ono-Kondo lattice model and so on (Chareonsuppanimit et al., 2012; Tian et al., 2016; Zhou et al., 2019). However, the most commonly used model in shale gas reservoirs is still the classic Langmuir equation, which assumes that the surface of the solid adsorbent is homogeneous with constant adsorption heat and methane is monolayer-adsorbed (Langmuir, 1918; Yu et al., 2014; Zhang et al., 2012). Although this model is very simple, it can accurately evaluate the adsorption capacity of shale (Gasparik et al., 2014; Zhang et al., 2012; Zhou et al., 2019). Thus, in this study, we use the Langmuir-based excess adsorption model to fit the observed isotherms assuming a constant adsorbed-phase density.
In this study, the methane adsorption isotherms of Longmaxi shales are obtained at pressures up to 30.0 MPa and different temperatures based on the gravimetric method. The objectives of this study are to (1) evaluate the applicability of the C-C equation to calculate the IHA of shale and (2) establish a suitable method for determining the true IHA of supercritical methane on shale under high pressure.
IHA calculation method
Supercritical methane adsorption model
The critical temperature of methane is only 190.65 K, so the adsorption of methane in shale is supercritical adsorption under formation temperature and pressure (Do and Do, 2003; Zhou et al., 2000, 2019). Under supercritical conditions, the excess adsorption capacity and absolute adsorption capacity have great differences, and these differences cannot be ignored at high pressures. Their relationship is defined as

Previous studies have shown that the Langmuir-based excess adsorption model can still fit the high-pressure adsorption isotherms very well (Tian et al., 2016; Zhou et al., 2019). The classic Langmuir adsorption model is


So the supercritical adsorption model base on Langmuir equation can be derived by equations (1) to (3), as shown by equation (4)


This model (equation (4)) can be used directly to fit the high-pressure adsorption isotherms measured in the experiment and obtain the unknown parameters
IHA determination
The IHA is defined as the differential change in energy that occurs when an infinitesimal number of molecules are transferred at constant pressure, temperature, and adsorbent surface area from the bulk gas phase to the adsorbed phase, and it can be expressed as (Chakraborty et al., 2006; Murialdo et al., 2015; Pan et al., 1998).

For real gas, its volume should be calculated by equation (7). If we assume methane is ideal, its volume can be calculated by equation (8).



The above equation is the classic C-C equation (Askalany and Saha, 2015; Pan et al., 1998; Whittaker et al., 2013;). It is obvious that two approximations are introduced in deriving this equation: (1) the bulk gas phase is considered ideal and (2) the adsorbed-phase volume is neglected. Integration of equation (9) leads to

Here,
If the absolute methane adsorption can be characterized by the Langmuir model (equation (2)), we can get the following differential formulas by equations (3) and (5).




Combining the above four equations, we can obtain

Substituting equation (15) into equation (6), the IHA can be calculated by the following equation

Therefore, for the IHA of methane on shale, the following four conditions can be considered (Table 1):
The isosteric heat of adsorption determination equations and its applicable conditions.

Assuming methane is ideal and neglecting the volume of adsorbed phase, 
Assuming methane is real and neglecting the volume of adsorbed phase, 
Assuming methane is ideal and without neglecting the volume of adsorbed phase, 
Assuming methane is real and without neglecting the volume of adsorbed phase, 
Experiments and data analysis
High-pressure methane adsorption experiment
Shale samples were collected from the Lower Silurian Longmaxi Formation in southern Sichuan Basin, China. The high-pressure adsorption isotherm experiments were conducted on Rubotherm gravimetric adsorption instrument (Dreisbach et al., 2002). Its core component is the magnetic suspension balance (MSB) with high precision of 10 μg. The maximum test pressure and the temperature are 35 MPa and 150°C, respectively. The long time fluctuation range of temperature can be controlled within 0.2°C.
The experimental procedure includes three steps: blank test, buoyancy test, and adsorption test. The first step is to test the mass and volume of sample container. After the shale sample is put in the adsorption cell, the second step is to test the mass and volume of sample in the container. And then the third step is to test methane adsorption amount at a constant temperature and increasing pressures. The observed adsorption quantity can be calculated by the following equation (Zhou et al., 2019).

Here,
Supercritical methane adsorption isotherms
In this study, four high-pressure adsorption isotherms of methane on shale were measured at 303.15 K, 313.15 K, 323.15 K, and 333.15 K, as shown in Figure 1, which shows that the excess adsorption capacity (
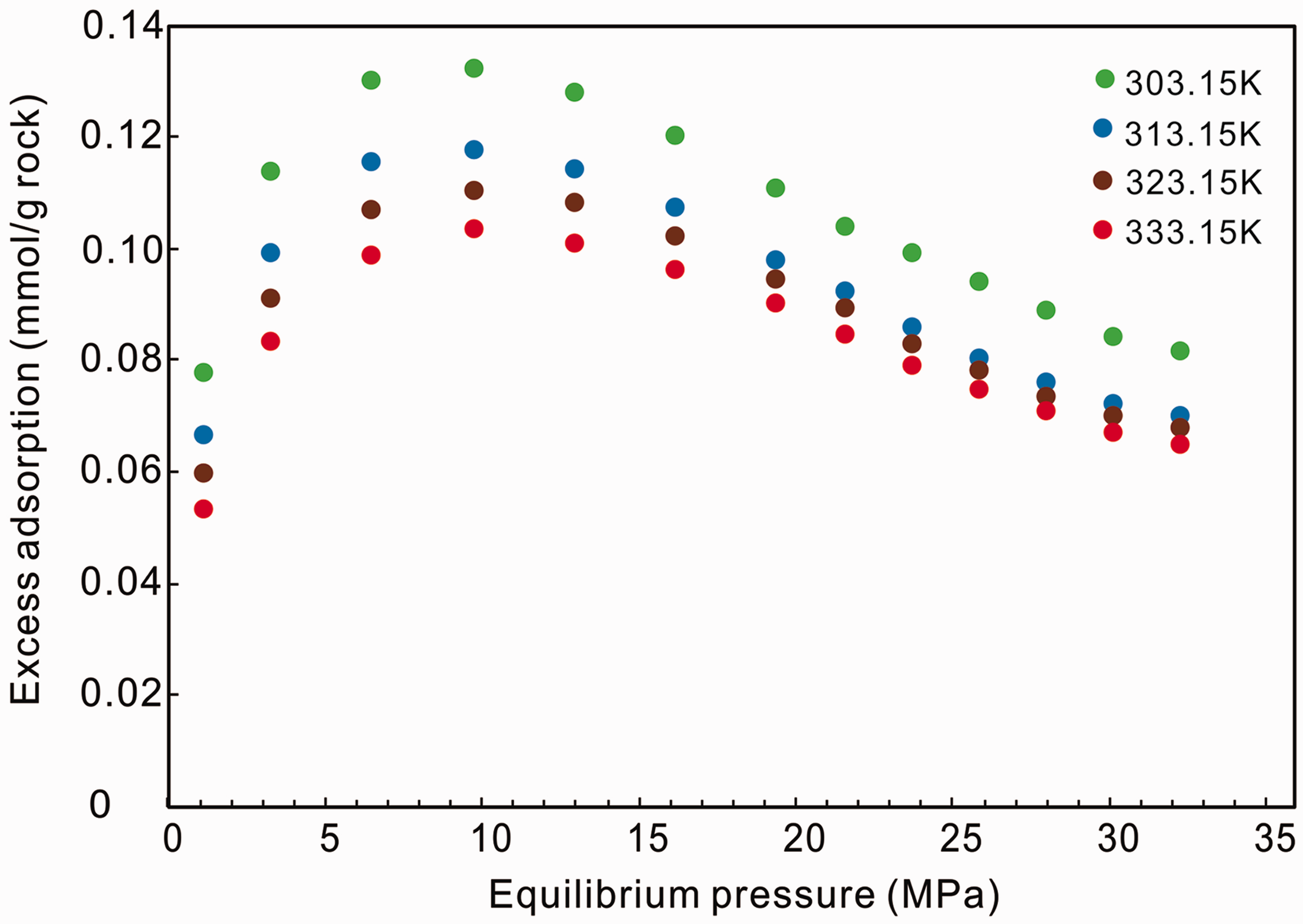
The observed excess adsorption isotherms at 303.15 K, 313.15 K, 323.15 K, and 333.15 K.
Fitted parameters and absolute adsorption
All four isotherms were then fitted simultaneously with the Langmuir-based excess adsorption model (equation (4)) using a least-squares residual minimization algorithm. The four independent fitting parameters were varied to achieve the global minimum of the residual-squares value within the following limits: 0 <
Figure 2 and Table 2 show the fitted results by the Langmuir-based excess adsorption model (equation (4)). It can be seen that this model fits the high-pressure adsorption isotherms quite well. The maximum methane adsorption capacities (
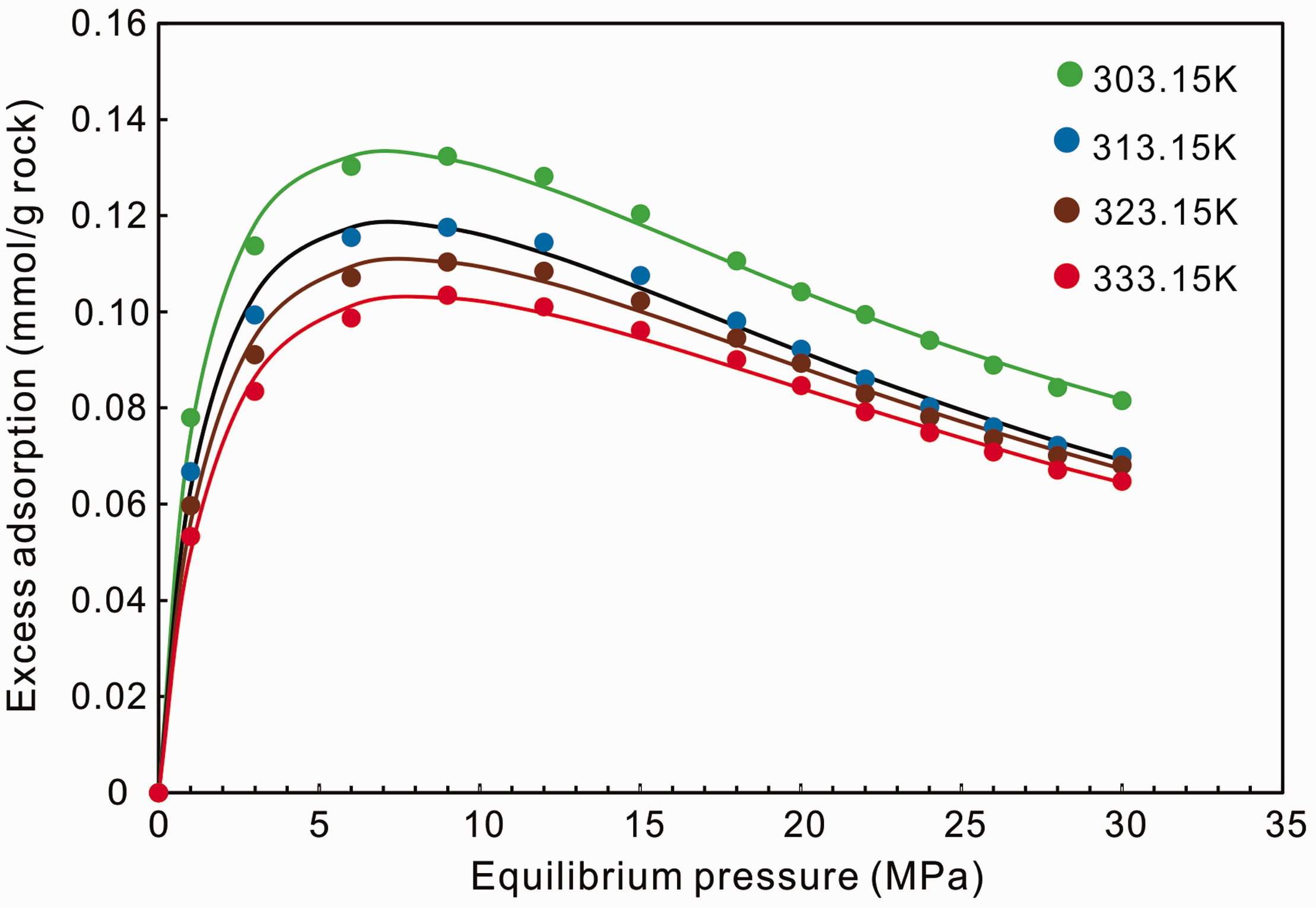
The fitting results of the excess adsorption isotherms of shale sample at different temperatures using Langmuir-based excess adsorption model (solid lines). The solid points represent the observed adsorption capacity under different pressures.
Fitted parameters by Langmuir-based excess adsorption model (equation (4)) at four temperatures.

After the adsorbed-phase density is determined (Table 2), we can transform the excess adsorption to the absolute adsorption by equation (1). And then the volume of adsorbed phase also can be obtained at different pressures and temperatures by this equation, which will be used for analyzing the IHA. Figure 3 shows the absolute adsorption isotherms at different temperatures, indicating the absolute adsorption capacity increases monotonically as pressure increases. When the experimental pressure reaches 25 MPa, the adsorption capacities tend to be saturated and absolute adsorption capacities hold at maximum values. Moreover, it clearly shows that the adsorption amount decreases with increasing temperature, which proves that the adsorption is an exothermic process (Zhang et al., 2012).
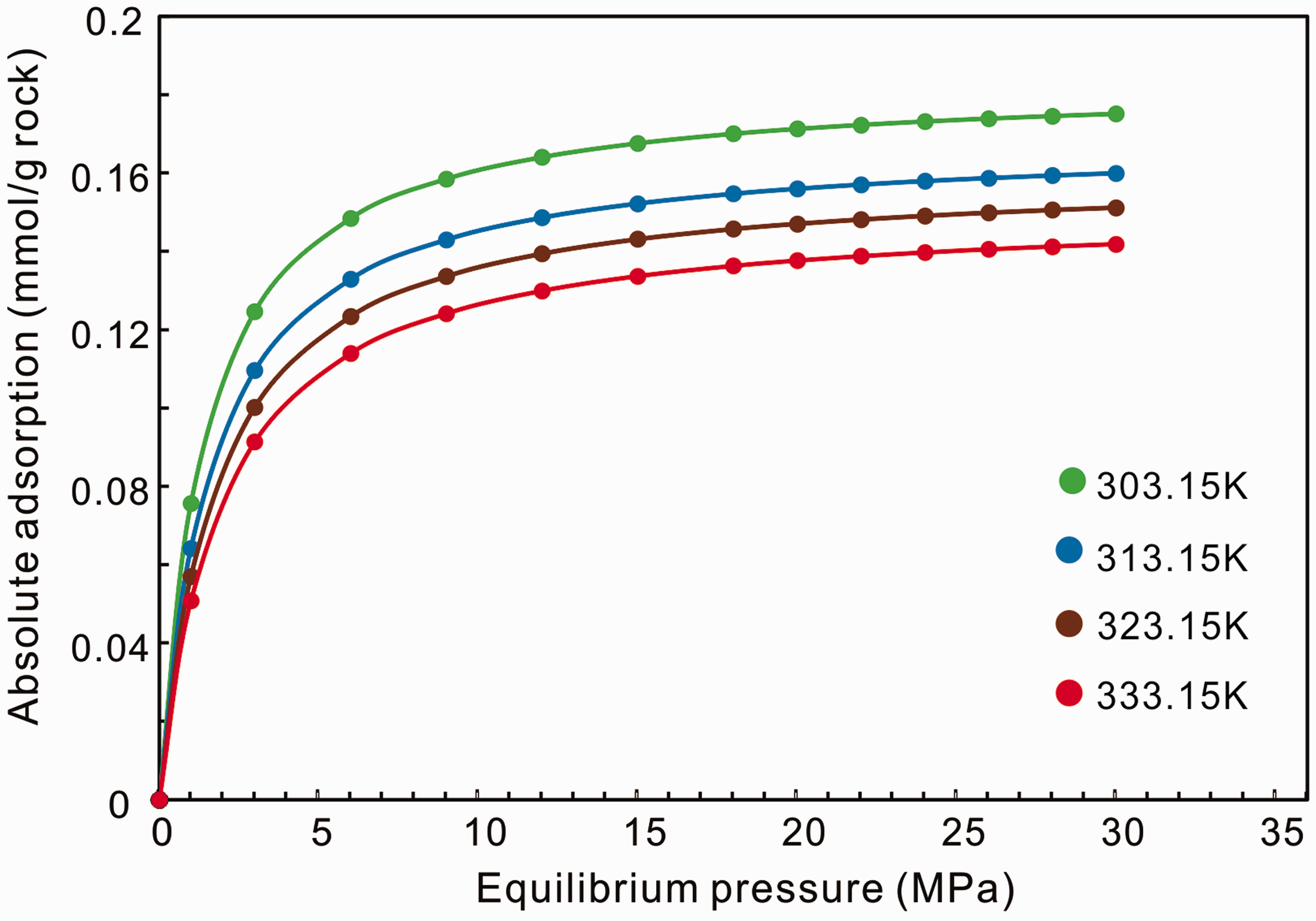
The transformed absolute adsorption isotherms at different temperatures.
Discussion
IHA calculated by C-C equation
Based on the absolute adsorption amount (

(a) The linear relationship between ln (
Generally, there are two reasons to explain the variation trend of IHA with the increase of
Volume of bulk gas and adsorbed phase
Four different conditions have been listed in “IHA determination” section and their corresponding IHA can be calculated by equations (17) to (20), where the key parameters are the volume of bulk gas (

Methane compressibility factor under different pressures and temperatures.
As the compressibility factor is determined, we can calculate its real volume of bulk gas by equation (7), as shown in Figure 6. We can see

Variation trend of the volume of bulk gas and adsorbed phase during the process of adsorption.
Comparison of IHA under four conditions
A comparison of the IHA calculated under four conditions (equations (17) to (20)) is shown in Figure 7. Overall, the different IHA follow a similar behavior at different temperatures, and their relationship can be expressed as

Effect of temperature on IHA of methane on shale under four different conditions calculated by equations (17) to (20), respectively. (a) qst,i-va, (b) qst,r-va, (c) qst,i+va and (d)qst,r+va.
In general, regardless of the gas law employed, the adsorbed-phase volume significantly affects the IHA, especially under high pressure conditions: without considering this effect leads to a higher IHA. And for cases considering the finite volume of the adsorbed phase, the difference between the real gas and ideal gas density also affects the IHA significantly: the ideal gas law always corresponds to a higher IHA. Moreover, it can be seen clearly that the commonly used C-C equation caused a significant overestimation of the IHA of methane in shale, proving that this equation is not applicable in shale gas reservoir.
A separate analysis of the change of IHA with temperature under each condition is shown in Figure 8. In all cases, temperature generally has a negative effect on the IHA: the higher the temperature, the lower the IHA. When pressure is low the IHA has not changed much, but there will be a significant change under high pressure conditions. For shale gas reservoirs, its formation pressure is about 35 MPa given a pressure coefficient of 12 MPa/km and a depth of 3000 m in Longmaxi formation in China, where the assumptions of ideal gas and neglecting adsorbed-phase volume are certainly not valid. Therefore, only the IHA calculated under the fourth condition can represent the actual IHA of methane on shale.

Effect of temperature on methane on shale under four different conditions at different temperatures calculated by equations (17) to (20), respectively. (a) 303.15k, (b) 313.15K, (c) 323.15K and (d) 333.15K.
IHA at zero coverage
As can be seen from the above discussion, IHA is a variable and therefore cannot be used as a uniform evaluation criterion for adsorption capacity evaluation. Steele (1973) proposed an IHA in Henry region at zero coverage (


Therefore, 
Employing the isotherms data,

Determination of the Henry’s coefficient (
Analysis results of the isosteric heat of adsorption at zero coverage.

Conclusions
The IHA is one of the key thermodynamic variables for evaluating the interaction between shale and methane. In order to analyze its characteristics under high pressure, methane adsorption isotherms at pressures up to 30 MPa and different temperatures were measured on shale samples collected from Longmaxi formation in Sichuan Basin, China. The conclusions drawn from this study can be summarized as follows:
The commonly used C-C equation for IHA determination was established assuming that methane is ideal and neglecting the volume of adsorbed phase. IHA calculated by this equation overestimates the true isosteric heat of shale, especially under high pressure. IHA under four conditions were analyzed, yielding a fixed order as IHA at zero coverage (
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Science and Technology Major Project (No. 2017ZX05035002-002).
