Abstract
The first ONTOX Hackathon of the EU Horizon 2020-funded ONTOX project was held on 21–23 April 2024 in Utrecht, The Netherlands (https://ontox-project.eu/hackathon/). This participatory event aimed to collectively advance innovation for human safety through the use of Artificial Intelligence (AI), and hence significantly reduce reliance on animal-based testing. Expert scientists, industry leaders, young investigators, members of animal welfare organisations and academics alike, joined the hackathon. Eight teams were stimulated to find innovative solutions for challenging themes, that were selected based on previous discussions between stakeholders, namely: How to drive the use of AI in chemical risk assessment?; To predict or protect?; How can we secure human health and environmental protection at the same time?; and How can we facilitate the transition from animal tests to full implementation of human-relevant methods? The hackathon ended with a pitching contest, where the teams presented their solutions to a jury. The most promising solutions will be presented to regulatory authorities, industry, academia and non-governmental organisations at the next ONTOX Stakeholder Network meeting and taken up by the ONTOX project in order to tackle the above-mentioned challenges further. This report comprises two parts: The first part highlights some of the lessons learnt during the planning and execution of the hackathon; the second part presents the outcome of the ONTOX Hackathon, which resulted in several innovative and promising solutions based on New Approach Methodologies (NAMs), and outlines ONTOX’s intended way forward.
Keywords
Introduction
ONTOX (ontology-driven and artificial intelligence-based repeated dose toxicity testing of chemicals for next generation risk assessment) is an EU Horizon 2020-funded project (https://ontox-project.eu/) that will deliver a strategy to create innovative New Approach Methodologies (NAMs) to predict the systemic, repeated dose toxicity effects of any type of chemical that, upon combination with tailored exposure assessment, will enable human risk assessment. The project focuses on six specific NAMs addressing adversities in the liver, kidneys and developing brain. The NAMs will consist of an ontology-driven and Artificial Intelligence (AI)-based computational system, fed by available physiological human data and targeted in vitro and in silico testing.
The first ONTOX Stakeholder Network Meeting took place on 13–14 March 2023. The discussion centred on identifying specific challenges, barriers and drivers in relation to the implementation of NAMs and probabilistic risk assessment (ProbRA). The participants identified several issues to be addressed toward the full implementation of NAMs and ProbRA in chemical risk assessment, and ranked them according to their level of complexity. It was concluded that there is a continued need for stakeholder engagement. The full report of the first ONTOX Stakeholder Network Meeting was published in Alternatives to Laboratory Animals (ATLA) in January 2024. 1 Based on the discussions at this first meeting, ONTOX identified the need to organise a ‘hackathon’ to tackle the most difficult challenges.
Hackathon as a working method in ONTOX
Why a hackathon?
During the first ONTOX Stakeholder Network Meeting, the invited stakeholders confirmed ONTOX’s proposal that new perspectives should be firmly included when addressing challenging obstacles in an innovative environment.
The purpose of the hackathon was to address so-called ‘wicked problems’. 2 Some of these problems are difficult but can be solved, while others are controversial or require expertise from outside the ONTOX consortium — thus, they are potentially suitable for a ‘pressure cooker’ approach, as envisaged in a ‘hackathon’. The organisation of a hackathon was consequently considered to be the most suitable way forward, in order to address identified obstacles in the ONTOX project.
Hackathons are short-term and intense events, ranging in size from small to huge, in terms of the number of participants. Traditionally, hackathon groups consisted solely of computer programmers who needed to solve a technological problem or produce a prototype. However, the scope of hackathons has broadened, and nowadays diverse interdisciplinary groups gather to solve a defined problem that meets a specific need, as determined by the event organiser. 3 The ONTOX partners contributed their own expert knowledge and experience, as well as their experience from other hackathons.
Selection of the overall theme and issues to address
Initially, the focus was on selecting an overall theme and the related challenges to be addressed during the ONTOX Hackathon. Conclusions from the first ONTOX Stakeholder Network Meeting created the foundation for this focus, including that the present modus operandi for testing chemicals is not sufficient to secure the better protection of (European) citizens from chemicals in the future.
According to the World Health Organisation (WHO): “More than 160 million chemicals are known to humans. About 40,000 to 60,000 of them can be found in commercial applications; 6,000 of these account for more than 99% of the total volume of chemicals in commerce globally. In 2017, the chemical industry was the second largest manufacturing industry in the world, and the trend is going upwards — chemicals sales are projected to almost double from 2017 to 2030”. 4 Approximately 20,000 chemicals are registered in the EU under the REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) regulation — however, the number of chemicals is constantly increasing, and people and the environment are constantly exposed to a mixture of chemicals that could potentially lead to health effects. 5
NAMs are alternatives to traditional animal testing for toxicity. They include in vitro, in chemico and in silico computational methods — which can be used alone or in combination — and they have the potential to be quicker, cheaper and more reliable than traditional animal-based methods. 6 There is a growing interest in the use of NAMs to assess the impact of chemicals on human health and the environment, and the transition from traditional in vivo (animal) testing to NAMs is considered necessary.
The ONTOX Hackathon addressed the use of AI in toxicology and in animal-free testing methods (e.g. NAMs), and aimed to pursue a combination of these two. The overall idea in the ONTOX project is to exploit the use of AI in NAMs to the utmost extent, within the ONTOX approach to ProbRA. AI is, however, considered as an accelerator — i.e. a co-pilot — rather than a standard-alone strategy. The purpose of the hackathon was a reality check on the development and implementation of such an ‘accelerator’ strategy. This is reflected in the title of the hackathon event, as well as in the underlying issues put forward to the participants. ONTOX previously identified the following four overall issues/obstacles which are considered important, not only for ONTOX, but also in general for product development and safety assessment:
Organisation of the ONTOX Hackathon
The hackathon participants were, by intent, people from within and outside the risk assessment domain and with a mix of genders, nationalities and experiences. The group included more-senior participants, students and early career researchers (primarily from ONTOX and the other two projects under ASPIS 7 ). This enabled an ‘out-of-the-box’ approach to solving tough challenges, and allowed the use of interdisciplinary expertise in an issue-focused format, as promoted by Heller et al.: “Issue-oriented hackathon events aim to solve broader business or social problems. […] These appeal to a wider range of participants from all walks of life and often contain more diverse teams from different disciplines. Issue-oriented events typically include between 30 and 50% non-tech participants compared to about 10 to 20% in tech-centric events”. 3
The Breakout Group participants.
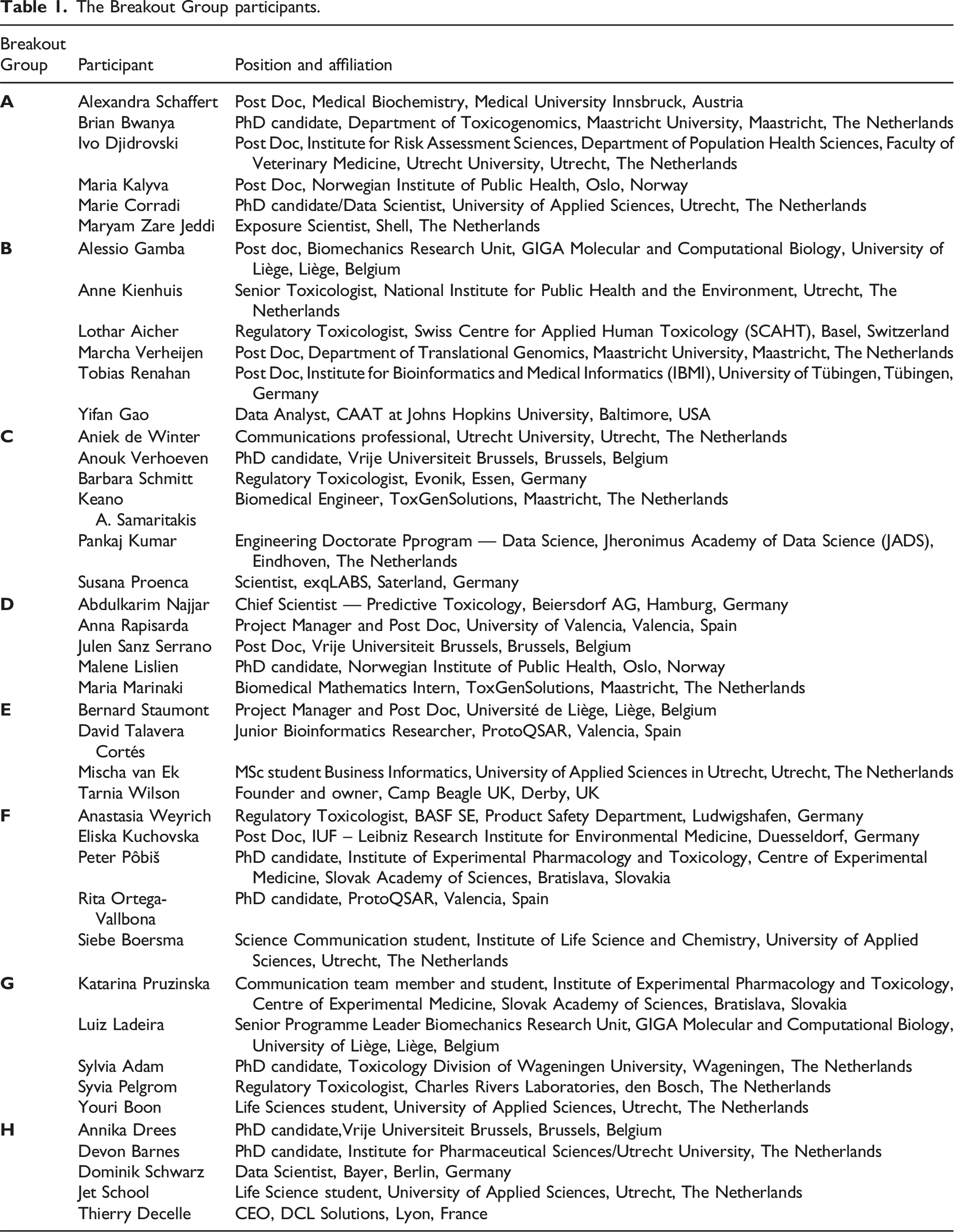
When organising the composition of each breakout group, the aim was to optimise the group diversity and take into consideration the participants’ issue preferences. In each group, early career researchers from ONTOX were included, to ensure that some knowledge of the project was available. Among the ONTOX-affiliated participants, a rapporteur was appointed within each breakout group, to ensure that the work and output from each group was formally recorded. Issues 1 and 2 were addressed by two breakout groups each, Issue 3 was addressed by one breakout group, and Issue 4 was addressed by three breakout groups. In total, 48 participants joined in the hackathon.
Detailed programme of the ONTOX Hackathon.
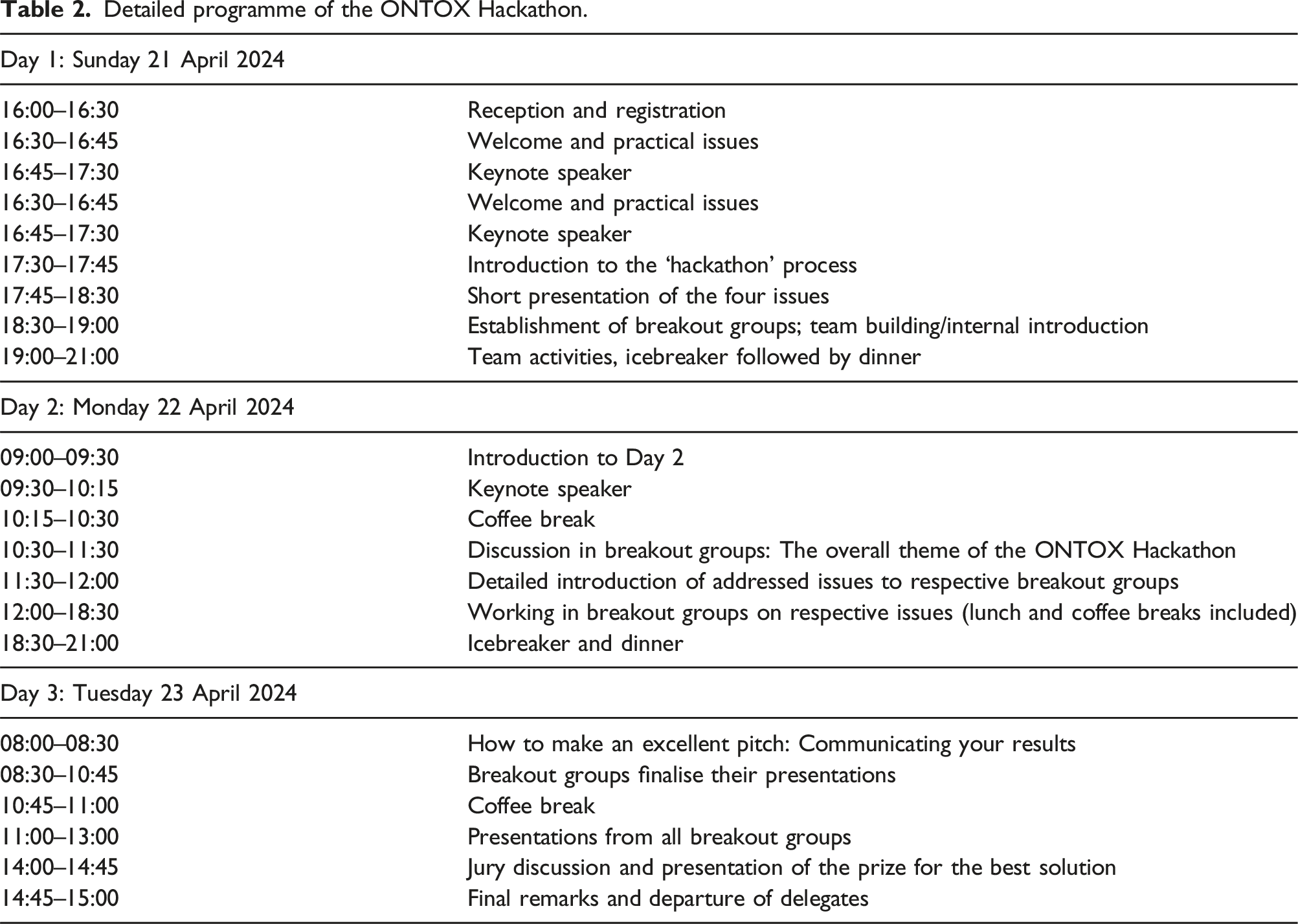
Three aspects needed to be addressed, in order to facilitate an innovative hackathon. Firstly, a venue that invited ‘out-of-the-box’ thinking and working was required. A modern university environment, with ample joint working space and an auditorium that could be used for the introduction and keynote speakers, was specified. To this end, this event was kindly hosted by the University of Applied Sciences in Utrecht (The Netherlands). Space for some parallel social activities was also available, e.g. computer games and board games addressing NAMs, networking, coffee breaks and dining (Appendix, Figure A1). Secondly, facilitators (26 in total) with different roles were assigned, to support the hackathon process (see Table 3): — Overall guidance during the hackathon was provided by two professional coaches. They introduced the hackathon as a working method, facilitated several teambuilding activities, organised breaks during the intense working sessions, intervened in the sessions whenever coaching guidance was necessary, and kept track on the timings. — All participants were given an overall food-for-thought on the idea and purpose behind the intended paradigm shift (i.e. going from traditional deterministic risk assessment using animal tests to a fully human-relevant, NAMs-based probabilistic risk assessment) by two invited keynote speakers. — For each of the four issues, an issue owner was appointed to give a short presentation to introduce the respective issues to all participants. This presentation was supplemented with a more detailed and tailored presentation to the groups that signed up for the specific issues, to provide a deeper and more detailed understanding of that particular issue. — During the hackathon, a panel of experts was available as a key resource for knowledge and feedback for the participants. All experts were available on a consulting basis for all four issues, if requested by the groups; they were entitled to react when approached, but not to act/interfere in discussions on their own initiative. The four issue owners were part of the expert panel, together with six additional experts covering different fields of expertise such as AI, data science, toxicology and stakeholder engagement. — A jury of four experts (representing academia and industry) monitored the work in progress, gave initial feedback to each of the eight breakout groups on Day 2, and examined the eight pitches on Day 3. An award ceremony was part of the hackathon conclusion, and written post-event feedback was given to all eight breakout groups. ESTIV (European Society of Toxicology In Vitro) sponsored participation in the 22nd ESTIV Congress in Prague June 2024
8
to the best pitch presenters. — Additionally, a team of ten people were responsible for covering the following areas: lessons identified/lessons learnt; coordination with coaches; communication; secretary for the jury; logistics support; video documentation and parallel activities. Event facilitators.

Lessons learnt
During the hackathon, interviews with representatives from the participants and the various facilitator groups led to the identification of a number of points that could be taken on board for the planning of future such events.
Feedback from the participants
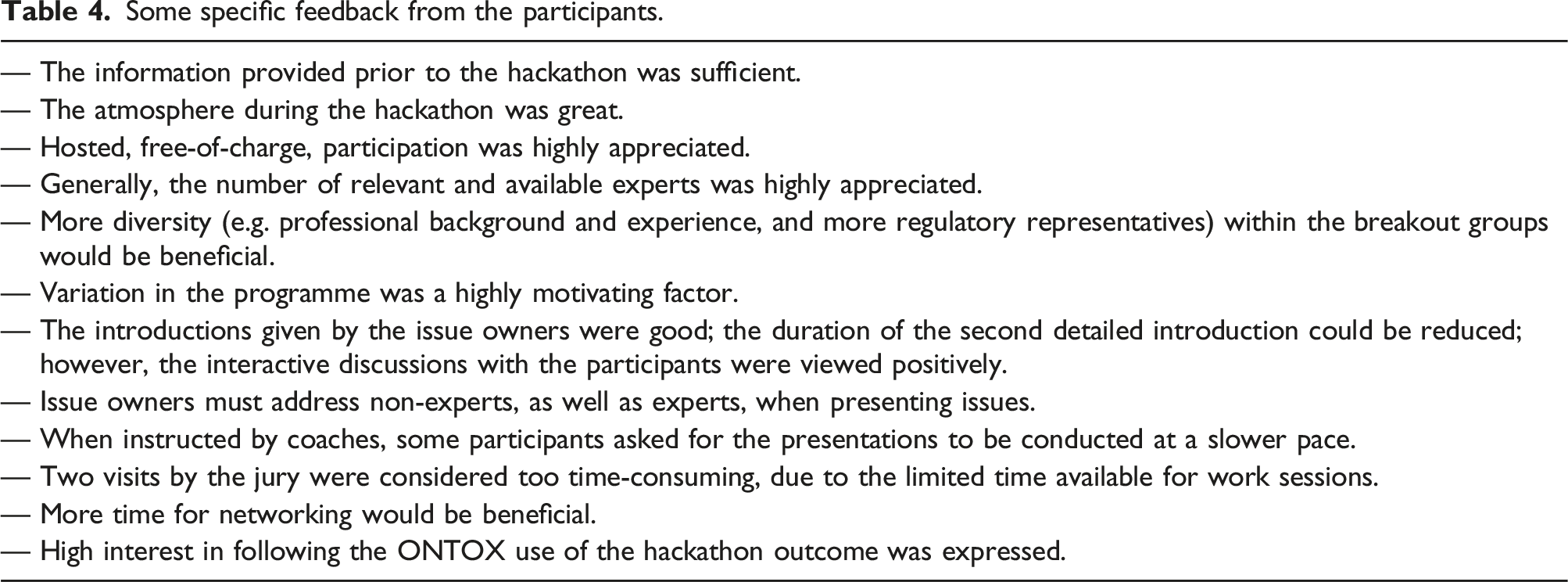
Some specific feedback from the participants.
Feedback from the professional coaches
Recommendations from the professional coaches for consideration when planning similar events in the future.

Feedback from the expert panel (including issue owners and keynote speakers)
This group expressed satisfaction with the pre-hackathon meeting for facilitators. However, it was felt that an additional meeting might be of value, as it would permit reflection on selected issues before discussing them in detail with issue owners and affiliated experts.
This group also mentioned that the duration of keynote presentations could be reduced, in order to prioritise work sessions where timing is relatively limited. One expert expressed the notion that there could be a potential disadvantage of involving participants with different approaches and experiences (i.e. experts from a range of fields, as well as laypersons without detailed scientific knowledge), and that this might negatively influence the output.
Feedback from the jury members
From a jury perspective, their presence during the entire hackathon was helpful, and the initial meetings with the respective breakout groups on Day 2 were useful for the participants, as well as for the jury preparations. However, the discussions during those meetings might have influenced the participants too much in terms of providing direction and advice with the purpose of putting them on the right track when needed.
There was a general concern that some groups tended to jump to conclusions far too quickly. Jury members suggested that future hackathon programmes should include a 2-hour brainstorming session on the first evening, followed by a full working second day and then a third day for the presentations. It might be an idea for the respective groups to have the opportunity to present their pitch to another group, as a ‘dress-rehearsal’ prior to their presentation to the jury. Dinner should preferably be arranged within the breakout groups.
Organisational take aways
From an organisational point of view, the preparation and execution of the ONTOX Hackathon required a substantial amount of time (more than a year), and this needs to be carefully considered when organising such an event. Overall, all participants and facilitators were very positive about the content and execution of the ONTOX Hackathon, with the majority finding that their participation in the event was cost-effective, both for ONTOX and for themselves as individuals.
General conclusion on the hackathon as a working method
Overall, the hackathon was identified as a very fruitful working method, when new solutions to complicated problems are required. The variety of perspectives was valuable; however, we failed to attract some of the identified expertise among the participants (e.g. politicians, policy makers, representatives from various regulatory authorities, and those from other scientific areas of interest that use AI), which might have added additional viewpoints and increased the value of the discussions.
Outcome of the ONTOX Hackathon
As already mentioned, ONTOX requested proposals on four issues from the hackathon participants. The following section provides more detailed background on each issue, the resulting proposals from the respective breakout groups, and the jury assessments.
Issue 1: How to drive the use of AI in chemical risk assessment?
Artificial Intelligence (AI) is expected to bring a wide array of economic and societal benefits to a wide range of sectors, and this includes supporting the ambition of ensuring a toxin-free environment. AI is assumed to be a game-changer, capable of utilising available big data with relevance for toxicological assessments, and doing so very rapidly, efficiently and with a very high certainty. However, to maximise explainability and confidence in the outcomes of AI-based models for chemical analysis and assessments, we need to ensure transparency in the algorithms employed — meaning that future implementation must fulfil the intention described in future regulations, e.g. the EU AI act 9 and related future national regulations. Additionally, it is necessary to find solutions for the sharing of data to create ‘big data’ as well as upholding industrial intellectual property rights (IPR) in a globally competitive world.
The participants were asked to find solutions on how to drive the use of AI in chemical risk assessment. They were expected to address the drivers (e.g. explainability, trust, transparency, etc.) and the barriers (e.g. proprietary AI, mistrust, uncertainties, etc.), not only from a European perspective but also from a global perspective/market.
Breakout Group A proposal
Identified problem
The lack of a harmonised and user-friendly platform that connects NAMs with their associated Adverse Outcome Pathways (AOPs) hinders the adoption of NAMs in chemical risk assessment.
Proposed solution
To develop the ‘NAMs Wizard’, a tool that uses an open-source large language model (LLM) to interpret user questions, search a database of NAM documentation, and link it to the AOP wiki to extract relevant key events and adverse outcomes. The proposed output of the tool would consist of a table, listing the AOP, NAM, description and reference (Figure 1). Proposal by Breakout Group A. AO = adverse outcome; AOP = adverse outcome pathway; KE = key event; MIE = molecular initiating event.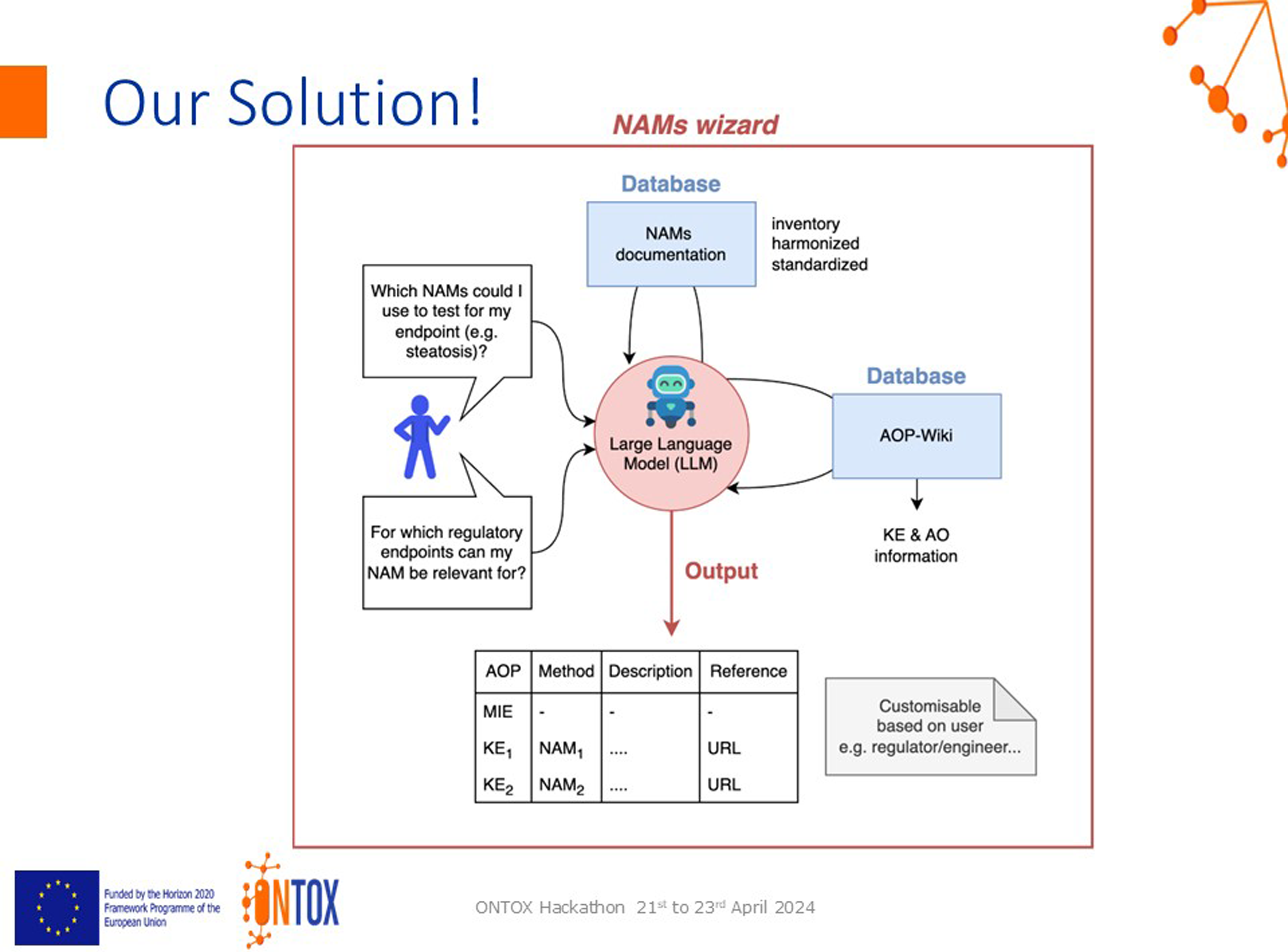
The jury concluded that the solution was pragmatic, very feasible and executable. It is a good combination of high tech and a viable problem, with a clear problem statement and a concrete, do-able solution using AI — the group already worked on a prototype (it was the only group that did this!).
Potential improvements included expanding the wider applicability, and clearly defining the roadmap and the long-term vision for the product, including how to keep it up to date. A little more novelty/out-of-the-box thinking was requested.
Breakout Group B proposal
Identified problem
The need to understand the toxicity and associated risks of mixtures — which is unfeasible with animal testing.
Proposed solution
To use AI models to predict the toxicity of mixtures, starting with a proof-of-concept study using pharmaceutical databases of human data on the adverse effects of taking multiple medicines. It was proposed that the data would be analysed with interpretable classic Machine Learning models, such as Random Forest, Logistic Regression or XGBoost, and compared to deep learning models (Large Language Models). The model performance would then be improved, and the simplest and most effective version selected. Learning could then be extrapolated to other, less data-rich domains such as pesticides (Figure 2). Proposal by Breakout Group B. LCA = lifecycle assessment; IATA = integrated approach to testing and assessment; SSbD = safe and sustainable by design; AOP = adverse outcome pathway.
The jury decided that the solution was very relevant and addressed a big problem worth solving — chemical mixture toxicology. It is a very ambitious vision but well defined, with a clear need for regulators to get involved to see the added value of AI. The solution addresses a very important problem in toxicology and the problem is clearly outlined. The group demonstrated a sense of urgency with a clear solution, and has started to mine data (they have worked on the solution).
Potential improvements included: more details on the solution, a more detailed roadmap with timelines, suggestions on how to get from pharmaceutical data to chemicals, and a step-by-step process to reach the solution. The presentation did not fully convince the jury of the feasibility of this proposal, due to the time limit for the pitch being exceeded.
Issue 2: To predict or protect?
Traditionally, toxicological testing has been hazard-oriented. It has been about predicting whether a chemical substance is toxic or non-toxic, following exposure. This approach has not included the possibility that chemical substances might be used in different applications and, therefore, that the probability of an adverse outcome from an exposure might differ in view of this.
If there is no exposure associated with the anticipated use of a chemical, then there is no significant risk. If that is the case, it is possible to reduce the number of tests needed for chemicals that will be used in these specific ‘no exposure’ applications. However, for those chemical substances where there is possible risk due to a certain level of exposure during their use, it is necessary to assess the chemical hazard probability. The next step is then to determine the probability of the risk (where risk = hazard × exposure). If the hazard probability is low (with low uncertainty), it can be concluded that there is minimal risk associated with the use of that specific chemical in that specific application. Conversely, if the hazard probability is high (with low uncertainty), then it can be concluded that there is a high level of risk associated with the chemical’s use, which would require risk management. Further, if the hazard probability is characterised by moderate probability and high uncertainty, more testing with human-relevant tests, such as NAMs, would be required.
The participants were asked to discuss whether it would make sense to change from the current ‘prediction’ approach outlined above, to a ‘protection’ approach based on the evaluation of potential adverse outcomes from an exposure in a specific application. If a transition from ‘prediction’ to ‘protection’ is deemed desirable, the solution should include ideas on how to make the transition, address the drivers (e.g. risk communication and risk perception) and the barriers (e.g. uncertainty and risk aversion), and consider the extent to which safety must be proven.
Breakout Group C proposal
Identified problem
Bridging the gap between predicting the adverse outcome (what happens at a molecular initiating event) and protection (preventing the adverse outcome) by exposure below a threshold (Figure 3(a)). Proposal by Breakout Group C. AOP = adverse outcome pathway; KE = key event; KER = key event relationship; MIE = molecular initiating event.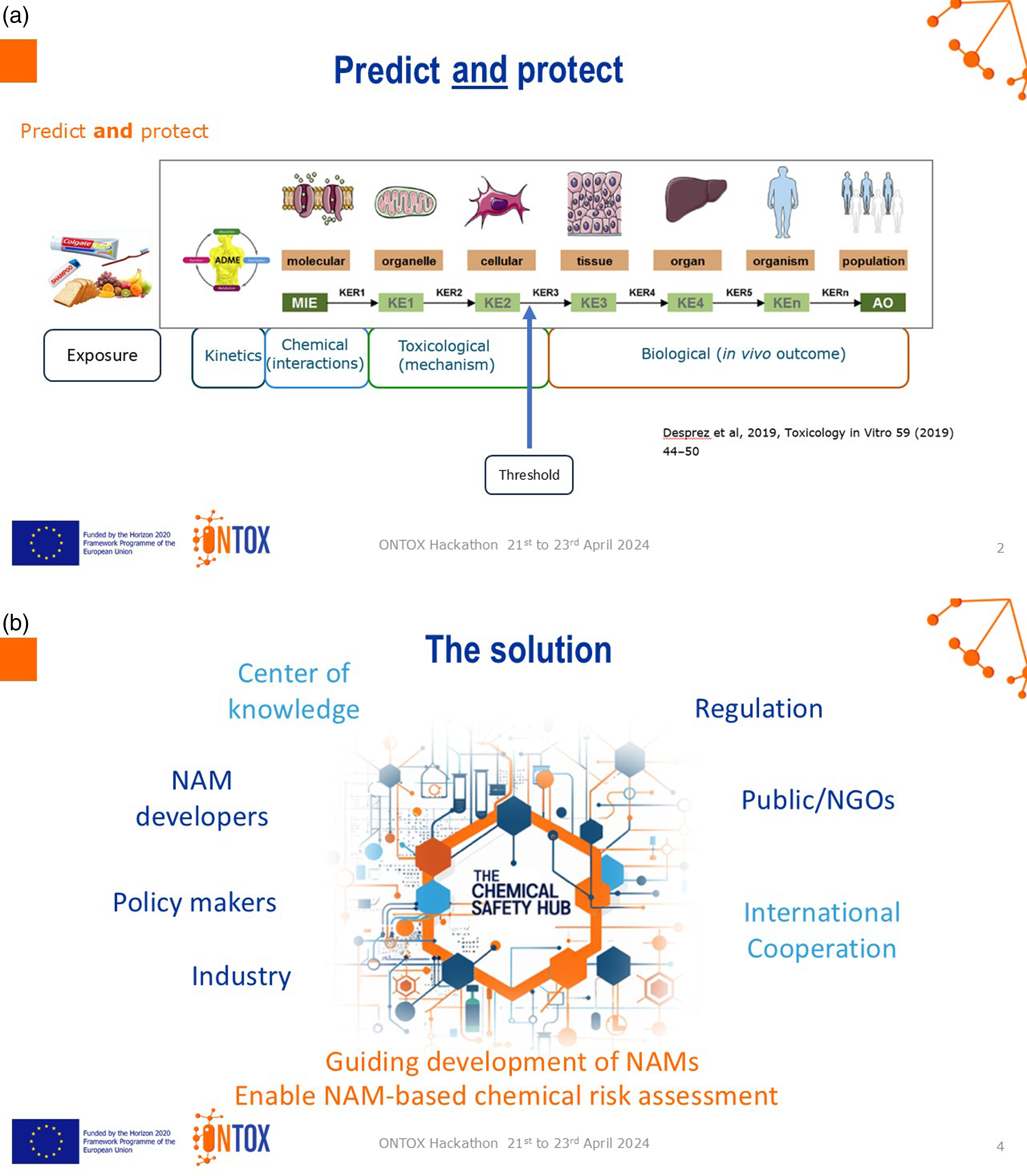
Proposed solution
To establish a European ‘chemical safety hub’ to centralise knowledge on safety assessment and NAMs. The hub connects regulators, policymakers, NAM developers, industry, international groups, public and non-governmental organisations. It identifies needs for NAM development, enables NAM-based risk assessment and communicates the findings to the public (Figure 3(b)).
The jury determined that it is a tangible solution focused on communication, impact and NAM development. The chemical safety hub brings all the key players to the same table, promoting collaboration. This will help with sustainability and long-term funding, facilitating the continuity of different projects.
Potential improvements included initiatives to avoid overlap with existing hubs or similar initiatives, and clearly defining this solution to differentiate it from other existing solutions. The feasibility of the proposal was questioned because it was uncertain how to get everybody involved and the approach implemented at different locations. A little more novelty/out-of-the-box thinking was requested.
Breakout Group D proposal
Identified problem
Chemical mixture risk assessment is needed, but it is challenging.
Proposed solution
A predictive approach that also protects, based on the use of exposure and hazard data on existing chemicals to guide future actions. If exposure data only are available, then the threshold of toxicological concern (TTC) should be used; if hazard data only are available, then Physiologically-based Pharmacokinetic (PBPK) modelling should be used. If neither are available, then AI should be used to define the desired chemical properties, generate candidates, and select safer compounds (Figure 4). An example of the Safe by Design approach is outlined below, for Chemical U: 1. Start by defining the properties of Chemical U. 2. Ask AI: To list substances with the desired properties (200,000 hits). 3. Ask AI: To filter for those that can be synthesised (50,000 hits). 4. Ask AI: To screen for involvement of the substances in AOPs. 5. Ask AI: To classify the substances according to the likelihood of involvement in AOPs as high/medium/low. 6. Select your Top 10. 7. Improve the weight-of-evidence. 8. Go-on! Synthesise and produce your Safe by Design Chemical U! Proposal by Breakout Group D. DNT = developmental neurotoxicity; PBPK = physiologically-based pharmacokinetic modelling; TTC = threshold of toxicological concern.
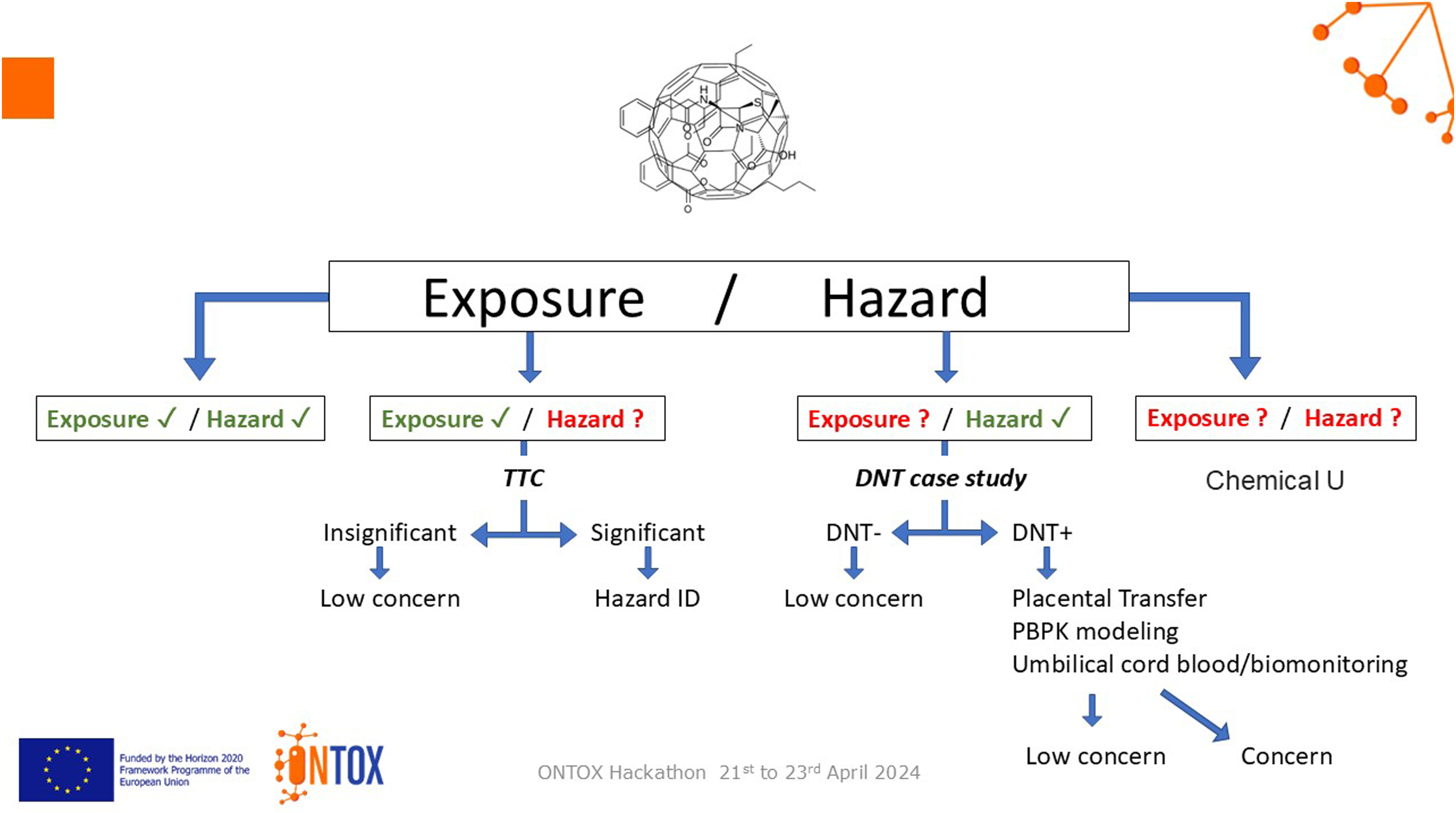
Potential improvements include ensuring broad applicability, and performing more feasibility assessments on the solution. Some questions around future deployment need to be answered, such as funding, IPR and AI strategy. The proposal appears to be similar to existing solutions. Developing the Safe by Design/AI application further would have been interesting.
Issue 3: How can we secure human health and environmental protection at the same time?
The EU is striving to be the world’s first climate neutral region, and has agreed on a future strategy, The European Green Deal. 10 This strategy describes how to protect the environment while ensuring healthy food for the population, as well as safe and sustainable transport, energy and industry. The question is whether securing health and environmental protection are opposing interests, or whether they are in fact converging objectives. The European Green Deal addresses both human health and the protection of the environment from a holistic perspective. However, there might be areas of interest or some other aspects, where a clear answer on potential contradictions is not forthcoming. As an example: Is it possible to produce healthy food in an economically sustainable way without any negative effect on the environment? As a result of such dilemmas, what should be prioritised or how should mitigation be approached?
The participants were asked to discuss how to prevent a situation whereby securing human health and environmental protection results in contrary interests. They were expected to address the drivers (e.g. safer and more sustainable by design, tools to reduce costs in production) and the barriers (e.g. requirements for commercial openness hampering business — i.e. IP-related issues, competence, and expertise — and potentially excessive costs), and to identify relevant stakeholders for involvement.
Breakout Group E proposal
Problem
Driving the use of AI in risk assessment while securing human and environmental health.
Solution
To develop a NAM Index to score and classify companies according to their use of NAMs, animal testing, transparency, etc. It is proposed that the index would be published on a designated website and used on product labelling, to raise consumer awareness. It would help bring stakeholders together and incentivise the transition to NAMs. Assessments would be performed by a third party, which would require the creation of a foundation to identify and invite independent third-party experts to perform this task (Figure 5(a)). The use and dissemination of the index would thus comprise: — labelling on relevant products, aimed at end-users, in the form of a green, orange or red label (see Figure 5(b)). — a mobile app for the public and end-users. — a report with details on the scores, made available to all stakeholders (including regulators), and which would also be publicly available. Proposal by Breakout Group E.

Potential improvements included the compatibility with other labelling and clear timelines for implementation. More details on the development process are needed and, to make it feasible, this process should be focused on a step-by-step approach. However, the feasibility was questioned due to a lack of answers to the following question posed by the jury: “There are already so many labels. How is this creative, and where will we get the data to build it?”
Issue 4: How can we facilitate the transition from animal tests to full implementation of human-relevant methods?
The EU citizens’ initiative, Save cruelty-free cosmetics — commit to a Europe without animal testing, has pressured the EU Commission to accelerate the implementation of non-animal and human-relevant testing of chemical substances in the future. 11 However, scepticism related to NAMs — which include the use of AI, models based on human cell culture (in vitro test methods), and computer-simulated models (in silico test methods) — remains, with claims that uncertainty is a specific problem related to the use of NAMs. Nonetheless, traditional laboratory tests based on the use of living animals (in vivo test methods) do have similar uncertainties, and there are several examples of adverse effects caused by chemical exposure which are specific to certain species (e.g. comparing rats versus human beings with regard to thalidomide causing malformations in babies), as well as species-specific chemical exposure-related diseases such as some neurodegenerative diseases, cancers and stroke.
The present traditional testing capacity is insufficient and the acceptance of animal use for testing is decreasing. However, the industry has experienced several situations where the present regulations have required an increase in the number of animal studies before chemicals are approved for use in specific applications (pharma, food, cosmetics, biocides, etc.). Alternative test methods (e.g. NAMs) are fully accepted in relatively few areas, and regulatory authorities have traditionally required validation of NAMs — which includes benchmarking with in vivo studies — before acceptance of these new methods. The assumption for this issue is that the implementation of NAMs by industry is driven by full acceptance from regulatory authorities.
The participants were asked to discuss how to drive the transition from animal-based chemical testing to the full implementation of human-relevant methods (e.g. NAMs). They were expected to address the drivers (e.g. aligning current incentives to apply a probabilistic approach which includes assessing exposure, identifying hazard, and perform risk assessment) and the barriers (e.g. are current guidelines and/ political agendas hampering the transition?). They were also asked to consider the following question: If we need a risk assessment paradigm shift, how will it be possible to enforce the equal validation of traditional animal in vivo tests and NAMs, with regard to the uncertainties inherent in both?
Breakout Group F proposal
Identified problem
The reluctance of European regulators to accept NAMs for risk assessment.
Proposed solution
To create the ‘Ultimate Sandbox’ — a safe space for NAM developers, users and regulators to discuss approaches. Attempts should be made to persuade organisations such as ECHA to join, via stakeholder pressure and an EU Commission mandate. A two-part ‘sandbox challenge’ should be planned for 2026, comparing traditional animal-based versus NAM-based assessments of chemicals selected by ECHA; a jury would evaluate the results (Figure 6). The proposed ‘ECHAllenge’ agenda is as follows: Proposal by Breakout Group F. ECHA = European Chemicals Agency.
Part 1 (2 February 2026): — Opening celebratory ceremony; — ECHA provides a list of test chemicals; — Preliminary data (in silico approaches).
Part 2 (2 April 2026): — First pitch: Traditional approaches (in vivo tests); — Second pitch: NAMs approaches; — Sandbox feedback (from the jury).
The jury assessed the solution as being very specific, pragmatic and a new way to involve ECHA via small steps. It is an important problem to solve, and the idea of a ‘sandbox playground’ for idea generation and testing was supported — getting ECHA to the table is important, and the sandbox concept is a great idea.
Potential improvements included solutions on how to build the sandbox and keep it up to date. Uncertainties and feasibility must be addressed. More focus on the sandbox solution and less focus on ECHA was recommended. A little more novelty was requested.
Breakout Group G proposal
Identified problem
The need for education on NAMs, to facilitate the transition from animal testing.
Proposed solution
To target science communication and education toward the general public, especially young people, leveraging their high social media engagement (Figure 7(a)). Ideas include: establishing an ‘Observatory for the Transition from Animal Testing’ to track progress; disseminating engaging NAM content through channels such as Instagram reels, video games, and TV (Figure 7(b)) — people spend an average of six hours and 40 minutes per day on their various screens; and developing an information campaign, targeting the general public by using, for example: — TV shows, films, series, etc.; — Interviews; — Podcasts; — YouTube; — Social media; — Games. Proposal by Breakout Group G.

Potential improvements included a structured plan to develop the solution further, as well as defining who would be responsible for its implementation. It was considered that it might be slightly passive in its approach, with the jury asking: How will the scheme really change mindsets and educate people?
Breakout Group H proposal
Identified problem
Facilitating the transition from animal testing to human-relevant methods (Figure 8(a)). Proposal by Breakout Group H.
Proposed solution
To develop a web portal to connect stakeholders (public, industry, regulators, academics). The portal should enable engagement (e.g. through debate), fundraising, idea sharing and regulatory guidance, and promote NAM development with modern audience outreach (Figure 8(b)).
The jury assessed that the proposal is easy to use for all stakeholders, reasonably easy to execute and highly feasible. It is low-cost with regard to its implementation, with potential high impact. Consequently, it is deemed to be an efficient source of funding for future NAM development.
Suggested improvements included addressing in more detail the question of ‘how to reach the audience’. The task was judged to be huge, and it was put forward that the focus needs to be on a step-by-step approach. Different regulators would have different views, and this issue must also be addressed. More details on the presented solution would have been useful, including a detailed plan for how to kick-start the initiative, which should have been worked out. The group was also asked how they would recognise when a cultural change has been achieved.
Conclusions on the hackathon outcome
The jury concluded that all of the solutions were highly relevant for ONTOX, and the high level of involvement of the young investigators was recognised and appreciated. The jury’s evaluation of each solution aimed to address the following aspects: — The solution is clear and well-defined. — The solution is achievable within the next five years. — The solution pushes creativity but remains realistic. — The achievement of the solution is measurable. — The solution is timely. — The solution is broadly applicable. — The young investigators were involved in creating the solution. — What are the strengths of the solution? — What should be improved? — The performance of the pitch presenter.
Yifan Gao (representing Breakout Group B), was judged to have presented the best pitch; the prize was a hosted participation in the ESTIV 2024 Congress in Prague, Czech Republic (Appendix, Figure A2). The three best solutions were awarded small souvenirs from Utrecht. The third prize was awarded to Breakout Group H (Issue 4), for the ‘NAMs kick-starter’ idea. Their solution was assessed as being easy to use for all stakeholders, an efficient and transparent source of funding for future NAM development, and potentially high impact and readily implementable. The second prize was awarded to Breakout Group B (Issue 1), for the ‘mixtox.ai’. Their solution was assessed as very relevant and ambitious, well-defined and with high added value. It was a timely solution, and prototype development was ongoing. The winning solution was awarded to Breakout Group A (Issue 1) for the ‘NAMs Wizard’ (Appendix, Figure A3). Their proposal was assessed as being an effective combination of high-tech methods, and a viable and innovative solution.
General conclusions and next steps
The format of a hackathon stimulates innovative ideas, especially for so-called ‘wicked problems’. 2 These types of problems are complex and difficult to solve, because they often expose underlying societal issues. For example, the ONTOX Hackathon is not only about the development and implementation of complicated technical solutions (such as AI) for probabilistic risk assessment, but also — perhaps more importantly — it is about how we, as a society, think about safety. What level of uncertainty are we willing to accept in a risk averse society? How can we trust something we do not fully understand? There are also dilemmas behind these challenging issues, such as wanting to respect nature and animal welfare, while simultaneously wanting to protect our health to the maximum extent. In this hackathon, we brought together people with different expertise, ages, and from both public and private domains. These combinations of people have led to new ideas, some closely aligned with the technological developments within ONTOX, such as the use of AI to predict mixture toxicity and to design safe chemicals, but also ideas more focused on the involvement of the end-users, the general public and stakeholders who are not directly represented in the ONTOX consortium.
In general, the hackathon participants considered that the programme, facilities and support were suitable. Some adjustments proposed by the participants will be considered during the planning of future similar events. With respect to the definitions of the four issues, we learnt that some were too-openly worded and, consequently, it took longer than expected before the team could start hacking the problem. It is a balance between, on the one hand, encouraging out-of-the-box thinking and increasing the chance of truly surprising and innovative ideas and, on the other hand, somewhat guiding the direction of the solutions so that their feasibility can be better assessed. The level of implementability of the solutions varied between the teams. What could help is rapid prototyping, which is a proven method often used in the hackathon set-up to provide more insight into the potential impact that an idea could have.
The outcome of the hackathon will be analysed and managed by ONTOX in the near future, enabling us to present the most promising solutions at the next ONTOX stakeholder network meeting. ONTOX will follow-up on a couple of these proposals, which have been assessed as having the potential to support the implementation and/or sustainability of the ONTOX AI-supported ProbRA approach.
Footnotes
Acknowledgements
ONTOX expresses a great thank you to all participants and facilitators for a very enthusiastic involvement (Appendix, Figure A4). The credits for the photographs used by Breakout Group H for their presentation slide (Figure 8(a)) are: bus couple, from the EC website (© iStock); man in suit, model photograph on bucco.us; grad couple, Cumberland University, Lebanon, TN, USA (https://www.cumberland.edu/academics/); meeting table (![]() ).
).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: European Union’s Horizon 2020 Research and Innovation programme, 963845. This work was performed in the context of the ONTOX project (https://ontoxproject.eu/) that has received funding from the European Union’s Horizon 2020 Research and Innovation programme under Grant Agreement No. 963845. ONTOX is part of the ASPIS project cluster (![]() ). The Hackathon was made possible in part by a financial contribution from the VHP4Safety project (NWA grant 1292.19.272).
). The Hackathon was made possible in part by a financial contribution from the VHP4Safety project (NWA grant 1292.19.272).
Correction (February 2025):
Disclaimer
This output reflects only the authors’ view, and the EU cannot be held responsible for any use that may be made of the information contained therein. Proposed initiatives expressed in this publication do not necessarily represent the regulatory position of the institutes or organisations whose representatives participated in the ONTOX Hackathon. All participants have given consent to be photographed or recorded during the ONTOX Hackathon event for promotional purposes.
