Abstract
This study introduces a novel in vitro methodology that employs the 3-D reconstructed tissue model, EpiOcular, to assess the irritation and phototoxicity potential of medical devices and drugs in contact with the eye. Our study evaluated diverse test materials, including medical devices, ophthalmological solutions and an experimental drug (cemtirestat), for their potential to cause eye irritation and phototoxicity. The protocols used in this study with the EpiOcular tissue model were akin to those used in the ultra-mildness testing of cosmetic formulations, which is challenging to predict with standard in vivo rabbit tests. To design these protocols, we leveraged experience gained from the validation project on the EpiDerm skin irritation test for medical devices (ISO 10993-23:2021) and the OECD TG 498 method for photo-irritation testing. The predictions were based on the tissue viability and inflammatory response, as determined by IL-1α release. By developing and evaluating these protocols for medical devices, we aimed to expand the applicability domain of the tests referred to in ISO 10993-23. This will contribute to the standardisation and cost-effective safety evaluation of ophthalmic products, while reducing reliance on animal testing in this field. The findings obtained from the EpiOcular model in the photo-irritation test could support its implementation in the testing strategies outlined in OECD TG 498.
Introduction
Vision is often considered the most vital of our senses, profoundly impacting our quality of life. The eyes may be exposed intentionally or accidentally to various products, including pharmaceuticals and medical devices. Like most medical device fields, ophthalmic devices are either diagnostic (e.g. retinal imager) or therapeutic (e.g. retinal prostheses, lasers, stents and intraocular lenses (IOLs)). Therapeutic ophthalmic medical devices and ophthalmological drugs are intended to maintain or restore vision, or relieve a condition. Medical devices operate through physical, mechanical, or sometimes electrical means. Importantly, they do not rely on a chemical action within or on the body to achieve their primary intended purpose.
On the other hand, drugs typically work at a molecular or biochemical level. They interact chemically with the body’s cells, tissues or organs to elicit a response aimed at treating, preventing or diagnosing diseases. Their effects are often systemic, meaning that they can affect the entire body, depending on their nature and how they are administered.
This broad spectrum of ophthalmic medical devices and drugs means that standardising the evaluation of their safety and efficacy is a distinct challenge. This challenge, stemming from a lack of harmonised, robust testing protocols, not only escalates testing costs but also introduces delays in regulatory approvals. Addressing these inconsistencies and harmonising testing approaches is crucial for advancing the field, and ensuring the timely availability of safe and effective ophthalmologic medical devices. 1
In the current feasibility study, we explored whether the reconstructed human cornea-like model, EpiOcular, can correctly predict the irritation and photo-irritation potential of some selected medical devices and pharmaceuticals. The protocols used in this study were akin to those used in ultra-mildness testing of cosmetic formulations, which is challenging to predict with standard in vivo rabbit tests. The predictions were based on the tissue viability and inflammatory response, as determined by IL-1α release. To design the protocols, we built on the knowledge and experience obtained in the development and validation of reconstructed 3-D epidermal human models for biocompatibility testing included in the International Organization for Standardization (ISO) 10993-23:2021 and the Organisation for Economic Co-operation and Development Test Guideline (OECD TG) 498 on phototoxicity.2–5
Materials and methods
The test system
In this study, we utilised an in vitro 3-D reconstructed cornea-like tissue model, the EpiOcular (OCL-200), which is produced by MatTek Life Sciences (MatTek IVLSL, Bratislava, Slovakia) under Good Manufacturing Practice (GMP) guidelines. These tissues are constructed from non-transformed human epidermal keratinocytes (NHEK) sourced from individual donors. During manufacturing, the cells and models undergo rigorous screening for infectious disease agents, yeast, fungi and bacteria, including mycoplasma.
6
The EpiOcular tissues (see Figure 1) closely resemble the morphological and barrier properties of the human cornea. Their structure comprises a layer of organised basal cells, topped with five to six layers of superficial cells that gradually flatten toward the apical surface.
6
The tissues are produced in a GMP-compliant environment to guarantee high reproducibility, and each tissue lot undergoes rigorous quality assessment through quantitative quality control (QC), including barrier property testing and histology evaluation procedures. Only lots that meet established QC standards are dispatched. The EpiOcular tissue model is a validated and regulatory accepted test system under OECD TG 492 for the eye irritation testing of chemicals.6–8 Histology of the reconstructed human tissue cornea-like model, EpiOcular, after haematoxylin & eosin staining. Scale bar = 100 μm.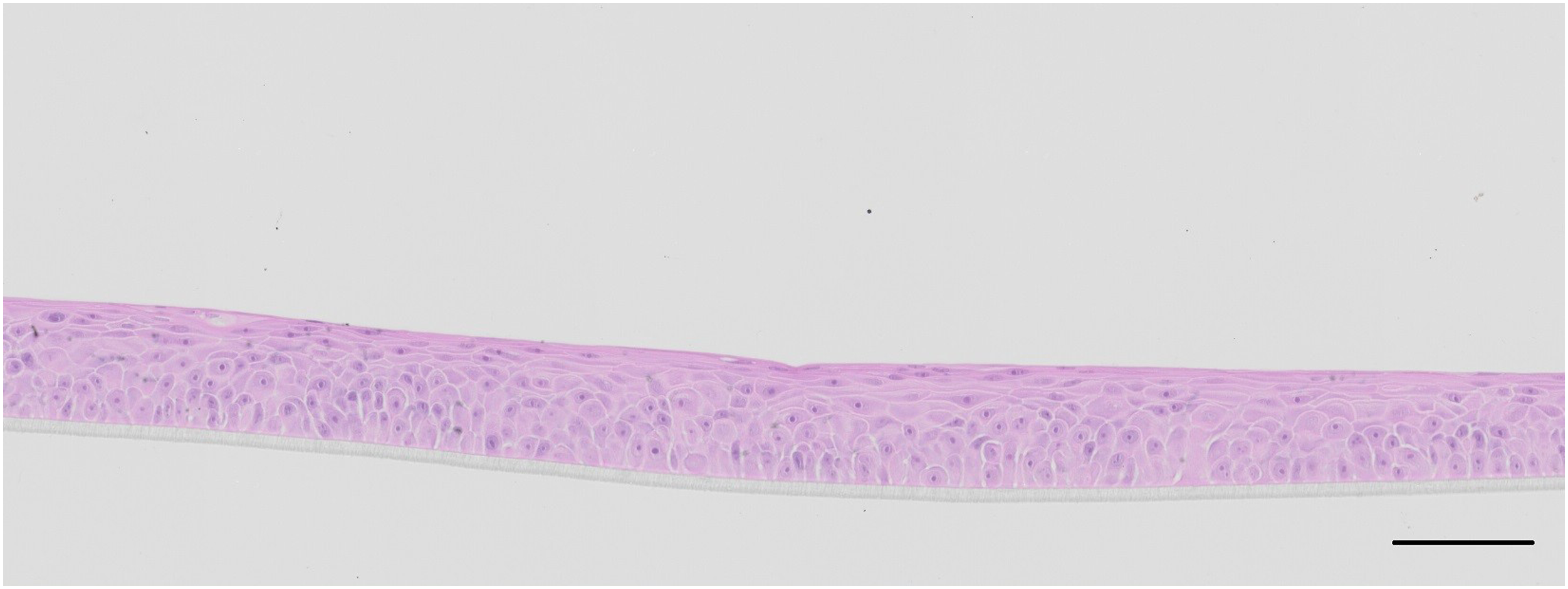
The test materials
Procuring test materials to help develop a highly sensitive, medical device-oriented protocol for eye irritation testing posed a significant challenge, primarily due to the unavailability of medical devices or biomaterials with irritating properties. Understandably, products suspected of causing such problems are usually quickly withdrawn from the market. Therefore, we expanded our sample pool to include eye drops and contact lens-cleaning solution (classified as medical devices) and non-prescription pharmaceuticals, anticipating some positive reactions due to the potential presence of active substances known to mildly irritate sensitive individuals. Thanks to a collaboration with two biotechnology companies (BioSurfaces LLC, USA and InoCure s.r.o., Czech Republic) we acquired novel electro-spun polymer scaffolds featuring various surface characteristics and fibre compositions. These electro-spun materials have diverse applications, from vascular grafts and perianal fistula plugs, to cell chambers for delivering bioactive materials. 9 The Polymer Institute of the Slovak Academy of Sciences (SAS) provided samples of medical grade polydimethylsiloxane (PDMS) and PDMS loaded with quantum dots (PDMS-QD), an experimental self-decontaminating material.10,11 PDMS is commonly used in ophthalmology for IOLs, contact lenses, corneal onlays and corneal epithelial pocket formation systems. 12 The inclusion of two types of IOLs, which are classified as medical devices, complemented this set. The Dr Vesely Očná Klinika (Eye Clinic), Slovak Republic, kindly provided these lenses.
Additionally, our experimental framework incorporated cemtirestat, an experimental drug developed by the SAS. Cemtirestat is an aldose reductase inhibitor and antioxidant, potentially capable of preventing cataracts in diabetic patients. 13 For the phototoxicity testing protocol, we selected chlorpromazine hydrochloride (CPZ) as a positive control material, because it accumulates in the eyes and often causes side effects as a result of its phototoxic nature. This well-reported characteristic makes it an ideal positive control for in vitro phototoxicity tests. 14
Materials used in the development of the protocol.

Test sample preparation
The electrospun scaffolds and IOLs were extracted in accordance with ISO 10993-12:2021, by incubating for 24 hours at 37°C in a water bath with shaking. 5 Saline and sesame seed oil were used as the extraction solvents, and the extraction ratio of solvent to material being extracted was chosen based on the guidance provided in Table 1 of ISO 10993-12:2021. Eye drops and contact lens solutions were tested undiluted. IOLs were also tested in direct contact with the EpiOcular tissue. Cemtirestat was diluted in Dulbecco’s phosphate-buffered saline (DPBS) without Ca2+ and Mg2+, and tested at the assumed therapeutic concentrations indicated by the drug developer (0.2 mg/ml and 2.0 mg/ml).
Triton™ X-100 at 0.3% w/v (supplied by MatTek in its final concentration) was used as a positive control in the eye irritation protocol. CPZ was used as the positive control in the phototoxicity testing protocol, to demonstrate the correct response of the EpiOcular tissue model to a phototoxic chemical. Five concentrations of CPZ were tested, in order to obtain dose–response curves for the irradiated and non-irradiated samples in the experiment, as suggested in OECD TG 498. 4 DPBS was used as the negative control.
Protocol for assessing irritation potential of medical device extracts and pharmaceuticals with low eye irritation potential with the EpiOcular tissue model
The protocol described in this study is based on insights gained through the data analysis of the MatTek EpiOcular ‘ET-50’ and ‘Sub-Draize’ eye irritation protocols, which are used by several cosmetic companies to evaluate the ultra-mildness of their products.15–17 In the Sub-Draize protocol, materials classed as non-irritating caused no significant decrease in 3-D EpiOcular tissue model viability, even after extended exposure. A similar approach to predicting the low irritation potential of medical devices was used when developing the ISO 10993-23:2021 protocol for skin irritation testing.3,18
Upon receipt of the EpiOcular test kit containing 24 inserts with reconstructed cornea-like tissue models, the inserts were visually inspected and transferred into 6-well plates containing 1 ml of the culture medium provided by the manufacturer. The plates with tissues were placed into a humidified incubator (5 ± 1% v/v CO2, 37 ± 1°C, 95 ± 5% relative humidity, referred to as Standard Culture Conditions; SCC). The EpiOcular tissues were pre-incubated for 1 hour, then the medium was replaced with fresh medium, and the tissues were further pre-incubated overnight under SCC. The following day, the tissues were transferred into fresh cell culture medium (1 ml/well in 6-well plates) and exposed to approximately 100 μl of the medical device extract or test material (neat or diluted, as specified in Table 1), as applied to the apical surface of the tissue. The tissues were exposed for approximately 21 hours (± 3 hours), under SCC. The protocol timeline is outlined in Figure 2a. Standard protocol timelines for medical device and pharmaceutical product testing with EpiOcular. a) Irritation testing; b) phototoxicity testing. MD = medical device.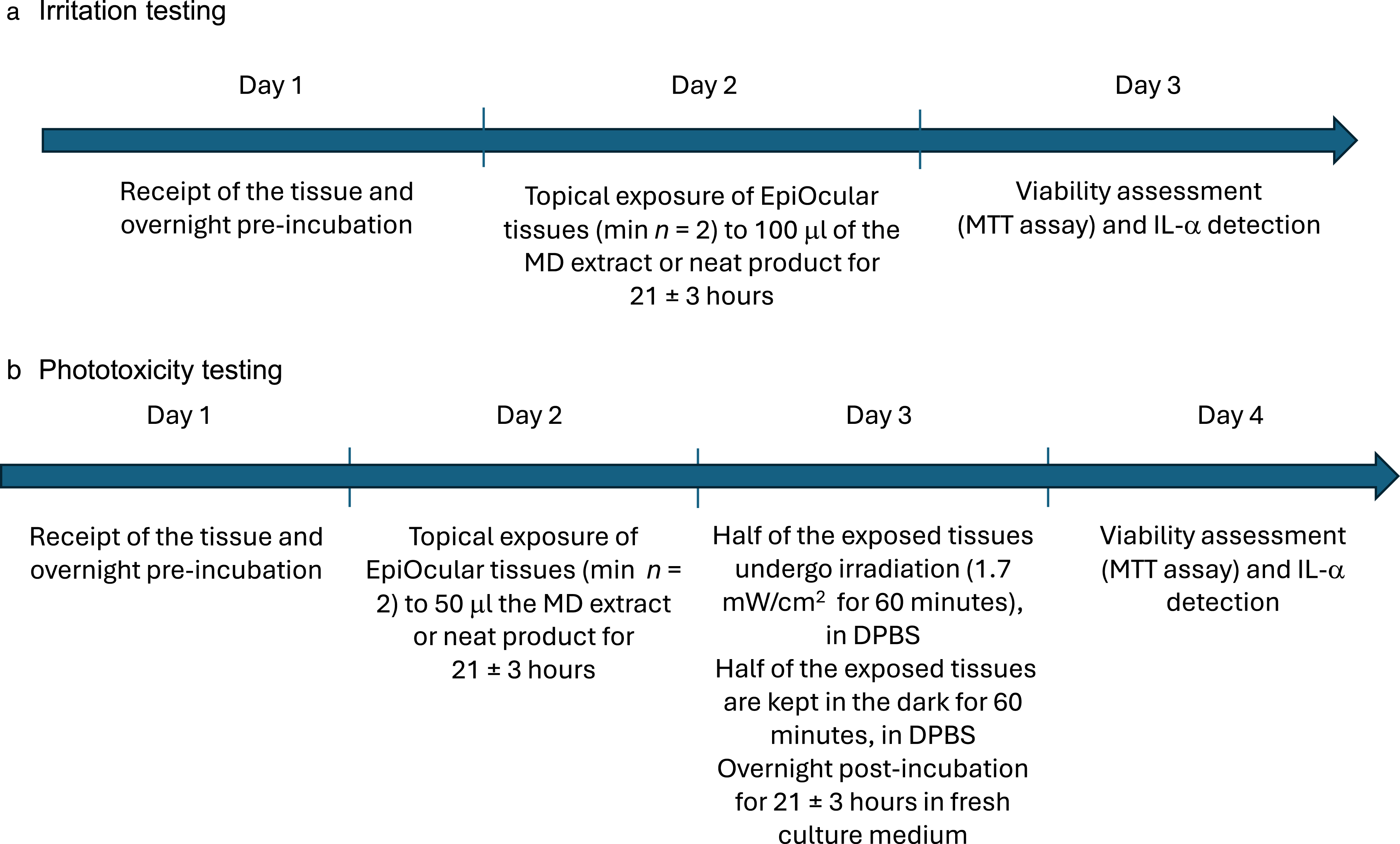
Following the 18-hour exposure period, the tissues were transferred into a 24-well plate pre-filled with 0.3 ml of MTT (1 mg/ml) and incubated at 37°C for 3 hours at SCC. The culture medium from the exposure phase was also collected for IL-1α analysis by Enzyme-Linked Immunosorbent Assay (ELISA) using the Human IL-1α/IL-1F1 Immunoassay Quantikine® ELISA kit (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s protocol. After the MTT incubation, inserts with tissues were transferred into a 24-well plate, and 2 ml of isopropanol were added to each well to completely submerge the inserts. The plates were sealed and tissues were incubated at room temperature for two hours on a plate shaker or overnight in the refrigerator (2–8°C) to extract the MTT–formazan product. Next, 0.2 ml of the extract were transferred to a 96-well plate (two aliquots per tissue sample), and the optical density (OD) was measured at 570 nm with isopropanol as blank. The relative viability of each tissue was determined according to the equation:
Protocol for assessing photo-irritation potential of medical device extracts and pharmaceuticals with low eye irritation potential with the EpiOcular tissue model
Firstly, the optimal irradiation dose for the EpiOcular tissue model was determined according to the ZEBET Standard Operating Procedure (SOP)20–22 and OECD TG 498. 4 On the day of the experiment, the height of the solar simulator SOL500 (Hönle) was set to 60 cm from the irradiated area to achieve an irradiation intensity of 1.7 mW/cm2 UVA, as measured with a calibrated UV radiometer (UVP Ltd, UK) through the lid of the 6-well plate. A dose of 1 J/cm2 every 10 minutes was delivered by using this experimental setting. After the overnight pre-incubation, the tissues were transferred to new 6-well plates pre-filled with 1 ml/well DPBS, and irradiated with a periodically increasing dose of UVA + visible light, from 1 to 11 J/cm2 (maximal time of irradiation was 110 minutes). After irradiation, the tissues were transferred to a fresh assay medium and further incubated overnight (for 18–24 hours). The tissues were subsequently assessed for viability by using the MTT assay (as described in the previous section). Irradiated tissue viability was compared to a non-irradiated control (n = 2) and calculated by following the equation described in the previous section. A dose–response graph (intensity of irradiation versus viability) was constructed to analyse the sensitivity of the tissues to increased doses of UVA + visible light (with any effects resulting from the UVA part of the spectrum only).
Following the irradiation dose optimisation, the phototoxicity testing was performed according to the ZEBET SOP ‘Phototoxicity protocol for use with EpiDerm™ model (EPI-200)’21–23 with minor modifications required for the EpiOcular tissues. EpiOcular tissues were transferred from the shipping agar into the 6-well plates containing 1 ml/well assay medium and were pre-incubated in a humidified incubator for one hour under standard culture conditions (SCC; 37 ± 1°C, 90 ± 10% RH, 5 ± 0.5% CO2) to allow the release of metabolites accumulated during storage on agar. After this 1-hour incubation, the tissue models were transferred into 6-well plates pre-filled with fresh assay medium and pre-incubated overnight under SCC.
The following day, the tissues were exposed to 50 μl of the test chemical or formulation, or the 24-hour extracts in sesame seed oil (CAS No. 8008-74-0; Sigma-Aldrich, pharmaceutical grade) or in saline. The 50 μl test sample aliquots were applied to the apical surface of the tissues. Each sample was tested on four tissues: two tissues per extract, chemical or formulation were irradiated, and two tissues were not irradiated (i.e. kept in the dark). The tissues were exposed to the test samples for approximately 21 hours (± 3 hours) under SCC. The following day, the tissues were transferred from the medium to new 6-well plates containing 1 ml/well DPBS. Then, one set of tissues was immediately irradiated according to the outcome of the optimisation experiments, and the second set kept in the dark under standard culture conditions. The protocol timeline is outlined in Figure 2b.
After irradiation, all tissues (irradiated and non-irradiated) were gently washed in three beakers containing approximately 150 ml of DPBS. The post-exposed tissues were transferred to fresh culture medium and were further incubated for 21 ± 3 hours under SCC. The next day, viability was determined with the MTT assay as described earlier, and the medium was collected to determine the release of the inflammatory cytokine IL-1α. In some samples, TNF-α was also determined by using the relevant Quantikine® ELISA kit from R&D Systems, according to the manufacturer’s protocol.
Photo-irritation testing with the 3T3 mouse fibroblast cell line
For comparison with the EpiOcular-based protocol outlined in the present study, the phototoxic potential of ophthalmological solutions and the experimental drug, cemtirestat, were also assessed by using the protocol outlined in OECD TG 432, employing the 3T3 Balb/c mouse fibroblast cell line. 24
Briefly, BALB/c 3T3 cells were maintained in culture for 18–24 hours to form monolayers. Two 96-well plates per test chemical were pre-incubated with eight concentrations, ranging from 100% to 0.001% (v/v), prepared from the neat products in DPBS. The cells were exposed to the test materials for one hour. After that, one of the two plates was exposed to a dose of 5 J/cm2 (50 minutes, 1.7 mW/cm2), whereas the other plate was kept in the dark. In both plates, the treatment buffer was replaced with fresh culture medium, and the plates were incubated overnight (18 ± 3 hours). Subsequently, the 3T3 Neutral Red Uptake phototoxicity assay (3T3 NRU PT assay) was conducted following the procedures outlined in OECD TG 432. Dose–response curves were constructed for the irradiated (Irr+) and non-irradiated (Irr–) cells. The concentration reducing cell viability to 50% (IC50) was calculated with MS Excel®. A photo-irritation factor (PIF) was calculated by following the equation below: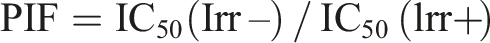
Results
Ocular irritation
In the pilot testing phase of the protocol, we exposed EpiOcular tissues to polar and non-polar extracts from five different scaffolds, based on polycaprolactone (PLC) and polyethylene terephthalate (PET): Inocure MF (PCL), Inocure NF (PCL), CS-200 Bio-Spun™ PET, CS-200 (–) Bio-Spun PET (negatively charged) and CS-200 (+) Bio-Spun PET (positively charged). None of these five materials had a negative effect on tissue viability, as shown in Figure 3 and Table 2. Furthermore, when the samples were assessed for their potential to induce inflammation, as determined by IL-1α release, the results indicated that these materials did not cause any significant effects that would classify them as inflammatory (see Figure 4). Viability of the EpiOcular tissues exposed to extracts from five different PCL-based and PET-based electrospun scaffolds. EpiOcular tissues were exposed to 100 μl of the polar and non-polar extracts from five different electrospun scaffolds (based on polycaprolactone (PCL) or polyethylene terephthalate (PET)) for 21 ± 3 hours. None of the tested extracts were classified as irritating based on the tissue viability, which remained above 100% compared to the negative control (ultra-pure water). The positive control was 0.3% Triton X-100. (s) indicates saline extraction; (o) indicates sesame seed oil extraction. Results obtained with the EpiOcular tissue model for the seven formulations and five scaffolds tested. The pilot phase involved the testing of a variety of scaffolds; the second phase involved the testing of a range of ophthalmic medical devices and products, and ophthalmological drugs. The negative controls were ultra-pure water; the positive controls were 0.3% Triton X-100. (s) indicates saline extraction; (o) indicates sesame seed oil extraction; SD = standard deviation (when n = 3); Diff = difference between duplicates (when n = 2). IL-1α release from EpiOcular tissues after exposure to the ophthalmological products and scaffold extracts. EpiOcular tissues were exposed to 100 μl of a range of samples (ophthalmological products and scaffold extracts; (s) indicates saline extraction; (o) indicates sesame seed oil extraction) for 21 ± 3 hours. The presence of IL-1α in the medium was quantified to determine whether the samples had the potential to induce inflammation. None of the tested samples, except for Ophthalmo-Septonex and the positive control (0.3% Triton X-100), elicited a significant IL-1α response. The response observed with Ophthalmo-Septonex can be attributed to the product’s composition and its mechanism of action against ocular sepsis.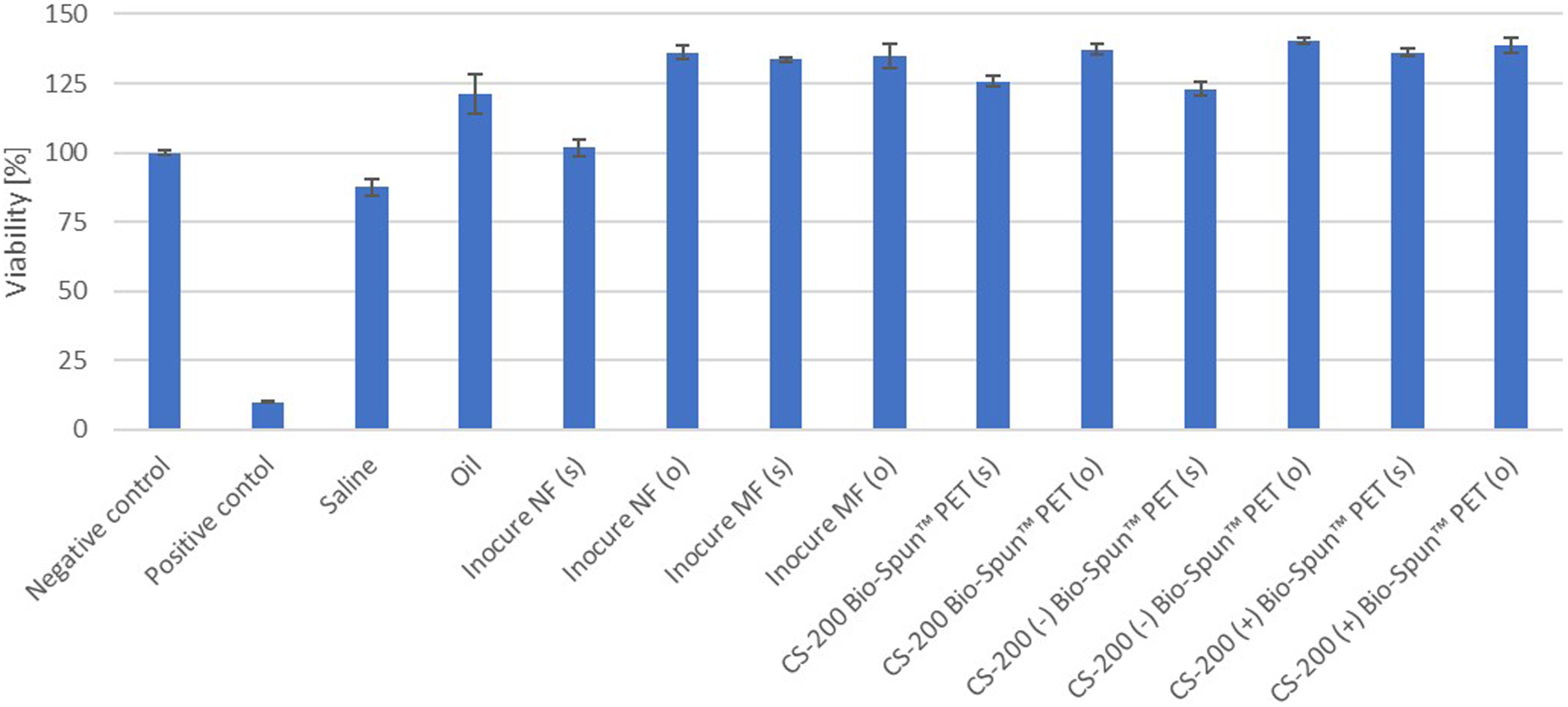
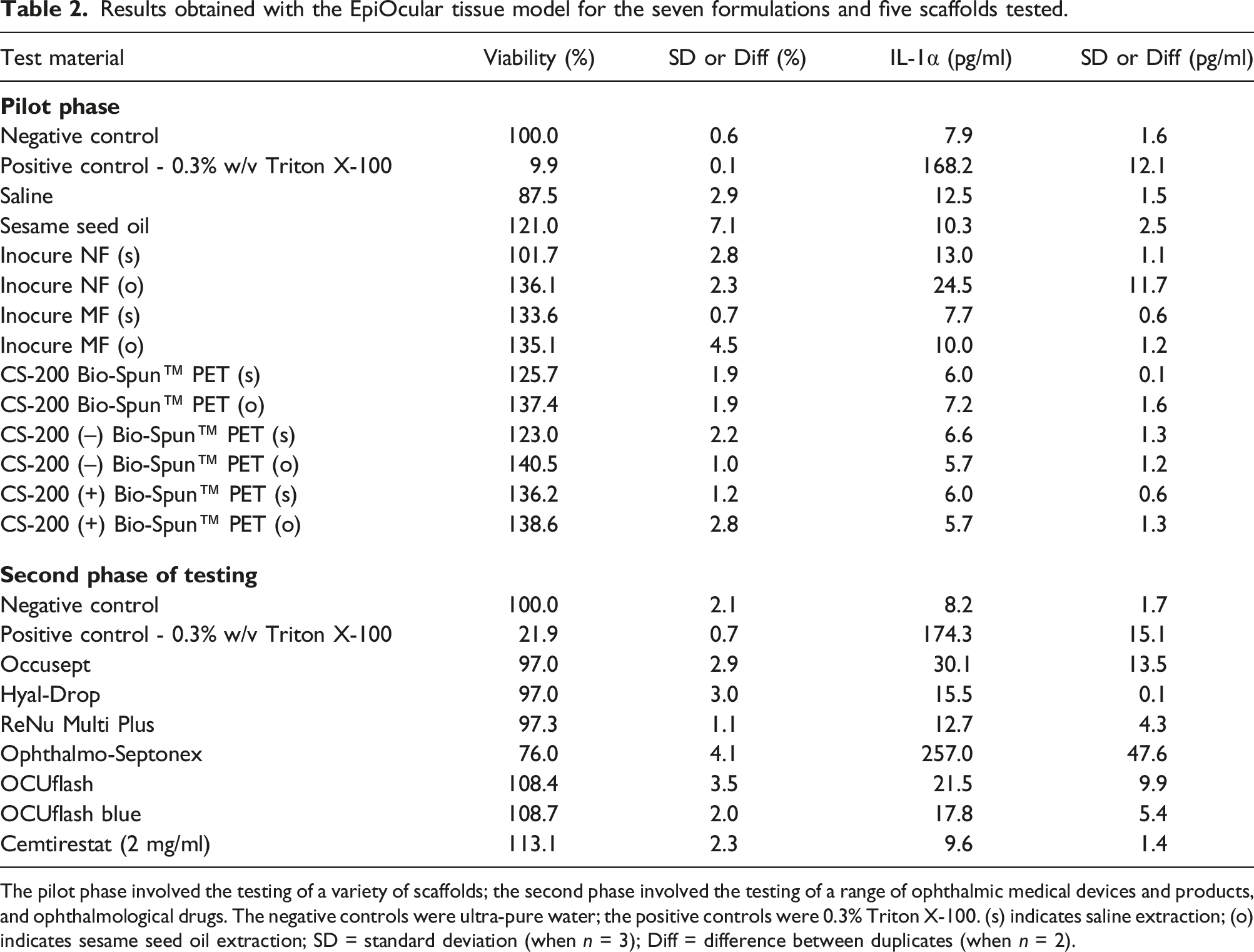
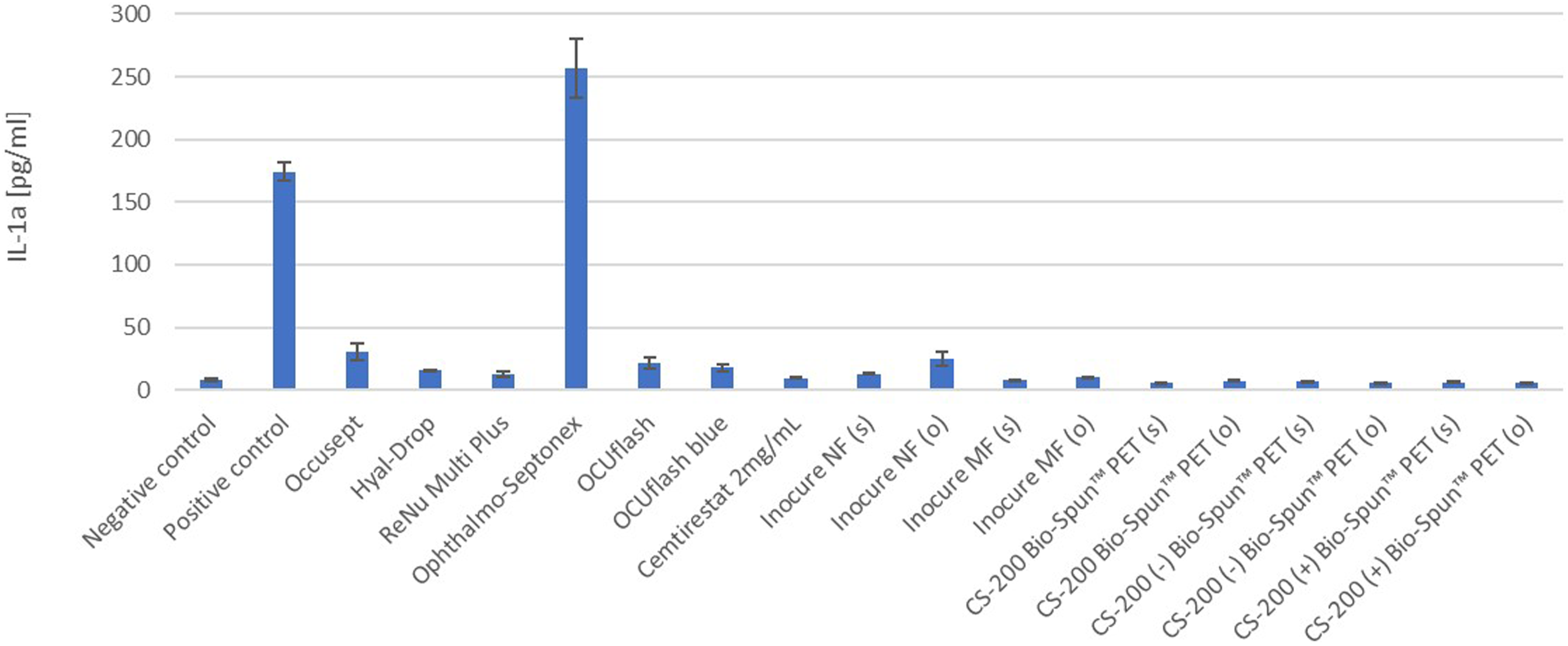
In the next testing phase, we expanded our protocol to include a carefully selected set of ophthalmological products, such as eye drops and contact lens-cleaning solution (classified as medical devices), an over-the-counter drug (Ophthalmo-Septonex) and an experimental drug, cemtirestat. As shown in Figure 5, the viability of the EpiOcular tissues exposed for 21 ± 3 hours to 100 μl of the ophthalmological solutions was strongly comparable to that of the negative control. The exception was Ophthalmo-Septonex, which decreased tissue viability by approximately 25% as compared to the negative control. In the IL-1α analysis (Figure 4), this sample elicited high levels of IL-1α release (257.0 pg/ml), suggesting that it may be a mild to moderate irritant to sensitive eyes or upon repeated exposure. Details on the viability and IL-1α release for all tested samples can be found in Table 2. Viability of the EpiOcular tissues exposed to eye drops and other ophthalmological products. EpiOcular tissues were exposed to 100 μl of the test samples, comprising four different eye drop formulations and a contact lens-cleaning solution (classified as medical devices), one over-the-counter drug (Ophthalmo-Septonex) and one experimental drug (cemtirestat), for 21 ± 3 hours. None of the test samples were classified as irritating based on viability, which, in six out of the seven samples, remained close to 100% (the value assigned to the negative control (ultra-pure water)). The positive control was 0.3% Triton X-100. Although Ophthalmo-Septonex decreased tissue viability by approximately 25%, it remained above the classification threshold for irritation (i.e. a 50% reduction in tissue viability).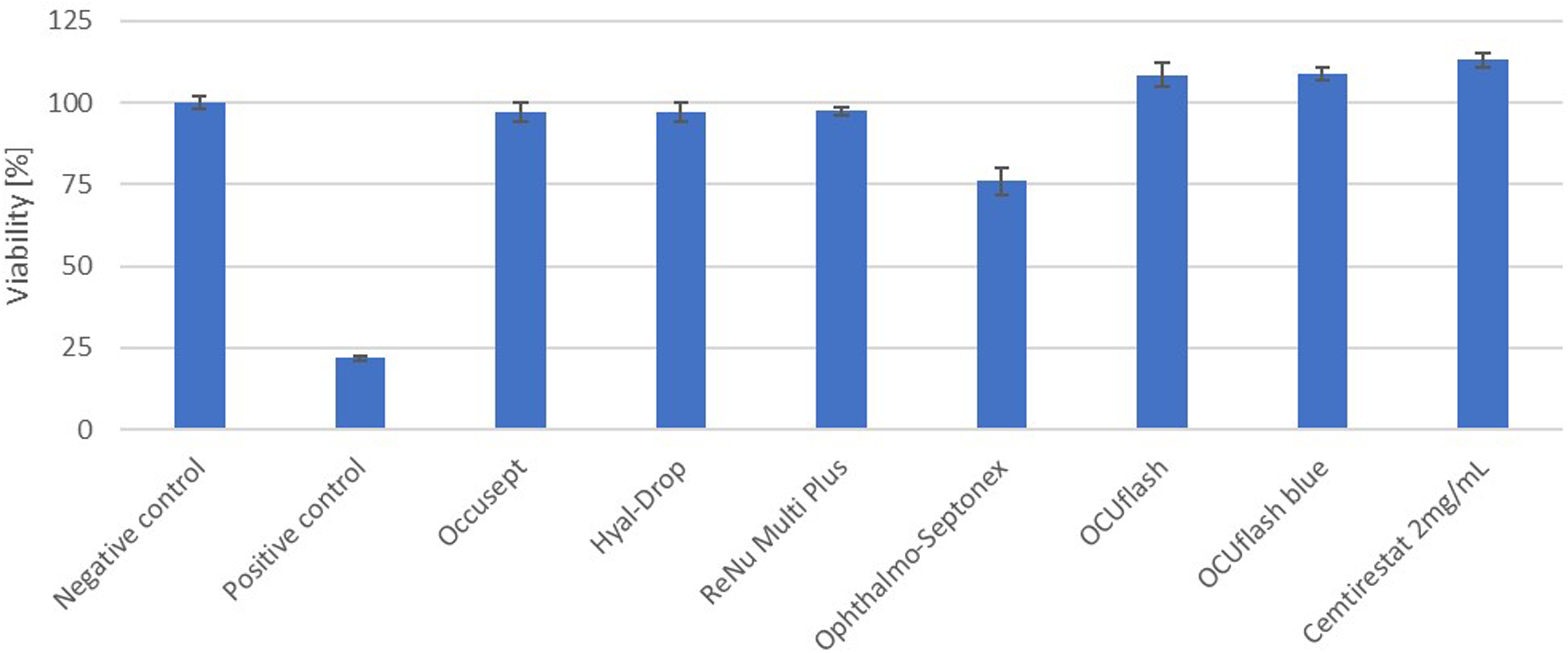
Optimisation of the irradiation dose for the phototoxicity testing protocol
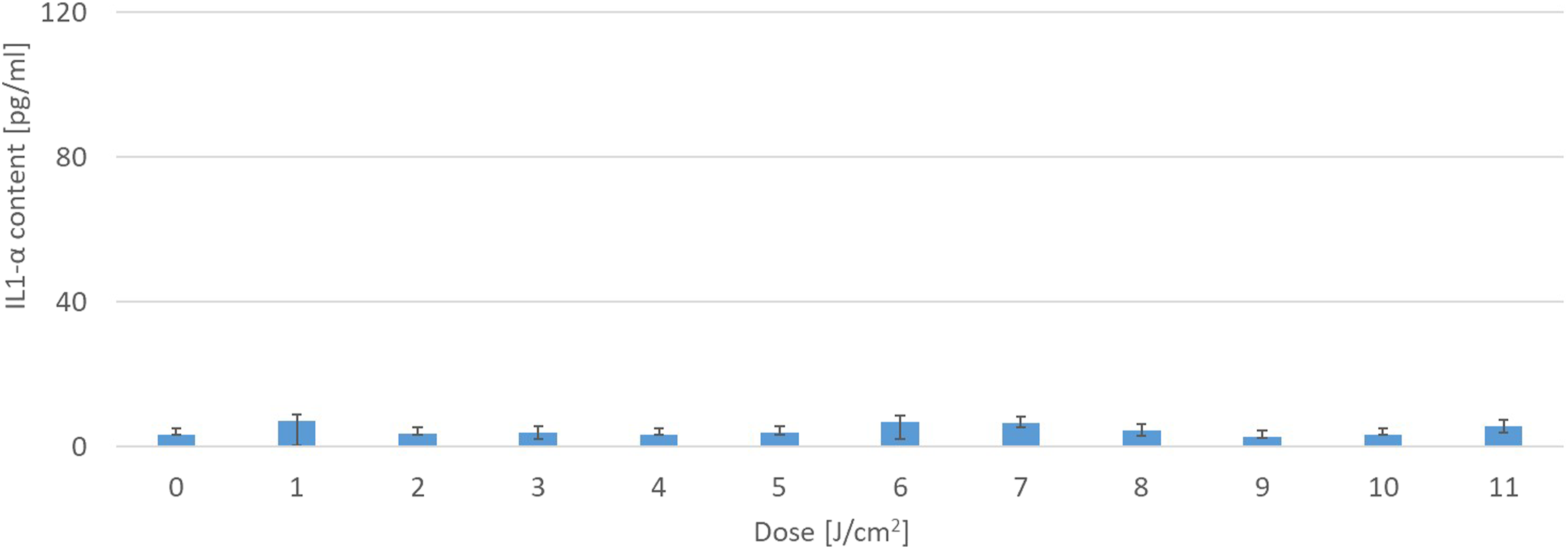
The sensitivity of EpiOcular tissue to increasing doses of UVA + visible light was an essential parameter that needed to be established for the subsequent phototoxicity assessment conducted in this study. Two different production lots of the EpiOcular tissue model were used in the irradiation dose optimisation. The tissue tolerated increasing irradiation (calibrated to UVA irradiance of 1.7 mW/cm2) at all doses tested, as demonstrated by the high viability recorded (Figure 6) and no significant increase in the IL-1α levels (Figure 7). The experiments were conducted with doses of up to 11 J/cm2 UVA (110 minutes of exposure). For subsequent experiments with the EpiOcular tissue, a dose of 6 J/cm2 was chosen, which also corresponds to the recommendations in OECD TG 498
4
(i.e. 6 J/cm2 for reconstructed human skin models). It is noted that a recommendation of 5 J/cm2 for the 3T3 Balb/c fibroblast cell line is given in OECD TG 432.
24
The viability of EpiOcular tissues exposed to increasing doses of UVA + visible light. EpiOcular tissues were exposed to increasing doses of the UVA + visible light (irradiation intensity of 1.7 mW/cm2 of UVA) for up to 110 minutes. Tissue viability was not impacted, even at the highest dose tested. A dose of 6 J/cm2 (60 min of 1.7 mW/cm2) was chosen for the subsequent experiments. IL-1α release from EpiOcular tissues after irradiation with increasing doses of UVA + visible light. EpiOcular tissues were exposed to increasing doses of the UVA + visible light (irradiation intensity of 1.7 mW/cm2 of UVA) for up to 110 minutes. The presence of IL-1α in the medium was quantified to determine if samples had potential to induce inflammation after exposure to light. No significant increase in IL-1α levels was observed compared to the negative control (i.e. non-irradiated tissue), which provided a mean IL-α value of 3.3 pg/ml.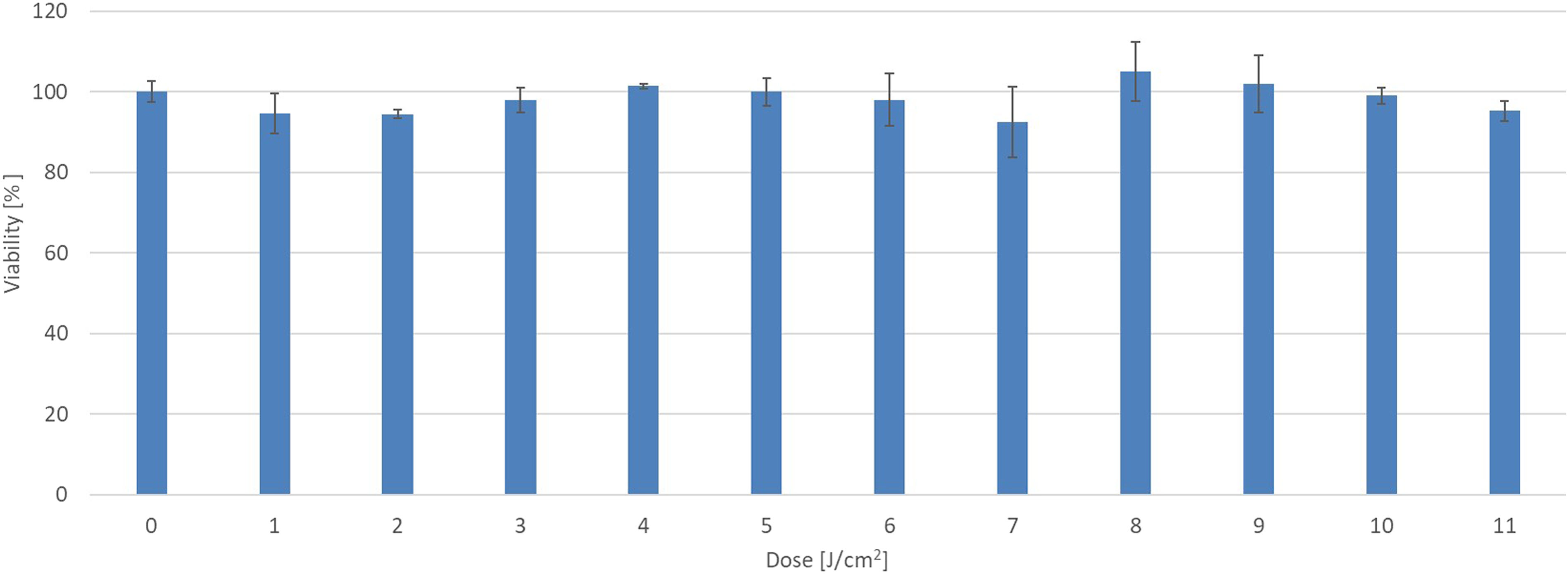
Assessment of the benchmark chemical, chlorpromazine hydrochloride (CPZ)
The benchmark chemical, CPZ, was selected to evaluate the phototoxic response predicted by the EpiOcular tissue model, as well as being used as the positive control. This substance yielded a clear dose–response outcome in the 3T3 NRU PT assay, resulting in a PIF value of 109 and an IC50 of 0.7 mg/ml (data not shown). CPZ has significant absorbance in the UVA range and partly in the UVB range (270–370 nm, with a maximum peak at 300–310 nm).
Cytotoxicity (i.e. treated tissue viability without subsequent irradiation) and phototoxicity (i.e. treated tissue viability after subsequent irradiation) were evaluated in the EpiOcular tissue model after 21 ± 3 hours of topical CPZ application (Figure 8). Cytotoxicity and phototoxicity were both observed at a concentration of 0.1% w/v (Figure 8). Phototoxicity was defined according to the established prediction model (PM) in OECD TG 498
4
— i.e. a > 30% difference between the non-irradiated and irradiated samples treated with the same test substance concentration. By this definition, phototoxicity was evident at a concentration of 0.01% w/v CPZ (Figure 8), which is ten times lower than the cytotoxic concentration of 0.1%. However, the first significant decrease in viability of the irradiated samples occurred at a concentration of 0.001%. This experiment confirmed that the selected dose of 6 J/cm2 was sufficient to induce the expected phototoxic response in the EpiOcular tissue model. Additionally, alongside viability, the presence of IL-1α and TNF-α in the medium was assessed. However, no significant increases in cytokine release were observed for either. The response in terms of cytokine release, at all CPZ concentrations tested, was comparable to that of non-treated tissues (data not shown). The phototoxic effect of chlorpromazine hydrochloride on EpiOcular tissues. Chlorpromazine hydrochloride (CPZ), a known phototoxic chemical, was chosen for evaluating the prediction of a phototoxic response. EpiOcular tissues were treated with a range of concentrations of CPZ for 18 hours, prior to exposure to UVA + visible light (6 J/cm2). The viability of treated and irradiated (UVA-VIS +) tissues was compared to that of treated but non-irradiated (UVA-VIS –) tissues. NC = negative control (DPBS).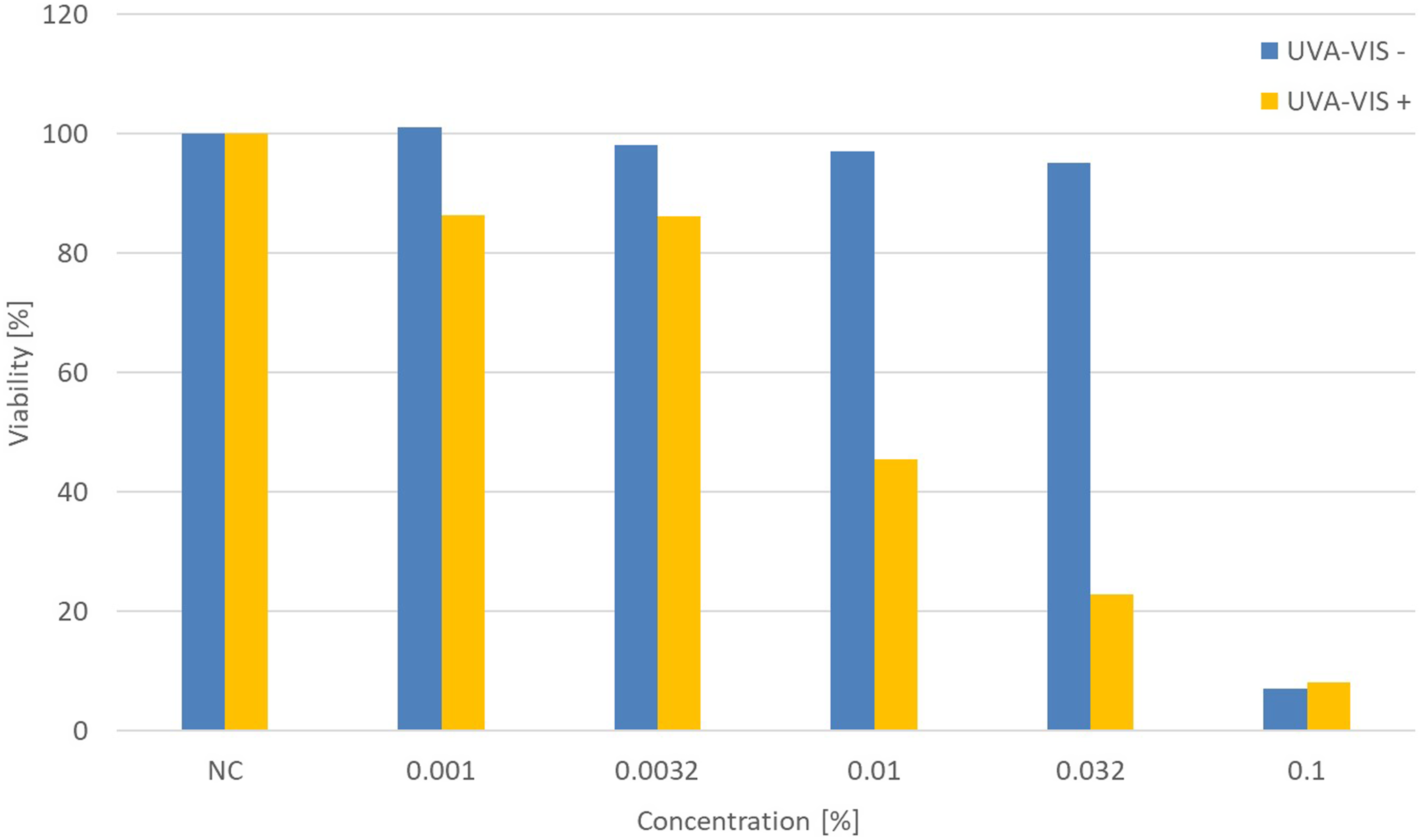
The phototoxic potential of selected medical devices and pharmaceuticals
Ophthalmological products and drugs
Summary of the results obtained with the 3T3 NRU PT assay and EpiOcular photo-irritation test for the selected ophthalmological products and drugs.
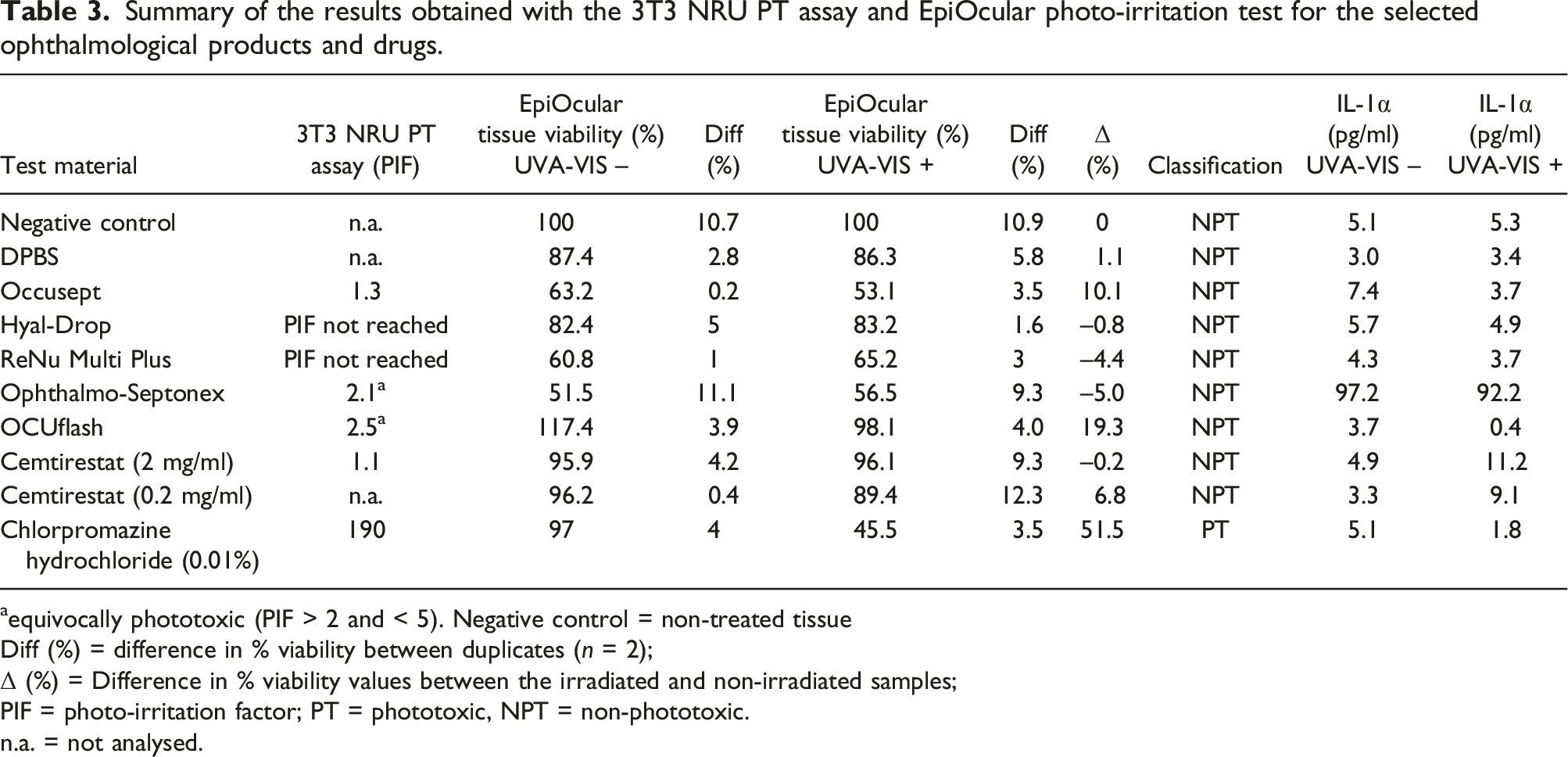
aequivocally phototoxic (PIF > 2 and < 5). Negative control = non-treated tissue
Diff (%) = difference in % viability between duplicates (n = 2);
Δ (%) = Difference in % viability values between the irradiated and non-irradiated samples;
PIF = photo-irritation factor; PT = phototoxic, NPT = non-phototoxic.
n.a. = not analysed.
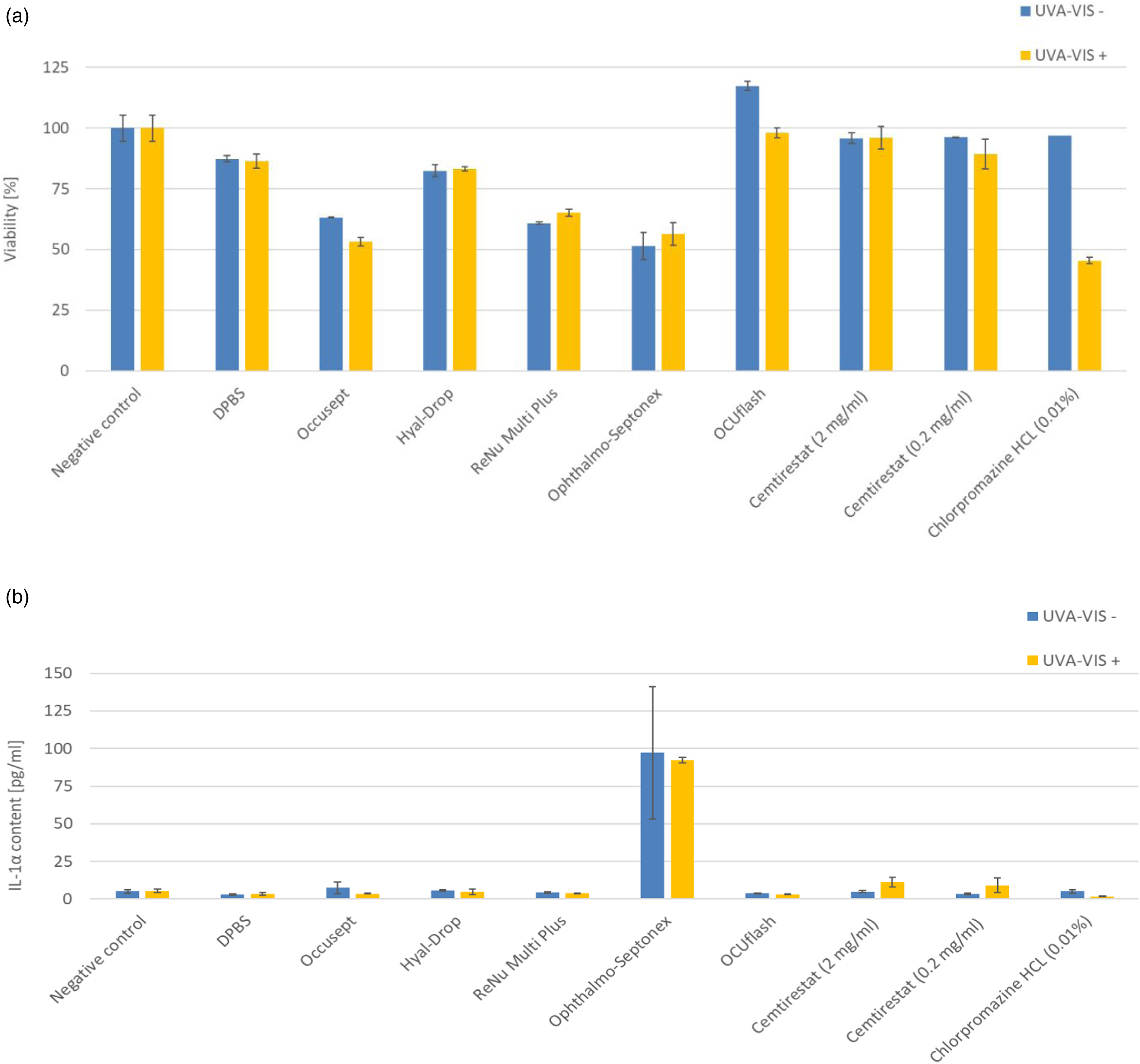
The effect of UVA + visible light irradiation on the viability and inflammatory response of EpiOcular tissues treated with selected ophthalmological products and drugs. EpiOcular tissues were treated for 21 ± 3 hours with selected ophthalmological products and drugs, prior to exposure to UVA + visible light (irradiation of 1.7 mW/cm2 for 60 min resulting to 6 J/cm2). a) The viability of treated and irradiated (UVA-VIS +) tissues was compared to that of treated but non-irradiated (UVA-VIS –) tissues. The only phototoxic material in this set of samples was chlorpromazine hydrochloride, with a 50% drop in viability between the irradiated and non-irradiated tissues. b) The levels of IL-1α in the medium of treated and irradiated (UVA-VIS +) tissues and treated but non-irradiated (UVA-VIS –) tissues were assessed. The findings revealed that Ophthalmo-Septonex might induce inflammation in both irradiated and non-irradiated tissues. NC = negative control, non-treated tissues.
None of the tested ophthalmological products, nor the experimental drug cemtirsetat, produced a positive response in the EpiOcular phototoxicity test. Compared to the simple (i.e. non-irradiated) irritation protocol, the photo-irritation test resulted in an overall decrease in tissue viability for most products. This trend could be explained by the additional post-exposure incubation period of 21 ± 3 hours, which is not performed in the irritation testing protocol. Ophthalmo-Septonex caused a significant release of IL-1α in both the irradiated (92.2 pg/ml) and non-irradiated (97.2 pg/ml) samples. The suspected phototoxicity of OCUflash (PIF = 2.5) and Ophthalmo-Septonex (PIF = 2.1) was not confirmed in the EpiOcular phototoxicity test.
Scaffolds, polymers and intraocular lenses (IOLs)
Despite the minimal possibility of scaffolds, polymers and IOLs causing phototoxicity, we tested extracts of these materials in the EpiOcular photo-irritation test for completeness of the presented data. The protocol (including the irradiation conditions of 6 J/cm2) was the same as that used for testing the ophthalmological products. Additionally, IOLs were tested in direct contact with the tissue under exaggerated irradiation conditions (11 J/cm2 UVA + visible light). The results are shown in Figure 10. None of these materials elicited a phototoxic response in the test. An interesting observation was made when the IOLs were tested in direct contact with the tissue, where a decrease in viability was observed between the irradiated and non-irradiated samples. This reduction in viability was limited to the central portion of the tissue, as observed in the MTT assay, and was likely caused by the mechanical and thermal effects of the IOLs, concentrating the wavelength energy on the central part of the tissue during the prolonged irradiation step (110 minutes). Evaluation of the possible phototoxic potential of selected medical devices, scaffolds and polymers extracted into saline or tested with direct contact. The EpiOcular tissues were exposed to 50 μl of the extracts for 21 ± 3 hours and then subjected to dose of 6 J/cm2 UVA-VIS (irradiation of 1.7 mW/cm2 for 60 min). For these samples and conditions, no significant decrease in viability was observed between the irradiated and non-irradiated tissues. However, when intraocular lenses (IOLs) were in direct contact with the EpiOcular tissue surface and subjected to an exaggerated UVA-VIS dose of 11 J/cm2 (1.7 mW/cm2 irradiation for 110 min), a reduction of approximately 30% in viability was observed between the irradiated and non-irradiated tissues. NC = negative control, non-treated tissues.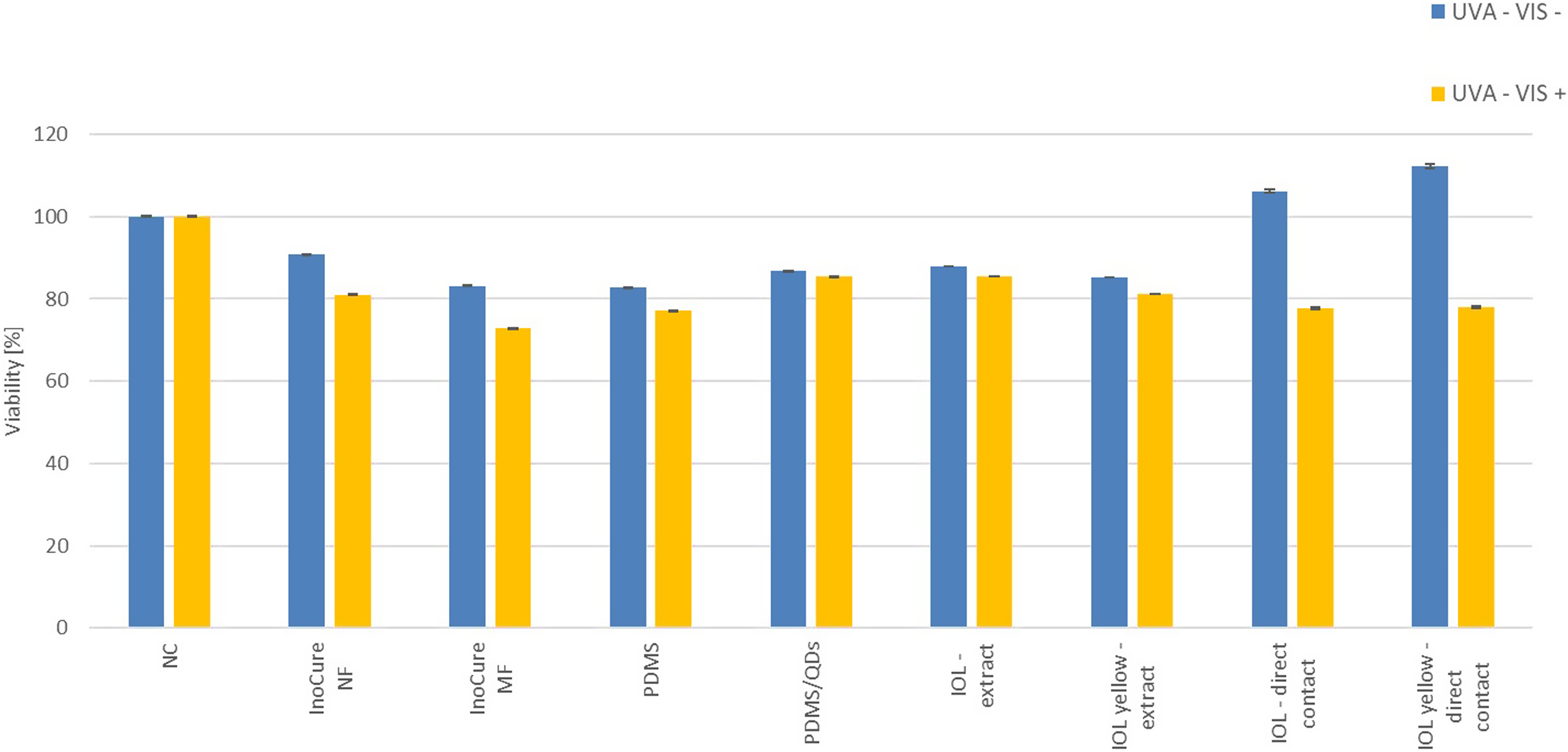
Discussion
This study introduces a novel in vitro methodology utilising the EpiOcular tissue model to evaluate the irritation and phototoxicity potential of medical devices and pharmaceuticals intended for ophthalmological use. The chosen experimental approach builds upon the experience gained during the development and validation of the EpiDerm skin irritation test for medical devices testing,2,3,18,25 as outlined in the ISO 10993-23:2021, 5 and the photo-irritation protocol for topically applied chemicals and pharmaceuticals detailed in OECD TG 498.4,20,22,23
During the development of the eye irritation test protocol, we analysed numerous published datasets related to eye irritation testing of chemicals, cosmetic products and pharmaceuticals. The protocols used to generated these data varied significantly, depending on whether the test had been developed for hazard evaluation or safety testing. Hazard-oriented protocols, designed for the classification and labelling of chemicals, typically involved testing the neat substances with short exposure times.6,8,26,27 In contrast, protocols for the safety assessment of cosmetic or pharmaceutical products (which may contain materials with low irritation potential, such as highly diluted irritants or very mild irritants active at low concentrations) used exaggerated exposure times and application doses to detect any signs of irritation or inflammation.15,16,28
Extracts derived from medical devices and formulations classified as medical devices (e.g. eye drops) present a toxicological profile akin to water-based cosmetic products with negligible irritation potential. Consequently, test protocols that involve prolonged exposure durations and integrate additional endpoints have been developed to identify potential inflammatory responses that could lead to irritation and photo-irritation.
A major challenge when evaluating the performance of the two protocols in the present study was the lack of well-characterised medical device samples that could irritate the cornea and for which corresponding in vivo data were available. The sourcing of photo-irritating samples presented an even more complicated situation. Committed to avoiding animal testing, we used eye drops and medical device polymers for which clinical data were available. Companies and collaborators interested in testing their products using animal-free approaches donated some of these materials. Other materials were either purchased from local pharmacies or obtained through contract research agreements.
Except for Ophthalmo-Septonex, none of the tested materials were classified as irritating in the present study, according to the irritation protocol. However, Ophthalmo-Septonex exhibited a notable effect, reducing tissue viability by 25% compared to the negative control and inducing a release of IL-1α (> 250 pg/ml) that was comparable to that caused by Triton X-100 (174 pg/ml). According to the manufacturer’s information leaflet, these eye drops are generally well-tolerated. However, in sensitive individuals, the product may cause increased blood flow (resulting in eye redness) and itching of the conjunctiva. This observation aligns with data obtained for some cosmetic ingredients tested by using the Sub-Draize EpiOcular protocol, and subsequently tested in human volunteers around the eye area. In these cases, panelists reported effects like swelling or itching of the eyes, which were not identified in animal testing but were predicted in the EpiOcular test.15,17 In the EpiOcular test, classification of Ophthalmo-Septonex as a mild irritant would be based on a combination of the MTT assay results and IL-1α assessment.
Based on discussions with professionals from eye clinics who are interested in the presented research, we concluded that photo-irritation testing might also be of significant interest to this professional community. It is feasible that OECD TG 498, 4 which features an EpiDerm-based test method, could be adapted to include the use of the EpiOcular tissue model. In the current study, the methodology described in a book chapter by Kandarova and Liebsch in 2017 22 was followed. However, it was necessary to adjust some technical parameters, such as the volume of the incubation medium (from 0.9 to 1.0 ml), the washing procedure to prevent mechanical damage of the EpiOcular tissue surface, and assessment of the EpiOcular tissue model’s resistance to increased doses of UVA light. CPZ was used to evaluate the protocols, as it represents a benchmark phototoxic chemical and is recommended as a standard positive control in the phototoxicity protocols of OECD TG 432 and TG 498. The phototoxic effects of CPZ were accurately predicted by using the MTT assay, aligning closely with the results previously published for the EpiDerm tissue model.21,23 Interestingly, we observed no release of IL-1α or TNF-α, which may be attributed to the drug’s anti-inflammatory properties, or the specific tissue batch used.
After this initial protocol evaluation with CPZ, the EpiOcular tissue model was exposed to various ophthalmological products (which were also assessed in the 3T3 NRU PT assay; OECD TG 432 24 ) and extracts from several scaffolds and polymers. Two products, Ophthalmo-Septonex and OCUflash, resulted in elevated PIF values of 2.1 and 2.5, respectively, in the 3T3 NRU PT assay, suggesting equivocal phototoxic potential. However, given the known oversensitivity of the 3T3 NTU PT assay, the absence of phototoxicity and no IL-1α release in the current EpiOcular-based phototoxicity test was not surprising.
In the current EpiOcular-based test, none of the materials — including ophthalmological products, scaffolds, polymers and IOLs — exhibited phototoxicity. Furthermore, the sensitivity of the EpiOcular tissue model to potentially irritating products, such as Ophthalmo-Septonex, was confirmed by using this testing approach. The photo-irritation testing protocol included a 21 ± 3-hour post-exposure incubation, which is essential for the development of a potential phototoxic response after irradiation. In the case of the Ophthalmo-Septonex, a significant release of IL-1α was notably observed in both irradiated and non-irradiated samples.
Conclusions
Despite the limited number of samples tested in both protocols evaluated in the current study, and considering research involving prolonged exposures by previous research groups focusing on cosmetics, we hypothesise that the EpiOcular eye irritation test, as it currently stands with its prediction models based on viability and IL-1α release, might be adequately sensitive to detect even subtle indications of potential eye irritation from ophthalmic medical devices and products, and ophthalmological drugs. Our research team is keen to extend collaboration with industrial partners to validate these protocols for further use in regulatory toxicology, especially for ophthalmological applications, and to promote their inclusion in ISO 10993 and ICH standards.
Footnotes
Acknowledgements
The researchers would like to acknowledge the following companies for the provision of the test samples: InoCure s.r.o., BioSurfaces LLC, the Dr Vesely Očná Klinika (Eye Clinic) and the Polymer Institute of the Slovak Academy of Sciences (research team of Dr Špitálsky).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the funding: Vega Grant 2/0153/20 — Evaluation of biocompatibility of medical devices and materials using reconstructed 3D tissue models, and APVV Grant APVV-19-0591 — In vitro evaluation of biocompatibility of medical devices and innovative materials for medical devices.
