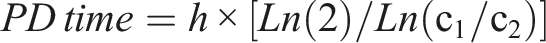Abstract
The likelihood that potential new drugs will successfully navigate the current translational pipeline is poor, with fewer than 10% of drug candidates making this transition successfully, even after their entry into clinical trials. Prior to this stage, candidate drugs are typically evaluated by using models of increasing complexity, beginning with basic in vitro cell culture studies and progressing through to animal studies, where many of these candidates are lost due to lack of efficacy or toxicology concerns. There are many reasons for this poor translation, but interspecies differences in functional and physiological parameters undoubtedly contribute to the problem. Improving the human-relevance of early preclinical in vitro models may help translatability, especially when targeting more nuanced species-specific cell processes. The aim of the current study was to define a set of guidelines for the effective transition of human primary cells of multiple lineages to more physiologically relevant, translatable, animal-free in vitro culture conditions. Animal-derived biomaterials (ADBs) were systematically replaced with non-animal-derived alternatives in the in vitro cell culture systems, and the impact of the substitutions subsequently assessed by comparing the kinetics and phenotypes of the cultured cells. ADBs were successfully eliminated from primary human dermal fibroblast, uterine fibroblast, pulmonary fibroblast, retinal endothelial cell and peripheral blood mononuclear cell culture systems, and the individual requirements of each cell subtype were defined to ensure the successful transition toward growth under animal-free culture conditions. We demonstrate that it is possible to transition (‘humanise’) a diverse set of human primary cell types by following a set of simple overarching principles that inform the selection, and guide the evaluation of new, improved, human-relevant in vitro culture conditions.
Keywords
Introduction
Over 90% of the drugs in clinical development currently fail. 1 Of these failures, 40 to 50% are due to a lack of clinical efficacy, around 30% are because of overt toxicity, and the remainder are due to poor pharmacokinetic properties or lack of commercial interest. 2 Insufficient efficacy may result from many factors, but the choice of in vitro preclinical model systems may play a large part in this. Although animal and human diseases share some aetiology, they are often not synonymous. The underpinning biology of disease can differ between animals and humans at the systemic, cellular and molecular level. This may raise issues in the clinical translation of findings from medical research models that involve animals, animal cells, or human cell models that are exposed to animal-derived biomaterials (ADBs).3–6
Mice are the model system most frequently used for early-stage preclinical research, 7 but they differ from humans in terms of genetics, physiology and immunology, with mice not naturally exhibiting many of the human diseases studied.3,8 A variety of methods are used to induce human-like disease in murine models, including genetic modification and exposure to biological or chemical agents.9–11 However, there can be substantial disparity between the onset, presentation, treatment and resolution of a disease in preclinical animal models and in humans. This leaves significant room for failure when moving from preclinical animal studies to clinical trials. Even before preclinical work is carried out in animal models, the presence of ADBs in in vitro cell culture models might negatively impact the translation of the results — and current evidence supports the premise that cell culture methods could be improved by the reduction and replacement of ADBs.12–14 Simple in vitro culture models form the underpinnings of various avenues of basic research and early phase drug development. Rapid and efficient refinement of these models could enhance early-stage drug development by enabling the identification and targeting of species-relevant genes or proteins, ultimately improving translational success.
The majority of in vitro culture systems rely on animal sera, such as fetal bovine serum (FBS), or other animal-derived supplements such as bovine pituitary extract (BPE) or bovine serum albumin (BSA), to provide growth factors and other mediators necessary for cell metabolism, viability and proliferation. 15 FBS is routinely used in cell culture, predominantly for its availability and tradition, rather than its specificity or physiological relevance. In fact, it is likely that there are more suitable alternatives for the culture of human cells. 16 ‘Animal-free’ alternatives rely on sufficient characterisation of the active mitogenic components in traditional sera to maintain basic cellular functions and permit proliferation. Although synthetic serum alternatives exist and may be a better solution, 17 specialised culture media also typically contain a cocktail of additional growth factors that are specific to the maintenance of a particular cell phenotype. Recombinant growth factors are available, but are still commonly stabilised with BSA, unless specifically stated otherwise. 18 At present, protocols to ensure that human primary cell culture systems are completely animal component-free are often bespoke, which means that the culture requirements need to be adapted for each cell and tissue type.
In the current study, a variety of different cell types — namely, dermal fibroblasts, pulmonary fibroblasts, uterine fibroblasts, retinal endothelial cells and peripheral blood mononuclear cells — were adapted to completely animal-free culture conditions. To assess the protocol suitability and determine whether commonalities in ‘humanisation’ protocols could be realised, we compared cell viability, survival and growth parameters of cells cultured under human-relevant conditions with cells cultured under standard conditions. For the five human cell types tested, it was concluded that transition to animal-free culture conditions was possible, without overt adverse effects on cell kinetics or phenotype. Although there is not a one-size-fits-all approach to this transition, some commonalities exist. We demonstrate the process used for decision-making and troubleshooting that informed the transition of multiple cell types, therefore showing the possibility of animal component-free cell culture as a step toward improving the translation potential of current preclinical tissue and cell culture models.
Materials and methods
The omission of ADBs from in vitro cell culture systems cannot be a one-size-fits-all approach, as each cell type will have individual requirements to ensure a successful transition to animal-free culture conditions. In view of this, a number of crucial steps should be undertaken prior to any laboratory experimentation, namely: — the identification of ADBs used in the existing systems; — investigation into, and sourcing of, appropriate alternative reagents; and — the selection of appropriate outcome measures.
These steps are explored extensively in the thesis of Dr L.R. Bramwell,
19
which is available as a practical guide (https://teamrna.wixsite.com/harrieslab/animal-free-research). The process that was followed, to identify, transition and validate the culture of multiple human cell types in animal-component free (ACF) media, is outlined in Figure 1. A flowchart outlining the process of transitioning human cells from their recommended ADB-containing medium to an ACF medium. ACF = animal component-free; animal-derived biomaterial = ADB. N = No; Y = Yes.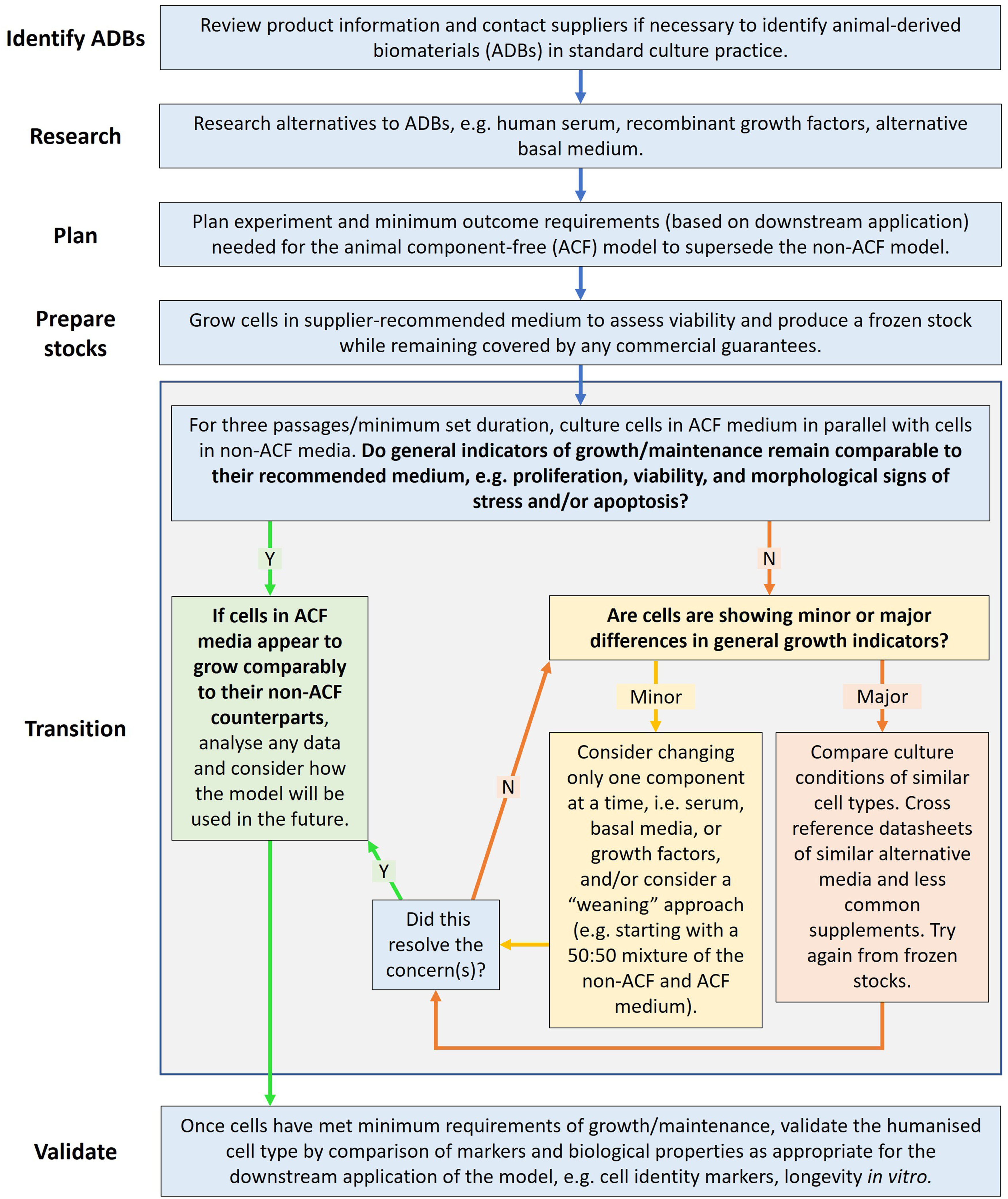
Following a literature review and planning phase, the initial cell cultures for expansion and transition experiments were prepared according to the vendors’ recommendations, to maintain any commercial guarantees of initial cell viability. We then carried out a parallel comparison of growth and survival parameters under animal-free culture conditions and in the original standard baseline media. Adherent cells were cultured over four passages, and suspension cells were maintained in vitro, with the measurements taken during the culture period.
Identification of ADBs in the studied in vitro culture systems
The initial stage of transition to animal-free culture conditions requires identification of the ADBs used in the standard culture conditions for each cell type. Product information and/or personal correspondence with company representatives were used to confirm whether the recommended medium for each cell type contained ADBs. Where ADBs were present in the medium, vendors were initially asked to recommend a suitable ACF alternative. Following this initial enquiry, we carried out a literature analysis to identify alternative animal component-free culture protocols that were already in use, by consulting resources such as NCBI PubMed, (https://pubmed.ncbi.nlm.nih.gov/), the FCS-free Database (https://fcs-free.org/) and Cellosaurus (https://www.cellosaurus.org/), as well as contacting alternative suppliers to determine the suitability of other commercially available media of a similar nature.
The cell lines used in the study
Details of the cell lines used in the study.
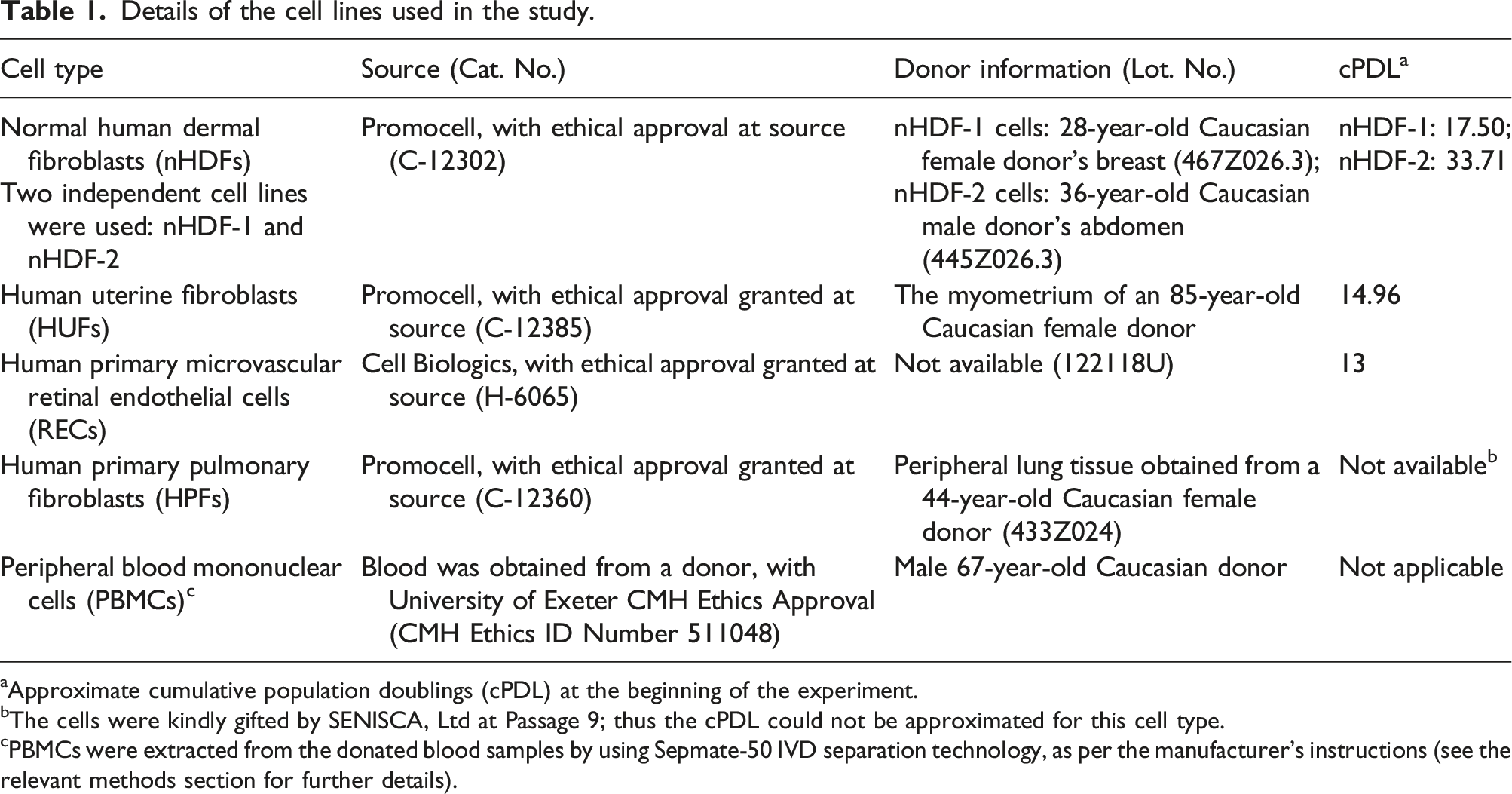
aApproximate cumulative population doublings (cPDL) at the beginning of the experiment.
bThe cells were kindly gifted by SENISCA, Ltd at Passage 9; thus the cPDL could not be approximated for this cell type.
cPBMCs were extracted from the donated blood samples by using Sepmate-50 IVD separation technology, as per the manufacturer’s instructions (see the relevant methods section for further details).
The PD time value was then used to calculate the cPDL, according to the following formula: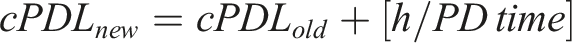
Dermal fibroblasts
Two independent cell lines of normal human dermal fibroblasts (nHDFs) were commercially sourced from Promocell (Heidelberg, Germany), for inclusion in the study. These are referred to hereafter as nHDF-1, which was used to compare a medium containing ADBs with an ACF medium, and nHDF-2, which was used to compare a medium containing a new type of non-animal derived supplement with human serum-containing medium.
Uterine fibroblasts
Human uterine fibroblasts (HUFs) were commercially sourced from Promocell (Heidelberg, Germany), for inclusion in the study. These cells were derived from the myometrium of an 85-year-old Caucasian female.
Retinal endothelial cells
Human primary microvascular retinal endothelial cells (RECs) were purchased from Cell Biologics (Chicago, IL, USA); no donor information was available.
Pulmonary fibroblasts
Human primary pulmonary fibroblasts (HPFs) were commercially sourced from Promocell, for inclusion in the study. These cells were derived from peripheral lung tissue obtained from a 44-year-old Caucasian female donor. The cells were kindly gifted by SENISCA, Ltd at Passage 9.
Peripheral blood mononuclear cells
Peripheral blood mononuclear cells (PBMCs) were obtained from the donated blood of a 67-year-old human male of Caucasian descent. Blood was obtained from the donor with University of Exeter CMH Ethics Approval (CMH Ethics ID Number 511048). The PBMCs were extracted from the blood samples by using Sepmate-50 IVD separation technology, as per the manufacturer’s instructions (Cat. No. 85450 and Cat. No. 07851; Stemcell Technologies, Vancouver, Canada). The PBMCs were not maintained for long-term growth due to their naturally short-term viability and so were cryogenically preserved after extraction. The cells were thawed rapidly at 37°C, added to 5 ml of pre-warmed medium and then centrifuged at 240g before resuspension in fresh medium. Then, they were seeded into a 24-well plate at a cell density of 1 × 106 cells/ml. As cells only need passaging when the medium is nutritionally depleted, or when the maximal seeding density within the culture vessel is exceeded, passaging was not required during this study.
The control (baseline) media and the animal component-free (ACF) trial media
The control (baseline) culture media and animal component-free (ACF) trial media.

nHDFs = normal human dermal fibroblasts; HUFs = human uterine fibroblasts; HPFs = human pulmonary fibroblasts; RECs = retinal endothelial cells; PBMCs = peripheral blood mononuclear cells.
DMEM = Dulbecco’s Modified Eagle Medium; FBS = fetal bovine serum; EGF = epidermal growth factor; FGF = fibroblastic growth factor; bFGF = basic fibroblastic growth factor; VEGF = vascular endothelial growth factor.
Dermal fibroblasts
In the ACF trial medium for nHDF-1, the 10% v/v FBS in the original baseline medium was substituted for 10% v/v filtered human serum. In the ACF trial medium for nHDF-2, a new type of non-animal derived supplement based on human platelet lysate (HPL) — GroPro Cell Culture Growth Supplement (SER-HPL-GROPRO, Zen-Bio, Durham, NC, USA), at a concentration of 10% v/v — was used instead of the 10% FBS.
Uterine fibroblasts
The 10% FBS in the original baseline medium was substituted for 10% filtered human serum in the ACF trial medium.
Retinal endothelial cells
The recommended basal medium for the RECs, Cell Biologics Complete Human Endothelial Cell Medium (Cat. No. H-1168), contained ADBs. An alternative basal medium from Promocell, Endothelial Cell Growth Medium MV2 (Cat. No. C-22221), was therefore considered for use as the baseline medium. The supplement kits supplied with both of these basal media (i.e. from Cell Biologics and from Promocell) contained BSA, so alternative animal-free growth factor sources were sought. However, as the supplement kits contained different combinations and concentrations of growth factors, direct substitutions with animal-free alternatives could not be made without firstly considering which growth factors should be included and at what concentrations. Therefore, a wider range of commercially available endothelial cell media supplement kits were compared, in order to identify common growth factors and thus formulate an appropriate ACF trial medium for use in the current study (see Table 2).
Pulmonary fibroblasts
Initially, the 10% FBS in the original baseline medium was substituted for 10% filtered human serum in the ACF trial medium. However, this caused a substantial reduction in cell proliferation compared to that observed in the original ADB-containing medium (data not shown). This impaired proliferation was resolved by the addition of 1 ng/ml basic fibroblastic growth factor (bFGF) to the initial ACF trial medium, thus ensuring that the proliferation rate of the newly humanised cells was comparable to that obtained in the control medium.
Peripheral blood mononuclear cells
The ACF trial media for the PBMCs were based on RPMI-1640 medium containing 1% v/v 10,000 units/ml penicillin–10,000 μg/ml streptomycin and were supplemented with either 10% filtered human serum or 10% GroPro Cell Culture Growth Supplement (see Table 2).
Assessment of growth kinetics, cell survival and morphological characteristics
Suboptimal culture conditions can result in changes to the cells that can be obvious on visual inspection with a light microscope, such as gross effects on apparent cell survival, or more subtle changes in cell morphology. Unless otherwise stated, for the experiments in this study, a DeNovix CellDrop™ machine with Acridine Orange/Propidium Iodide (AO/PI) viability staining (Cat. No. A8097 and Cat. No. P4864; Sigma Aldrich, Merck) was used to count the cells, calculate population doubling time, measure the average cell diameter and assess cell viability. An AO stock solution (at a working concentration of 5 μg/ml in sterile ddH2O) and a PI stock solution (at a working concentration of 100 μg/ml in sterile ddH2O) were combined in equal volumes, vortexed and stored in black-walled tubes at 4°C until required. Immediately prior to cell counting, a 10 μl sample of the cell suspension was diluted 1:1 with 10 μl of the combined AO/PI stain, mixed by gentle pipetting and 10 μl pipetted onto the CellDropTM machine for measurement.
Primary human dermal and uterine fibroblast cells were counted with a Hirschmann haemocytometer, because their inherent uneven morphology renders automated counting less accurate than manual counting. Assessments were carried out by light microscope (Zeiss AxioCam ERC55 PrimoVert) to identify any obviously visible stress and cell death responses — for example, appearance of stress granules, membrane blebbing, production of cell detritus and apoptotic cells.
Costs
A comparison of costs for the control (baseline) media and the animal component-free (ACF) trial media.
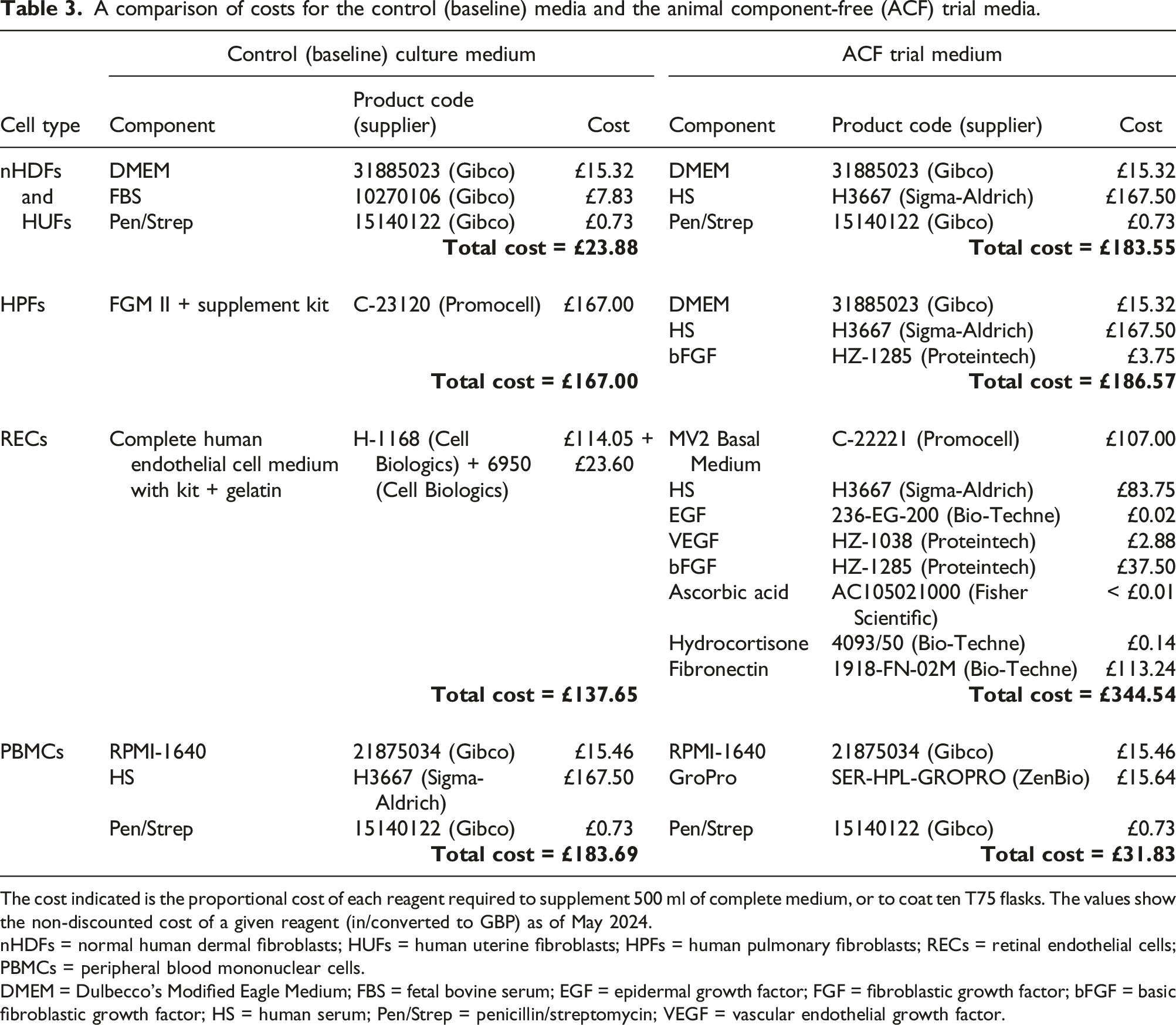
The cost indicated is the proportional cost of each reagent required to supplement 500 ml of complete medium, or to coat ten T75 flasks. The values show the non-discounted cost of a given reagent (in/converted to GBP) as of May 2024.
nHDFs = normal human dermal fibroblasts; HUFs = human uterine fibroblasts; HPFs = human pulmonary fibroblasts; RECs = retinal endothelial cells; PBMCs = peripheral blood mononuclear cells.
DMEM = Dulbecco’s Modified Eagle Medium; FBS = fetal bovine serum; EGF = epidermal growth factor; FGF = fibroblastic growth factor; bFGF = basic fibroblastic growth factor; HS = human serum; Pen/Strep = penicillin/streptomycin; VEGF = vascular endothelial growth factor.
Statistics
Independent t-tests were used to compare the means for all experiments, except the comparisons of PBMCs grown in parallel in three different experimental media, which used a one-way ANOVA with Tukey’s post-hoc test. p values below 0.05 were considered statistically significant for a difference between experimental groups. n indicates the number of individual measurements taken across one experiment. Statistical tests and graphs were produced using Graphpad Prism version 9.4.1 for Windows (GraphPad Software, San Diego, CA, USA, www.graphpad.com). Error bars on the graphs represent the standard error of the mean (SEM).
Results
Transition to animal-free culture conditions does not affect population doubling time for adherent cell lines
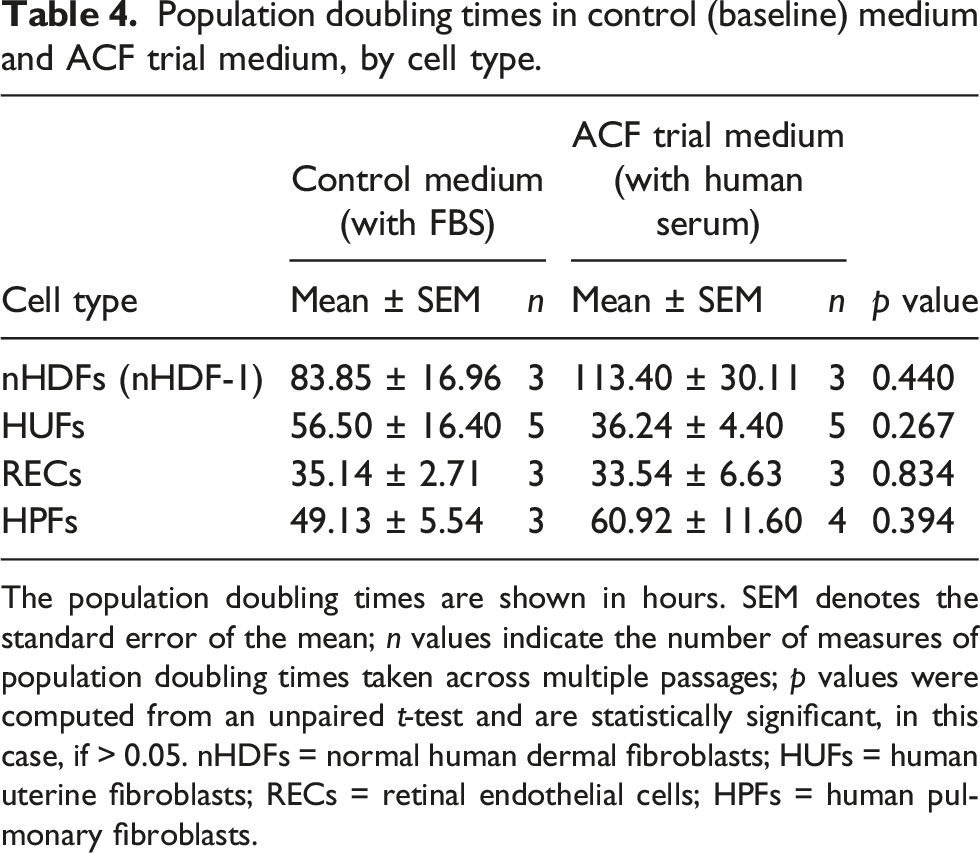
Figure 2 shows that the adherent cells grown in media containing human serum (i.e. nHDF-1, HUF, REC and HPF cells) grew at the same rate as cells grown in media containing FBS. Population doubling (PD) times (hours) for the adherent cell lines under the control (baseline) culture media and the ACF media are shown in Table 4. Population doubling times for four different cell types. The graphs show the population doubling (PD) times for cells cultured in medium supplemented with either fetal bovine serum (FBS) or with human serum (HS). a) Normal human dermal fibroblasts (nHDF-1; n = 3 for both media); b) human uterine fibroblasts (HUFs; n = 5 for both media); c) retinal endothelial cells (RECs; n = 3 for both media); and d) human pulmonary fibroblasts (HPFs; n = 4 for both media). The error bars denote the mean ± standard error of the mean (SEM); ns = not significant for an unpaired t-test. Population doubling times in control (baseline) medium and ACF trial medium, by cell type. The population doubling times are shown in hours. SEM denotes the standard error of the mean; n values indicate the number of measures of population doubling times taken across multiple passages; p values were computed from an unpaired t-test and are statistically significant, in this case, if > 0.05. nHDFs = normal human dermal fibroblasts; HUFs = human uterine fibroblasts; RECs = retinal endothelial cells; HPFs = human pulmonary fibroblasts.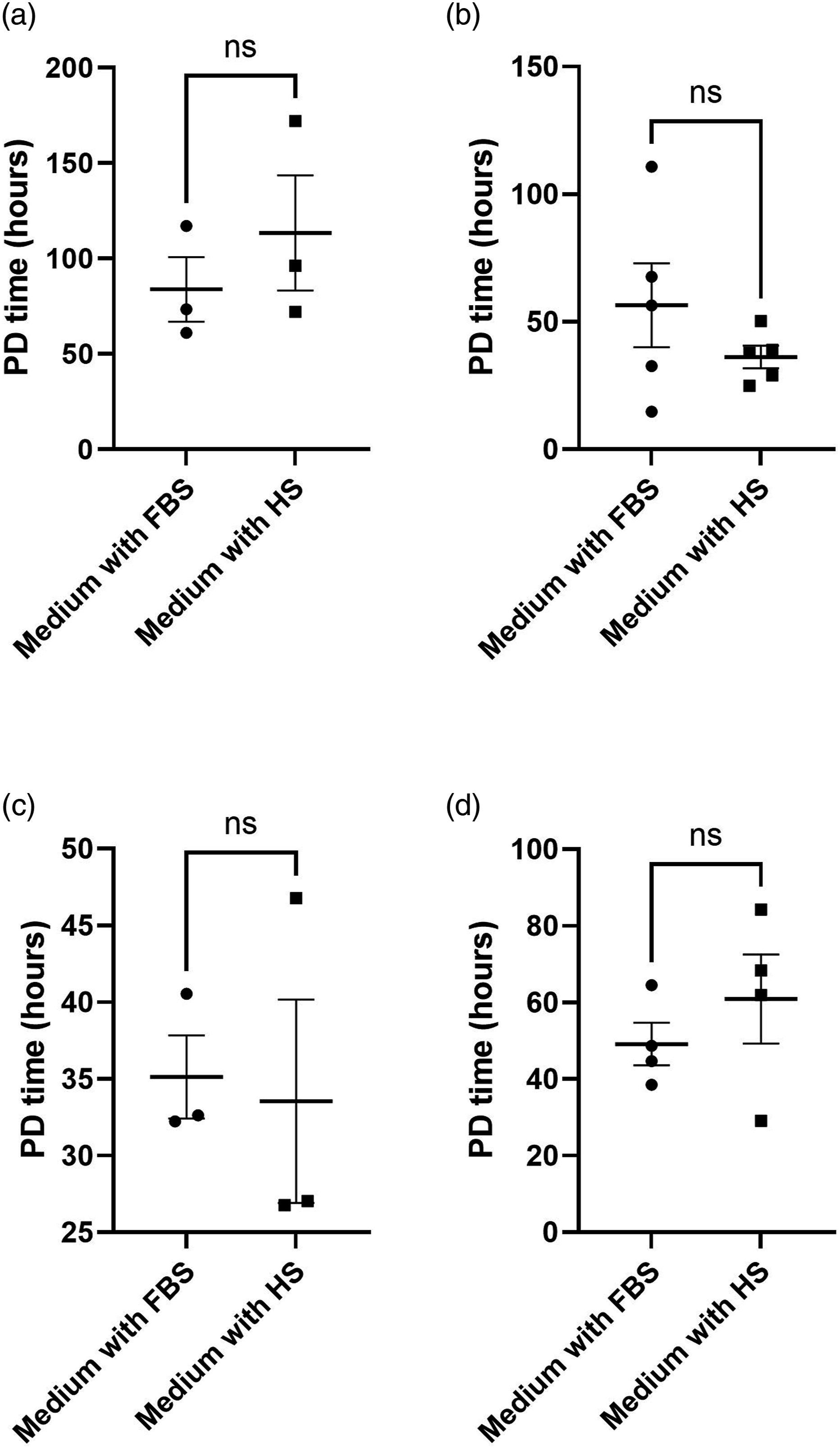
The effects of a new type of non-animal derived supplement on PBMC and dermal fibroblast survival in culture
Substitution of serum with a new type of non-animal derived supplement, GroPro, did not affect PBMC survival in in vitro culture, but it was unsuitable for use as a direct substitute for serum in dermal fibroblast cultures.
Peripheral blood mononuclear cells
As PBMCs are relatively non-proliferative in in vitro culture, cell survival is a more appropriate outcome measure than proliferation for this particular cell type. PBMC survival did not differ when the cells were subjected to different serum supplementation regimes in short-term culture. Cell survival decreased very slightly over five days in all of the media tested, but there were no significant differences between the cells cultured in the presence of FBS, human serum or GroPro (see Figure 3). On Day 5, viable PBMCs in medium containing FBS were at 92% of their original seeding density, whereas viable cells in media containing human serum or GroPro were at 98% and 95% of their original seeding density, respectively (p = 0.93). Short-term survival of PBMCs in media with different supplements. Cell counts were taken at three intervals over a five-day culture period. The circles represent the cells grown in medium supplemented with fetal bovine serum (FBS); the squares represent medium supplemented with human serum (HS); and the triangles represent medium supplemented with GroPro. The error bars show the mean ± standard error of the mean (SEM); n = 3 for all data points. PBMCs = peripheral blood mononuclear cells.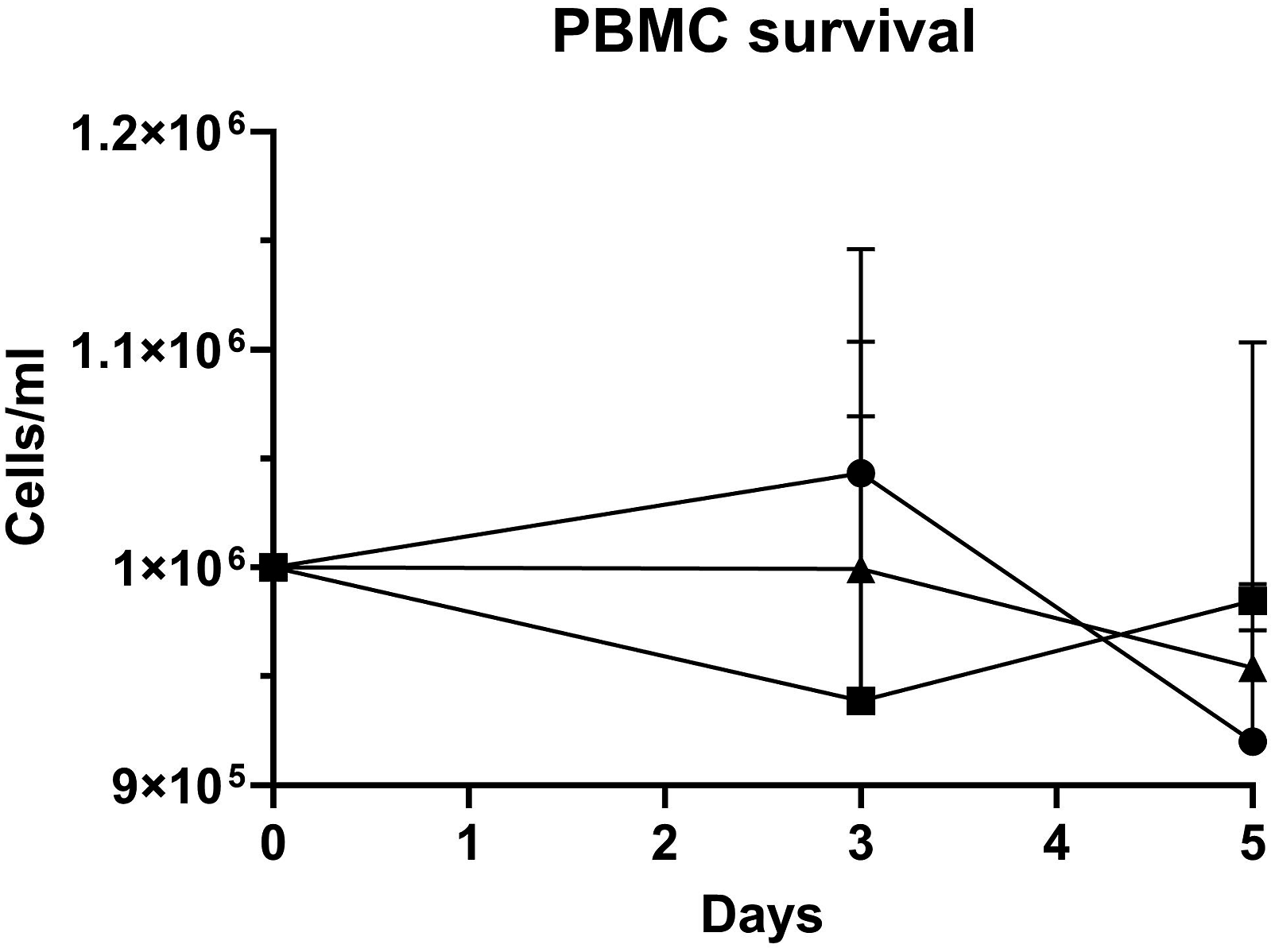
Dermal fibroblasts
Cell sizes during growth in control (baseline) medium and the ACF trial media.
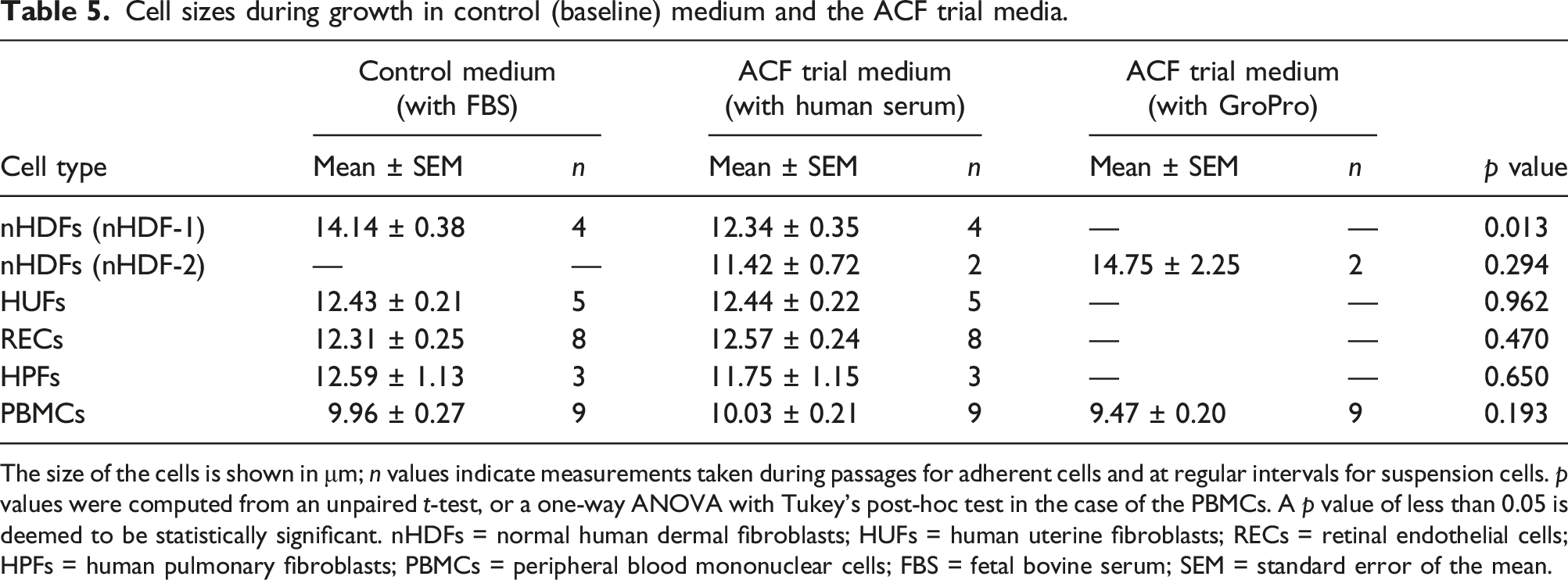
The size of the cells is shown in μm; n values indicate measurements taken during passages for adherent cells and at regular intervals for suspension cells. p values were computed from an unpaired t-test, or a one-way ANOVA with Tukey’s post-hoc test in the case of the PBMCs. A p value of less than 0.05 is deemed to be statistically significant. nHDFs = normal human dermal fibroblasts; HUFs = human uterine fibroblasts; RECs = retinal endothelial cells; HPFs = human pulmonary fibroblasts; PBMCs = peripheral blood mononuclear cells; FBS = fetal bovine serum; SEM = standard error of the mean.
The effect of the transition to animal-free culture conditions on cell size
The data in the current study indicates that the transition to animal-free culture conditions can affect the size of the cells, but this effect was cell-type specific. The mean cell diameter of nHDF-1 cells was reduced by 13% in the ACF medium containing human serum, as compared to the control (baseline) medium containing FBS (p = 0.013). However, the mean cell size of the HUFs, RECs, HPFs and the PBMCs was unchanged when the cells were transitioned to their respective ACF trial media. These results are illustrated in Figure 4, with the full data shown in Table 5. Size measurements for five different cell types. The graphs show the size measurements of cells cultured in medium supplemented with fetal bovine serum (FBS), human serum (HS) or GroPro. a) Normal human dermal fibroblasts (nHDF-1; n = 4 for both media shown); b) human uterine fibroblasts (HUFs; n = 5 for both media shown); c) retinal endothelial cells (RECs; n = 8 for both media shown); d) human pulmonary fibroblasts (HPFs; n = 3 for both media shown); and e) peripheral blood mononuclear cells (PBMCs; n = 9 for all three media shown). The error bars denote the mean ± standard error of the mean (SEM). Significance for either an unpaired t-test, in (a) to (d), or a one-way ANOVA with Tukey’s post-hoc test in (e), is denoted as follows: ns = not significant; *p < 0.05.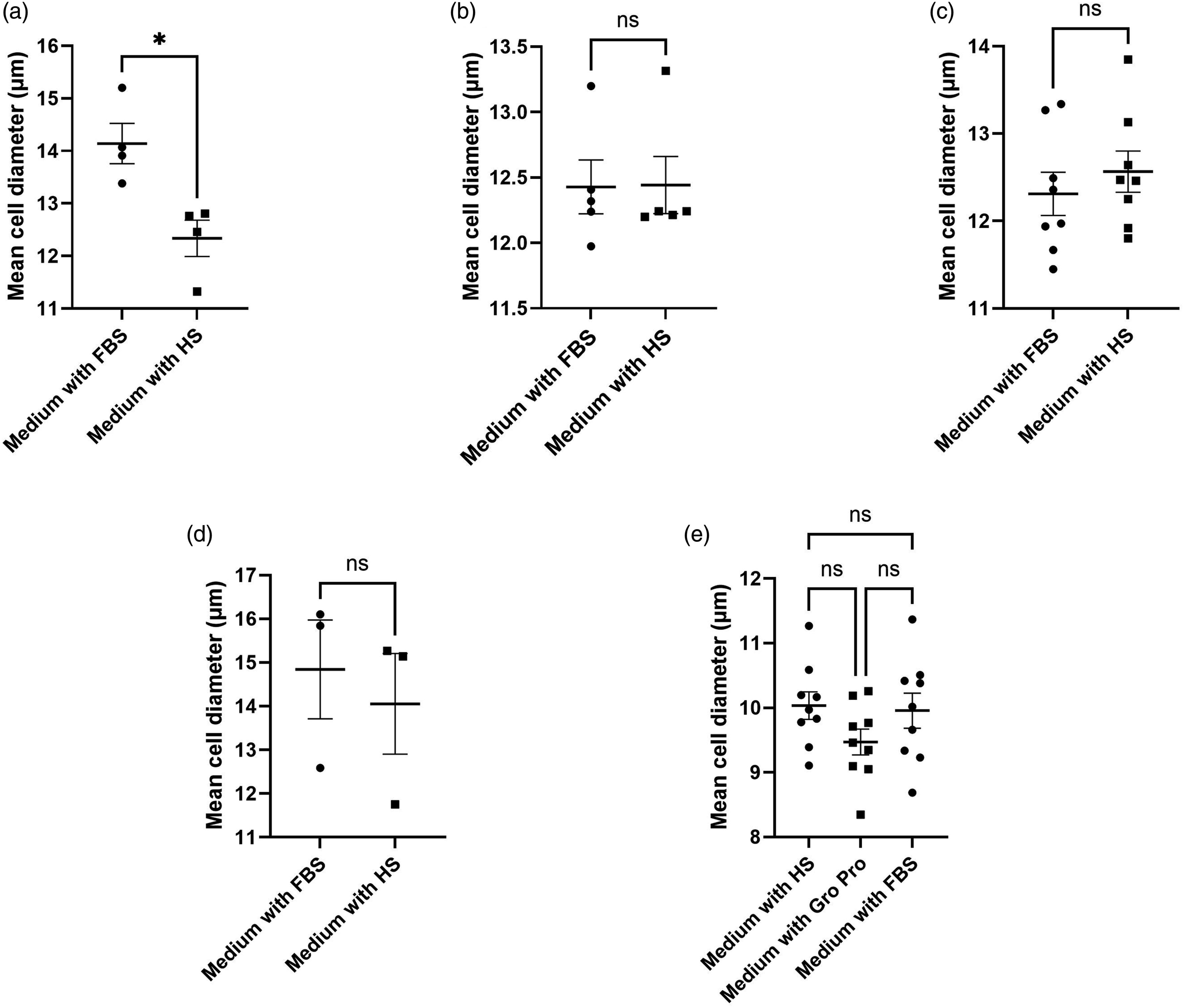
There was no apparent difference in size between nHDF-2 cells cultured in human serum-supplemented medium and GroPro-supplemented medium; however, the cells died too early when cultured in the presence of GroPro to draw any further conclusions in this respect.
The cells were consistently monitored throughout the transition process, to ensure that the cell morphology and viability remained sufficiently comparable between the two groups of cells (i.e. those growing in the original ADB-containing medium and those growing in the ACF trial medium). Example images of HPF cells, that illustrate the type of cell morphology and viability that was typically observed, are shown in Figure 5. After three passages in ACF medium, the HPFs appear as elongated spindles, with no visual signs of stress, debris or apoptotic bodies. Indeed, they are morphologically analogous to the cells cultured in the original FBS-containing medium. Light microscopy images of human pulmonary fibroblasts. Human pulmonary fibroblasts (HPFs) were grown in: a) the recommended FBS-containing medium; and b) the ACF trial medium. The images were captured at Passage 13, using the automatic settings of a Zeiss AxioCam ERC55 PrimoVert (image contrast, sharpness and colour balance were adjusted post-acquisition for display purposes).
Discussion
Scope
We have successfully formulated ACF alternatives to the medium traditionally used for several different human cell lines. Human serum was successfully used as an alternative to FBS, to supplement the culture media of dermal fibroblasts, uterine fibroblasts, retinal endothelial cells and PBMCs. Our results also showed that it was possible to substitute FBS with a new type of ACF supplement, GroPro, in PBMC cultures but not in nHDF cultures. The processes that were undertaken to successfully transition the cells to their respective ACF media, as well as various points for consideration when trialling different ACF media alternatives, are discussed in detail below.
In the US, the legal requirement for animal in vivo studies to be carried out as part of the clinical pipeline has recently been removed. 20 This echoes the consensus that there are cases in which in vivo animal models do not provide a meaningful contribution to the assessment of drug safety or efficacy in humans, due to the poor translatability of results from preclinical research, and further amplifies the notion that animal in vivo models are not strictly necessary for the study of human diseases. Not only do animal in vivo models pose inherent ethical issues, but they also possess biological drawbacks due to inter-species differences. The species-specific nature of some biological processes could mean that xeno-free human cell culture models would be more representative of human in vivo physiology than common culture models that contain ADBs.16,21–23
It may be argued that, at present, the main reason for upholding the use of ADBs as the ‘gold standard’ in human cell culture is that it represents ‘traditional practice’. Considering this, and the recent change in guidelines, we propose that moving away from ‘traditional practice’ may provide the first simple, yet often overlooked, step toward improving the physiological relevance of the cell culture conditions. This will, in turn, improve the translatability of the results obtained with the in vitro model. However, the transition of a cell line to growth in a different medium is not always straightforward, and many factors need to be considered to maximise the chances of success. Prior to starting the transition process for each model, it is important to identify the critical attributes of the existing culture conditions that must be maintained, and those that can be altered. This can be more challenging in the case of proprietary culture media, as the formulations are often undisclosed. Thus, they may include undefined concentrations and/or batches of growth factors, sera and/or other components. 24
Trial media transitioning studies
For most cell types, the initial approach for transitioning from growth medium containing FBS and other ADBs was the trial substitution of such media supplements with their non-animal derived equivalents. For all of the cell types tested in the current study, there were no significant differences observed in PD times between cells that were cultured in their recommended ADB-containing medium and cells that were cultured in the ACF equivalent. This supports the concept that ACF culture medium is a viable alternative to medium containing animal sera.
Only one of the five cell types studied exhibited a significant difference in average cell size when comparing growth in the control medium and in the ACF equivalent. During our experiments, cell morphology, viability and stress appeared similar in the FBS-containing and ACF media. Whilst morphology varies between cell types, the characteristic morphology remained consistent between cells from the same donor cultured in the two types of medium. This is indicated by the representative comparative images of HPFs that had been cultured in parallel for more than three passages in the different media. The ‘humanisation’ of the culture conditions was therefore deemed successful, allowing the cells to be taken forward for use in further experiments.
The wider application of animal-free in vitro cell culture
In addition to this study, further research from our group demonstrates the value of animal-free culture practices in wider research, with multiple discoveries being made in the fields of viral immunology, the biology of ageing, and diabetes.12,25–28 In the case of our research into the biology of ageing, human primary dermal fibroblasts were successfully cultured under animal-free conditions well beyond the manufacturer’s standard time-limit, until replicative senescence was eventually reached. This could potentially be more widely applied to a range of other cell types and human disease studies.
There has been a recent increase in the availability of xeno-free media for the culture of specialised cell types, including for the generation and maintenance of stem cells. This should provide further confidence that ACF media are not only suitable for the effective maintenance of cells but, due to the inherent reproducibility of such media, they could potentially improve the experiments carried out based on such specialised cell culture, as well as its wider applications.29–33 Variations in reprogramming potential and cell morphology, as well as gene and cytokine expression profiles, have been observed between iPSC cultures maintained in FBS-containing and xeno-free media.32,34 However, these differences were also anatomical site and donor dependent. 34 It has been widely reported that culture conditions can influence morphology, cytokine expression and gene expression in many types of cell, including iPSCs and human primary cells.35–37 Culture media, plasticware and passaging protocols can also affect cell phenotype, and hence expression profiles, regardless of whether ACF media are used. 38 Therefore, in order to conclusively determine the specific culture methods that are more phenotypically relevant, it would be beneficial to compare in vivo cellular expression profiles to those of in vitro-grown cells. However, donor-to-donor variability could impede appropriate conclusions from being drawn, unless large datasets from multiple donors were available.
Human serum considerations
When human serum is used, some additional considerations and preparation steps were found to be beneficial, prior to its routine implementation in cell culture. The commercial supplier provided sera that was heat inactivated, screened and obtained from healthy AB+ donors, minimising the risk of infectious contaminants, antibodies or inflammatory factors that could lead to damage or immuno-activation of the cells. In most cases, as with FBS, batch testing can provide a helpful means to identify serum that is most appropriate for the desired use. Human serum can be relatively turbid and can require additional filtering prior to use. Due to its human origin, there is a risk of cross-reactivity of its inherent antibodies and proteins in any downstream assays, but in most cases batch testing can help to mitigate this. However, for this reason, human serum is not suitable for use in culture when the experiments are intended for the detection of extracellular proteins/antibody production. Animal-free culture is more financially comparable to specialised cell culture than it is to ‘standard’ culture based on ADB-containing media. We demonstrate here that this is mainly due to the relatively high cost of human serum for ACF media supplementation.
Other non-animal derived media supplements
Other types of serum alternatives are promising avenues to explore for use in routine in vitro cell culture. However, these alternatives pose their own issues. As demonstrated in the current study, the non-animal derived supplement, GroPro, was not suitable for use as a comprehensive replacement for either human serum or FBS — even though it did support the in vitro survival of PBMCs, it was unable to support nHDFs. GroPro is derived from HPL, and therefore may exhibit batch-to-batch variability in a similar manner to animal-derived sera. A next step for optimising the use of GroPro supplement for fibroblast culture could be to add synthetic carrier-free growth factors to the mix. It is also worthwhile noting that, unless growth factors are specifically listed as ACF or carrier-free, synthetic growth factors often contain small amounts of BSA. Carrier-free growth factors can be reconstituted in human serum albumin (HSA) for stability.
A number of fully ACF and chemically defined media and supplements are now commercially available. These do not exhibit batch-to-batch variation in the same way as biological products. Weber et al. 29 detailed a reproducible (mostly chemically defined) ACF medium that can support a variety of human cancer and normal immortalised cell lines, as well as primary nHDF cultures. 29 This medium may represent a promising starting point for the animal-free culture of a variety of human primary cell types. The transition process and evaluation methods presented here are applicable to such fully defined alternatives. This approach could therefore be used to assess the benefits of incorporating various cell type-dependent recombinant growth factors into the culture media to optimally support the maintenance and differentiation of specialised primary cell types. This could save valuable protocol development time and, providing that the cells maintain their original phenotype, allow for the rapid and consistent transition of cells to ACF media for use in downstream experiments.
Final remarks
The inherent limitations of in vitro cell and tissue culture are present whether using ‘traditional’ or animal-free culture techniques. When transitioning cells to animal-free culture conditions, it is always important to consider which critical attributes of the existing culture conditions must be upheld in order to produce a physiologically relevant in vitro model. We have demonstrated that the evaluation of cell viability, cell morphology and proliferative capacity represents a realistic and positive first step toward the successful adoption of human-relevant cell culture practices.
Depending on the downstream applications, phenotypic markers or transcriptomics may need to be further assessed before the implementation of a new cell culture model system. A first port of call for evaluating phenotypic molecular markers after transitioning the cells toward growth in ACF media, would be to determine the expression of the cell surface protein markers outlined by the cell line supplier. For example, in the case of nHDF, HUF and HPF cells, this would be the mesenchymal surface marker CD90 and, for REC cells, the cell surface and cellular adhesion markers CD31 and VE-cadherin, respectively. PBMCs are a differentiated and heterogenous cell population and thus would require a more complex characterisation, incorporating a variety of markers. 33 In Figure 1, we outline the process followed in this study for the ‘humanisation’ of the culture conditions, which informed the successful transition of multiple cell types toward growth in ACF media. So far, our studies have helped to, at the very least, reduce the requirement for animal-derived components in human cell culture. Of equal importance, they have also potentially contributed toward the development of models that are more physiologically relevant to humans and will thus generate results that are more translatable to in vivo human diseases.
Conclusions
From the bench to the bedside, potential new therapeutics often encounter multiple barriers as they progress from basic research to clinical trials. Often, these issues are rooted in problems associated with poor physiological relevance of animal models and/or models that use animal-derived components. Making research models more human-relevant at every stage is therefore worth considering. The removal of ADBs from the in vitro culture process has been successfully performed for five human primary cell types (dermal fibroblasts, uterine fibroblasts, retinal endothelial cells, pulmonary fibroblasts and peripheral blood mononuclear cells), demonstrating the possibility of taking a simple step toward increasing physiological relevance in basic in vitro research. The process of ‘humanising’ these cell culture conditions highlighted just how commonly-used ADBs are in in vitro research. Human serum is a straightforward and strong candidate for the replacement of FBS, the most commonly used animal-derived supplement in human cell culture. Although non-animal derived media supplements are viewed as promising alternatives in view of their defined characterisation and reproducibility, they are not yet suitable for all cell types. The approach outlined in this paper guides researchers that are looking to remove ADBs from their day-to-day cell culture practices, and highlights some of the key considerations and current challenges faced in order to achieve this goal.
Footnotes
Acknowledgements
The authors wish to thank to Dr Nicola Jeffery for discussions regarding the humanisation process and SENISCA, Ltd for the kind provision of the human pulmonary fibroblast cells. This research was supported by the National Institute for Health and Care Research (NIHR) Exeter Biomedical Research Centre (BRC). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Harries is an inventor on patent PCT/GB2019/052125, and is a founder, chief scientific officer and research and development lead for SENISCA, Ltd. Dr Gould and Dr Davies are research scientists at SENISCA, Ltd. Dr Bramwell, Mr McMullan and Ms Trusler have no financial interests to declare in relation to the content of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge funding from Animal Free Research UK (grant numbers 18-001, 147, 149 and 182). No reagents containing animal-derived biomaterials were purchased with this grant.