Abstract
Phototoxicity testing is crucial for evaluating the potential harmful effects of pharmaceuticals and chemicals on human skin when exposed to sunlight. Traditional
Introduction
Phototoxicity can be elicited by exposure to certain chemicals — including pharmaceuticals, cosmetics or food ingredients — that are photoactivated by ultraviolet (UV) A/B light.1–3 Several classes of pharmaceuticals often cause phototoxic reactions in the skin and eyes at clinical doses, 3 and perceptible adverse effects would lead to a reduction in medication compliance. In addition to the clinical issues, drug-induced phototoxicity has also halted the development of new drug entities, and the pharmaceutical industry and regulatory agencies have struggled to predict and/or avoid phototoxic liability.
Various
Given the considerable expense and time requirements associated with traditional
Materials and methods
A set of 57 non-proprietary compounds were analysed by using the
Compounds were entered into Derek using SMILES strings. From the Derek Prediction Set Up, ‘phototoxicity’ prediction only was selected from the miscellaneous endpoints menu. When evaluating the results from Derek Nexus, various degrees of phototoxicity predictions were considered — such as ‘probable’, ‘plausible’ or ‘equivocal’ — as positive indicators for phototoxicity. Conversely, a negative result was assigned in cases where the query compounds did not match any phototoxicity alerts.
The OECD Toolbox was used in Classic User Interface mode. Compounds were entered into the Toolbox using the CAS number. For each compound, data available in the Toolbox from the photosensitivity database, was sought (under the category of Human Health Hazards, photoinduced toxicity). Information on this database (as stated in the OECD Toolbox) reports that this database contains photosensitisation data for chemicals evaluated by weight-of-evidence. The weight-of-evidence approach involves using a combination of information from several independent sources to give sufficient evidence to fulfil an information requirement. According to the information provided on the OECD Toolbox model, the weight-of-evidence evaluation is based on the following: “(1) Negative in phototoxicity test (
Results
Our study yielded significant insights into the predictive performance of Derek Nexus and of the QSAR Toolbox for phototoxicity assessment. A summary of our results can be found in Table 1, where we compiled the phototoxicity alerts provided by Derek Nexus and the QSAR Toolbox. Based on the compiled information, the compounds were classified as True Positive (TP), False Positive (FP), True Negative (TN) and False Negative (FN), as presented in Figure 1. Phototoxicity prediction by using Derek Nexus and the QSAR Toolbox. TP = true positive; FN = false negative; FP = false positive; TN = true negative; +ve = positive, i.e effect observed; –ve = negative, i.e no effect observed; PPV = Positive predicted value; NPV = Negative predicted value. Phototoxicity data including aThis column refers to the collective results obtained with the 3T3 NRU PT assay, 3-D tissue models and historical bCall for phototoxicity, based on the outcome of the Derek cCall for phototoxicity, based on the outcome from the OECD Toolbox, where +ve = a positive result as recorded in the photoxicity database within the Toolbox, or a positive prediction based on the phototoxicity QSAR model available in the Toolbox; –ve = a negative result as recorded in the photoxicity database within the Toolbox or out of domain of the QSAR model (please refer to the main text). PT = phototoxicity; NPT = no phototoxicity; +ve = positive, i.e effect observed; –ve = negative, i.e. no effect observed.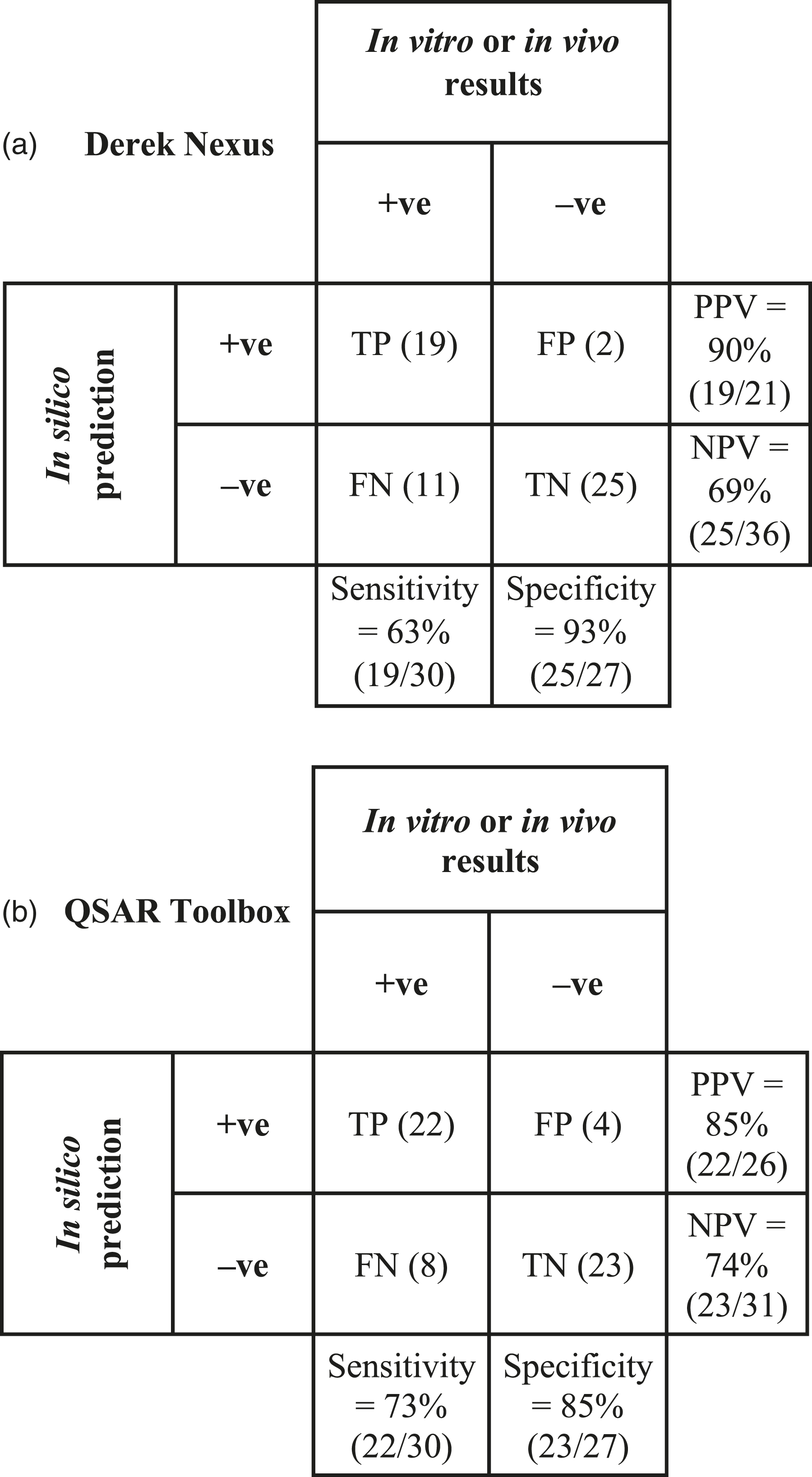
Derek Nexus showed a sensitivity of 63%, specificity of 93%, Positive Predictive Value (PPV) of 90%, Negative Predictive Value (NPV) of 69%, overall accuracy of 77% and balanced accuracy of 78%. Similarly, the QSAR Toolbox showed a sensitivity of 73%, specificity of 85%, PPV of 85%, NPV of 74%, overall accuracy of 79% and balanced accuracy of 79%. The formulae employed for calculating these predictive parameters were as follows: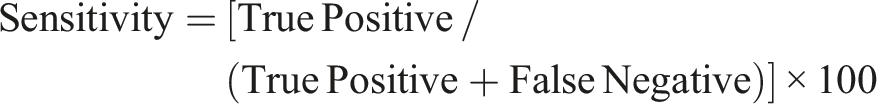
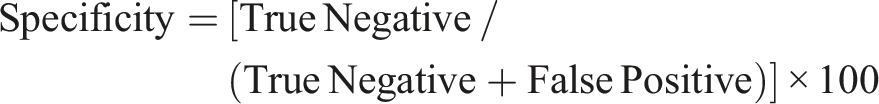

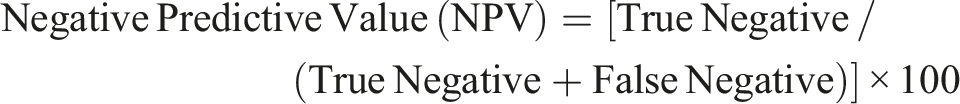

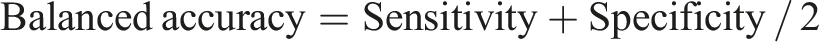
It is essential to acknowledge that both Derek Nexus and the QSAR Toolbox predictions are applicable only to substances with known chemical structures. In contrast, predicting the phototoxic potential of mixtures, formulations and oils (such as plant extracts and cosmetics) with unknown compositions, remains difficult using
Our findings highlight the potential of incorporating QSARs into the workflow for assessing phototoxicity in pharmaceuticals, particularly within the context of the ICH S10 guidelines. Additionally, QSARs can be valuable for the initial phototoxicity evaluation of chemicals.
Discussion
Exogenous compounds that reach the skin after topical application or after oral exposure, such as pharmaceuticals and personal care products, can be activated by solar radiation and may contribute to adverse effects in the skin, such as photoirritation, photo-induced ageing or photocarcinogenicity.2,11,12 As a result, photosafety testing has become a mandatory regulatory requirement for consumer products that absorb light in the range of 290–700 nm and with (relevant) partitioning in the skin or eyes. Guidance documents outlining the strategy and approaches for the photosafety evaluation of pharmaceutical products are provided by both the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) Center for Drug Evaluation and Research (CDER).13,14 In addition, an OECD guidance document on phototoxicity testing of chemicals is in preparation.
Testing of pharmaceutical compounds and personal care products may include evaluating acute phototoxicity (photoirritation), photoallergenicity, photogenotoxicity and photocarcinogenicity. 15 A limited number of knowledge-based expert systems are currently available for predicting photo-induced toxicity. This is not surprising, since establishing the phototoxic potential of chemical entities is a complex task, due to the various mechanisms involved in photoirritation, photogenotoxicity or photoallergenicity reactions. 16 Derek Nexus is one of the tools containing structural alerts for photo-induced toxicity. 17 However, Ringeissen and coworkers found that Derek was not successful at identifying the phototoxicity of compounds tested with the 3T3 NRU PT assay. 10
The QSAR paradigm is endorsed by one of the most widely used
In the current study, we obtained a balanced accuracy of 78% by using Derek Nexus and 79% with the QSAR Toolbox. Our analysis revealed that Derek Nexus (v6.1.0) and QSAR Toolbox (v4.5) are effective in predicting phototoxicity. We conducted an
Conclusions
In conclusion, our study demonstrates the value of Derek Nexus and QSAR Toolbox in accurately predicting the phototoxicity of drugs and chemicals. Derek achieved sensitivity of 63%, specificity of 93%, a Positive Predictive Value (PPV) of 90%, a Negative Predictive Value (NPV) of 69%, an overall accuracy of 77% and a balanced accuracy of 78%. Similarly, the QSAR Toolbox achieved commendable predictivity, showing a sensitivity of 73%, specificity of 85%, PPV of 85%, NPV of 74%, overall accuracy of 79% and balanced accuracy of 79%. Notably, to the best of our knowledge, Derek provided predictive alerts for phototoxicity that were unavailable through any other
However, for robust validation, further assessment of Derek Nexus and the QSAR Toolbox with a broader range of test compounds is warranted in order to address the broader spectrum of applicability domains, e.g. medical devices, pesticides, food and animal feed.
Despite this need for further assessment, the current results strongly support the incorporation of both Derek Nexus and of the QSAR Toolbox into the workflow of phototoxicity testing for pharmaceuticals, some raw materials used in cosmetics and medical devices, and other chemicals. The predictive capabilities exhibited by these tools offer a valuable means of streamlining phototoxicity evaluation, contributing to enhanced safety assessments in drug and chemical development while avoiding animal use.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly supported by APVV Research Grant APVV-19-0591:
