Abstract
The Institute for In Vitro Sciences (IIVS) is sponsoring a series of workshops to develop recommendations for optimal scientific and technical approaches for conducting in vitro assays to assess potential toxicity within and across traditional tobacco and various tobacco and nicotine next-generation products (NGPs), including Heated Tobacco Products (HTPs) and Electronic Nicotine Delivery Systems (ENDS). This report was developed by a working group composed of attendees of the seventh IIVS workshop, ‘Approaches and recommendations for conducting the mouse lymphoma gene mutation assay (MLA) and introduction to in vitro disease models’, which was held virtually on 21–23 June 2022. This publication provides a background overview of the MLA, and includes the description of assay conduct and data interpretation, key challenges and recommended best practices for evaluating tobacco and nicotine products, with a focus on the evaluation of NGPs, and a summary of how the assay has been used to evaluate and compare tobacco and nicotine products.
Keywords
Introduction
Specific guidelines and recommended genotoxicity test batteries for tobacco and nicotine products are currently limited. To date, these include: the Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) in vitro toxicology testing sub-group recommendations on the toxicity testing of combustible tobacco products; 1 and the recommendations for conducting and submitting genotoxicity evaluations of electronic nicotine delivery systems products under the US Food and Drug Administration (FDA) Guidance Premarket Tobacco Product Applications for Electronic Nicotine Delivery Systems (ENDS). 2
It is common practice, however, that tobacco and nicotine products — including combustibles, Heated Tobacco Products (HTPs), Electronic Nicotine Delivery Systems (ENDS) and oral products — are evaluated by using established guidelines for consumer goods and pharmaceuticals, such as the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) S2(R1) Guideline, 3 the UK Committee on Mutagenicity of Chemicals in Food, Consumer Products and the Environment, 4 the US FDA CFSAN Redbook, 5 and published guidance by Health Canada. 6 All of these guidelines follow a similar strategy for genotoxicity assessment, and include assays to detect gene mutation and chromosomal effects. These assays are addressed in Organisation for Economic Co-operation and Development (OECD) Test Guidelines (TGs) and include: the Ames test for bacterial mutagenicity; 7 the in vitro micronucleus test 8 for the determination of genotoxicity in mammalian cells (which includes aneugenic and clastogenic events); and the mouse lymphoma thymidine kinase (TK) assay, 9 a mammalian gene mutation assay that detects both gene and chromosomal mutations. From a regulatory and scientific standpoint, the mouse lymphoma TK assay and the in vitro micronucleus assay are considered equally appropriate and interchangeable for the measurement of chromosomal damage. In addition to these genotoxicity assays, the assessment of cytotoxicity for tobacco products is also recommended by CORESTA. 1
From 2019, the Institute for In Vitro Sciences (IIVS) has sponsored a series of workshops to identify, discuss and develop the recommendations for the optimal scientific and technical approaches for conducting in vitro assays to evaluate the potential toxicity of traditional cigarettes, HTPs and ENDS. 10 The primary focus of these workshops has been on the genotoxic evaluation of these products by using regulatory genetic toxicology assays, and has included a discussion of various methods to generate and test the types of samples that can be used for these evaluations. 11 The importance of dosimetry and considerations for conducting dosimetry evaluations has also been discussed. 12 Other workshops are in preparation to address the issues, challenges and recommendations identified in the first workshop. 10
As a part of the IIVS workshop series, working groups of experts held discussions, prepared background papers and developed recommendations for discussion during the workshops. The current report was prepared by the working group focusing on the mouse lymphoma TK assay, hereafter referred to as the MLA. 9
The mouse lymphoma TK assay
Overview
The MLA is one of the recommended regulatory assays for evaluating the potential for test materials to induce genetic damage. It detects both gene mutations and chromosomal events and, therefore, has the potential to determine the mutagenic and clastogenic potential of test articles. 9 The MLA has advantages over other mammalian gene mutation assays, such as the hypoxanthine phosphoribosyl transferase (HPRT) assay, as it is able to detect a broader spectrum of genotoxic effects. Therefore the MLA, but not the HPRT assay, is considered equivalent to the cytogenetic assays for detecting chromosomal events.
The OECD Genetic Toxicology Guidelines were revised in 2014–2015. The MLA assay was separated from OECD TG 476 to become a new guideline, OECD TG 490. 9 This was done to incorporate the internationally harmonised recommendations from the MLA expert working group of the International Workshop for Genotoxicity Testing (IWGT), and includes recommendations for assay acceptance criteria and data interpretation.13–17
The MLA requires the use of a TK heterozygous mouse lymphoma cell line (L5178Y tk+/– clone 3.7.2C) developed by Clive et al. 18 The TK enzyme in this cell line is encoded by a heterozygous gene that is part of the salvage pathway for pyrimidine nucleic acid breakdown products. A forward mutation occurring in the TK gene results in a change from tk+/– (heterozygous) to tk–/– (mutated). The loss of TK enzyme activity causes resistance to the toxic analogue trifluorothymidine (TFT), meaning that the mutant cells (tk–/–) are unable to uptake TFT and can therefore survive in its presence. 19 Mutations detected in the assay are those that lead to the loss of TK functionality of the TK+ allele which is located on chromosome 11b. These events can include point mutations, chromosomal deletions, rearrangements, mitotic recombination and some aneuploidy.20,21 Unlike the Ames (bacterial reverse mutation) test, the MLA detects forward mutations.
Detailed methods for conducting the MLA can be found in several publications and a brief overview is provided below.14,22–24 A schematic overview is also shown in Figure 1. The test articles may be formulated in a number of vehicles but, as is the case for most cell culture assays, organic solvents should not exceed 1% (v/v) and aqueous solvents should not exceed 10% (v/v) in the final treatment medium. The use of organic solvents should be supported by data demonstrating their compatibility with the assay and the lack of genotoxic effect at the concentration used. The Mouse Lymphoma Assay (MLA) procedure. TFT = trifluorothymidine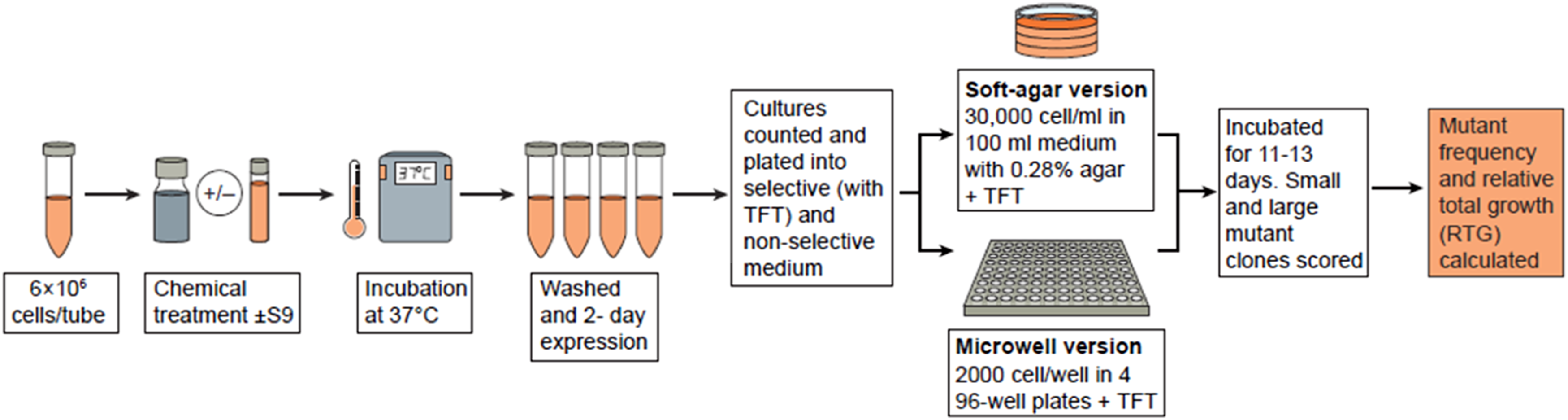
Test concentrations are selected to cover a range of cytotoxic effects, as recommended in OECD TG 490. 9 The cell cultures are typically treated for short-term time points (typically 3 to 4 hours), both in the presence and absence of metabolic activation. These treatment conditions have been shown to lead to the detection of most mutagenic and genotoxic effects — however, it may also be useful to include a 24-hour treatment in the absence of S9, particularly for poorly soluble substances, nucleoside analogues or any test articles capable of causing cell cycle delay. It is noted that, while the 24-hour treatment is recommended in the ICH guidelines for evaluating pharmaceuticals intended for human use, the use of a 24-hour treatment is optional in OECD TG 490. 9
Following treatment, a 2-day expression period allows for newly generated mutants to lose functional TK activity, so that mutants can be enumerated by using TFT selection. There are two versions of the MLA, based on the media used for mutant enumeration (one using soft agar cloning in Petri dishes, and the other using liquid media and microwell plates). The frequency of mutants is determined by comparing the cloning efficiency of cells in culture medium with and without the selective agent. Cytotoxicity is measured by using Relative Total Growth (RTG), which is a parameter developed by Clive et al. 18 that includes a comparison between the treated and control cultures by using cell growth during treatment, expression and cloning efficiency. The TK mutants detected in the assay form colonies of two distinct sizes (small and large), as defined in OECD TG 490. 9 The induction of large colony mutants is associated with the induction of point mutations, while the induction of small colony mutants is associated with the induction of chromosomal mutations. Colony sizing should be conducted for each assay (see the Methods sections in the cited publications for details). This assessment is carried out in order to ensure that the growth conditions were also optimal for small colony mutant formation and enumeration. Colony sizing may also provide some insight as to whether the test material induces point mutations or chromosomal mutations or, as is generally seen, both types of mutations (Figure 1).
Assay acceptance criteria
Prior to evaluating the results from an individual experiment, the data from the assay should be evaluated to assure that the experiment is acceptable. The following criteria must be met: — two experimental conditions (short-term treatments, in the presence and absence of S9) were conducted; — an adequate number of cells and concentrations were analysed; and — the highest concentration treatment resulted in an adequate level of cytotoxicity, as determined by a RTG of nominally 10–20% of the vehicle control value, or otherwise meeting the concentration selection recommendations in OECD TG 490.
9
The vehicle or solvent control mutant frequency should be 50–170 × 10–6 (i.e. between 50–170 mutants per million cells) (for the microwell version) or 35–140 × 10–6 (for the agar-based version), and should be within the historical control ranges of the test laboratory. Additional acceptability criteria are defined in OECD TG 490.
9
The positive controls used in each independent experiment should show an increase in total mutant frequency of at least 300 × 10–6 above the concurrent vehicle control mutant frequency, with at least 40% of the induced mutant frequencies being small colonies and/or an increase in small colony mutant frequency of 150 × 10–6 above the concurrent vehicle control value. This last criterion is to assure adequate mutant colony growth conditions and the optimal recovery of small colony mutants.
Evaluation criteria
Providing that all acceptability criteria are met, the experimental results can be evaluated to determine whether the test material is mutagenic in the MLA.
The IWGT MLA expert group developed an approach for defining positive and negative responses, to ensure that responses defined as positive had increases in mutant frequency that were biologically relevant. This approach relies on the use of a predefined induced mutant frequency (i.e. increase in mutant frequency above that of the concurrent vehicle control), which is designated as the Global Evaluation Factor (GEF). This value is based on the analysis of the distribution of the negative control mutant frequency data from several laboratories.15,16 For the agar-based version of the MLA, the GEF is 90 × 10–6 and for the microwell version of the MLA the GEF is 126 × 10–6. For example, if the concurrent negative control in a microwell assay is 80 × 10–6, then the test chemical must have a mutant frequency of at least 206 × 10–6, in order to meet the criteria for a positive response. The GEF values apply to short-term treatments in the absence and presence of S9, and also to long-term treatments in the absence of S9.14,16 Positive responses are thus defined as those that show concentration-related increases that reach the GEF — and the test material is then considered to be mutagenic in the MLA. A test material is considered to be negative if, under all experimental conditions used, there is no concentration-related response or, if there is an increase in mutant frequency, it does not exceed the GEF. In such cases, the test material is considered unable to induce mutations in this system. This approach is included in the recommendations of OECD TG 490 for the MLA.
Generally, the maximum concentration of the test material used is based on its cytotoxicity (other situations are described in OECD TG 490), and in such cases the highest concentration should aim to achieve between 10 and 20% of the vehicle control RTG. 9 Care should be taken when interpreting positive results (i.e. where the GEF is exceeded) that are only apparent when the test material concentration results in a RTG value of less than 20%; also, a result should not be considered positive if the increase in mutant frequency occurred only at test material concentrations leading to RTG values of 10% or less.9,14
The testing of single compounds
The MLA is frequently used for determining the mutagenic potential of pharmaceuticals, agrochemicals and industrial chemicals, for both regulatory submissions and research purposes. The assay has primarily been used to determine whether or not a test substance is mutagenic, as per the criteria discussed above. Additionally, it has been used to compare the relative mutagenicity of compounds. For example, in Dearfield et al. 25 and Moore et al., 26 nine acrylate/methacrylate compounds were tested for mutagenicity in L5178Y mouse lymphoma cells, exposed to a 4-hour treatment in the absence of metabolic activation (i.e. S9), by using the agar-based method. The results demonstrated that, of the nine acrylates tested, seven were clearly positive, whereas two were equivocal. Of the seven clear positives, there were differences in their mutagenic potencies. That is, some compounds required lower concentrations than others to induce maximal cytotoxic, mutagenic and clastogenic responses. Additionally, the authors also concluded that acrylates were generally more potent than their corresponding methacrylates.
This shows that the MLA has potential to be used not only for binary mutagenic/non-mutagenic determinations, but also to rank the potencies of mutagenic compounds.
The use of the MLA for the evaluation of tobacco products and Next Generation Products
A variety of different products, such as traditional cigarettes, HTPs, ENDS and oral products, have been tested in the MLA. Additionally, different sample types (test matrices) can be generated from some of these products. Figure 2 provides an overview of the different product types and sample matrices that are commonly tested in the MLA. An overview of the different product types and sample matrices that are commonly tested in the MLA. ACM = Aerosol Collected Mass; ENDS = Electronic Nicotine Delivery Systems; GVP = Gas–Vapour Phase; TPM = Total Particulate Matter.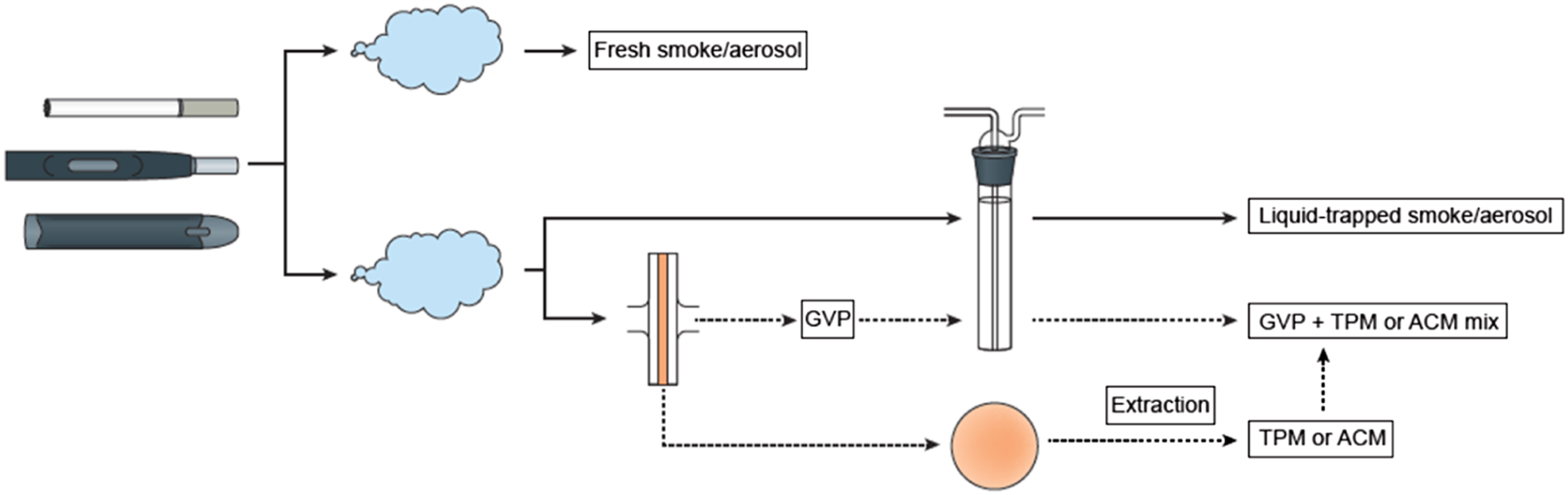
The testing of combustible tobacco products
The MLA has been used for the testing of combustible tobacco products, to assess the mutagenic potential of traditional cigarettes as part of a battery of other in vitro assays. 1 The inclusion of the MLA may help in a weight-of-evidence approach, to address key questions when changes to the product are made. The assay can address the question as to whether the inclusion of additives to a tobacco blend, or cigarette filter, alters the mutagenic profile of the product relative to a control cigarette that is constructed of the same materials but does not contain the additives. Also, because of the toxic nature of combustible tobacco products, efforts to reduce the toxicity of cigarettes have been explored. These efforts have included putting particular additives into the tobacco blend to reduce the tobacco content, and making modifications to the filter to enhance the filtration of volatile compounds. Assays have also been used to identify the relative mutagenic profiles of cigarettes containing different tobacco blend ratios or single blends, in order to rank their relative mutagenic potential. The MLA has been used, for example, to add to or complement the weight-of-evidence data gained from analytical chemistry, the Ames test and the in vitro micronucleus test. Studied changes to a combustible cigarette have included the addition of flavourings to the prototype cigarette, altered blends, or toxicant-reducing technologies.
A variety of matrices of combustible cigarettes have been tested in the MLA (Figure 2). Most studies to date have investigated the effects of Total Particulate Matter (TPM), where the cigarettes are smoked to a standardised puffing regime, and the particulate phase captured on a Cambridge filter pad (CFP). This is then extracted with dimethyl sulphoxide (DMSO) to a known stock solution, which is then subsequently diluted and applied to the cell cultures. 11 Other matrices, although less common, include the application of the Gas–Vapour Phase (GVP), where the aerosol that passes through the CFP is bubbled into a container containing a solvent to capture the soluble vapour phase, which can subsequently be applied to the cell cultures. Furthermore, the TPM and GVP can be combined into one test article from the same product and applied to the cell cultures. Additionally, the MLA has also been attempted with whole aerosol exposures. Therefore, the MLA is amenable to a variety of test article matrices.
TPM is the most commonly used test article from combustible tobacco products. However, more recently, the GVP has also been used. Methods to adapt the MLA to accommodate whole aerosol exposures have been investigated. The methods typically used to collect TPM, GVP and/or TPM + GVP extracts fractionate the mainstream smoke from its native form. Whole aerosol methods may offer a more representative form of exposing cells — however, these cells would need to be exposed at the air–liquid interface (ALI) to allow exposure to the cigarette whole aerosol, whereas the L5178Y cells are grown in suspension. This can be overcome by reverting to exposures by means of a soft agar-based method.
The general findings of published studies that used the MLA for the testing of combustible tobacco products.

aC3B was defined (from the dose–response curve) as the equal mutagenic effect concentration for three-times the background mutant frequency. 35
BMD = Benchmark Dose; GEF = Global Evaluation Factor; GVP = Gas–Vapour Phase; HCI = Health Canada Intense; ISO = International Organization for Standardization; MDPH = Massachusetts Department of Public Health; MF = Mutant Frequency; MLA = Mouse Lymphoma Assay; RTG = Relative Total Growth; TPM = Total Particulate Matter; WSS = Whole Smoke Solutions.
There are some clear messages from studies employing the MLA to make comparisons between combustible tobacco products, in order to investigate the potency of different blends, harmful/potentially harmful constituent (HPHC) reducing technologies, or changes to the format of the product (including the addition of particular ingredients).
All studies listed in Table 1 utilised the L5178Y TK+/– 3.7.2C cell line, and the majority used the microwell format of the assay. Some of these studies compared the soft agar-based method to the microwell version, and the overall conclusions were that the two assay formats are interchangeable.
All of the listed studies reported a concentration-related increase in cytotoxicity, as indicated by a reduction in the RTG. Where authors explicitly commented on the RTG values achieved, the combustible tobacco products (where TPM was used as the test article) often achieved an RTG of between 10 and 20%. Additionally, the TPM from the combustible products induced dose-related increases in mutant frequency in each study reviewed. These increases above the background mutant frequency also exceeded the GEF, indicating that TPM is positive in the MLA. However, there were some instances where the authors reported that either only the top one or two concentrations exceeded the GEF, and that these concentrations were in the RTG range of 10–20%. Some authors also reported that the surpassing of the GEF was not always reproducible between experiments.
There were also some differences between the number of replicate cultures used in the MLA. Some authors used single replicates in multiple experiments, while most authors used duplicate cultures in a single experiment. Other authors used four replicate cultures, based on statistical power calculations, in order to increase the power of the assay to detect differences between combustible TPMs. 28 According to OECD TG 490 recommendations, the use of duplicate cultures is advisable in a standard assay — however, single cultures are also permissible.
Differences in the number of concentrations tested in the assay were also apparent. Most authors had followed the OECD TG recommendations and used only the minimum number of four concentrations, ranging from high to low RTG values. However, this may make interpretation of a study difficult, if the highest concentrations need to be removed due to excessive cytotoxicity, and/or if there is steep concentration-related toxicity between closely-spaced concentrations. Additionally, as the concentration-related response needs to be well characterised, it may be of value to increase the number of concentrations tested.
All of the listed studies were based on short-term treatments, with common exposure times being 3 or 4 hours, in the presence or absence of S9. Some of the studies also included experiments to look at the effects of a long-term exposure period of 24 hours, in the absence of S9.
Some best practices are recommended when conducting the MLA with combustible tobacco products. Concentration range-finding experiments should be performed, in order to identify the optimum concentrations to use in the main experiments, as most, if not all, combustible products are cytotoxic and mutagenic in this assay. It is generally advisable to start performing the assay with more test concentrations than the minimum four recommended under the OECD TG 490 guidance. This allows for the subsequent elimination of concentrations from the scoring process, due to either no cytotoxic effects or, conversely, excessive toxicity being observed. It also helps to select cultures for cloning that exhibit responses covering the desired cytotoxicity range, in order to best characterise the concentration-related response. This selective step usually results in at least four usable concentrations remaining, for mutant enumeration. The use of single cultures is practiced by some laboratories, in order to set up a large number of concentrations which will assist with data interpretation (positive or negative). When the goal is to make comparisons between combustible products, it is recommended that at least duplicate cultures are used and statistical power calculations are performed to determine the appropriate level of resolution to differentiate between products.
Heated Tobacco Products
A summary of the general findings of published studies that used the MLA for the testing of Heated Tobacco Products (HTPs).

aC3B was defined (from the dose–response curve) as the equal mutagenic effect concentration for three-times the background mutant frequency. 35
CFP = Cambridge Filter Pad; GEF = Global Evaluation Factor; GVP = Gas–Vapour Phase; HCI = Health Canada Intense; ISO = International Organization for Standardization; HTP = Heated Tobacco Product; MDPH = Massachusetts Department of Public Health; MLA = Mouse Lymphoma Assay; PBS = Phosphate-Buffered Saline; RTG = Relative Total Growth; THP = Tobacco Heating Product; TPM = Total Particulate Matter.
In the early years of testing HTPs, the TPM or GVP collected from the HTPs was treated in the same way as that derived from a traditional cigarette. The TPM or GVP from the comparator cigarette, and from the HTP, were both extracted at the same stock concentration and the test concentrations used in the assays were also the same. With this approach, some key findings were made:
Firstly, the studied HTPs were discovered to be less cytotoxic than comparator traditional cigarettes, typically only reaching the 60% RTG mark, which does not meet the required RTG specified by OECD TG 490. 9 Secondly, from the mutant frequency evident after exposure to TPM or GVP from a HTP, when tested at the same extract concentrations as those from a cigarette, the HTPs were not considered to be mutagenic (see Table 2). This is in contrast to the responses seen with the comparator cigarettes, where typically the TPM reduces the RTG to 10–20% and the GEF is exceeded (see Tables 1 and 2).
Over recent years, HTP testing has continued and, as a result, the testing approach has greatly improved. This is mainly based on the recognition that HTPs score as being considerably less toxic than comparator cigarettes. These improvements have included increasing the stock concentration of the HTP TPM extract, allowing the final concentration in the assay to be at least double that from a cigarette. This approach led to a dose-related increase in the measured cytotoxicity resulting from the HTP TPM exposure (see Table 2). Indeed, these increased HTP TPM extract concentrations also reduced the RTG to approximately 40% (see Table 2).
For HTPs, most of the listed studies reported that exposure to the TPM or GVP did not result in the GEF being reached. From these studies, it was also evident that dose–response relationships for mutant frequency and cytotoxicity were lacking. However, some of the other studies reported dose-related increases at significantly higher concentrations than those from a cigarette comparator, although the actual doses tested were not specified (see Table 2). This suggests that, based on a dose-to-dose comparison, the induced mutant frequency from the studied HTPs is considerably less than that of comparator cigarettes.
The common theme of testing HTPs in the MLA is that high concentrations, often in excess of 500 μg TPM/ml (or μg TPM equivalent, when testing GVP) as the final concentration, are required to induce a mutagenic response. This is much higher than the concentrations required to induce responses from a comparator cigarette. Indeed, this is not unexpected, as HTP aerosol composition is considerably different to that of a traditional cigarette. Fewer toxicants are present in the emissions of a HTP, and those that are present occur at lower concentrations, primarily due to the reduced operating temperature and absence of combustion, which also accounts for the most significant differences between the two product types.
Best practice for HTP testing
There are certain recommendations to be made when testing HTPs in the MLA. Dose range-finding experiments should be performed, in order to identify the optimum concentrations to use in the main experiments. However, it must be acknowledged that the concentrations required are likely to be significantly higher than those used for analysing traditional cigarettes, as discussed previously. The highest concentration used should ideally result in a 10–20% RTG, but it should be noted that this may not be possible, as the maximum feasible concentration may be below the concentration required to reach the recommended RTG, which may also be dependent on the treatment condition used. The number of concentrations used in the assay should be increased from the minimum four, as recommended in the OECD TG 490 guidance, in the event that concentrations need to be eliminated where there are cases of excessive toxicity and to characterise the dose–response relationship. The choice between the use of single and duplicate cultures is similar to that for combustibles (see above). Depending upon the goals of the study, it may not be appropriate to use single cultures — generally, a minimum of two replicates should be used when the goal is to make comparisons, either with other HTPs or with traditional cigarettes. Additionally, HTPs contain higher level of humectants (such as propylene glycol and/or glycerine) which may have an adverse effect on the assays. Therefore, it is also recommended that the osmolality of the culture is monitored, to ensure that it keeps within the specified range until the end of the treatment period.
Given that studied HTPs are less cytotoxic relative to traditional cigarette comparators, there will be instances where the RTG limit cannot be achieved. For this scenario, the recommendation could be to test the highest feasible concentration for the test matrix type, or to alter the vehicle, where appropriate, from an organic solvent (e.g. DMSO) to an aqueous solvent (e.g. phosphate-buffered saline; PBS) that can usually be added at larger volumes to the cell culture system, and thereby potentially increase the concentration of a test substance in the assay.
Most of the data regarding HTPs reviewed for this publication demonstrate that, on an equivalent dose level to combustible cigarettes, the studied HTPs are significantly less cytotoxic and therefore require much higher concentrations of emissions to produce potential toxic and/or mutagenic responses. Indeed, even at these higher concentrations, mutagenicity is still absent or less than comparator combustible cigarette responses. There may be some instances, under some treatment conditions, where the required RTG may not be achieved and the level of mutagenicity fails to reach the induced GEF. In these instances, the maximum achievable concentration should be tested in the assay.
Electronic Nicotine Delivery Systems
A summary of the general findings of published studies that used the MLA to assess the mutagenic potential of Electronic Nicotine Delivery Systems (ENDS).

ACM = Aerosol Collected Mass; ENDS = Electronic Nicotine Delivery Systems; GEF = Global Evaluation Factor; HCI = Health Canada Intense; MF = Mutant Frequency; MLA = Mouse Lymphoma Assay; RTG = Relative Total Growth; TPM = Total Particulate Matter.
Despite there being so few published studies on the assessment of ENDS in the MLA, there are some clear findings from the studies that are available. Firstly, the compositions of the ENDS aerosols studied are reported as being much simpler and containing reduced toxicant levels, as compared to both traditional cigarettes and HTPs — which is consistent with these ENDS products being much less toxic than comparator cigarettes and HTPs. This is reflected in the RTG values achieved, and the considerably higher ENDS aerosol concentrations necessary for the MLA to show any cytotoxic effect. However, this will be dependent on the composition of the aerosol. The need for higher aerosol concentrations may also introduce other challenges. For instance, ENDS devices typically use base formulations containing propylene glycol and glycerine, which are known to have osmolality effects within the assay system and may cause artefactually positive results. 48 Therefore, caution should be taken when testing ENDS in the MLA, with osmolality and pH measurements ideally being monitored throughout the treatment period.
Oral products
Oral products represent a distinct product category as compared to cigarettes, HTPs and ENDS, as they are placed in the mouth rather than inhaled. Oral products have traditionally been composed of tobacco contained in a pouch or loose, and placed in the user’s mouth to absorb nicotine and flavour. More recently, oral products have been developed that contain no tobacco, but use other materials as a substrate to contain nicotine.
A summary of the general findings of published studies that used the MLA as part of a mutagenic assessment of oral tobacco products.

GEF = Global Evaluation Factor; MF = Mutant Frequency; MLA = Mouse Lymphoma Assay; RTG = Relative Total Growth.
Discussion
The MLA is a well established in vitro assay, for the assessment of mammalian mutagenicity and genotoxicity in L5178Y cells. Since its establishment in the 1970s, it has undergone further development, refinements and improvements, facilitated by the International Working Group on Genotoxicity Testing and leading to its standalone OECD TG.
Classically, the MLA is used to assess the mutagenic and/or genotoxic properties of pharmaceuticals, agrochemicals and industrial chemicals, and is commonly interpreted as a binary ‘yes/no’ prediction on the mutagenic potential of the studied substances. However, the MLA has also been used to rank the mutagenic responses of industrial chemicals. For example, Dearfield et al. 25 used the MLA to assess the mutagenic potential of nine acrylate/methacrylate compounds.
For the current report, the assembled working group conducted a literature review, in order to identify key studies that have used the MLA as part of the assessment of tobacco and nicotine products. The literature search revealed that the assay has been used in the testing of several product categories, including combustible cigarettes, HTPs, ENDS and oral products. The MLA has typically been used with combustible products to study the effects of particular changes that aim to reduce toxicant emissions (for example, altering the blend of the tobacco rod or the filter design), and to investigate whether any such reductions translate into an in vitro biological effect. The assay has also been used to evaluate the mutagenicity of different blends of tobacco. Furthermore, the MLA has been used to rank the relative mutagenicity of different cigarette types. The TPM from traditional cigarettes is by far the most common tobacco product test article used in the MLA. The assay has also been used to test GVP and whole smoke, obtained by bubbling the smoke through a solvent to trap particulates and vapour phase elements and applying this extract to the cultures. Adaptations to the MLA have also attempted to expose MLA cells directly to whole aerosol at the air–agar interface. Although this last method was successful and dose-related increases in mutant frequencies were observed following the exposure of the L5178Y cells to whole smoke, the method did not meet the requirements of the OECD TG and thus may not be amenable to routine testing use due to technical and logistical challenges.
More recently, the MLA has been used in the testing of NGPs that include a variety of HTP variants, ENDS and oral products. In these studies, the respective NGP has been compared to a traditional combustible cigarette, in order to investigate the mutagenic profile of the NGP relative to a comparator traditional cigarette. The test matrix used with the NGPs is typically TPM or GVP compared to the same fraction from a cigarette. ENDS MLA tests usually use ACM compared to TPM from a comparator cigarette, and oral products are extracted in a solvent such as DMSO, PBS or a more biologically relevant matrix (such as artificial saliva). One of the key findings of the testing of NGPs in the MLA — although this is not unique to the MLA, and is based only on the results of the studies reviewed here — is that the tested NGPs are considerably less toxic and less mutagenic than the cigarette comparators. Therefore, in order to achieve the required level of cytotoxicity, as determined by a reduction in the RTG with the aim of achieving a RTG between 10–20%, considerably higher concentrations of the test article need to be applied. Often, this is more than double the concentration relative to the comparator cigarette, which also introduces challenges. Extracts of TPM and GVP from NGPs can be prepared to much higher concentrations than those from a cigarette, and these NGP extracts are often prepared to the highest feasible concentration. However, there may be instances (depending upon the treatment condition used) where, despite preparing extracts to the highest feasible concentrations, the RTG of 10–20% may still not be reached. Also, the higher levels of humectants used in NGPs may interfere with the osmolality of the in vitro system, and cause artefactual results.
Recommendations
Based on this literature review and the collective experience of the working group members, there are several best practices that can be recommended for the testing of cigarettes and NGPs in the MLA.
For the testing of cigarettes, the recommendations by the working group are: 1. A concentration range-finding experiment should be performed, in order to identify the optimum range of concentrations to use in the main experiments, as most, if not all, combustible products are cytotoxic and mutagenic in this assay. 2. The number of concentrations used in the assay should be increased from the minimum four recommended under the OECD TG 490 guidance, in the event that some concentrations need to be eliminated due to observed cases of excessive toxicity, and to facilitate characterisation of the concentration-related response. 3. The use of single cultures has limitations when it comes to making comparisons, but it is an option when the goal is to identify whether a test article is positive or not in the assay. For comparisons between combustible products, it is recommended that at least duplicate cultures are used and statistical power calculations performed to determine the appropriate level of resolution to differentiate between products. 4. The evaluation criteria to determine a positive or negative response, as discussed previously and documented in OECD TG 490, should be followed. 1. A dose range-finding experiment should be performed, in order to identify the optimal range of concentrations to use in the main experiments. However, the concentrations required are likely to be significantly higher than that relative to a cigarette. 2. The highest concentration tested should ideally achieve the 10–20% RTG, but this may not be possible, as the maximum feasible concentration may be below the concentration required to reach the recommended RTG. 3. The number of concentrations used in the assay should be increased from the minimum four recommended under the OECD guidance, in the event that concentrations need to be eliminated due to observed cases of excessive toxicity and to characterise the dose-response. 4. Unless the goal of the evaluation is to determine whether the NGP is mutagenic or non-mutagenic, where it can be appropriate to use single cultures, a minimum of two replicates should be used. 5. NGPs may contain high levels of humectants, such as propylene glycol and/or glycerine, which may have an adverse effect on the assay. Therefore, it is also recommended that the osmolality of the culture is monitored and kept within the specified range throughout the treatment period. 6. The evaluation criteria to determine a positive or negative response, as discussed previously and documented in OECD TG 490, should be followed.
For the testing of NGPs, the recommendations by the working group are:
Conclusions
All of the publications reviewed herein demonstrate that, on an equivalent dose to comparator combustible cigarettes, the studied NGPs are considerably less cytotoxic and less mutagenic. Indeed, even at higher NGP concentrations, mutagenicity is still absent or less than the combustible cigarette responses. This literature review also highlighted that the MLA is amenable to the use of several different test matrices, including TPM, GVP, ACM, aqueous and organic solvents, and even aerosols. If used as part of a weight-of-evidence approach for the assessment of tobacco products and NGPs, the MLA complements other tests comprising an in vitro battery — including the Ames test, the in vitro micronucleus test, and a suitable test for cytotoxicity (e.g. the Neutral Red Uptake assay), as well as potentially biologically relevant analytical chemistry findings.
Footnotes
Disclaimer
This article has been reviewed by the organisations of the authors and approved for publication. The views expressed in the manuscript do not necessarily reflect the policy of these organisations. Any mention of commercial products is for clarification only and is not intended as approval, endorsement or recommendation. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Food and Drug Administration.
Author contributions
Dr Moore, a co-chair of the workshop series, facilitated the workgroup meetings and discussions leading to the recommendation which are presented in this manuscript. Dr Curren, a co-chair of the workshop series, participated in this workgroup discussions and helped edit the manuscript. Dr Crooks was responsible for coordinating the compilation of the manuscript and conducting all the final editing. The other authors all contributed to the discussions, activities of the workgroup and the content of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This publication was supported by the Food and Drug Administration (FDA) of the US Department of Health and Human Services (HHS) as part of a financial assistance award (FAIN) (1R13D007386), and by a non-government source, the Education and Outreach Program of the Institute for In Vitro Sciences (IIVS), Inc. IIVS is a 501(c)(3) non-profit that receives funding from NGOs, industry, trade associations and individual donors.
Ethical statement
Informed consent
Informed consent was not required for the preparation of this article.
Appendix
The abbreviations used in the main text.
ACM
Aerosol collected mass
ALI
Air–liquid interface
B[a]P
Benzo[a]pyrene
CFP
Cambridge filter pad
CORESTA
Cooperation Centre for Scientific Research Relative to Tobacco
DMSO
Dimethyl sulphoxide
ENDS
Electronic Nicotine Delivery Systems
FDA
US Food and Drug Administration
GEF
Global Evaluation Factor
GVP
Gas–Vapour Phase
HPHC
Harmful/potentially harmful constituent
HPRT
Hypoxanthine phosphoribosyl transferase
HTP
Heated Tobacco Product
ICH
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use
IIVS
Institute for In Vitro Sciences
ISO
International Organization for Standardization
IWGT
International Workshop for Genotoxicity Testing
MLA
Mouse Lymphoma Assay
MMS
Methyl methanesulphonate
NGP
Next-generation Product
OECD
Organisation for Economic Co-operation and Development
PBS
Phosphate-buffered Saline
RTG
Relative Total Growth
TFT
Trifluorothymidine
TG
Test Guideline
THP
Tobacco Heating Product
THS
Tobacco Heating System
TK
Thymidine kinase
TPM
Total Particulate Matter
