Abstract
The failure rate for the translation of drugs from animal testing to human treatments remains at over 92%, where it has been for the past few decades. The majority of these failures are due to unexpected toxicity — that is, safety issues revealed in human trials that were not apparent in animal tests — or lack of efficacy. However, the use of more innovative tools, such as organs-on-chips, in the preclinical pipeline for drug testing, has revealed that these tools are more able to predict unexpected safety events prior to clinical trials and so can be used for this, as well as for efficacy testing. Here, we review several disease areas, and consider how the use of animal models has failed to offer effective new treatments. We also make some suggestions as to how the more human-relevant new approach methodologies might be applied to address this.
Keywords
Introduction
It is claimed that research cannot function without reliance on animal models as proxies for healthy humans (in safety and toxicity testing) and for human diseases (in efficacy testing). This may be true to some extent, but only in that, historically, the ‘options’ were limited. Prior to the discovery and applications of human-relevant new approach methodologies (NAMs), such as induced pluripotent stem cells (iPSCs), computational modelling and dynamic 3-D culture systems, biomedical research was almost always forced to rely on animals as models. However, times have changed, research has evolved, and the use of more human-relevant tools is proving to be more predictive of human responses and human diseases than animals ever were, or ever could be.
Here we take several examples of human diseases, namely Parkinson’s disease (PD), Alzheimer’s disease (AD), respiratory tract diseases, rheumatoid arthritis (RA), and human immunodeficiency virus (HIV) infection/acquired immune deficiency syndrome (AIDS). These diseases were selected for the following reasons: — their high prevalence; — their severe burden on the patient and on healthcare systems; — the high numbers of animals used for their associated research, drug development and testing, as shown by the most recent EU statistics on the number of animals used for scientific purposes;
1
and — the relatively high translational failure rate in the drug development process.
In combination, basic, translational and applied research studies for respiratory tract, nervous system, immune system and musculoskeletal conditions account for almost one quarter of the total animal use across the EU. 2 This represents the use of over two million animals annually, with no indication of a decrease in animal use since 2015 (when online records began). Despite this sustained reliance on animals, for all the diseases described here — and indeed, for many other diseases — there is a paucity of effective treatments, either preventative or curative. This narrative review explores where the existing animal models of these diseases have shown promise in identifying apparent new treatments that ultimately fail to translate to the human patient. This range of diseases also provides examples across a range of organ systems — namely, neurological, respiratory tract, musculoskeletal and immune systems. By using these disparate examples, we illustrate how results from animal models have too often failed to translate into effective treatments, evidencing that this is not a failing for a specific tissue type or disease pathology, but is, in fact, a more widespread phenomenon related in part to insurmountable species differences.
Of course, the problems of translatability are not limited to the areas explored in this paper — there are issues with other disease areas as well. 3 We realise that oncology is one of the fields in which translatability problems are greater and the failure of traditional models is recognised.4–10 However, we were not able to include ‘cancer’ among the diseases explored because, with over 200 diseases classified as ‘cancer’, 11 this would be a huge undertaking that would, in itself, require a dedicated paper on the topic.
We show how a misguided reliance on animals has hindered our understanding of disease progression and treatment. For HIV, we show how animal models have failed to lead to the development of an effective vaccine. We also consider how the emerging NAMs offer a more predictive, human-relevant approach to understanding human diseases. For a more comprehensive review of available and emerging NAMs, we refer readers to the repository of the European Commission’s Joint Research Centre (https://publications.jrc.ec.europa.eu/repository/search?query=advanced+non-animal) and the Non-Animal Technologies (NAT) Database (https://nat-database.org).
Alzheimer’s disease (AD)
Every year, there are nearly 10 million new cases of dementia worldwide, with AD being the most common form, contributing to 60–70% of total dementia cases. 12 Dementia represents the seventh leading cause of death among all diseases. 12 Moreover, the failure rate in AD drug development is very high, with 99% of trials showing no drug–placebo difference. 13 There are huge global public health costs associated with this, given that more than 55 million people in the world suffer from dementia. Thousands of drugs have been tested for their potential as AD treatments by using traditional animal and non-animal approaches. However, since 1995, only six drugs have been approved by the FDA for AD treatment. 14 These drugs can help improve cognitive and behavioural symptoms, although they do not work for all patients.
The cholinesterase inhibitors donepezil, rivastigmine and galantamine, which increase acetylcholine availability in brain synapses, have shown efficacy in patients with mild to moderate AD. However, it is not possible to identify those patients who will be responsive prior to treatment. 15 A systematic review and meta-analysis assessed the effects of donepezil reported in 30 studies, with a total of 8257 participants. This reported that people with mild, moderate, or severe AD treated for 12 or 24 weeks with donepezil experienced small benefits in cognitive function and daily living activities. However, it should be noted that the evidence was declared to be of moderate quality and was downgraded due to study limitations such as lack of anonymising. 15 A 2022 meta-analysis of five randomised controlled trials, involving 2974 people, showed that, while high dose donepezil improved cognitive function, it also increased the risk of side effects such as heart problems. 16
Memantine is an N-methyl-
Aduhelm was approved by the FDA in 2021. It is an antibody that targets amyloid beta and was shown to clear plaques in mouse models. However, this approval is not without controversy, firstly due to the price, but also due to the lack of preclinical impact on cognitive improvement and possible deaths associated with amyloid-related imaging abnormalities (ARIA).18,19 In the past 15 years, aduhelm stands out as the only new approved drug, and there are still no proven, approved disease-modifying treatments for AD. 20
According to the most recent drug pipeline analysis by Cummings et al., 126 agents were tested in 152 clinical trials in 2021, with 28 treatments in Phase III trials, 74 in Phase II trials and 24 in Phase I trials. Compared to previous analyses, the 2021 pipeline review showed an increase of mechanistic approaches for the treatment of AD, with drugs targeting a variety of different targets, particularly in Phase II trials. 21 Since the 2020 pipeline review, nine trials in Phase I, 18 trials in Phase II and seven trials in Phase III had been either completed, terminated, suspended, or their status was unknown. Recent drug failures include: LTMX (TauRx), 22 Azeliragon, 23 Crenezumab, 24 Aducanumab (aduhelm), 25 Verubecestat, 26 Lanabecestat (LY3314814), 27 Atabecestat, 28 Pioglitazone, 29 the FYN inhibitor AZD0530, 30 and ITI-007 (lumateperone, originally developed for the treatment of schizophrenia).31,32 So far, 155 interventional clinical trials aimed at testing drugs for AD have been terminated (as of 5 July 2022, according to publicly available data in clinicaltrials.gov). The reasons behind the early termination of a study are multiple, and may encompass low study accrual, slow participant recruitment, assessment of futility analysis, or other administrative or strategic reasons. However, lack of efficacy or safety issues represent the most important concerns. Appraisal of drug development is further hampered by unsuccessful preclinical and clinical studies not being reported in the literature and results not released by sponsors. 33 Clearly, the latter is an extremely important aspect to consider, as regulatory authorities grant validation to NAMs such as in vitro models, upon demonstration of their ability to predict in vivo (animal) preclinical data. 34
The general failure in AD drug development includes drugs targeting amyloid deposition, but also drugs working through different mechanisms of action.35–39 In their 2019 perspective article, Mullane and Williams
14
highlighted some of the possible major reasons to explain such a formidable failure in drug development for AD. These include: — Failure to consider the multifactorial aetiology of the disease, i.e. drug development in AD has been heavily focused on the two classical hallmarks: amyloid plaques and neurofibrillary tangles deposition in the brain. Nevertheless, mounting evidence suggests that AD is a multifactorial syndrome, with inflammatory, immune and metabolic components. — The general assumption that familial/gene‐based early‐onset AD (FAD, accounting for about 1–3% of total AD cases) and late‐onset sporadic AD (LOAD, accounting for the remaining 97–99% of cases) are basically the same. It has been shown that, although FAD and LOAD are characterised by the same behavioural and pathological features,
40
animal models of FAD do neither recapitulate the genetics
41
nor the onset and progression of LOAD.42,43 — There is a propensity to neglect relevant aspects during preclinical drug assessment, e.g. efficacy, target selectivity, pharmacodynamic and pharmacokinetic properties of new compounds.44–47 As a consequence, several candidate compounds have progressed to later phases of clinical trials, even without compelling evidence of efficacy.48,49 — The limited construction or predictive validity of animal models of AD.
50
With regard to this last point, inbred/naturally occurring animal models may show accelerated senescence with elevated levels of hippocampal amyloids and behavioural impairments, such as the SAMP8 and SAMP10 mouse models, 51 while transgenic animals have been genetically modified to express some genetic variants identified in FAD. Transgenic mice may show the formation of amyloid plaques and NFTs, gliosis, some synaptic alterations, and some signs of cognitive delay. However, despite the generation of amyloid deposits, these mice often do not exhibit substantial loss of neurons. 52 Moreover, the reliance on models not relevant for humans may ultimately generate false negative results, which are more problematic in drug screening than false positives; 53 false negative results can lead to the exclusion from clinical trials of potentially effective therapeutic compounds. 54 Finally, none of these animal models reflect the pathogenesis of the disease as it occurs in humans,55,56 as the animals do not develop the comorbidities typically associated with LOAD, 14 such as cardiovascular, 57 inflammatory, 58 immune system 59 and sleep disorders, 60 and metabolic syndrome-associated risk factors. 61 Considering the key role that lifestyle plays in the aetiopathogenesis of AD, the use of non-human animal models (such as mice) to study the contribution of lifestyle factors and associated comorbidities becomes even more questionable, in light of the interspecies differences in metabolism, 62 gut microbiome, 63 immune system 64 and epigenetics, 65 which will always have an impact on external validity. 66
New approach methodologies for human-relevant Alzheimer’s disease research
Human-based studies, models, and readouts suitable for Alzheimer’s disease (AD) research and applicable for drug development.
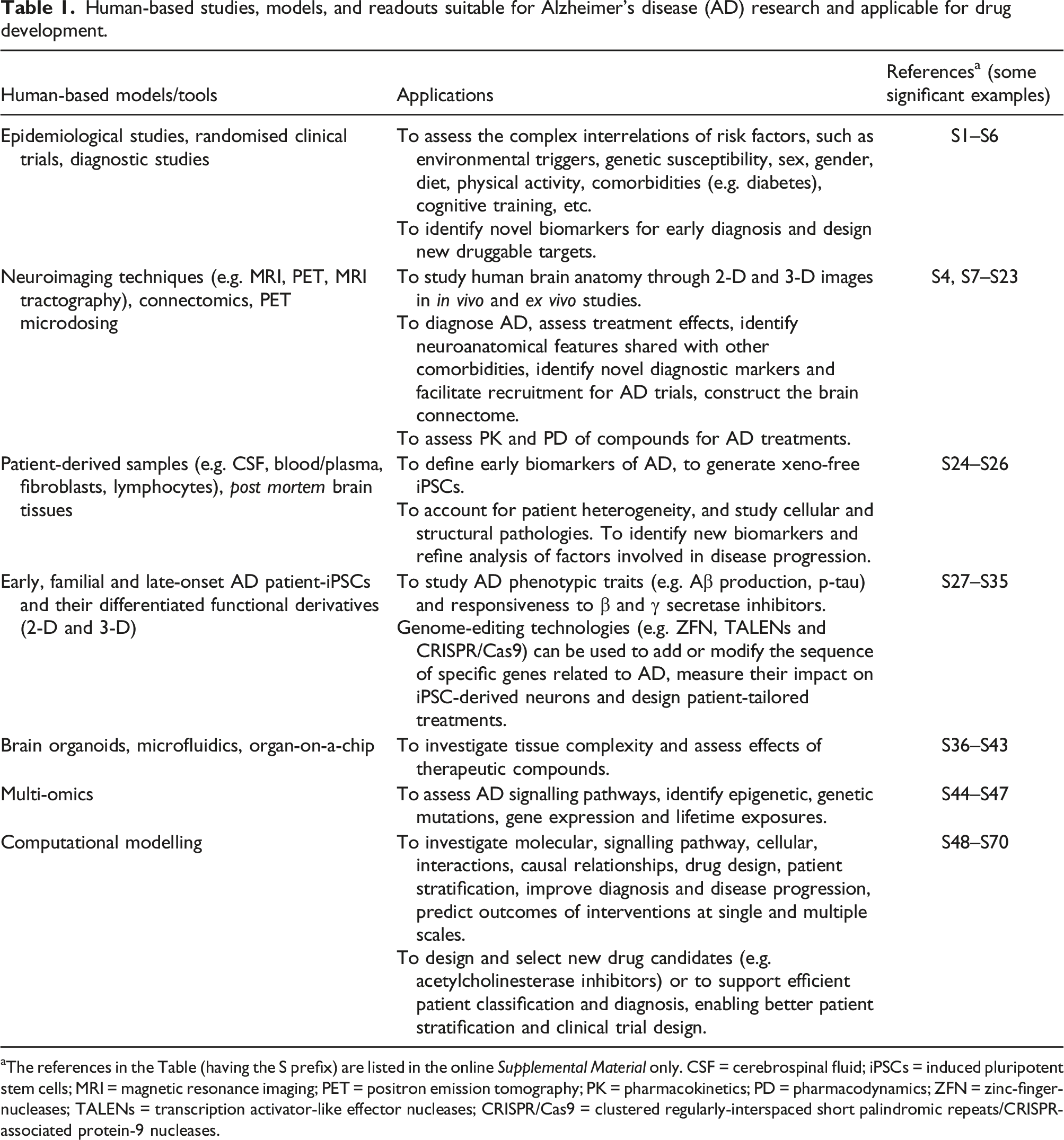
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. CSF = cerebrospinal fluid; iPSCs = induced pluripotent stem cells; MRI = magnetic resonance imaging; PET = positron emission tomography; PK = pharmacokinetics; PD = pharmacodynamics; ZFN = zinc-finger-nucleases; TALENs = transcription activator-like effector nucleases; CRISPR/Cas9 = clustered regularly-interspaced short palindromic repeats/CRISPR-associated protein-9 nucleases.
Ultimately, human-based observational and intervention studies are fundamental, in order to gather insights into environmental and lifestyle risk factors, discover novel biomarkers for early diagnosis, and to identify and eventually promote preventive intervention strategies that have proven to be effective in reducing the symptoms and progression of AD.72–76
Parkinson’s disease
Model generation, means of induction, disease features and shortcomings of models that are currently available for Parkinson’s disease (PD) research.
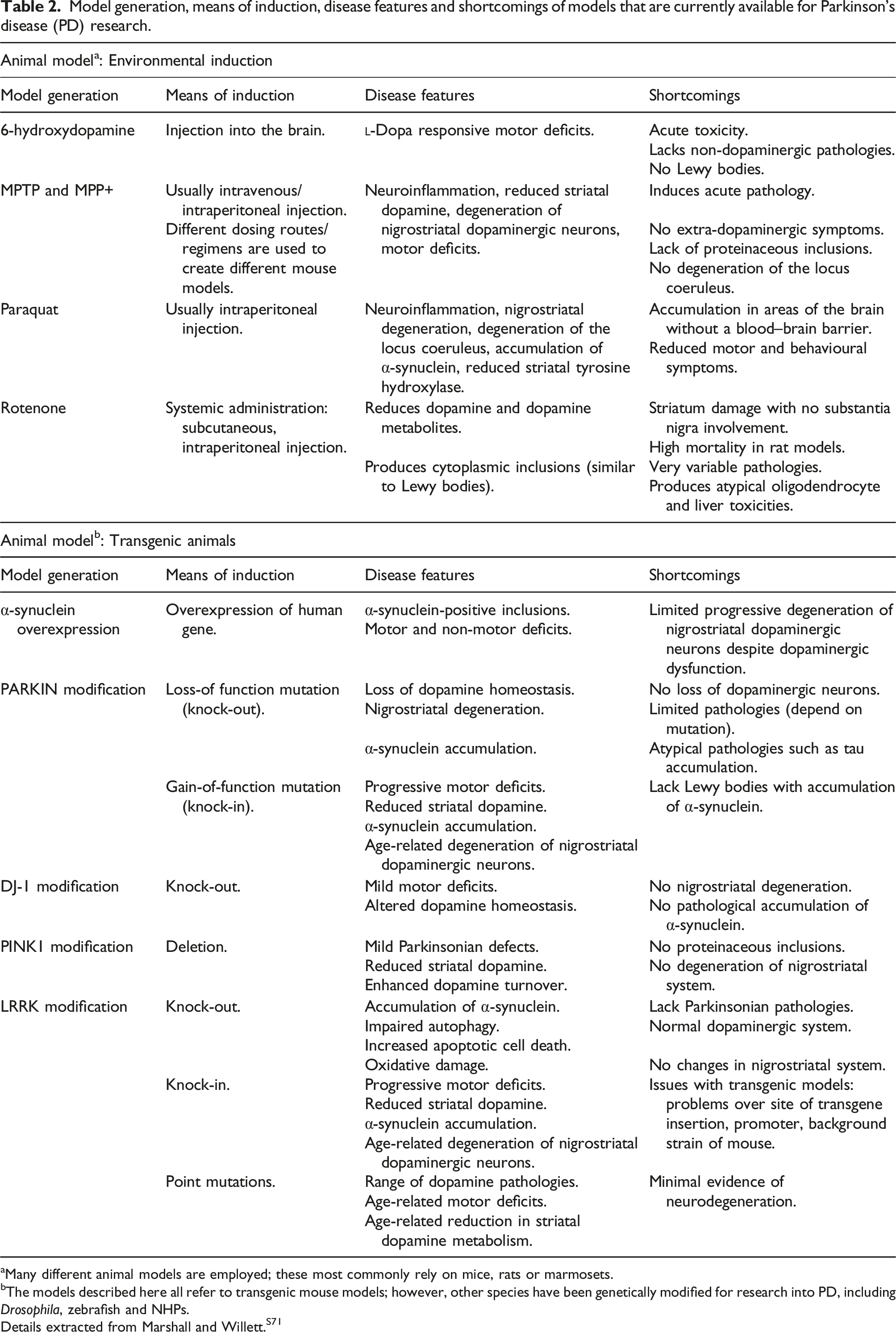
aMany different animal models are employed; these most commonly rely on mice, rats or marmosets.
bThe models described here all refer to transgenic mouse models; however, other species have been genetically modified for research into PD, including Drosophila, zebrafish and NHPs.
Details extracted from Marshall and Willett.S71
Beyond the impact on quality of life for patients and carers, there is a great financial need to address the requirement for better treatments for PD. Recent estimates put the total economic burden of PD at almost US$ 52 billion, 81 which includes the cost of medication, care requirements and also the loss of income for affected patients, given that PD accounts for around 3.2 million disability-adjusted life years (DALY; a measure of overall disease burden that takes into account the number of years lost due to ill-health, disability or early death). 79 Conservative estimates indicate that there will be at least 1.2 million people living with PD by 2030. 82 This is likely to be an underestimate due to the increase in the ageing population (ageing is the biggest risk factor for PD), 83 and taking into account better treatment management that is expected to extend the longevity of PD patients. In addition, the increasing prevalence and incidence of gait, behavioural and cognitive disorders — for example, dementia is around 4–6 times more common in people with PD 84 — could lead to an increasing need for institutional care, further straining limited public health budgets.
Treatment for PD remains symptomatic, and there are currently no neuroprotective or disease-modifying therapies available.
85
This is despite the preclinical success of neuroregenerative approaches in animal models,
86
suggesting that instigating disease through acute toxin administration in rodents, which fails to recreate the chronic, progressive neuronal cell death typifying the human condition, leads to an overestimation of the translational ability of the animal models. Analysis of the published literature reveals the continuing reliance on rodent models of this disease (Figure 1). Over one-third of the primary PD research available through PubMed is associated with mouse models, with a further third exploiting rat models of the disease (Figure 1a). It appears encouraging that around 20% of the papers retrieved focused on in vitro models, but a breakdown of the data to assess model use over time reveals that the use of in vitro models has remained basically unchanged over the past 30 years, while the use of mice is on the increase (Figure 1b). It is particularly interesting to note a recent study that transplanted human iPSC-derived neurons bearing a mutation associated with PD into mouse brain.87,88 The study investigated whether iPSCs may have another role in in vivo disease modelling. However, despite increased levels of α-synuclein in the transplanted human neurons, there was an absence of cell spreading or aggregation (which would typify the disease phenotype), and so the authors concluded that “our results support the hypothesis that there might be a species barrier between human to mouse concerning alpha-synuclein spreading.” Application of models used in Parkinson’s disease (PD) research over the past 30 years. (a) PubMed was searched using the MeSH term Parkinson’s AND [mouse] or AND [rat] or AND [in vitro] to give an approximate number of publications employing the different models. The data are presented as the percentage of papers retrieved for all model systems. Almost 70% of papers used rat or mouse models, indicating that PD research is still heavily reliant on rodent models of PD, despite their widely acknowledged drawbacks and failure to recapitulate many vital disease symptoms. (b) The publications retrieved in (a) were broken down further to five-year intervals, to investigate whether there were any shifting trends in model utilisation. Whilst there is a noticeable decline in the number of papers employing rat models over time (orange/middle bars), this is not compensated with an increase in more human-relevant, in vitro methods (grey/right-hand bars) and instead seems to reflect increased use of mice (blue/left-hand bars).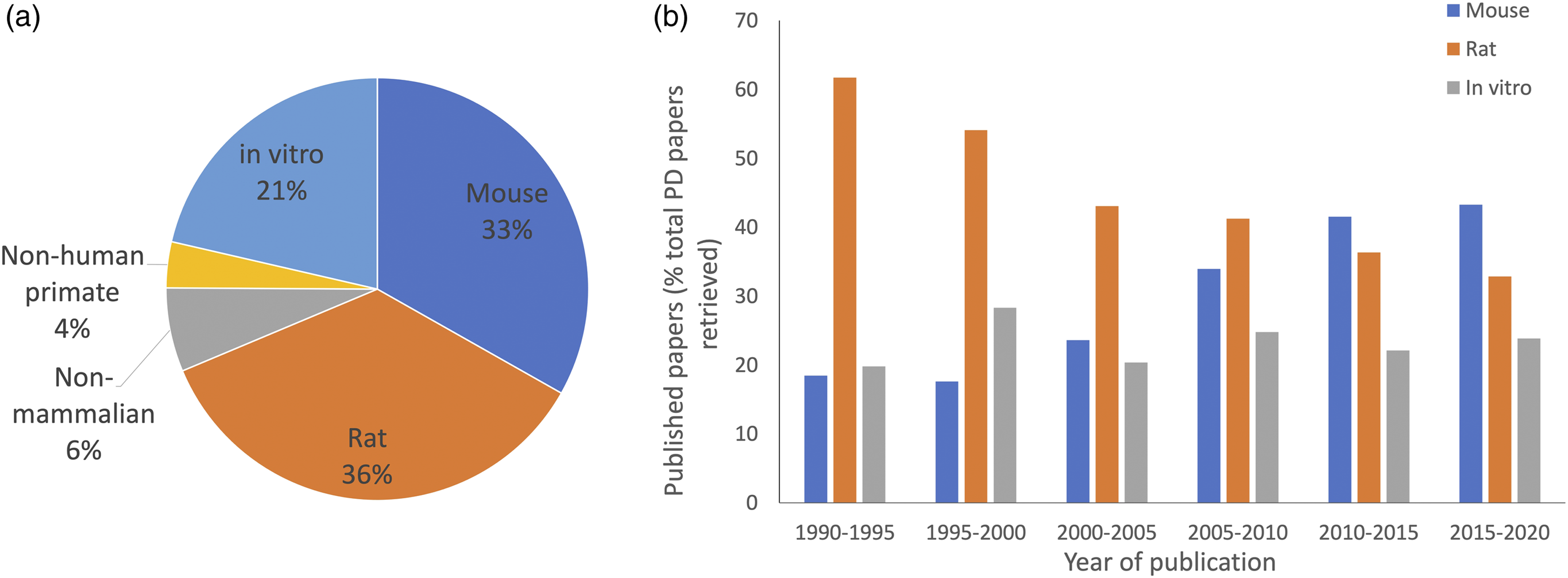
Several reasons underpin the failure of translation of animal-based preclinical trial results for PD drug treatments. As these often go beyond the disease itself, they are also relevant to consider in the context of the other conditions explored in this article. For PD, a recent retrospective analysis of preclinical data revealed that, for 266 studies of potentially disease-modifying interventions, improvement was apparent for 90% of studies on mice, 95% of studies on rats, and 67–80% of trials on non-human primates.
88
However, only 32% of human trials showed any clinical improvement.
88
This analysis reviewed the experimental details of the preclinical studies and reported several biases, namely that: — studies with non-human primates showed no gender bias, but tended to use younger adults; — studies using mouse models tended to use male animals, with one genetic strain predominating; and — studies with rats also showed a male bias, but used a wider variety of genetic strains.
Human-relevant, non-animal tools available for Parkinson’s disease (PD) research.
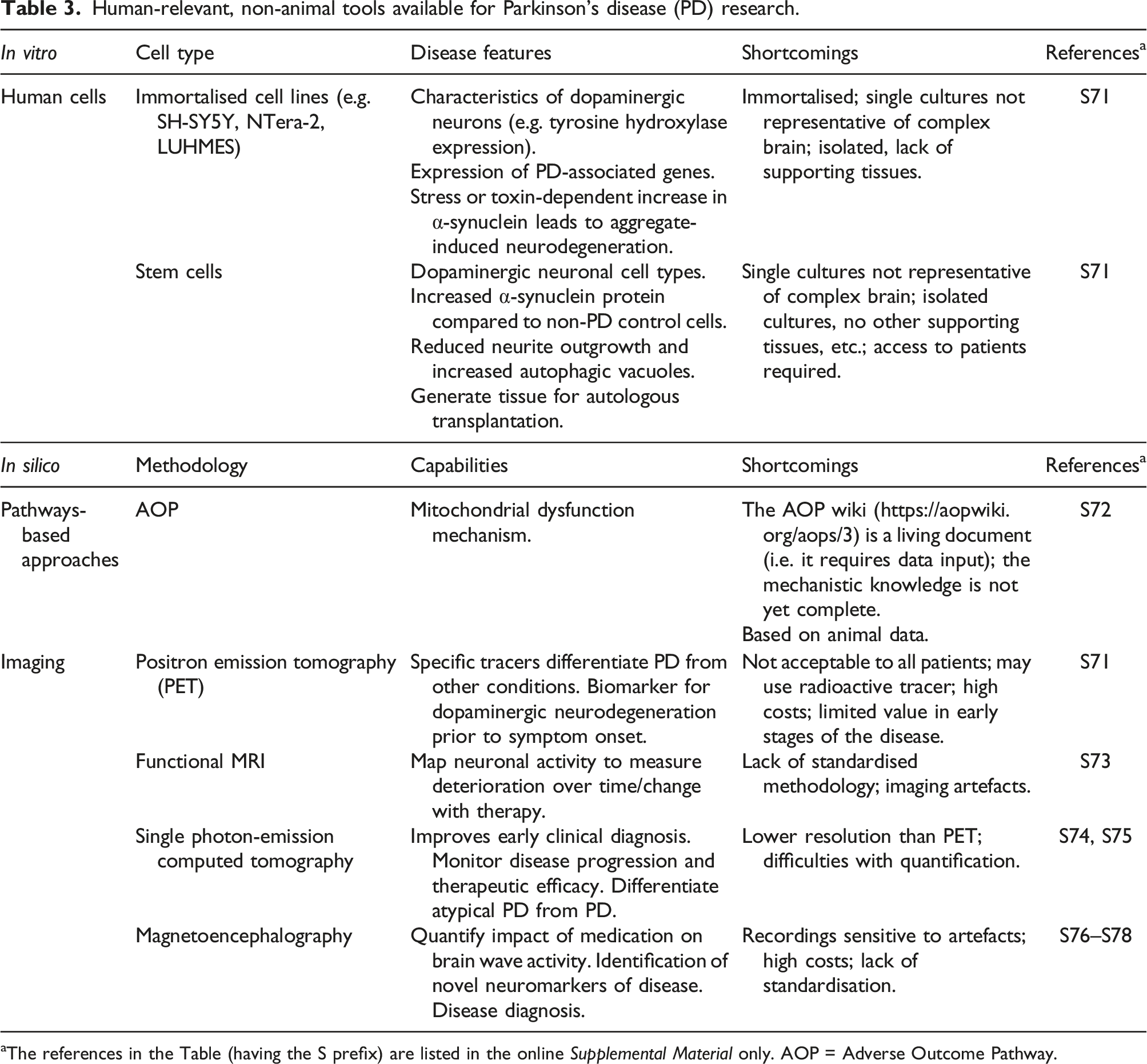
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. AOP = Adverse Outcome Pathway.
Rheumatoid arthritis
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterised by persistent synovitis, and various degrees of bone and cartilage erosion that eventually lead to the destruction of the joint, with resulting pain and disability. Systemic inflammation related to RA is associated with several extra-articular comorbidities (including cardiovascular disease), resulting in increased mortality in RA patients. 90 RA is regulated by both genetic and environmental factors, where approximately 50% of the risk for development of the disease is attributable to environmental factors, such as cigarette smoking. 91 In industrialised countries, RA affects approximately 1% of the population, making it the most common form of inflammatory arthritis. The disease most commonly begins in middle age, in the midst of working life, with significant social and economic impact, 92 despite currently available drug treatments.93–96
Current treatment options of RA involve non-steroidal anti-inflammatory drugs, glucocorticoids and disease-modifying anti-rheumatic drugs (DMARDs), such as methotrexate. For patients who fail to respond satisfactorily to these drugs, the additional use of biologic DMARDs, in particular tumour necrosis factor-α (TNF-α) inhibitors, offer greater chances for disease management. However, despite the undoubted success of therapeutic strategies employing anti-TNF-α, about 40% of patients treated with TNF-α blockers remain refractory to treatment, 97 and up to 50% of primary responders lose their response within 12 months of starting therapy.98,99 These drugs necessitate life-long treatment, but are associated with toxic effects, as well as the appearance of serious systemic adverse effects (such as increased risk of infections or cancer).100–103 With the increase in availability of biologic therapies, the outcomes for patients with RA have significantly improved, but it remains a debilitating chronic condition for which there is currently no effective cure. 104
Lack of knowledge of the disease-specific human pathophysiology and aetiology severely hampers the development of targeted drugs for RA. This could partly be a consequence of the use of too simplistic in vitro models, or an over-reliance on animal models that often cannot accurately recapitulate human RA aetiopathogenesis and drug responses, as well as the inadequate consideration and/or use of human-relevant research methods. So far, RA has generally been studied by using a variety of in vitro assays and animal models.105–112 Cell-based in vitro assays are based on relatively simple (co)culture systems, and the assays are generally used to investigate cell adhesion, cell migration, antigen presentation and lymphocyte activation.113,114 Cell and tissue models, especially those of human origin, are valuable tools in RA research. Traditional human synovial cultures have been crucial in the development of TNF-α blockers, which are, to date, the most successful therapy available for slowing disease progression and relieving symptoms. 115 However, culturing cells and tissues under static and non-physiological conditions (e.g. in plastic culture ware), or using cancer cell lines, could severely affect the relevance of the results.116,117
Many preclinical arthritis models have been developed, that are derived from a variety of species (e.g. mouse, rat, rabbit and monkey).118–121 Several murine models of arthritis have been established, 122 including models that require immunisation with antigens (e.g. proteoglycan-induced arthritis, 123 streptococcal cell wall arthritis, 124 collagen-induced arthritis (CIA)125,126 and antigen-induced arthritis); 127 models induced by adjuvants (e.g. oil-induced arthritis);128,129 spontaneous models (TNF-α transgenic mouse 130 and K/BxN T-cell receptor transgenic mouse 131 ); and humanised models. 132 While RA-like rodent models exhibit some of the typical features found in RA — i.e. joint swelling, synovitis, pannus formation and bone erosion — each model differs in the speed of disease onset, chronicity, severity, resolution and histopathology. 133 The rodent models each differ with regard to their histopathology, as well as being different again from human RA histopathology. 134
None of these models is truly RA, and none consistently predicts the effect of a therapeutic agent in patients. For instance, methotrexate (to date the first-line DMARD for RA treatment) is only marginally effective in the CIA model, and interleukin-6 deficiency has little or no effect in passive transfer models of arthritis or in TNF-transgenic mice. 125 Anti-CD20 antibodies (a next generation therapy widely employed in RA) only work in the CIA model when administered in the very early stages of disease induction, and are not effective in the later stages. For all these drugs, consideration of the preclinical (animal model) results, without clinical data, could have led investigators to abandon these effective therapeutic approaches. Conversely, positive data in rodents might lead to overestimation of the therapeutic effect in humans. For example, non-steroidal anti-inflammatory drugs are remarkably effective in rat adjuvant arthritis but provide only modest relief for RA patients.135,136
Although animal models have undoubtedly contributed to our understanding of the fundamental immunological mechanisms of RA, concerns about low clinical development success rates for investigational drugs,137–141 coupled with increasing awareness of the ethical issues surrounding the use of pain-inducing animal models, have led many to question their utility in the study of complex human conditions, as well as their effectiveness in drug target identification.142–146
Human-based advanced approaches for RA research
Human-based tools, models and approaches suitable for rheumatoid arthritis (RA) research.
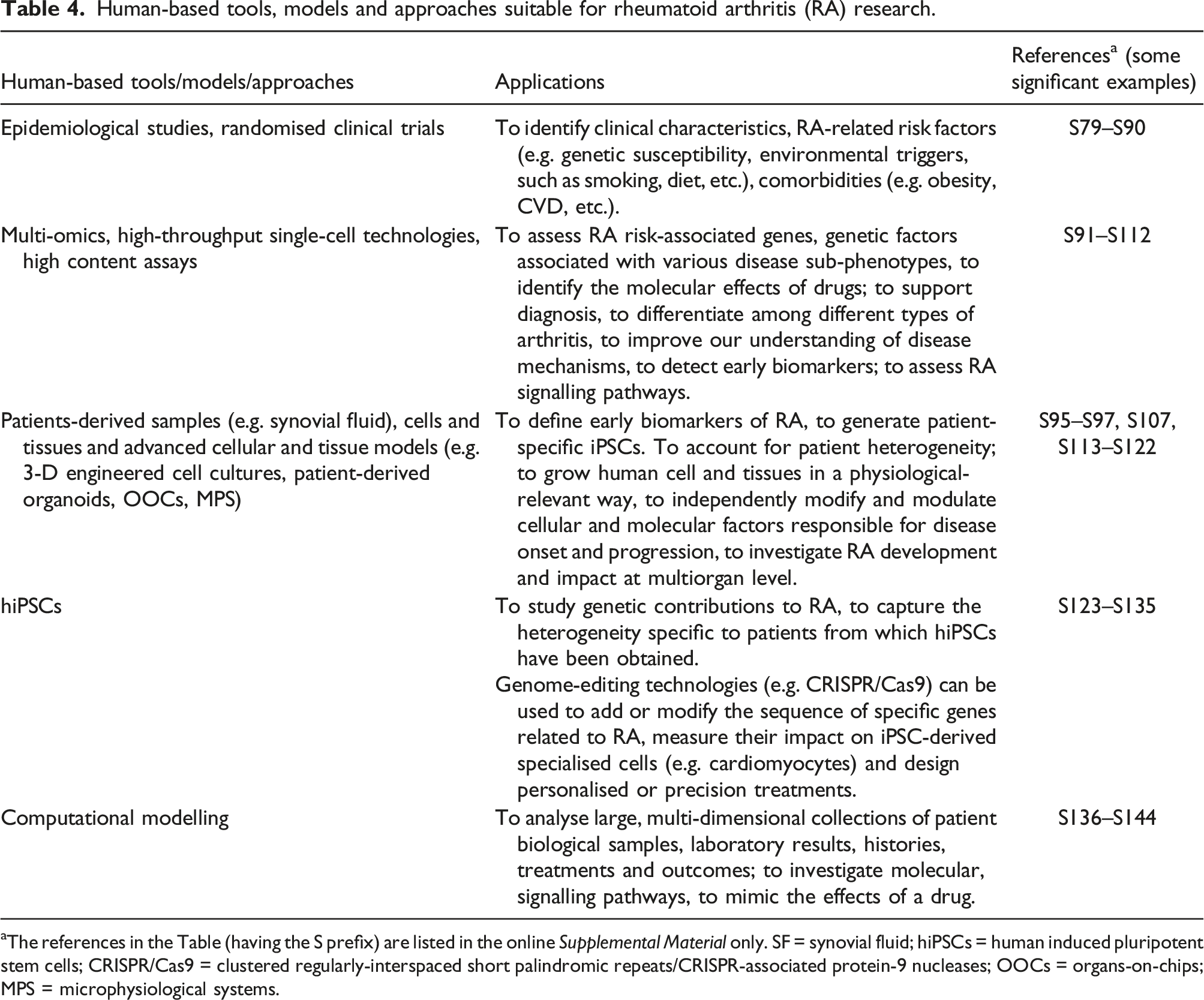
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. SF = synovial fluid; hiPSCs = human induced pluripotent stem cells; CRISPR/Cas9 = clustered regularly-interspaced short palindromic repeats/CRISPR-associated protein-9 nucleases; OOCs = organs-on-chips; MPS = microphysiological systems.
Respiratory diseases
In 2018, the World Health Organisation (WHO) announced their goal to reduce mortality due to non-communicable diseases (NCDs) by 35% by 2030. 147 Chronic respiratory diseases constitute a major proportion of these NCDs. The WHO is calling for better advocacy and research investment for such lung diseases, and this has led to the development of more human-relevant methodologies to ensure better translation, and to the more efficient use of research funding and expertise. Ultimately, these strategies will lead to a better understanding of these disease mechanisms in humans.
There is some evidence that science is moving away from the use of animal models — for example, the recent project from the European public–private partnership Innovative Medicines Initiative (IMI 3TR) calls for an analysis of clinical data from patients of various inflammatory and autoimmune conditions (before and after treatment), with the ultimate aim of shedding light on the factors that determine whether or not a patient is likely to respond to a given treatment. 148 However, there is still an over-reliance on animal models that fail to offer effective insight into disease mechanisms or provide targets that translate to successful human treatments.66,149–151 This is particularly apparent for respiratory diseases. For example, asthma and Chronic Obstructive Pulmonary Disease (COPD) represent major non-communicable respiratory diseases of epidemic proportions that affect people of all ages and have an increasing prevalence globally, but there are no cures for either.152,153 Research focused on developing animal models of these complex, human conditions has resulted in a plethora of options based on a wide range of non-human species (fruit flies, mice, rats, guinea pigs, rabbits, primates, dogs, horses, sheep),154–157 yet there have been few therapeutic breakthroughs and there remains a great unmet need for better therapies. Experts in the field call for a better understanding of the natural (human) progression of asthma and COPD, in order to identify new therapeutic targets, and to develop biomarkers that permit patient selection and enable mapping of the impact of long-term therapy. 158 Here we consider the translational failures of animal models of asthma and COPD; however, it is beyond the scope of this narrative review to cover all of the non-animal approaches available. For this, we refer to the recent project from the European Commission Joint Research Centre (JRC). The JRC sponsored the development of a collection of non-animal models for respiratory tract disease. 159 This snapshot of the currently available non-animal models for asthma revealed 41 different model systems under development, looking at various salient aspects of the disease — including inflammation, bronchoconstriction and airways remodelling. 159 Focusing funding and research efforts into further development of these human-relevant non-animal methodologies will be crucial to the better understanding of asthma disease mechanisms that is required for more effective treatments and the ultimate goal of a cure. 160
Asthma
Asthma is characterised by variable and recurring symptoms, reversible airflow obstruction and easily triggered bronchospasms manifested as recurrent attacks of breathlessness, wheezing, coughing and chest tightness, which vary in severity and frequency between individuals. 152 The cause(s) of asthma are still not completely understood, but are thought to be a combination of genetic predisposition and environmental exposure to inhaled particles that may irritate the airways or cause allergic reactions. Diagnosis is typically based on the pattern of symptoms, response to therapy over time and spirometry, and the disease may be categorised as intermittent, mild, moderate or severe (where severe disease is defined as “uncontrolled despite maximal optimised therapy and treatment of contributory factors, or that worsens when high dose treatment is decreased”). 161 Disease burden is associated with disease severity — despite relatively few people with severe asthma (less than 25% of the adult asthmatic population; data from a Dutch survey 162 ), the associated costs are great163,164 and, on an individual basis, are almost five times more for severe asthma than for people with mild disease. 165 The direct and indirect healthcare costs are vast, averaging around US$5000 per patient per year, but these costs are higher for people with severe asthma 165 and are on the increase. Global asthma-related costs are estimated to exceed those of tuberculosis and HIV combined, and developed economies can expect to spend up to 2% of their overall healthcare budget on the disease. 166
It is therefore clear that asthma represents a huge societal and economic burden, as it is associated with emergency care, multiple hospital admissions, missed days of school and work, as well as potential permanent disability or early death. In high income countries, asthma is the most frequent reason for hospitalisation (responsible for 30% of paediatric admissions), but it occurs in all countries, regardless of their level of development. 167 Variation in incidence across countries has led to estimates of between 1 and 18% of the global population being affected, 168 with the WHO estimating that 235 million people are currently living with asthma. 152 It remains the top-listed chronic condition, accounting for 15 million DALY lost every year. 169
Disease features, model characteristics and limitations of animal models used in asthma research.
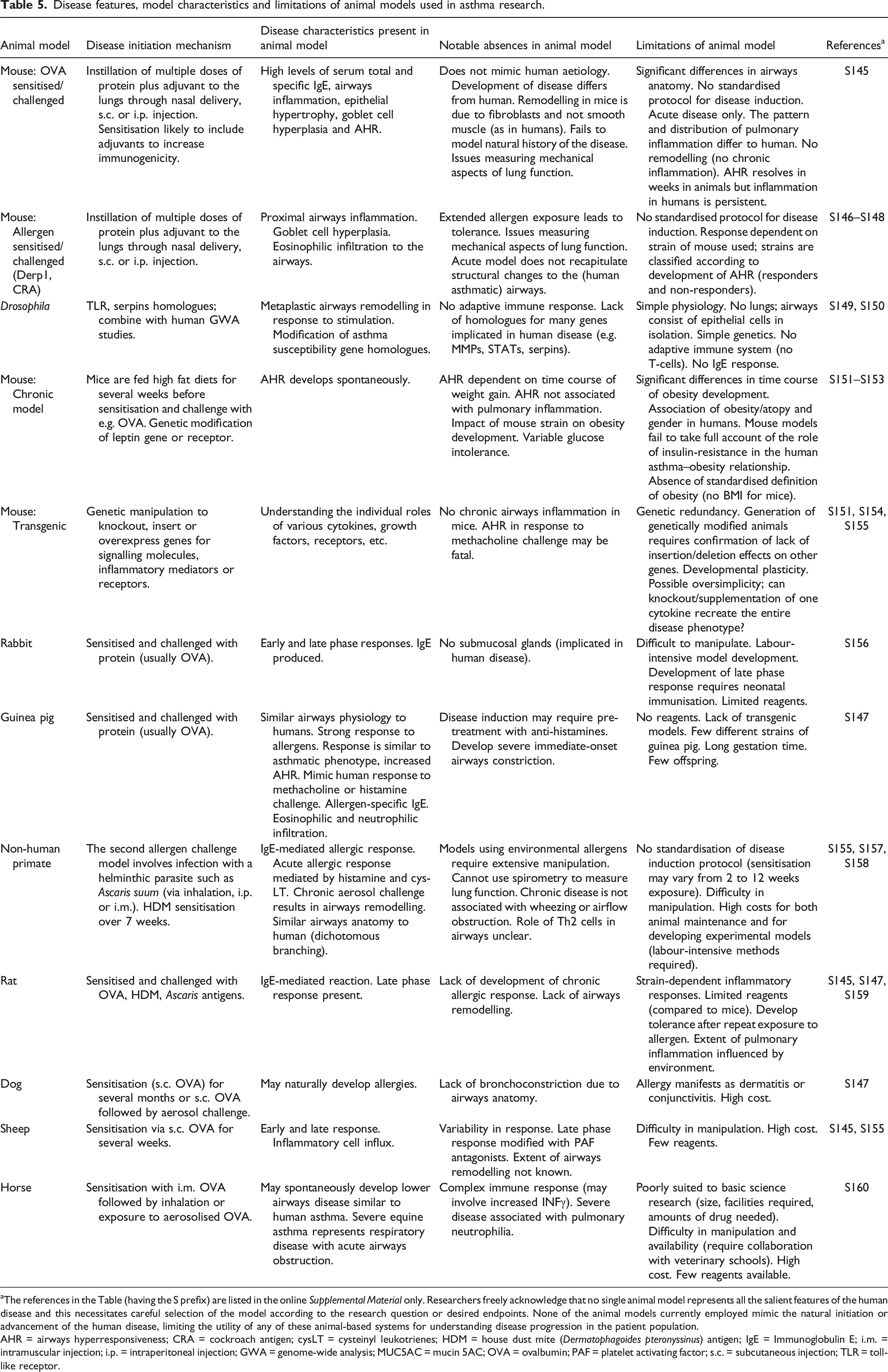
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. Researchers freely acknowledge that no single animal model represents all the salient features of the human disease and this necessitates careful selection of the model according to the research question or desired endpoints. None of the animal models currently employed mimic the natural initiation or advancement of the human disease, limiting the utility of any of these animal-based systems for understanding disease progression in the patient population.
AHR = airways hyperresponsiveness; CRA = cockroach antigen; cysLT = cysteinyl leukotrienes; HDM = house dust mite (Dermatophagoides pteronyssinus) antigen; IgE = Immunoglobulin E; i.m. = intramuscular injection; i.p. = intraperitoneal injection; GWA = genome-wide analysis; MUC5AC = mucin 5AC; OVA = ovalbumin; PAF = platelet activating factor; s.c. = subcutaneous injection; TLR = toll-like receptor.
Asthma targets that showed preclinical success in animal models of the condition, but failed in patients.
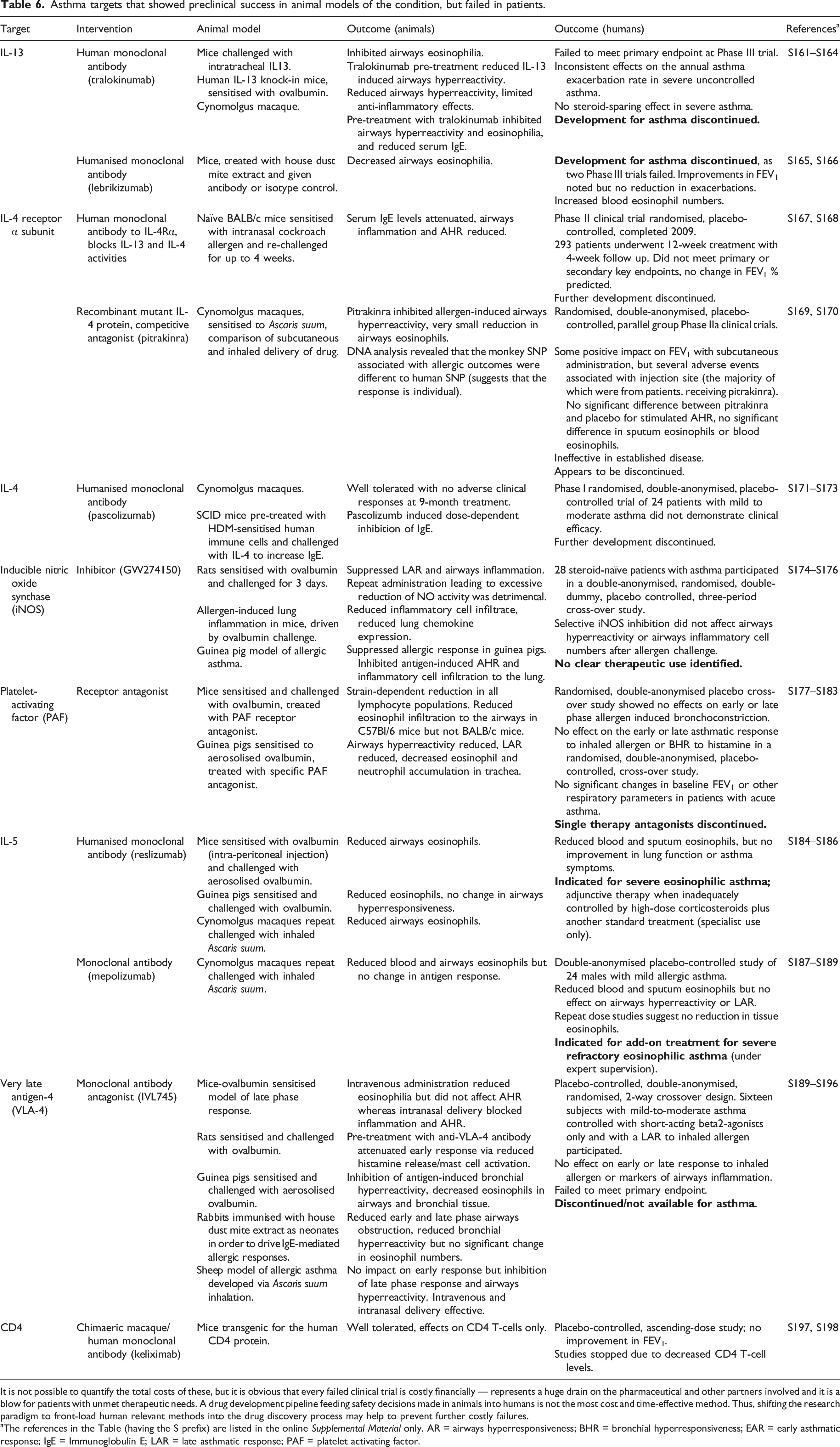
It is not possible to quantify the total costs of these, but it is obvious that every failed clinical trial is costly financially — represents a huge drain on the pharmaceutical and other partners involved and it is a blow for patients with unmet therapeutic needs. A drug development pipeline feeding safety decisions made in animals into humans is not the most cost and time-effective method. Thus, shifting the research paradigm to front-load human relevant methods into the drug discovery process may help to prevent further costly failures.
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. AR = airways hyperresponsiveness; BHR = bronchial hyperresponsiveness; EAR = early asthmatic response; IgE = Immunoglobulin E; LAR = late asthmatic response; PAF = platelet activating factor.
It is apparent that, in order to effectively model a uniquely human disease — with such a complex natural history, progression and individual subtypes 176 — a new approach is required that does not rely on animals. Indeed, in their review of 2011, Holmes and colleagues declared that: “The best model to use to study human asthma are patients.” 177 Of course, there remains a need for preclinical research, but this research does not have to be based on the use of animals. Indeed, a shift away from failing animal models does now seem possible, as outlined below and throughout this review.
The new non-animal technologies outlined here, as well as those described in more detail in the JRC project, 159 are all applicable to furthering our understanding of respiratory diseases. These include: patient-centred approaches that focus on biobanking samples for biomarker analysis; 178 ‘omics’ analyses to identify subtypes of disease; 179 and other patient cohort studies. 180 In addition, advances in tissue engineering now permit incorporation of the mechanical stresses of breathing into lung-on-a-chip models; 181 improvements in human tissue procurement are enabling the development of specific disease models, such as those based on precision-cut lung slices; 182 and various efforts are underway to create in silico models of the airways to model airflow and drug deposition in healthy lungs and in people with complex pathophysiologies (e.g. the EU-funded AirPROM). 183
Chronic Obstructive Pulmonary Disease
Chronic Obstructive Pulmonary Disease (COPD) is number one in the so-called ‘big five’ respiratory diseases. 184 COPD is an umbrella term used to describe progressive lung disease, including emphysema and chronic bronchitis. It is characterised as a chronic inflammatory lung disease that develops slowly and usually becomes apparent after 40 or 50 years of age. The most common symptoms are breathlessness, chronic cough and sputum production. 153 The leading cause of COPD is tobacco smoking, although long-term exposure to chemical irritants can also be involved. Diagnosis usually involves multifactorial assessment, including clinical presentation, spirometry to assess lung function, and assessment of exposure to risk factors. At the moment, there is no cure for COPD, but treatments are available that aim to ease symptoms, lower the chance of complications and generally improve quality of life. 185
The WHO estimated a prevalence of 251 million cases of COPD globally in 2016, causing approximately 3.17 million deaths in 2015 (representing 5% of all deaths globally that year). 153 The severity and prevalence of this condition make the economic costs extremely high — around 6% of the EU’s total healthcare expenditure is dedicated to COPD — equating to around €38.6 billion annually. 186 In the USA, where COPD is the third most common cause of death, the condition cost the economy around US$50 billion in 2010, 187 and these costs rocket when comorbidities are taken into account. A recent study of over 250,000 people in Canada revealed that the co-incidence of cardiovascular disease doubles the economic cost of COPD. 188 More recently, a systematic review of global ‘per patient’ costs revealed annual costs of US$17,219 in the USA, around US$3700 in Asia and almost US$6300 in Europe. 189 Since increasing severity also increases costs, it is vital to reduce disease progression and prevent exacerbations that require hospitalisation, to reduce the economic burden of COPD management.
Animal models used for research into chronic obstructive pulmonary disease (COPD).
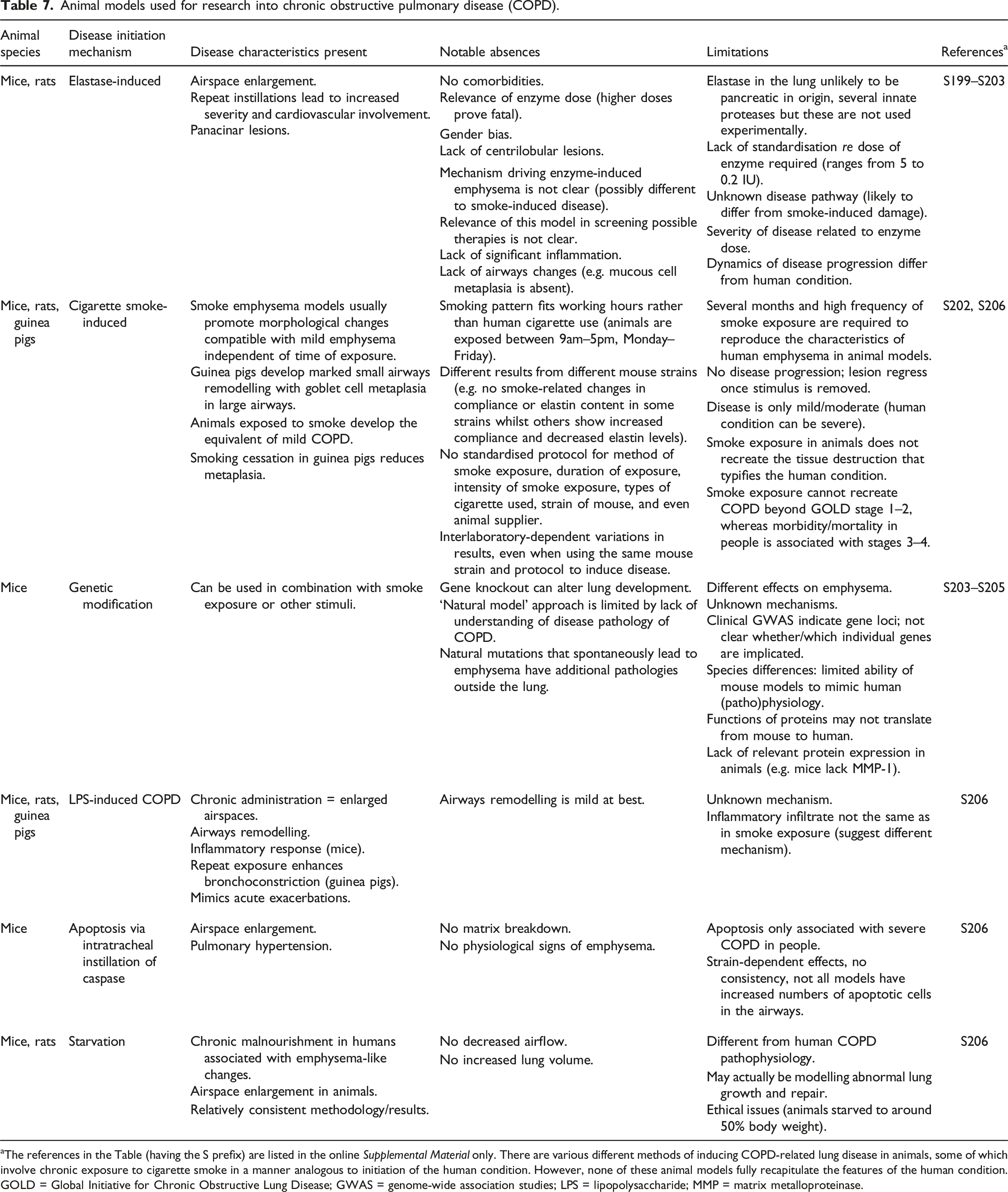
aThe references in the Table (having the S prefix) are listed in the online Supplemental Material only. There are various different methods of inducing COPD-related lung disease in animals, some of which involve chronic exposure to cigarette smoke in a manner analogous to initiation of the human condition. However, none of these animal models fully recapitulate the features of the human condition.
GOLD = Global Initiative for Chronic Obstructive Lung Disease; GWAS = genome-wide association studies; LPS = lipopolysaccharide; MMP = matrix metalloproteinase.
It must be noted that none of the animal models used, irrespective of the mechanism employed to initiate disease, accurately describe the human situation. Substantial differences exist in the architecture of the airways of other mammals and humans, and in the types and ratios of cells present throughout the respiratory tract. For example, mouse airways lack mucus-producing goblet cells — instead, the main secretory cell type is the club cell found throughout the trachea, bronchi and bronchioles. 190 In contrast, healthy human tracheobronchial airways contain goblet cells, estimated at around 3% of the total epithelial cells per mm of basement membrane, 191 and goblet cell hyperplasia is associated with airway disease. 192
Overall, animal models do not reflect the variable pathology and different stages of COPD severity in humans, and thus are restricted to modelling a limited number of characteristic features of COPD in the clinic. Also, several studies have shown that different strains of mice show various levels of sensitivity to CS challenge. 193 Additionally, for respiratory research and indeed, human disease modelling in general, the animals used tend to be young, healthy males and this is obviously not the case for human patients. Disease initiation often bears little resemblance to its natural counterpart in humans, and researchers are exquisitely aware of the time period that has elapsed between disease initiation in the animal models and the treatment interventions — this is again not the case for human patients. Particularly for progressive conditions such as asthma and COPD, the disease may not be diagnosed for some time and treatment may start at varying intervals for different individuals, impacting on outcomes.
Non-animal methods for human-relevant respiratory research
Unfortunately, the development of non-animal approaches to replace the use of animals in the study of human respiratory diseases has been slow. Human tissue and tissue engineering approaches show promise, but are limited by access to both normal and diseased human tissue. 194 The development of 3-D air–liquid interface (ALI) cultures has enabled the creation of more physiologically accurate models that recapitulate the pseudostratified mucociliary phenotype observed in vivo. Culturing cells at the ALI was shown to induce differentiation to a polarised, pseudostratified epithelial layer that is composed of multiple cell types and thus has the resultant functionality, including apical ciliary activity and mucus secretion. 195 Mathematical models can provide new insights to improve our understanding of asthma, and a number of these are under development. In silico approaches have been used, mainly for deposition modelling of inhaled therapies. 196
Precision-cut lung slices (PCLS) offer a phenotypically-accurate model of lung tissue behaviour and have been used to study the effects of allergens, bronchodilators, cigarette smoke and infectious agents. 197 PCLS represent a particularly physiologically relevant model, as they maintain the native cellular architecture. They thus permit the study of multiple cell types and functionality (e.g. macrophages, airway smooth muscle contractility, etc.) that is not possible in other, more reductionist cell-based models. However, as with many human tissue-based models, the availability of high quality tissue for normal and diseased airways can be problematic, and variability across donors can be an issue.198,199
For pulmonary disease research, computational (in silico) methods have become more prominent in the investigation of drug deposition and absorption, including the development of Physiologically-based Pharmacokinetic (PBPK) modelling and Quantitative Structure–Activity Relationships (QSARs) to aid the screening of promising drug candidates. For example, the IPRLu is a novel QSAR model, built using data from isolated, perfused rat lung, that accurately predicts pulmonary absorption. The authors claim that “The novel QSAR model described here can replace routine generation of IPRLu model data for ranking and classifying compounds prior to synthesis” offering a potential replacement for animal models. 200 Computational models can also aid the development of an overall prediction framework to provide information over and above what can be derived from direct measurement techniques. 201 For example, bronchoconstriction involves complex interactions among the small airways, resulting in regions of poor ventilation. Mathematical modelling of the mechanisms involved in bronchoconstriction allows insight to be gained into this complex airway behaviour 202 and better mechanistic understanding could lead to the identification of novel druggable targets. Computational models are available which can assess patient response to acute disease, 203 and these are being developed to generate patient-specific computational models of chronic disease. 204
The development of better and more accurate models will potentially lead to the better diagnosis of different types of asthma and COPD, allow better monitoring of the disease, provide tools for more targeted research into disease mechanisms, and ultimately assist the development of personalised medicine and the matching of appropriate treatments to each patient.
The continued need for HIV/AIDS vaccines
In 2008, a review of HIV/AIDS vaccine research and testing 205 formed part of a successful campaign to end invasive chimpanzee research in the USA, following an enquiry by the US Institute of Medicine (IOM) in 2011. 206 This review focused on the translatability of HIV/AIDS vaccine testing in chimpanzees to humans — the chimpanzees having been bred in large numbers in the USA from the 1980s, specifically for HIV/AIDS research. It was found that 85 different vaccine products showing encouraging results in NHPs (both chimpanzees and macaques) had led to 197 clinical trials, but they did not lead to a successful human vaccine, despite around 20 years of effort. This lack of translation could certainly be attributed, in part, to the widely acknowledged difficulty of the task — there is no shortage of evidence-based appreciation of the enormity of the challenge, given the nature of HIV and AIDS — but it must also be placed squarely at the door of species differences between NHPs and humans, and between HIV and the SIV (simian immunodeficiency viruses) used in research, which manifest in different pathologies.
Now, it is timely to summarise this 2008 review 205 and importantly to offer an update of what has happened in the years since. Has there been any success, or hope, or do animal models still fail to translate to human benefit? Infamously, in 1984, the US Health and Human Services secretary declared that a vaccine would be available within two years, and President Clinton set a goal of 2007 for the same. Both came to pass unfulfilled, and now, many years later, while a vaccine remains elusive, similar promises continue to be made. Optimism that NHPs infected with SIV and/or hybrid SHIV will eventually bear fruit persists, though it seems more guarded.
Crucially, it is clear that a vaccine is no less needed now than it has been for any of the past 30-plus years, even with the advent of controlled anti-retroviral therapy (cART), for example. This is because there are serious issues with regard to cART, which must be taken for life and has associated morbidities, making new therapeutic vaccination strategies “more than necessary”. 207 It is accepted that treatment alone will not put an end to AIDS, and that greater efforts are needed to accelerate the development of a preventive vaccine. 208 Furthermore, it is believed that vaccines are “urgently needed” and the “aggressive pursuit” of them is justified, 209 and that even a vaccine which is 70% effective could reduce infections by up to two-thirds over time and save millions of lives. 210 Since 1981, when AIDS was first recognised, more than 70 million people have been infected by HIV, of which 36 million have died. 210 Perhaps 40 million or so are living with HIV infection presently, and in 2018 alone, more than 770,000 people died from HIV and almost 1.7 million became infected worldwide. 211
In 2020, the HIV Vaccine Trials Network (hvtn.org) listed 19 ongoing vaccine trials under its auspices, involving almost 12,000 participants. Of these, 12 were in Phase I, and only one — the most advanced — was in Phase II/III. In July 2022, there were 11 ongoing trials, with just over 600 participants; again, all but one were in Phase I, with one in Phase III. The US NIH’s clinicaltrials.gov website listed 823 trials at the time of writing (July 2022), up from 197 in the cited 2008 paper by Bailey, though a greater proportion have now progressed to Phases III and IV (15%). These trials involved different vaccine subunits, various pox vectors, DNA, adeno and adeno-associated viruses, alphavirus, measles virus and vesicular stomatitis virus vectors, various prime-boost combinations, and a variety of different adjuvants and modes and routes of administration. 212 Nevertheless, it is a similar picture to 14 years ago: most trials were in Phase I; a small percentage had progressed to Phase III; and, crucially, there was still no licensed effective vaccine of any type.
The current/recent vaccine situation: Efficacy trials and DNA vaccines
A 2016 review reported that more than 250 Phase I and II vaccine clinical trials had been conducted worldwide, though “a clear correlate of protection remain[ed] elusive”. 210 Another contemporary review reported that four vaccine concepts had been tested in six efficacy trials (typically, Phase III) at the time of writing. 209 The results of four of these were “no efficacy” (Vax003, AIDSVAX B/E; Vax004, AIDSVAX B/B; HVTN 502 ‘Step trial’; and HVTN 503 ‘Phambili trial’), of which two actually showed an increased rate of HIV infection (Step and Phambili trials). Another trial was “stopped for futility” (HVTN 505) and the other (RV144) is the only efficacy trial to date that showed even modest efficacy, of 31.2%. 209 Regarded as the most promising candidate ever, at least until very recently, this vaccine’s protective effect nevertheless “waned over time”, 213 with vaccine efficacy falling from 60% to 32.1% after 3.5 years. 214 Some researchers were quick to point out that, in spite of it being another failure, there were some important outcomes from the lessons learned: in particular, the development of the HVTN 702 vaccine, which is similar to RV144 but involving antigens from a different clade of HIV. This HVTN 702 vaccine was mentioned in a late-2019 review, reporting an unprecedented level of late-stage clinical activity. Three efficacy trials were ongoing in Africa, one of which was the HVTN 702 Phase IIb/III trial, also known as ‘Uhambo’ (NCT02968849). The others were HVTN 705/HPX2008 (Imbokodo, NCT03060629) involving 2600 women; and HVTN 706/HPX3002 (MOSAICO, NCT03964415) involving 3800 participants, similar to the regimen in the Imbokodo trial, but with a mosaic gp140. Others included a Phase IIb trial involving a DNA vaccine combined with pre-exposure prophylaxis, PrEPVacc (NCT04066881); and two Phase IIb trials of passive antibody immunity, HVTN 703 and 704. 215 The authors conveyed some lack of confidence in preclinical studies, stating: “As results from efficacy-stage studies are eagerly awaited (as soon as 2021–22), all possible outcomes must be anticipated.” This was prescient. At the time of writing, news came through of another failure of a large-scale efficacy trial. Sponsored by the National Institute of Allergy and Infectious Diseases (NIAID), HVTN 702 (or Uhambo) which had enrolled almost five-and-a-half thousand HIV-negative volunteers in South Africa in 2016, was terminated due to no difference in HIV infection between individuals who had received the vaccine, and those who had received a placebo. 215 This was despite “great hopes” for it, and it resulting from a “lesson” from the failure of the RV144 vaccine trial. 215
Another recent review noted a focus on DNA vaccines, with more than 160 in clinical trials; one third of which were for HIV/AIDS. 211 The DNA vaccines were based on multiple antigens, and had shown promising results in multiple species, including macaques and baboons. Other recent reviews have highlighted DNA prime/modified vaccinia Ankara (MVA) boost vaccinations, which have been an area of research for around 20 years. Optimism remains, based largely on evidence of robust immune responses in animals and humans. However, while there is evidence of protection from mucosal infection in NHPs, there remain stubborn issues around protection from infection against high-dose challenges, CD8+ T-cell responses in humans, and poor and inconsistent immunogenicity of the DNA component in both NHPs and humans. 216 Given the latest in a line of failed efficacy trials amid high levels of optimism, the results of salient ongoing trials must be awaited with some trepidation and anticipation of further disappointment.
Reasons for failure: Biological differences between NHPs and humans, and between HIV and SIV
In 2008, detailed descriptions of the failure of chimpanzee research in this field, in terms of empirical lack of success to translate to an effective human vaccine, as well as interspecies differences in HIV pathology and immune function, highlighted the fact that this failure was likely, if not guaranteed, to continue. 205 Subsequently, general reviews of the genetic differences between chimpanzees and humans, 217 and also between monkeys and humans, 218 have revealed underlying differences in all aspects of gene expression that specifically affect SIV/HIV infection and pathology. These are compounded by differences in the SIV and (artificial, hybrid) SHIV viruses used to model human HIV infection in NHPs (see below, and Bailey 205 ). All of this was supported by referenced opinions of researchers on the poor relevance of chimpanzee HIV/AIDS research to humans, which expressed concern and doubts over the human relevance of the SIV/SHIV-infected macaque model for HIV vaccine development, which had been used since the turn of the century.
These species differences, and their important consequences for SIV/HIV pathology, still remain. Other differences have been reported more recently, including: — significantly different transcriptome profiles in CD4 and CD8 cells between cynomolgus macaques and humans;
219
— sex differences in SIV and HIV disease progression and morbidities between species, including altered expression levels of interferons and other immune response genes,
220
and differences in vaccine-induced antibody species and their functions in SIV-infected rhesus macaques;
221
— differential expression of the α4β7 integrin in CD4 T-cells of different NHPs used in HIV/AIDS research, which are one of the major targets of infection and which affect the course and rate of progress of the disease;
222
— differences in several interferon-induced transmembrane protein (IFITM) genes between different NHPs used in HIV/AIDS research and humans, which can restrict SIV/HIV infection and replication;
223
and — species specificity of Nef and Vpu genes/proteins in overcoming restriction of SIV/HIV, mapped to just a small number of amino acids.
224
There have been a number of notable issues recently reported that could affect the translation of data from NHPs to humans,
225
namely that: — the human routes of HIV transmission are relatively poorly understood, and this has consequences for their modelling in NHPs; — many transmission and viral challenge studies in NHPs have involved cell-free virus stocks, and potentially more relevant use of infected cells is omitted from most NHP studies; — the intravenous transmission route is often used in NHPs, but is the least clinically relevant for HIV; — NHP protocols often involve large volumes of fluid and numbers of virions, which is not a good model for human transmission; and — concerns have been raised over the human-relevance of the mucosal challenges that are used in NHPs.
One review 226 noted that no transgenic animals, despite being genetically engineered to express proteins known to be necessary for HIV replication (absent in non-genetically modified monkeys and in small animal models used in research), could support robust viral replication or development of the disease — presumed to be due to other cofactors essential for HIV replication and other proteins that inhibit HIV. It also noted important differences between the types of NHPs used in HIV/AIDS research: each of the main species used (rhesus, pig-tailed, and cynomolgus macaques) has unique traits “that can profoundly affect the outcome of SIV infection”, a deep understanding of which is necessary “to avoid pitfalls that can lead to confusing or uninterpretable results”. For instance: Indian-origin rhesus macaques develop AIDS within 1–2 years of infection, compared to 8–10 years for humans; human and macaque MHC genes differ in several ways (see also Bailey 218 ); Chinese and Burmese rhesus macaques show very different pathogenesis to the Indian rhesus macaques (probably due to important immunogenetic differences); pig-tailed macaques develop AIDS in just over half the time of rhesus macaques; cynomolgus macaques, when infected with SIVs/SHIVs that are pathogenic to Indian-origin or Chinese-origin macaques, show less pathogenicity, and viral loads that are several logs lower and more variable; Chinese rhesus macaques have acute and chronic viral loads of SIV that can be several orders of magnitude lower than macaques of other origins, and the underlying cause of this may well impact viral transmission. 225 The suggestion that simply using different types of SIV more “specifically adapted” to Chinese-origin macaques seems superficial — why would this be more human relevant?
Differences between the viruses used in ‘modelling’ human HIV infection in monkeys appear to have been taken more seriously, at least by some. “Renewed enthusiasm” for a move away from using SIV has been prompted by the resistance to neutralisation evident in many SIVs, the limited availability of antibody reagents to SIV epitopes of interest, and the fact that many broadly reactive neutralising antibodies to HIV envelope protein (Env) do not cross-react with SIV. Unfortunately, the response by many is not to focus on human research, but to ‘tweak’ the monkey approach — thus, instead of using SIV, many are developing hybrid SHIV viruses. Even so, shortcomings with SHIV are acknowledged, with Del Prete et al. stating that they “represent limited genotypic breadth… and [thus] do not represent the most clinically-relevant Envs for transmission studies and vaccine assessment”. 225 Other concerns are that HIV vaccine immunogens cannot be tested by challenge with SIV, and that SIV is not sensitive to many HIV inhibitors, and that the viruses may have some similar co-receptors, but they have different ones too. 226 Often, SHIVs created to try to overcome these problems have been disappointing — some replicated poorly in NHPs and, even if some began to replicate more efficiently with time, they can be “paradoxically easy to protect against by vaccination”. Taking the other approach — i.e. manipulating HIV to replicate in NHPs — has been less problematic, but it is acknowledged that much more engineering needs to be done to produce a virus that reliably replicates in macaques, that this process is still in its infancy, and that no animal model may ever capture all the features of human HIV infection. 226
Update on HIV/AIDS vaccine clinical trials.

In 2008, a search of the National Library of Medicine’s clinicaltrials.gov database returned 197 clinical trials involving 85 different vaccines of many types.S78 A new search (“HIV vaccine” on 3 February 2020) to update this produced 766 results, detailed in the table.
Importance of human-specific discoveries and data
On top of this caution over NHP models, there is significant optimism surrounding the capabilities of, and an appreciation of the need for, human-specific research. Recent illustrative examples include the demonstration that intestinal microbiota antigens (which differ between individuals) can alter immune responses to HIV, blocking the induction of antibodies that could protect against HIV infection (these studies predated similar research in NHPs). 227 The critical role of CD8+ cells in viral clearance has been attributed to NHP studies (for example, see Schmitz et al. 228 ) but has also been demonstrated (and elaborated upon) in studies of human elite controllers, which remain extremely important229,230 (see also Seddiki et al. 207 ). The importance of studying acutely infected humans — particularly their CD8+ cell responses and how these change over time and affect reservoir size and viral diversity, and critical host–virus interactions — was conveyed in a 2016 review. 231 The authors believed that this would be crucial in informing interventions for both preventive and therapeutic vaccines. 231
The isolation of broadly neutralising antibodies (bNAbs) from HIV-infected people has increased our understanding of the sites of vulnerability that they target on HIV itself. These bNAbs develop in a large proportion of HIV patients after 5+ years, and are able to effectively neutralise many (and diverse) strains of HIV. Our understanding of these antibodies, alongside studies on the fine structure of Env and on antibody–virus co-evolution, are important in informing the current and future directions of vaccine design.232,233 This was reiterated by a 2017 review, ‘Lessons learned from human HIV vaccine trials’, 234 which noted that, while no bNAbs had been induced by any vaccine trials in humans at the time of writing, the detailed analysis of memory B-cell repertoires and the evaluation of antibody effector functions in humans had greatly expanded what could be learned from human trials, with particular regard to antibodies with potential neutralising properties.234,235 The study of bNAbs in the small percentage of HIV-infected people with elite serum neutralisation has indicated a variety of ways in which bNAb activity may be elicited, and several major bNAb sites have been identified. 236 Meanwhile, though some researchers who use genetically modified mice are advocating their use to further the study of the role of bNAbs in HIV vaccination, it is questionable whether they can solve the previous lack of any animal model capable of identifying limiting factors in bNAb production and vaccine strategies capable of promoting it. 235
It is, of course, acknowledged that the development of an effective preventive or therapeutic HIV/AIDS vaccine is an ambitious scientific undertaking of immense challenge. The reasons for this challenge are well documented, and involve factors such as the variability of the virus, its immune system evasion, and particular aspects of its life cycle. In view of these acknowledged difficulties, it is of paramount importance to utilise the best tools and models available, instead of a continued reliance on SIV and SHIV-infected NHPs and a defence of using ‘every tool in the toolbox’ — however poor some of those tools appear to be. We must ensure that any and every tool is as relevant to human infection, disease and biology as possible. Increasing numbers of scientists are opining that NHP use in this field is at best unreliable, and probably so poor as to be misleading and a waste of human, animal and financial resources. Increasingly, researchers have highlighted the promise and necessity of novel and human-specific technologies, including mass cytometry, structural analyses, immunogen design, omics techniques, clinical research, studies of elite controllers, 237 and this is where the future endeavours and focus should lie.
Discussion
We have used several distinct human diseases to illustrate where animal models are failing to offer translational power to understand the condition, or to reveal safe and effective drugs.
We accept that, historically, biomedical research and testing relied on animals as the only option for assessing safety and efficacy, but this is no longer true. There are many areas where the non-animal new approach methodologies are proving more predictive than animals. For example, significant advances in the field of toxicology were initiated over a decade ago, with the advent of Tox21 and the seminal report, Toxicity testing in the 21st century: A vision and a strategy, 238 which articulated a vision for toxicity testing based on human cell-based assays, and so reducing reliance on animals, the financial costs and testing time. Since then, we have seen a rapid evolution of NAMs, in a manner incomparable to that of animal models, and the increasing application of non-animal approaches for chemical safety testing — including the formal inclusion of NAMs into international test guidance documents.239–241
We are also seeing the increased evidence of the utility of NAMs in predicting human toxicity. For example, a recent study used 27 compounds to test the ability of human liver chips to predict hepatotoxicity and found specificity of over 80% and sensitivity of 100%. 242 Beyond this use for toxicity testing, it is encouraging to see NAMs increasingly being applied in biomedical research. The acceleration in focused research activity that came about as a consequence of the recent pandemic illustrated how NAMs can rapidly be applied for understanding disease progression and for repurposing previously approved drugs to provide much-needed treatment options.243,244 Recently, the US Food and Drug Administration (FDA) accepted efficacy data from an organ-on-a-chip as part of an Investigational New Drug (IND) submission for clinical trial. 245 We recommend that readers access Ingber’s recent article on organs-on-chips, 246 for a more comprehensive review than is possible in this current paper, where we provide just one example to explore the promise of this technology.
Throughout this review, we offer several examples where non-animal approaches are in development, or already being used, to address the issue of high drug attrition rates. This issue needs to be urgently addressed, in order to meet the currently unmet need for new drugs, and to get medicines to patients faster, through a more efficient process. To achieve this goal, it seems that biomedical research and drug development need to (continue to) shift away from the failures of animal use and move toward these more promising, predictive approaches.
The integration of non-animal, human-based approaches is key. In particular, the integration of complex in vitro models with computational approaches could allow the identification of new therapeutic targets and the evaluation of efficacy and toxicity of new drugs, reducing costs and testing time. It is also essential to allocate funding for human-relevant approaches and invest resources in the optimisation and qualification of these new in vitro models, as well as in the improvement of the quality of human tissues (from deceased or living donors) via the provision of, and ongoing support for, efficient tissue and cell banks. Another important aspect is the need to invest much more in primary prevention, considering the importance of lifestyle in the onset especially of AD, cancer, cardiovascular diseases, pulmonary disorders, and autoimmune disorders.
We suggest that more focus on human-relevant methods is required to fulfil the needs of patients and pharma, and we urge readers to take a closer look at the respiratory tract disease resource developed by the JRC 159 and also to explore the other resources available. These collate non-animal models for breast cancer, 247 immuno-oncology, 248 immunogenicity testing for advanced medicinal therapy products, 249 and neurodegenerative diseases. 250 We encourage the development of more collections and are looking forward to the JRC’s planned creation and curation of a database of these methods (see also the NAT Database, https://nat-database.org) — and, importantly, the dissemination and implementation of the methods therein. For biomedical research, this could be through the animal welfare/ethical review process where Three Rs ‘champions’ could raise awareness of non-animal approaches and make suggestions as to how and where these methods may offer reduction and replacement opportunities.
There is also the need for new regulatory approaches, and to persuade industry, research funding bodies and the scientific community at large, that there is a need for a change in biomedical research approaches — for example, striving to increase the level of multidisciplinary collaboration. Greater transparency and timeliness in the sharing and communication of results obtained in the various phases of preclinical and clinical experimentation are also desirable, so that even regulators and members of the scientific community who are not directly involved in such studies can be informed quickly about any possible failures. 251
Conclusion
A paradigm shift in biomedical research is clearly still needed. This shift should increasingly converge toward human-based and human-relevant approaches, in order to tackle the growing prevalence of human diseases. The hope is that, through the integration of different areas of competence and investigation, it will be possible to encourage primary prevention initiatives and support scientific research activities that are based on the use of methods and models that will deliver human-relevant results for the ultimate benefit of human patients.
Supplemental Material
Supplemental Material - Poor Translatability of Biomedical Research Using Animals — A Narrative Review
Supplemental Material for Poor Translatability of Biomedical Research Using Animals — A Narrative Review by Lindsay J. Marshall, Jarrod Bailey, Manuela Cassotta, Kathrin Herrmann and Francesca Pistollato in Alternatives to Laboratory Animals
Footnotes
Acknowledgements
The authors would like to thank Prof. John Gluck for early review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The references listed in the Tables (having the S prefix) are listed in the online Supplemental Material only.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
