Abstract
Background
Zinc deficiency (ZD) is one of the most prevalent nutritional deficiencies reported in celiac disease (CeD). Oral supplementation and dietary modifications are the primary strategies for addressing it. However, it is unclear whether individuals with CeD would prefer to make additional dietary modifications or opt for supplementation to treat ZD.
Aim
This study aimed to explore patient preferences for the treatment of ZD.
Methods
Adult patients (≥18 years) with a confirmed diagnosis of CeD who were already adopting a gluten-free diet (GFD) and had untreated ZD were recruited from the Adult Celiac Disease Clinic at McMaster University. After providing informed consent, participants completed study questionnaires at baseline and three months after initiating zinc supplementation. Data were collected using REDCap. Statistical analyses were performed using IBM SPSS software (V22, Chicago, USA).
Results
From March 2022 to January 2024, 44 participants were enrolled in the study. All completed baseline questionnaires, of them 25 (57%) completed questionnaires at follow-up. At baseline, 34% of participants preferred to treat nutrient deficiencies through dietary changes, compared with 28% at follow-up. Plasma zinc level normalized in all participants after 3 months of supplementation. At follow-up, the proportion of participants with significant gastrointestinal symptoms increased from 72% at baseline to 80% (p = 0.01).
Conclusions
One-third of patients with CeD preferred to manage nutrient deficiencies through dietary modifications. A zinc-optimized GFD may represent a promising approach to address zinc deficiency in this population. These findings provide preliminary evidence to inform the design of future randomized clinical trials.
Background
Celiac disease (CeD) is a chronic immune-mediated condition driven by gluten in genetically predisposed individuals (Catassi et al., 2022; Pinto-Sanchez et al., 2024). The only established treatment for CeD is lifelong adherence to a strict gluten-free diet (GFD). While a GFD is effective in promoting disease remission and mucosal healing, it presents significant challenges due to its social restrictiveness and nutritional inadequacies (Cardo et al., 2021). One notable concern is the frequent occurrence of micronutrient deficiencies, even among individuals adhering to a GFD (Chao, 2023; Fathi et al., 2013; Rondanelli et al., 2019; Russell et al., 2025; Wierdsma et al., 2013). As a result, guidelines from the American College of Gastroenterology and the National Institute for Health and Care Excellence recommend routine screening for micronutrient deficiencies in newly diagnosed patients with CeD (I.C.G.T.Coeliac Disease; National Institute for Health and Care Excellence: London, UK., 2015; Rubio-Tapia et al., 2013).
Zinc deficiency (ZD) is among the most prevalent nutritional deficiencies in CeD, affecting up to 30% worldwide and 48% of treated patients at a tertiary care Celiac Disease Clinic in Ontario (Jivraj et al., 2022; Russell et al., 2025). The prevalence of ZD is significantly higher than 8–17% reported in the world's population (Roohani et al., 2013). While the prevalence of ZD in the Canadian general population is not well established, it is estimated that 10–35% of Canadians consume less than the recommended dietary allowance (RDA) of zinc (Zn) (Health Canada, 2012). Zn levels may be reduced in individuals with CeD primarily due to impaired intestinal absorption and the limited micronutrient fortification of gluten-free (GF) foods (Maxfield et al., 2025). GF products are often low in essential vitamins and minerals and tend to be higher in fats and sugars (Shepherd and Gibson, 2013). Moreover, dietary constituents such as phytic acid found in legumes, seeds, nuts, and whole grains can bind to Zn and impair absorption (Maxfield et al., 2025; Roohani et al., 2013).
Zn is the second most abundant trace element in the human body and plays a crucial role in numerous physiological processes, including cellular turnover, immune and reproductive function, neurotransmission, and wound healing (Ahmed et al., 2021).
ZD has significant health consequences, including delayed puberty, infertility, cognitive impairments such as reduced memory, attention, and learning capacity, increased susceptibility to infections, and impaired wound healing. Additionally, symptoms commonly associated with ZD, such as diarrhea, anorexia, altered taste and olfactory function, and fatigue, can contribute to substantial weight loss and malnutrition (Saper and Rash, 2009). The primary approaches to managing ZD include dietary modifications and oral Zn supplementation (Roohani et al., 2013). In clinical practice, oral supplementation remains the mainstay of treatment (Health Canada, 2012; Roohani et al., 2013). While generally well tolerated, Zn supplements can produce adverse effects, most notably gastrointestinal disturbances including abdominal pain, diarrhea, and nausea. Furthermore, high-dose or prolonged Zn supplementation may interfere with the absorption of other essential minerals, particularly iron and copper (Adams et al., 2002; Silva et al., 2021). Despite its clinical relevance, standardized guidelines for dietary Zn supplementation have yet to be established (Chao, 2023). Many individuals prefer dietary approaches over pharmacological treatments, often viewing them as more natural, sustainable, and better aligned with long-term health goals (Ahmed et al., 2021). In theory, a Zn-optimized diet could potentially address ZD while minimizing side effects and avoiding depletion of other nutrients (Adams et al., 2002; Saper and Rash, 2009; Silva et al., 2021). Such a diet would ideally consist of well-tolerated, GF foods rich in bioavailable Zn and low in phytates, and should be tailored to individual preferences and tolerances (De Leon Morilla et al., 2022). However, evidence on the practical implementation of a Zn-rich, low-phytate diet in individuals with CeD remains limited.
To date, it is unclear whether individuals with CeD, already navigating the complexities of a strict GFD, would prefer to make additional dietary modifications or opt for supplementation to treat ZD. Therefore, this study aimed to explore patient preferences for the treatment of ZD and assess the effect of Zn supplements on CeD symptoms.
Methods
This study was conducted at the Celiac Disease Clinic, McMaster University Medical Centre, Hamilton, ON, Canada. Adult patients (≥18 years of age) with a confirmed diagnosis of CeD were included in the study. Diagnosis was established based on positive CeD-specific serology—anti-tissue transglutaminase IgA (anti-tTG ab), anti-deamidated gliadin antibodies, and/or anti-endomysial antibodies and confirmed by duodenal biopsies demonstrating villous atrophy (Marsh grade ≥3a). Participants were eligible if they had untreated ZD, defined as a plasma Zn (pZn) concentration ≤9.4 µmol/L, identified during routine blood work. pZn levels were measured as part of their laboratory routine, using Inductively Coupled Plasma Mass Spectrometry (ICP-MS) at the Core Laboratory of the Clinical Chemistry and Immunology Department, Hamilton General Hospital, Hamilton, Canada. Demographic data were collected using REDCap software (US, v11.1), including age and gender, disease activity, CeD-specific serology, duodenal biopsies, current medications and supplements, clinical medical history, weight, height, and serum levels of micronutrients. All participants provided written informed consent prior to enrollment. Data were collected at baseline and at follow-up, which occurred three months after participants were prescribed 25 mg Zn gluconate to be taken once daily during their visit to the clinic. A sample bottle containing 100 tablets of 25 mg Zn gluconate (Jamieson Laboratories, Canada) was kindly provided by their physician during the clinic visit.
The primary outcome of this study was to explore patient preferences regarding treatment of ZD, while secondary outcomes included assessment of the effects of Zn supplementation on CeD-related gastrointestinal and extraintestinal symptoms. We also explored associations between symptom burden and pZn levels.
Outcome measures
Each participant completed three questionnaires to evaluate treatment preferences and gastrointestinal and extraintestinal symptoms at baseline and follow-up.
Patient preferences were assessed using a 13-item questionnaire designed to explore their views based on knowledge and treatment preferences related to nutrient deficiencies in general and ZD (see Supplementary Material 1). The questionnaire included items addressing the perceived importance of various aspects of nutrient deficiencies and ZD management. Responses were recorded using a five-point Likert scale, with options ranging from “Extremely important” to “Not at all important.” Symptoms associated with CeD were evaluated using the Celiac Symptom Index (CSI), a validated 16-item questionnaire employing a five-point Likert scale (Leffler et al., 2009). CSI scores ≤30 were considered indicative of clinical remission, while scores >30, specifically within the ranges of 30–35 and ≥45, were interpreted as suggestive of ongoing or active disease. The CSI can be used to track symptom burden over time and assess the effectiveness of treatment, primarily the gluten-free diet. Extraintestinal symptoms were assessed using the Extraintestinal Symptom Visual Analogue Scale (ES-VAS). This 10-item tool evaluates the severity of common extraintestinal manifestations in CeD, including skin rashes, headaches, cognitive symptoms (“brain fog”), fatigue, joint and muscle pain, feeling anxious, and feeling depressed. Each item is rated on a scale from 0 to 10, where 0 indicates “no symptoms at all” and 10 indicates “severe symptoms.” Patient Preferences Survey and the ES-VAS are provided in the supplementary material.
Statistical analysis
Statistical analyses were conducted using the IBM Statistical Package for Social Sciences (SPSS) software version 22 (Chicago, USA). GraphPad Prism 10 Software version 10.5.0 (Boston, USA) was used to create the graphs. Continuous data were expressed as mean and SD or median Interquartile Range (IQR). Comparison of categorical variables between the groups was performed using the McNemar, Fisher's correction. As quantitative variables were not normally distributed according to the Shapiro test, Wilcoxon signed rank test was used to compare median values of symptoms at baseline and follow-up. To find the impact of patient dropout in the study, a subgroup analysis was conducted to assess the demographic characteristics, of those who had follow-up, and those who dropped out. Spearman correlation was used to assess the relationship between the pZn level and extraintestinal symptoms. A p value <0.05 was considered statistically significant.
Results
Demographics
From March 2022 to January 2024, 211 CeD patients were eligible for the study. Of those, 44 patients agreed to participate and completed the questionnaires at baseline (Figure 1). Of them, 25 (57%) patients completed the study questionnaires at 3-month follow-up.
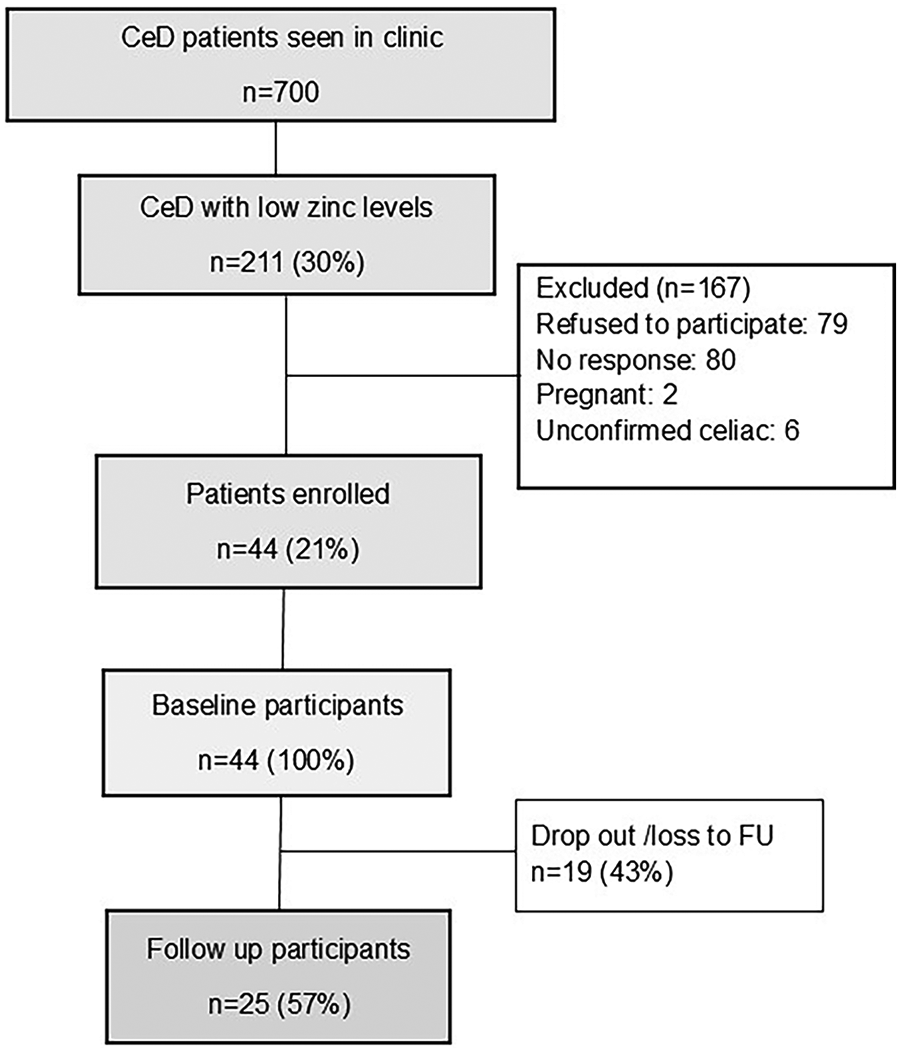
Flow diagram of study recruitment.
Of all 44 participants, 37 (84%) were female, and the median age (IQR) was 35 (27) years. Table 1 shows the demographics of the patients who completed study questionnaires at baseline and follow-up. Demographics of patients who did not complete the follow-up are shown in Supplementary Table 1. Given that the proportion of missing data in our study was substantial (43%), multiple imputation was considered inappropriate. Therefore, a complete-case (per-protocol) analysis was performed, including the 25 participants who completed follow-up. At baseline, 86% of the patients were taking oral supplements, and 42% of them included a multivitamin containing a low amount of Zn (<10 mg). None of the participants were taking Zn supplements before enrollment.
Demographics of patients recruited in the study.

Note: N/A = not assessed.
*Median (IQR range).
Patients completed study questionnaires at baseline.
Patients completed study questionnaires at baseline as well as at follow-up.
Patients completed study questionnaires at follow-up.
Patient knowledge of nutrient deficiencies and treatment preferences
Overall, 34.1% of participants preferred to treat nutrient deficiencies through dietary changes, 61.4% would prefer a supplement over diet, and 4.5% had no specific preference. Patients’ preferences remained unchanged at follow-up: 28% preferred dietary changes to treat nutrient deficiencies, 68% preferred supplementation, and 4% had no preference (p < 0.001) (Figure 2).
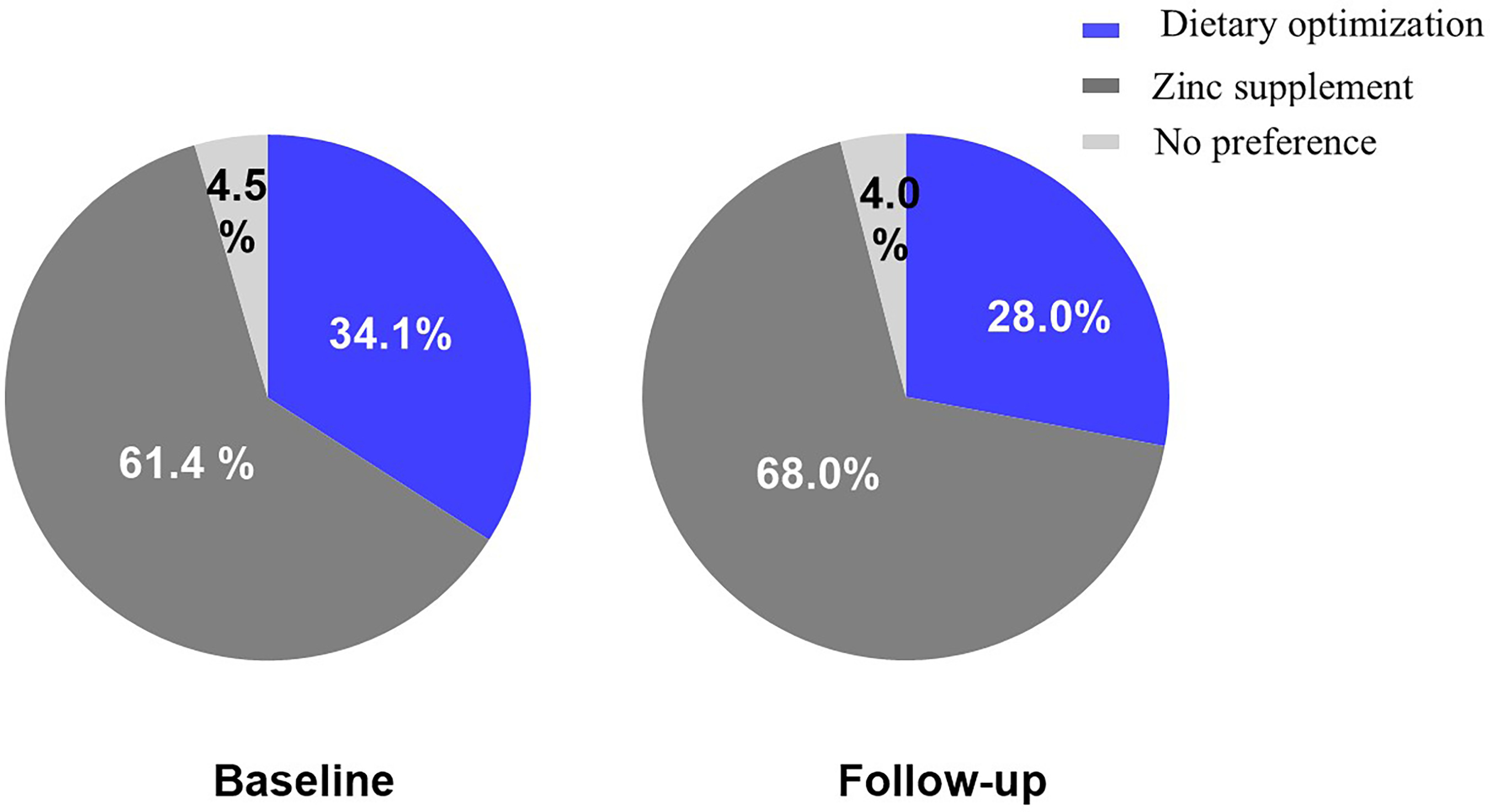
Patients’ preference for treatment of nutrient deficiencies at baseline and follow-up.
In two separate questions, patients were asked how likely they were to treat Zn deficiency (ZD) with a supplement and with dietary changes, including optimizing nutrient intake from a GFD. At baseline, 95.5% reported they were highly likely to use a Zn supplement, increasing to 100% at follow-up. In contrast, 89% indicated they would use dietary modifications at baseline, but this declined to 68% after 3 months of Zn supplementation (p < 0.001). We also explored perceived costs associated with treatment. Although 66% of patients expected that a Zn-optimised GFD would be more expensive than the standard GFD, 50% of the participants reported that this would limit their ability to adopt such a diet. Table 2 provides details on the responses for each question.
Patient satisfaction survey responses at baseline (n = 44) and follow-up (n = 25).
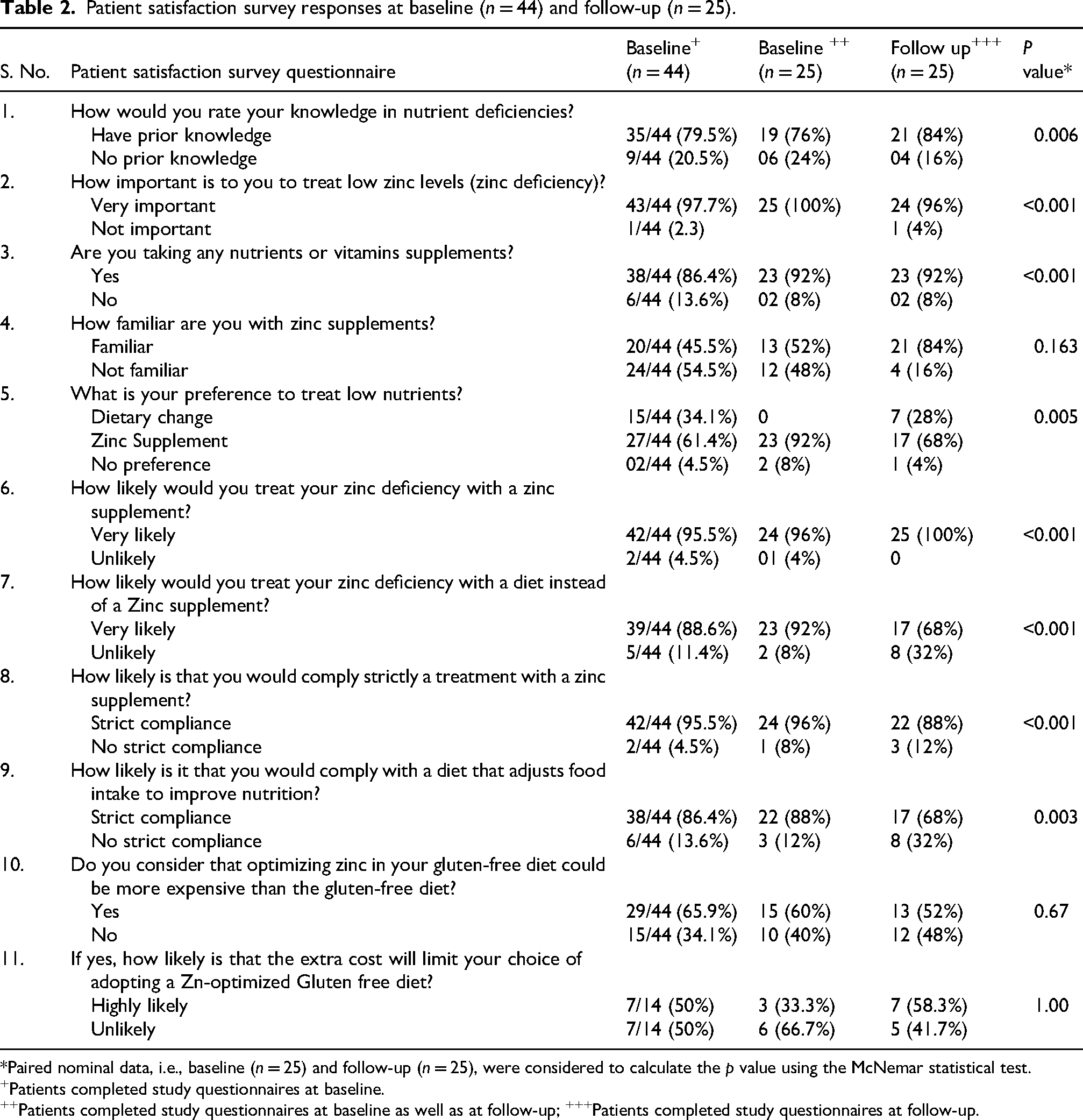
*Paired nominal data, i.e., baseline (n = 25) and follow-up (n = 25), were considered to calculate the p value using the McNemar statistical test.
Patients completed study questionnaires at baseline.
Patients completed study questionnaires at baseline as well as at follow-up; +++Patients completed study questionnaires at follow-up.
Celiac-specific and extraintestinal symptoms before and after Zn supplement
At baseline, celiac symptoms were reported by 72% of patients, and the most common symptoms were bloating (84%), abdominal pain (80%), and low energy (48%). Three months after Zn supplement, celiac symptoms were more commonly reported (CSI ≥30 baseline vs. follow-up, 72% vs. 80%; p = 0.01). More specifically, participants reported increased gastrointestinal symptoms such as abdominal pain (80% vs. 84%, p < 0.001), bloating (84% vs. 96%, p < 0.001), sensation of incomplete emptying of bowels (68% vs 72%, p = 0.06) after three months of Zn supplement. Some symptoms, such as diarrhea and low appetite, remained unchanged after treatment. Nausea (64% vs. 44%, p = 0.85), hunger pain (76% vs 68%, p < 0.001), and low energy (48% vs. 24%, p = 0.28) decreased at follow-up (Supplementary Figure 1).
The most common extraintestinal symptoms at baseline were feeling anxious (76%), fatigue (64%), brain fog (36%), and feeling depressed (40%). After 3 months of treatment, headaches (28% vs. 16%, p = 0.01), feeling anxious (76% vs. 60%, p = 0.13), and skin rash (16% vs. 8%, p ≤ 0.001) decreased in frequency. Other extraintestinal symptoms, such as fatigue (64% vs. 40%, p = 0.07), feeling depressed (40% vs. 28%, p = 0.1), brain fog (36% vs. 20%, p = 0.06), and joint and muscle pain (32% vs. 24%, p = 0.05) decreased after treatment, but these were not statistically significant. Table 3 shows the median of extraintestinal symptoms, and Figure 3 shows the improvement in the symptoms before and after Zn supplement.
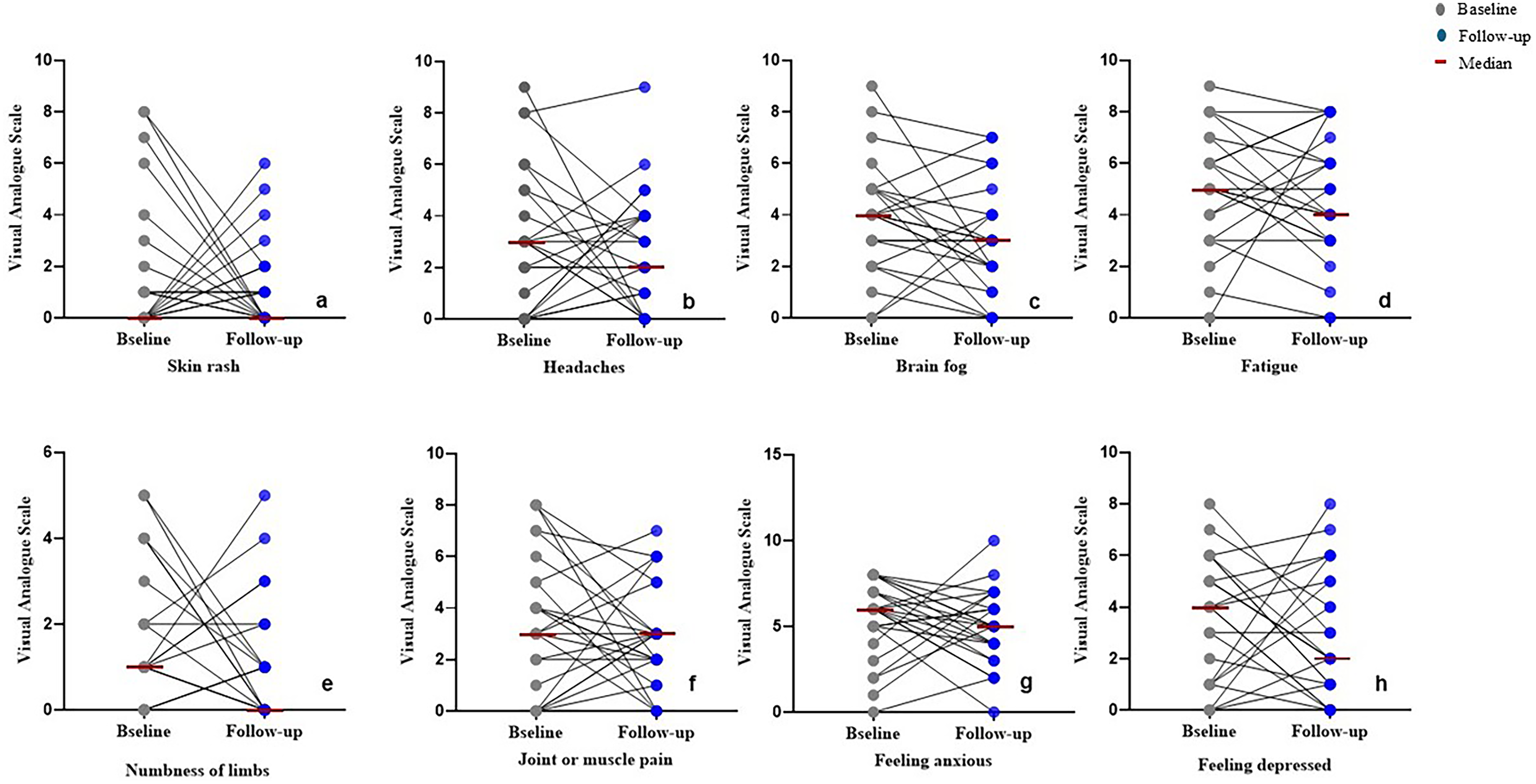
Improvement in the extraintestinal symptoms at baseline (n = 25) and after Zn supplement (n = 25). (a) skin rash; (b) headaches; (c) brain fog; (d) fatigue; (e) numbness of limbs; (f) joint or muscle pain; (g) feeling anxious; and (h) feeling depressed.
Extraintestinal symptoms of study patients at baseline and follow-up.
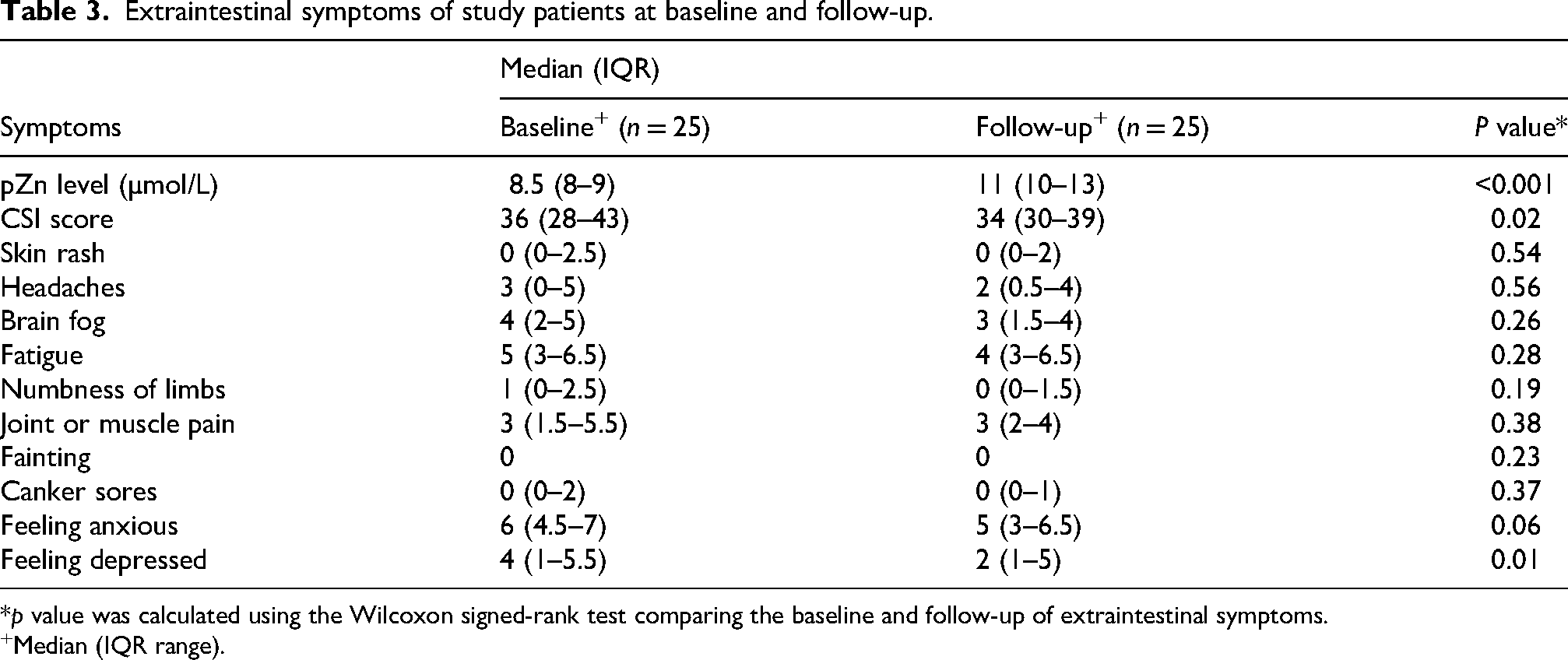
*p value was calculated using the Wilcoxon signed-rank test comparing the baseline and follow-up of extraintestinal symptoms.
Median (IQR range).
Status of plasma Zn levels (pZn) and the association of pZn with symptoms
Compared to the baseline, pZn level normalized in all patients after 3 months of treatment (median pZn baseline vs follow-up 8.5 vs. 11.0 µmol/L; p ≤ 0.001)
Reported side effects
Three months after Zn treatment, five participants reported three minor side effects (abdominal pain, bloating, and sensation of incomplete emptying). None of the participants reported stopping the supplements due to side effects (Supplementary Table 3).
Discussion
In this study, we explored patients’ preferences for treating nutrient deficiencies in CeD. Approximately one-third of participants preferred dietary therapy over supplementation. These findings are noteworthy given the high prevalence of ZD in CeD, which affects up to 50% of individuals even after treatment (I.C.G.T.Coeliac Disease; National Institute for Health and Care Excellence: London, UK., 2015). While previous studies have examined patient preferences for dietary interventions versus supplementation (Hsiao, 2013; Khubchandani and Batra, 2024; Yancy et al., 2013), this is the first study, to our knowledge, to report such preferences specifically in the CeD population.
Adhering to a strict GFD is inherently challenging, and the prospect of additional dietary modifications may increase this burden, potentially explaining why two-thirds of participants with CeD preferred supplementation over dietary changes. Moreover, when assessing participants’ knowledge of nutrient deficiencies, most were unfamiliar with ZD and its treatment options.
In our study, Zn supplementation was associated with less frequent extraintestinal symptoms, particularly headaches and joint and muscle pain. Additionally, improvements were noted in celiac-specific symptom scores, suggesting a broader potential impact of zinc repletion on overall symptom burden in individuals with CeD. While previous studies have identified that ZD may manifest with a range of intestinal symptoms (e.g., diarrhea) and extraintestinal features such as skin lesions, hair loss, anorexia, sensory impairments, and neurological disturbances (Ahmed et al., 2021; Health Canada, 2012; Hsiao, 2013), to our knowledge, no prior study has specifically evaluated the effects of Zn supplementation on symptom improvement in the CeD population.
An increase in abdominal pain and bloating was observed at follow-up despite normalization of pZn levels and coincided with the onset of new gastrointestinal symptoms after initiation of Zn supplementation. These adverse effects were infrequent and generally mild, with fewer than 10% of participants reporting worsening overall CeD symptoms after three months of supplementation. Although the observational design of our study precludes causal inference, gastrointestinal symptoms such as abdominal pain and bloating are recognized side effects of Zn supplementation, and an association with supplement intolerance cannot be excluded. This possibility warrants further evaluation in adequately powered randomized controlled trials, particularly given that side effects may limit adherence to supplementation regimens (Maxfield et al., 2025). Notably, a substantial proportion of participants withdrew from the study for unclear reasons; whether these withdrawals were treatment-related remains uncertain, underscoring the need for further investigation in the CeD population.
Our findings also identified perceived economic barriers to dietary management. At baseline, 66% of participants, and 52% at follow-up, believed that a Zn-optimized GFD—rich in foods such as sirloin beef, crab, gluten-free oats, pumpkin seeds, and pork chop would be more expensive than a traditional GFD (Zinc: Fact Sheet for Health Professionals, 2022). More than half reported that the high cost would ‘highly likely’ limit their ability or willingness to adopt such a diet. Consistent with previous reports, participants identified the high cost of gluten-free products as a major obstacle to maintaining a strict GFD, which may further reduce the feasibility of optimizing Zn intake through diet. While dietary optimization may offer a promising approach to address ZD, particularly for patients who prefer non-pharmacological strategies or are intolerant to supplements, no standardized or clinically validated Zn-rich diet currently exists (Maxfield et al., 2025; Roohani et al., 2013). As a result, this approach is not routinely recommended in clinical practice (Chao, 2023; Verma et al., 2023).
This exploratory study has several limitations, including a small sample size and low retention rates, both of which may affect the generalizability of the findings. The quality of the relationship between research staff and participants is a recognized factor influencing trial success; thus, the minimal interaction in this pragmatic study may have contributed to the low retention rate (Poongothai et al., 2023). Notably, participants who dropped out were younger and less likely to adhere to a GFD (Supplementary Table 1), a finding that may be important when designing future clinical trials. Additionally, among participants who completed the patient preference questionnaire at both time points, none chose dietary change as their preferred treatment for ZD at follow-up, which may reflect a potential population bias.
To our knowledge, this is the first study to investigate patient preferences specifically in individuals with CeD. While larger, more rigorous studies are needed, these findings provide a foundation for future research. Randomized controlled trials are particularly warranted to assess the feasibility, acceptability, and efficacy of a zinc-optimized GFD for treating ZD in CeD. Although dietary optimization of nutrients is an appealing strategy, the potential added cost and complexity of a Zn-optimized GFD may limit its adoption. A feasibility pilot study is therefore a logical next step prior to pursuing a larger efficacy trial.
Conclusions
One-third of patients with CeD in our study preferred managing nutrient deficiencies through dietary modifications. A zinc-optimized GFD may be a promising option to address zinc deficiency, particularly for those favoring non-pharmacological approaches or unable to tolerate supplements. However, its feasibility and effectiveness remain unclear. The results of this study offer preliminary data to guide the design of future randomized clinical trials, which are needed to determine the efficacy, feasibility, and clinical impact of a zinc-optimized GFD in improving zinc status within this population.
Supplemental Material
sj-docx-1-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-1-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-docx-2-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-2-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-docx-3-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-3-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-docx-4-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-4-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-docx-5-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-5-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-docx-6-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-docx-6-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Supplemental Material
sj-pdf-7-nah-10.1177_02601060261425465 - Supplemental material for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design
Supplemental material, sj-pdf-7-nah-10.1177_02601060261425465 for Patient preferences in managing zinc deficiency in celiac disease: Insights to inform future clinical trial design by Anil K Verma, Shilpa Tandon, Kennedy Graham, Jedid-Jah Blom, Lindsey Russell, Jane Morgan, David Armstrong and Maria Ines Pinto-Sanchez in Nutrition and Health
Footnotes
Acknowledgements
The authors acknowledge gratitude to all the patients who participated in this study.
Ethics approval and consent to participate
The study was approved by the Hamilton Integrated Research Ethics Board (HiREB#14108).
Consent for publication
The manuscript has been reviewed and approved for publication by all the authors.
Authors’ contributions
AKV: data curation, writing – original draft preparation, writing – review and editing, and statistical analysis. ST: patient recruitment, patient coordination, data curation, protocol development, statistical analysis, and writing – review and editing. KG: protocol development, patient recruitment, review and editing. JJB: nutritional assessment, review, and editing. LR: review and editing. JM: nutritional assessment. DA: review and editing. MIPS: conceptualization and methodology, writing – review and editing, supervision, project administration, and final approval of the version to be published.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M.I.P.S. was supported by the Farncombe Family Digestive Health Institute Nutrition Initiative and received an AFP Gastroenterology Division and HAHSO grant. The study received no funding from any funding body.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MIPS received research funding from Takeda and ProventBio; none of these are relevant to this manuscript. LR received an honorarium from Takeda and Baxter for presentations and holds a Napo Pharmaceuticals Inc research grant not related to this manuscript. D Armstrong has received honoraria from Takeda and Fresenius for consulting, presentations, and research funding from Nestlé© Canada; none are relevant to this manuscript. AV, ST, and KG declare that they have no conflicts of interest.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
