Abstract
Background
Increasing interest in probiotics for mental health has led to products marketed to consumers claiming psychological benefits. This study aimed to identify mental health claims on probiotic supplements and to provide a narrative review of the scientific evidence to support these claims.
Methods
Probiotic supplements with mental health claims on the product label were identified. A search of the scientific literature was performed using Google Scholar, PubMed and Cochrane Central Register of Controlled Trials. Studies were included if the primary outcome was mental health or a biomarker of mental health, if they investigated the exact probiotic strain or combination of strains as present in the product, and if the study was a trial of probiotics conducted in either humans or animals. Included studies were assessed for risk of bias.
Results
Fourteen probiotic products with mental health claims were identified containing five different strains or strain combinations. Eighteen studies relevant to the products and investigating mental health outcomes met inclusion criteria. Ten studies investigated the same combination of probiotics and provided evidence to support the claims made. Statistically significant results were not universally present for all products.
Conclusion
The amount and quality of evidence to support claims of mental health benefits made on probiotic products is variable. The results support the need for a grading system to facilitate informed consumer decision-making when choosing a probiotic supplement to support mental health.
Introduction
A rapidly expanding body of research suggests great promise in the field of probiotics for improved mental health (Berding and Cryan, 2022; Johnson et al., 2023; Madison and Bailey, 2023; Musazadeh et al., 2023). Alongside this research is a burgeoning market for probiotic supplements sold to consumers with claims of ‘mental health benefits’, forecast to be worth $85 billion USD by 2027 (Statista, 2022), and over $105 billion USD by 2029 (Dairy Industries International, 2024). Probiotics are live microorganisms that when ingested in sufficient amounts confer a health benefit to the host. Psychobiotics are microorganisms specific to the improvement of mental health (Dinan et al., 2013). Through actions in the gut, probiotics can alter mood and psychological outcome by modulating neurotransmitter synthesis including serotonin and GABA, reducing pro-inflammatory cytokines or acting on the hypothalamic–pituitary–adrenal axis to modulate the stress response (Misera et al., 2021; Ross, 2023). Despite a surge in commentaries and reviews promoting probiotics for health and well-being and multiple pre-clinical studies demonstrating putative mechanisms, probiotic supplementation in human trials does not always result in observable changes in mental health (Kelly et al., 2017; Le Morvan de Sequeira et al., 2022; Vasiliu, 2023), and there are conflicting interpretations of the available research evidence (Britton et al., 2021; Freedman et al., 2020; Suez et al., 2019). While some clinical trials demonstrate the positive benefits of probiotic consumption, other high-quality studies fail to find significant reductions in psychological symptoms. Heterogeneity in well-being outcome measured, analysis of biomarkers of disease in healthy participant populations, and diverse approaches to trial design and probiotic strain studied are some of the proffered reasons for variability in study results (Freedman et al., 2020; Suez et al., 2019). The presence of both positive and negative study outcomes leads to conclusions in review articles that can favour one perspective or the other. A predominance of review articles in the published literature alongside relatively fewer randomised controlled trials further perpetuates a discrepancy between claims of benefit and the evidence to support these claims (Freedman et al., 2020).
Probiotics are classified according to their taxonomy. In addition to their genus and species, probiotics are designated a specific strain identifier. The effects of probiotics are species and strain specific (McFarland and Goh, 2019; Sniffen et al., 2018). For consumers to achieve their desired health goal, it is essential that they select the probiotic that is suitable for the specific health improvement they are seeking. Evidence to support a product claim needs to be specific to the strain or formulation contained in the product rather than simply the species, or probiotics more generally. An examination of websites accessed by consumers for information about probiotics in gastrointestinal disorders identified that 94% of the websites contained information about probiotic species; however, strain-level information was not always provided (Brinich et al., 2013).
Regulatory systems around the world classify probiotic products depending on their intended use, and the associated health claims on product labelling (Freedman et al., 2020; Spacova et al., 2023). Provided the product does not claim to prevent or treat a disease, probiotics are typically classified as dietary supplements or functional foods. They are distinguished from medicines because they cannot contain controlled drugs or make disease treatment claims and are often governed by legislation related to food and food labelling as opposed to medicines. Qualified health claims require scientific evidence to support them and an appropriate disclaimer or qualifying statement with approved wording (Food and Drug Administration, 2024a). Structure-function claims describe the role of an ingredient in the human body in general terms. For example, general well-being claims make a statement about the role of the ingredient in supporting general well-being (Food and drug Administration, 2024b). Adherence to regulatory requirements for labelling is one aspect of health claims made about probiotics. Scientific researcher, consumer and clinician expectations of the evidence supporting health claims are likely to differ. Evaluating the evidence to support mental well-being claims on probiotic products is an important step towards a shared understanding of potential benefit between industry, researchers, clinicians and consumers.
Consumers have a broadly positive perception of probiotics and believe that they can have health benefits (Kozirok, 2021). Consumers are more likely to take probiotics if they undertake healthy lifestyle behaviours, are university educated, have a higher socioeconomic status and have a greater awareness of gut health (Khalesi et al., 2021). Many people believe that health claims made on product labelling have been evaluated to substantiate the claims made on products (Harrison et al., 2015). Nevertheless, consumers retain some skepticism around claims found on labels (Avila et al., 2020; Kozirok, 2021). Participants in focus groups investigating perspectives of supplement use were not universally convinced about the validity of health claims made on dietary supplements although this did not preclude their use (Meer et al., 2005). A review of the scientific literature to support probiotic product claims found sufficient evidence for 22 products from 249 trials reviewed and moderate to strong evidence for conditions such as antibiotic-associated diarrhea and diarrhea in children. However, many products lacked confirmatory analysis and the authors highlighted the strain and disease specific nature of probiotics including that the efficacy of a strain may differ according to whether it is taken to prevent or treat a condition (Sniffen et al., 2018).
Physicians are increasingly likely to encounter questions from their patients about probiotics, particularly in relation to gastrointestinal problems (Brinich et al., 2013). Almost half of Australian consumers report that they would only use probiotics if recommended by a clinician (Khalesi et al., 2021). Like consumers, clinicians hold positive attitudes about probiotics, with the majority of health practitioners believing they are safe and effective (Fijan et al., 2019; Van der Geest et al., 2020). However, there is limited knowledge among clinicians regarding the effects of probiotics on non-gastrointestinal conditions. While 80% of general practitioners in Europe had previously recommended probiotics to a patient, only 38% had ever recommended a probiotic for mental stress (Van der Geest et al., 2020). A survey of prescribing healthcare professionals found women and gastroenterologists were more likely to recommend probiotics, and this was more often done for bowel health or gastrointestinal problems. Patients often initiated discussion about probiotics and the choice of probiotic was often left to the patient (Draper et al., 2017). Clinicians are arguably more trained to evaluate scientific evidence behind claims and have easier access to the scientific literature than consumers. However, clinicians face a large amount of information both in the general media and scientific literature making it time-consuming to evaluate an expanding amount of information to determine which probiotic formulations are indicated for different health conditions (Khoruts et al., 2020).
Evaluation of health claims and attempts to synthesise the plethora of studies into guidelines to assist clinicians have largely focused on gastrointestinal disease and symptomatology where much of the research has traditionally been focused. Psychobiotics referring to the use of probiotics for mental health from the consumer perspective are less well understood, and there is a need to investigate mental health claims made on probiotic dietary supplements available over the counter and examine the evidence to support such claims.
Aim
The aim of this study was to evaluate the scientific evidence for health claims purporting to improve mental health made on probiotic supplement products available for consumer purchase.
Methods
Design
In a two-step process, probiotic products claiming mental health benefits were identified. A narrative review of the literature relevant to each product formulation was then undertaken.
Selection of probiotic products
Probiotic supplement products in capsule or tablet form were identified by searching online retail suppliers of supplements, supermarkets and probiotic retailers. The following search terms were used to capture products relevant to mental health: ‘mental health probiotics’, ‘mental wellbeing probiotics’, ‘mood probiotics’, ‘anxiety probiotics’, ‘depression probiotics’, ‘anti-depression probiotics’ and ‘stress probiotics’. Searches were conducted using Google.
Each supplement product was screened and included if the following inclusion criteria were met; the product was available for sale to New Zealand consumers, was in capsule or tablet form, included a specific claim related to the improvement of mental health (e.g., mood, anxiety or stress), and contained at least one listed probiotic strain.
The listed ingredients of each product were reviewed to identify the probiotic strains present. In instances where a product contained more than one probiotic strain, all strains present were recorded. In addition to the probiotics found in the product, the dosage in colony-forming units (CFU), other non-probiotic ingredients, and specific claims relating to mental health were recorded for each product. Fourteen products that met our inclusion criteria were identified from the search of online retailers. There were five distinct formulations used in these products. Table 1 shows details about the components of each product, and health claims pertaining to mental health that were made on the packaging or website of the product.
Probiotic products sold in New Zealand with mental health claims.
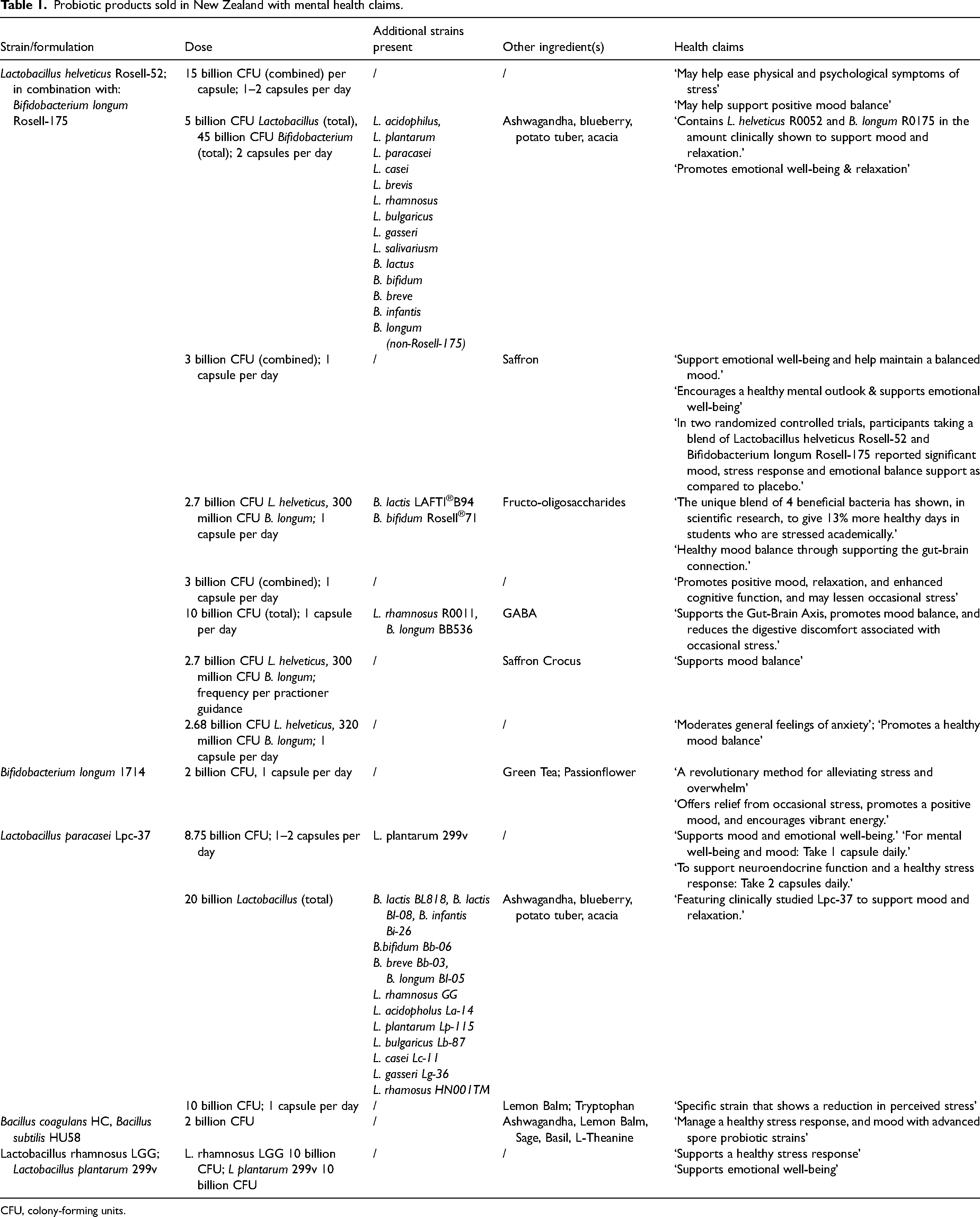
CFU, colony-forming units.
Review of supporting scientific evidence
A literature search was conducted to examine the relevant evidence to support the health claims made on each of the included products using Cochrane Central Register of Controlled Trials, PubMed and Google Scholar. Databases were searched from date of inception to December 15, 2023. Bibliographies of all relevant studies were reviewed to conduct a recursive search and identify any additional articles. Search terms included ‘mental health probiotic clinical trial’, ‘anxiety probiotic clinical trial’, ‘depression probiotic clinical trial’ and ‘stress probiotic clinical trial’. Additionally, specific strains listed in the supplements were included in search terms. Included studies were restricted to those that investigated the strain of probiotic alone or in the same combination as included in the product formulation. For example, if a strain included in one of the formulations was studied in combination with a different strain not used in the product formulation, the study was excluded because it is not possible to attribute results to the relevant strain. In some cases of multi-strain products, the label identified a single strain indicated for mental health improvement and the literature search and review focused on the specified strain.
Studies were included in the evaluation of evidence to support claims for the identified products if the study: reported results of a clinical trial in either animals or humans and included evaluation of a mental health outcome or biomarker of mental health as the primary outcome. Studies conducted in both populations of people with clinical diagnosis of a mental disorder, or healthy volunteers were included.
Studies without specific strain-level information were excluded. Evidence suggests that multi-strain probiotics may exert synergistic effects beyond the contribution of individual strains (McFarland, 2021; Su et al., 2020). Further exclusion criteria were: reviews, mechanism of action studies, safety studies, duplicate reports and trials with insufficient detail of the probiotic intervention.
Data extraction
Each study was reviewed and information capture followed a standard PICO (population, intervention, control, outcomes) procedure: (1) Patient population (healthy/diseased, age, gender), (2) Intervention (probiotic strain used, daily dose (CFU), duration, follow-up period), (3) Control (type of control, placebo or open, randomisation, blinding), (4) Outcomes (Outcome measure used, change in symptom, bio marker or psychological assessment scores, statistically significant or not).
Risk of bias
Risk of bias was determined following a standard Cochrane Risk of Bias 2 tool (RoB 2) for human studies (Higgins et al., 2019). For animal studies, the SYstematic Review Centre for Laboratory animal Experimentation (SYRCLE) Risk of Bias tool was used (Hooijmans et al., 2014). This assessment uses the same risk factors as the Cochrane tool adapted for use in animal studies. All five domains were considered: Domain 1: randomisation process, Domain 2: deviations from intended intervention, Domain 3: missing outcome data, Domain 4: outcome measurement, Domain 5: selection of the reported result. Following RoB 2 procedure, overall risk of bias was determined, and studies were categorised as either ‘high risk’, ‘some concerns’ or ‘low risk’. Studies in which all five domains are ‘low risk’ are considered low risk overall. Studies with one domain as ‘some concerns’ are judged ‘some concerns’ overall. Studies with multiple domains as ‘some concerns’ or one or more domains as ‘high risk’ are high-risk overall.
Results
The literature search identified 78 articles relating to probiotics and mental health conditions. After excluding duplicate reports on the same clinical trial (n = 3), review articles and studies where mental health was not the primary outcome (n = 49), pre-clinical mechanism of action or safety studies (n = 5), studies with insufficient description of the probiotic (n = 1) and strains not present in the products available to New Zealand consumers (n = 2), there were 18 articles that met the inclusion criteria. Table 2 summarises the evidence for each probiotic formulation from these 18 studies.
Published studies investigating probiotic strains and mental health outcomes separated by specific probiotics strains and/or combinations.
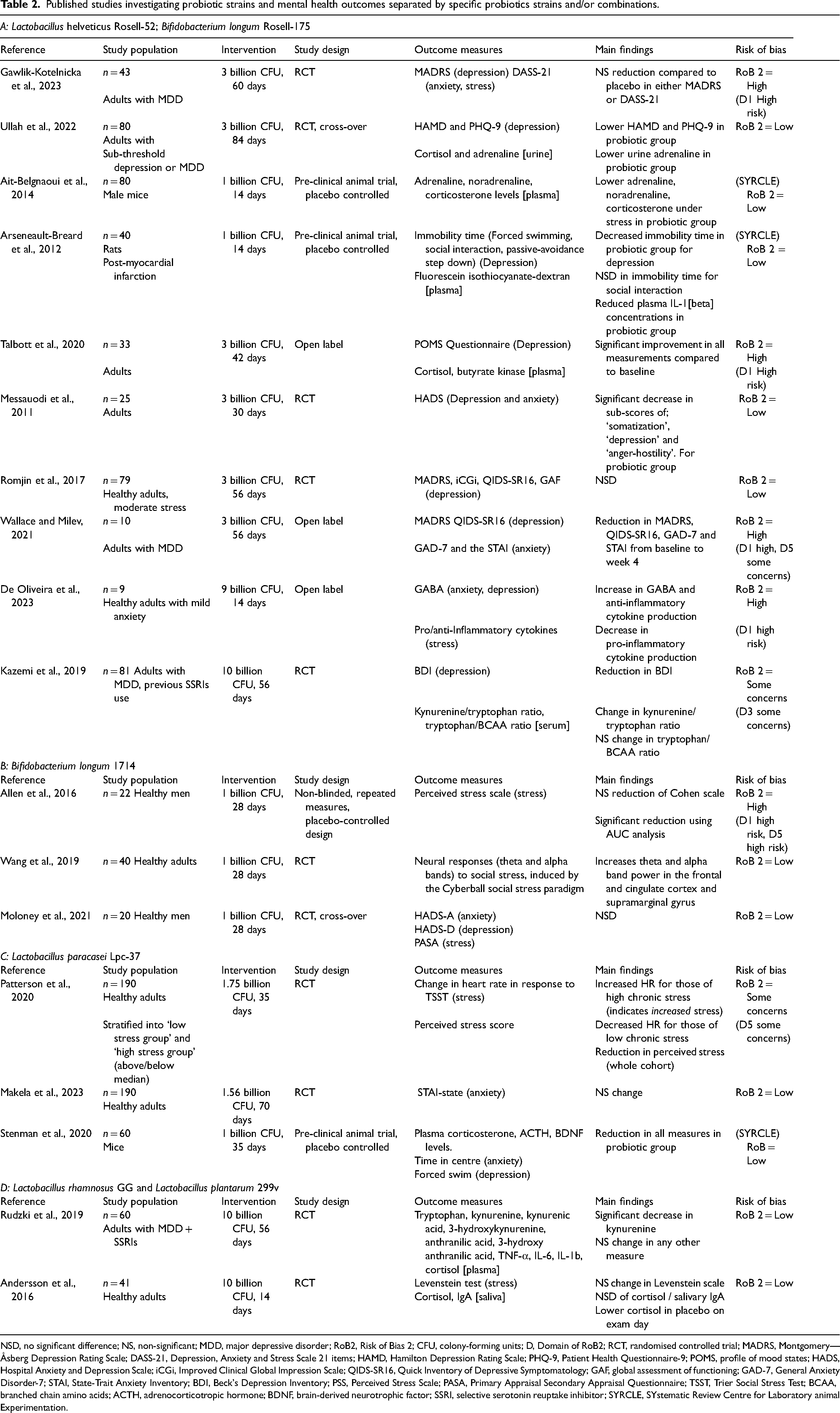
NSD, no significant difference; NS, non-significant; MDD, major depressive disorder; RoB2, Risk of Bias 2; CFU, colony-forming units; D, Domain of RoB2; RCT, randomised controlled trial; MADRS, Montgomery—Åsberg Depression Rating Scale; DASS-21, Depression, Anxiety and Stress Scale 21 items; HAMD, Hamilton Depression Rating Scale; PHQ-9, Patient Health Questionnaire-9; POMS, profile of mood states; HADS, Hospital Anxiety and Depression Scale; iCGi, Improved Clinical Global Impression Scale; QIDS-SR16, Quick Inventory of Depressive Symptomatology; GAF, global assessment of functioning; GAD-7, General Anxiety Disorder-7; STAI, State-Trait Anxiety Inventory; BDI, Beck's Depression Inventory; PSS, Perceived Stress Scale; PASA, Primary Appraisal Secondary Appraisal Questionnaire; TSST, Trier Social Stress Test; BCAA, branched chain amino acids; ACTH, adrenocorticotropic hormone; BDNF, brain-derived neurotrophic factor; SSRI, selective serotonin reuptake inhibitor; SYRCLE, SYstematic Review Centre for Laboratory animal Experimentation.
The literature search found a varied number of studies investigating each of the different strains. Lactobacillus helveticus Rosell-52® and Bifidobacterium longum Rosell-175® were always trialled together, and a total of 10 clinical trials investigating this combination met the inclusion criteria. Bifidobacterium longum 1714 and Lactobacillus paracasei Lpc-37® each had three clinical trials that met the inclusion criteria. Lactobacillus plantarum 299v® had two included clinical trials. Lactobacillus rhamnosus GG and Bacilus subtilis HU58/Bacilus coagulans HC did not have any studies that met all inclusion criteria.
Study characteristics
Overall, there was considerable heterogeneity in study sample size, design, population, probiotic dosage and intervention duration.
Study sample size ranged from 10 to 190 participants with most including between 25 and 75 subjects. Populations included animals (n = 3) (Ait-Belgnaoui et al., 2014; Arseneault-Breard et al., 2012; Stenman et al., 2020), healthy adults (n = 10) (Allen et al., 2016; Andersson et al., 2016; De Oliveira et al., 2023; Mäkelä et al., 2023; Messaoudi et al., 2011; Moloney et al., 2021; Patterson et al., 2020; Romijn et al., 2017; Talbott et al., 2020; Wang et al., 2019) and adults with a diagnosed mental health disorder (n = 5) (Gawlik-Kotelnicka et al., 2023; Kazemi et al., 2019; Rudzki et al., 2019; Ullah et al., 2022; Wallace and Milev, 2021). All human trials were conducted on adults. Most human trials (n = 13) involved both males and females, while two included only male participants. Of the studies in human participants, 11 were randomised, double-blind, placebo-controlled trials, three were open label and one study was randomised and placebo-controlled but not blinded. All animal trials were placebo-controlled and non-blinded.
Intervention periods ranged from 14 days to 84 days. Intervention dosages depended on the strain. For L. helveticus Rosell-52® and B. longum Rosell-175®, six studies used 2.7 billion CFU of L. helveticus and 0.3 billion CFU of B. longum Rosell®-17. Two studies tested a lower dosage (1 billion CFU total), and two studies tested a higher dosage. All studies on B. longum 1714 had a dosage of 1 billion CFU. All studies on L. paracasei Lpc-37® tested a dosage between 1 and 2 billion CFU. Both studies on L. plantarum 299v® had a dosage of 10 billion CFU.
The majority of studies were deemed low-risk of bias, following the Cochrane RoB 2 method. Eleven studies were ‘low-risk’, two had ‘some concerns’ and five were ‘high-risk’. Provision of funding or products for the research is not part of the RoB2 assessment process. Of the 18 studies, 13 were funded or had probiotics supplied by the manufacturer of the strain used in the trial. Five were independently funded.
Outcomes
Psychological outcomes were assessed in human studies using a combination of self-report questionnaires, responses to stress-inducing situations observed by researchers in research conditions, or physiological biomarker assays of metabolites implicated in stress response and mood. Primary mental health outcomes measured included depression, anxiety and stress either in isolation or combination. Of the 18 included studies, 12 reported a statistically significant change in one or more of the outcome measures in the intervention group compared to placebo. By strain, 8 of the 10 studies investigating the combination of L. helveticus Rosell-52® and B. longum Rosell-175® reported significant improvements in psychological outcome. Each of B. longum 1714. and L. paracasei Lpc-37® had two of three studies reporting significant improvements in mental health in the probiotic group. Neither of the two studies examining L. plantarum 299v® found significant group differences between the placebo and probiotic groups.
Table 3 outlines the number of studies that reported a significant finding in depression, anxiety or stress for each strain or formulation. Overall, studies that investigated L. helveticus Rosell-52® and B. longum Rosell-175® combined showed significant results in all three psychological outcomes of depression (n = 7 studies with significant results), anxiety (n = 2 studies with significant results) and stress (n = 1 study with significant results) (Table 3). For studies investigating L. paracasei Lpc-37®, B. longum 1714 or L. plantarum 299v®, significant findings were less common, being reported in one or two studies (Table 3).
Number of studies reporting a significant difference in outcome measure out of number of included studies by mental health outcome and probiotic strain.

Discussion
Variability in evidence to support mental health claims
We identified probiotic dietary supplement products making mental health claims and reviewed the scientific literature to evaluate support for these claims. Overall, the amount and quality of evidence to substantiate mental health claims made on probiotic products varied. Most of the strains had clinical trial results that underpinned the claim made. However, statistically significant results from clinical trials were not universally present for all strains. Study design factors including small sample size and choice of participant population, lack of mechanistic understanding of probiotic actions in the body, and the multiple translational steps between non-human and human studies explain why some studies fail to find evidence to support specific probiotic strains (Gibson et al., 2011). Our findings are consistent with existing literature suggesting that while certain probiotics can be beneficial for mental health, not all strains are effective (Le Morvan de Sequeira et al., 2022; Liu et al., 2018; Vasiliu, 2023).
A key finding is that the quantity of evidence is highly variable between strains. A review aiming to synthesise evidence to guide clinicians in which probiotics to recommend to their patients for different gastrointestinal symptoms also identified a variable quantity of evidence for different conditions (Sniffen et al., 2018). In our study, over half of the clinical trials that met the inclusion criteria were performed on one formulation, L. helveticus Rosell-52 and B. longum Rosell-175. Clinical trials examining the effect of this formulation on mental health are present in the literature from an earlier date than other strains. Researchers published the first trial of the Rosell combination for mental health in 2011 (Messaoudi et al., 2011). By contrast, B. longum 1714 and L. plantarum 299v® had clinical trials investigating mental health as the primary outcome published in 2016. The first study investigating L. paracasei Lpc-37® for mental health was published in 2020. Differences in publication date have possibly contributed to a time effect resulting in variation of study quantity. In the future, the effect of this lag time may diminish, allowing more accurate comparisons between formulations. The overall number of clinical trials was low. In total, 18 trials met the inclusion criteria across five formulations. The small number of studies limits the ability to compare strains. Previous perspectives on the probiotic regulatory environment have noted a trend for PubMed articles on probiotics to increase while the number of papers publishing results from human trials is decreasing (Freedman et al., 2020). Our results support the need for further clinical trials conducted in human populations to improve our understanding of probiotics for mental health (Liu et al., 2018; Misera et al., 2021; Morales-Torres et al., 2023; Ross, 2023).
Probiotic strain specificity in research and regulation
Our results support existing literature suggesting that the positive benefits of probiotics for mental health differ by strain and outcome measured. For example, of the included studies researching B. longum 1714 supplementation, positive effects on psychological stress were identified, but symptoms of depression were not significantly alleviated. In contrast, B. longum 175 coupled with L. helveticus 52 significantly reduced depressive symptoms. However, attributing this benefit solely to B. longum 175 is problematic given it was combined with L. helveticus 52. Literature suggests that B. longum alone has strain-specific effects (Jiang et al., 2021) and mental health improvements are strain-specific (Joseph and Law, 2019). The research supporting probiotics for gastrointestinal problems such as antibiotic-associated diarrhoea is also strain- and disease-specific (Sniffen et al., 2018). Meta-analyses or published reviews often pool probiotics of the same species but different strains, potentially obscuring the reader's ability to determine the evidence for a particular strain. The choice of a probiotic should be disease and strain-specific. There are differences in regulatory labelling requirements in different jurisdictions. The European Union Food Safety Authority (EU FSA) has stringent regulations regarding the health claims made on probiotic product labels, requiring extensive scientific evidence and pre-approval of product wording (Food Supplements Europe, 2021). As a result, most probiotic products in the EU cannot use the term ‘probiotic’ on the label as it implies a health benefit unless the EU FSA has approved specific claims. By comparison, The US Food and Drug Administration (FDA) allows broader use of ‘probiotics’ on labels with less regulatory scrutiny, provided the claims are general and include disclaimers. Medsafe, New Zealand's FDA equivalent, has regulations similar to those of the US FDA (Medsafe New Zealand, 2023). We reviewed probiotic products marketed to New Zealand consumers, and therefore, we are likely to have selected products with less stringent requirements for scientific evidence than products in the EU. These differences in regulatory standards for labelling may explain why some of the probiotic strains in the products we reviewed had limited scientific evidence behind them. The impact of regional differences in the regulation of the advertisement, sale and use of probiotics means that products can meet legal requirements. However, consumers may still find it difficult to appraise the level of evidence behind these claims.
We restricted our literature review to studies testing a single strain or the exact product formulation. Lactobacillus helveticus Rosell-52® and B. longum Rosell-175® were always combined in trials, allowing interpretation of results attributable to the same combination used in products marketed to consumers but making it difficult to isolate which strain or synergistic effect is responsible for reported benefits. Several of the products investigated in our study contained non-probiotic ingredients or other probiotics in addition to the specific strains referenced as conferring mental health benefits. Because evidence suggests that multi-strain probiotics may exert synergistic effects beyond the contribution of individual strains (McFarland, 2021), consumers and clinicians should be aware of the limitations of research specific to individual components of the formulation.
Heterogeneity in trial design limits conclusions.
Appraising evidence of health benefits is challenging, even when confined to assessing evidence for a specific strain (Khoruts et al., 2020). Differing sample populations, outcome measured, probiotic dose and the duration of intervention limit the accuracy of generalisable conclusions (Preidis et al., 2020). A panel of scientists recommended that supporting research should adhere to the following conditions to meet the regulatory standards for a claim to be approved by the European Union: trials should use a dose available in commercial products, the sample size should be adequate, duration should be appropriate for the end-point of the study, evaluation of multiple parameters should be minimised, participants should reflect the general population, detailed biological and genetic description of the probiotic strains used in studies should be the same as those in the product, multiple trials are present to provide supporting evidence, and studies should include biomarkers to investigate mechanisms of action (Gibson et al., 2011).
Ideally, evidence to support general well-being claims comes from clinical trials conducted in healthy populations (Britton et al., 2021; Gibson et al., 2011). Research in healthy volunteers requires large sample sizes because changes in well-being outcomes are likely to be smaller than in clinical populations. Of the studies included in our examination of support for specific mental health claims, the median sample size was 41 participants. This sample size is lower than is considered appropriate for claim approval; for example, a systematic review of the European Medicines Agency's licensing approvals found a median trial size of 113 participants in studies supporting approved claims (Djulbegovic et al., 2018).
Methodological considerations
Our method has some limitations. We conducted a narrative rather than systematic review of the probiotic literature. We excluded studies that did not include mental health as the primary outcome of interest, and studies that trialled strains in combination with other probiotics not present in the product formulations. Thus, we may have omitted evidence from secondary analyses that would support some of the health claims made. Secondly, we identified products sold to New Zealand consumers and therefore, other products available in different countries may have evidence to support mental health claims.
Our two-step process of first identifying formulations making claims about mental health improvement, followed by a review of literature specific to support the claims, contributes to understanding the relationship between marketing claims and scientific evidence. A strength of the study is the comprehensive search of multiple databases for studies pertinent to species and strain specificity rather than providing a review of the effect of probiotics more generally. A further strength of this review is the use of the risk of bias assessment.
Consumer perspectives on product claims
Probiotics are positively viewed by consumers and health practitioners (Fijan et al., 2019; Kozirok, 2021; Van der Geest et al., 2020). However, little is known about the level of scientific evidence consumers expect to be present to substantiate claims about mental health, and any assumptions consumers make about claims when choosing a probiotic. Awareness of the strain and dose-specific nature of probiotic effects may not be widely understood, suggesting the need for the construction of a multi-perspective independent group to evaluate the scientific evidence to support health and well-being claims (Britton et al., 2021). A similar framework was used to review probiotics for gastrointestinal conditions successfully (Preidis et al., 2020). The Gradings of Recommendations Assessment, Development and Evaluation system could be used by an expert interdisciplinary panel to guide the public and clinicians in choice of probiotics for mental health. This panel could assess the use of probiotics as adjunctive therapy, or as stand-alone use, as well as use in healthy populations for improvements in mental well-being and could be guided by the PICO (population, intervention, comparison, outcomes) format (Guyatt et al., 2011). This approach has previously been successfully implemented for probiotic use in several gastrointestinal conditions including IBS, ulcerative colitis, necrotizing enterocolitis and Crohn's Disease (Preidis et al., 2020). Guidelines aimed at bridging the gap between scientific research and consumer knowledge should be informed by an understanding of consumer interpretation of product claims.
Conclusions
The current evidence to support generalised claims that probiotics improve mental health is limited. The strain-specific nature of probiotics, heterogeneity in study design and differences in regulation of health claims on products, has resulted in a lack of clarity for consumers and clinicians attempting to evaluate whether a specific product can improve mental health. Further high-quality randomised controlled trials are required to advance understanding of probiotic benefits for psychological well-being. Synthesising available scientific evidence and translating it into a consumer-friendly ranking system could assist both clinicians and consumers in choosing probiotics for mental health.
Footnotes
Authors’ contributions
RS was involved in the manuscript concept and design, results interpretation, drafting, formatting and reviewing the manuscript. ND was involved in drafting, formatting and reviewing the manuscript. RD was involved in the identification of products, literature search and screening of articles and preparation of the first draft.
Availability of data and materials
All data collected is presented in the tables in the manuscript.
Consent for publication
All authors consent to publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
No data about, or collected from, humans or animals is used in this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding from the School of Medicine Faculty of Medical and Health Sciences, University of Auckland Summer Research Scholarship supported this work through funding for R.D. The funder had no role in the design, data collection, data analysis, and reporting of this study.
