Abstract
Keywords
Introduction
The World Health Organization (WHO) estimates that approximately 190 million children under five years of age are affected by vitamin A deficiency (VAD) (World Health Organization et al., 2011). Upward of 50% of children between the ages of 6 and 59 months are affected with VAD, which is still very common throughout most of sub-Saharan Africa (SSA). The highest rates of disability-adjusted life years and age-standardized incidence of VAD in 2019 were found in central, eastern and western SSA, with rates in western SSA being the highest at 15,571 per 100,000 and 37 per 100,000, respectively (Baker et al., 2023). Vitamin A is an essential nutrient for the human body and is crucial in maintaining the visual, physiological and immunological systems (Soares et al., 2019). The human body does not produce vitamin A, so daily intake is required to prevent any adverse health outcomes from micronutrient deficiency. It is essential for combating both communicable diseases, such as measles and respiratory infections, and non-communicable diseases, including certain types of cancer. Adequate levels of vitamin A are crucial for maintaining overall health and preventing disease (Soares et al., 2019; Underwood, 1994).
The vehicles for vitamin A programs can vary depending on the affected population. In some regions, where access to healthcare facilities is limited, or where populations are across remote areas, strategies such as campaigns may be used. Alternatively, Global efforts to prevent VAD have included vitamin A supplementation (VAS) programs that may be integrated into existing healthcare services, such as routine immunizations. Additionally, food fortification can serve as a complementary strategy to address VAD. VAS is a type of vitamin A program in which vitamin A is given to at-risk populations suffering from VAD by providing high-dose supplements, which are typically administered annually or bi-annually, for children aged 6–59 months (Dalmiya and Palmer, 2007). The WHO works with local governments to implement these high-dose capsules for children in this age group. In 2009, a WHO/United Nations Children's Fund (UNICEF) Steering Committee for Guidelines on VAS, which align with the WHO Essential Nutrition Actions (ENAs) was established. These evidence-based guidelines and recommendations for VAS programs are instrumental in helping local governments grasp the most effective practices and approaches for implementing such programs. For instance, WHO strongly recommends high-dose VAS in infants and children aged 6–59 months, particularly in regions where VAD poses a public health concern. This recommendation entails administering a dose of 100,100 IU of vitamin A to infants aged 6–11 months once and a dose of 200,000 IU to children aged 12–59 months every 4–6 months (World Health Organization et al., 2011).
In 2013, WHO classified VAD as a public health problem affecting about one-third of children aged 6–59 months, with the highest rates in SSA (48%) (UNICEF, 2023). In Africa, the gains of the past decades in promoting better vitamin A status, health and well-being of under-five children are being eroded. According to the UNICEF global nutrition database (2023), only 13 countries in West, Central, Eastern and Southern Africa had 80% or more coverage of VAS as of 2021. Coverage in countries like Nigeria, Zimbabwe, Gabon, the Democratic Republic of Congo and Botswana has declined over the past two to three years, as evidenced by the decrease in percentages from the figures representing the year 2017 to those of 2021: Nigeria (80% to 57%), Zimbabwe (44% to 16%), Gabon (33% to 27%), the Democratic Republic of Congo (74% to 56%) and Botswana (69% to 64%) (Figure 1). Without novel and appropriate strategies, this decline may continue considering the dwindling resource commitment to health across African countries and the development partners’ investment reduction. This underscores the urgent need for concerted efforts and commitment to combat VAD and ensure the optimal health and development of children in Africa.
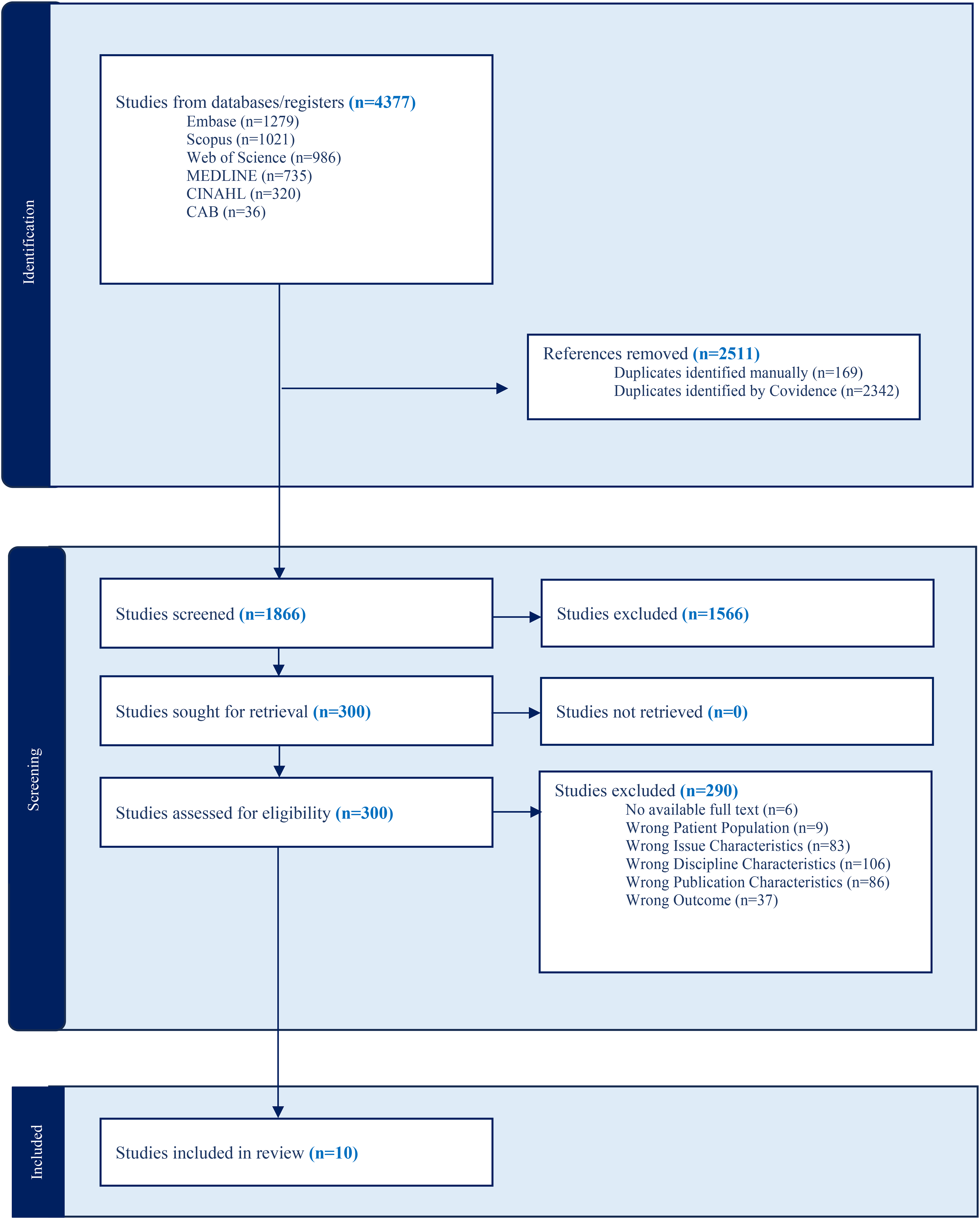
PRISMA diagram.
Evidence has shown disruption of the healthcare structures with limited access to and delivery of essential health services in SSA due to the COVID-19 pandemic (Assefa et al., 2021). In Guinea in 2020, the coverage of VAS was notably impacted by the pandemic. A significant proportion of caregivers reported that their children did not receive VAS due to the absence of health workers in their communities, indicating logistical constraints exacerbated by pandemic-related challenges. Similarly, Burkina Faso, Côte d’Ivoire, and Mali witnessed decreased accessibility during the pandemic, leading to diminished VAS coverage (Baker et al., 2023). With this significant decline, a sharp increase in the prevalence of VAD is imminent as various strategies addressing VAD seemingly perform far worse than VAS. For example, biofortification is a promising strategy adopting a food system approach to promote adequate intake of vitamin A through increasing pro-vitamin A content of commonly consumed staple foods. However, evidence from Nigeria showed that fewer than 10% of the population consumed biofortified vitamin A foods (NBS/UNICEF, 2022).
Communities suffering from weak health systems and infrastructure, such as distance from health facilities like those in Africa and South Asia, are unable to reach the most vulnerable groups, especially children (McLean et al., 2018). Sustainability is also an issue where countries face challenges maintaining long-term coverage rates due to limited funding and a lack of national planning (McLean et al., 2018). Furthermore, the critical role of adequate vitamin A nutrients in promoting immunity is well known; thus, there may be increased susceptibility to infections. This may be counterproductive as many of these countries are not fully recovered from the recent COVID pandemic. Preventing the impending health consequences of the declining VAS in SSA is urgent and vital.
Despite the strong recommendation of VAS programs as essential for children 6–59 months of age, and the significant scale up of VAS programs worldwide (Aghaji et al., 2019), there has been a continued drop in coverage and VAD is still very common in SSA (Baker et al., 2023). This review aims to understand the barriers and facilitators to VAS program implementation in Africa, specifically targeting strategies to counter the declining coverage and effectiveness. By illuminating these factors, the review seeks to pave the way for innovative approaches that can address the challenges and ensure the successful delivery of VAS programs.
Methods
This systematic review is presented according to the Preferred Reporting Items for Systematic Reviews and meta-analyses (PRISMA) 2020 checklist (Page et al., 2021). The protocol developed for this study has since been uploaded onto PROSPERO, the international prospective register for systematic reviews (CRD42023413057) (Ezezika et al., 2021).
Search strategy
A comprehensive search strategy was designed by a medical librarian (AM), alongside the research team (OE, OA, SQ) and included the following databases: Embase, Scopus, Web of Science, MEDLINE, CINAHL and CAB. Africa search filters developed by the University of Alberta were utilized (University of Alberta, 2023). The search was executed on 31 March 2023. No publication date limits were applied. The search strings used in each database are shown in Supplemental File 1 (S1).
Eligibility criteria
To be included in the review, studies needed to be in English-language, primary research articles, and needed to meet the following characteristics: report on VAS programs among children ages 6–59 months in Africa, only about high-dose vitamin A administered through capsules or droplets and discuss VAS programs through the perspective of public health and implementation science. We excluded articles if the study was not in Africa, and if they did not discuss VAS in the context of public health and implementation science among children ages 6–59 months. A summary of the eligibility criteria is presented in Table 1.
Summary of inclusion and exclusion criteria.

Study selection
Study screening was completed using Covidence, a systematic review management tool. First, all literature retrieved from the databases were imported into Covidence. After uploading all materials into Covidence, the automatic deduplication feature on Covidence removed all duplicates from the database searches. After this process, we underwent an abstract and title screening pilot test using the project's screening guidelines document to ensure consensus between team members. All article titles and abstracts were screened independently by two reviewers. Conflicts were resolved by a third author. Next, the full texts of each study were retrieved and imported into Covidence. Two reviewers were involved in full-text screening. We conducted another pilot testing of the inclusion and exclusion guidelines during the full-text screening stage on five articles to ensure consensus between team members. After a high level of reviewer agreement was reached, all full texts were screened independently by two individuals. During full-text screening, the screeners focused on ensuring that the studies were about barriers and facilitators of implementing the VAS program rather than adoption because the screeners had been over-inclusive during title and abstract screening on this criterion, and we wanted to ensure that the eligibility criteria were strictly followed. Following group discussions, a third author resolved conflicts between the reviewers. Each stage of our screening process can be found in our PRISMA diagram (Figure 1).
Quality assessment
We assessed all studies selected for data extraction using the Mixed Methods Appraisal Tool (MMAT). This tool is used for systematic reviews that involve a range of study types such as qualitative studies, randomized control trials, non-randomized studies, quantitative descriptive studies and mixed methods studies. Considering our systematic review involved various types of studies, the 2018 version of the MMAT was selected as a tool to assess the quality of each study. Included in the MMAT is a series of questions used to appraise the studies. Each study is first assessed using two screening questions regarding the clarity of their research questions and the appropriateness of the methods to address the research questions. Following the two screening questions, each study is assigned a study type (qualitative studies, quantitative randomized control trials, quantitative non-randomized studies, quantitative descriptive studies and mixed-methods studies), and then undergoes appraisal through five questions specific to their study type. Two authors individually appraised each study and then came together to reach consensus. Additional information about the tool and the outcomes of its application can be located in Supplemental File 2 (S2). This table encompasses the scores ranging from 0 to 4 for all 10 of our studies, classifying them as either low- or high-quality studies based on the MMAT.
Data extraction
Two reviewers independently performed data extraction for the included articles (n = 10). The template for data extraction captured the following items: Author, year, country, study design & methods, type of VAS, participant, age group, objectives, and barriers and facilitators to implementation of the intervention. The two reviewers met to reach consensus on the data independently extracted from the articles. After reaching consensus, the data extracted was verified by a third author. Supplemental File 3 provides descriptive objectives about each study compiled in a study characteristics table (S3). All data related to barriers and facilitators were compiled in a consensus document, which included the name of each barrier and facilitator extracted from each study, the sentence in which the barrier or facilitator was mentioned, and a longer excerpt from the study giving the barrier or facilitator more context.
CFIR application
The Consolidated Framework for Implementation Research (CFIR) is a conceptual framework created to guide the systematic assessment of factors that influence the implementation and effectiveness of interventions (Damschroder et al., 2022). The 2022 version of the CFIR was used to help organize and conceptualize the barriers and facilitators identified in the study and is a guide composed of five broad domains and 70 constructs, each associated with effective implementation (Damschroder et al., 2022). Each extracted facilitator and barrier was coded according to the CFIR framework. Incorporating the CFIR during the analysis phase of this review was beneficial, as integrating a conceptual framework increased the generalizability of the study results.
Two authors independently coded each facilitator and barrier according to the CFIR framework and then came together to identify and resolve any disagreements between the two sets of data that were coded. The third author was brought in to further resolve any remaining conflicts that the first two authors may have had when coding the collected data, by determining which CFIR code for each barrier and facilitator would be most appropriate based on the CFIR definitions. A consolidated document containing all relevant barriers and facilitators extracted from included articles with the associated CFIR constructs was developed.
Results
Type of study/methods
Various types of studies were used in the 10 articles included in this systematic review. Half of the studies were qualitative; one was a randomized control trial and four employed mixed methods. The studies that were classified as qualitative varied amongst each other in their data collection methods. The qualitative articles involved a combination of interviews (n = 2), structured questionnaires (n = 2), focus groups (n = 1), surveys (1) and statistic collection with follow-up supervision (n = 1). The one randomized control trial, randomized mothers to receive a mobile voice or text reminder on health facility attendance at five immunizations and VAS visits. The mixed methods studies employed a dual approach, integrating both qualitative and quantitative data to address their research objectives. In one study, quantitative data on costs and coverage were paired with qualitative insights into the delivery modalities of VAS, comparing mass campaigns to routine health services. Another study utilized a cross-sectional survey that combined both quantitative and qualitative data collection methods. Moreover, a different approach was taken, utilizing a combination of population-based sample surveys, administrative records and field observations. Additionally, a separate mixed-methods study gathered information through a diverse set of sources, including administrative data, interviews and focus groups.
Year of publication
Our search was designed to include articles from 1946 to present, as it marks the inception of several prominent databases like MEDLINE. This broader time range was chosen to ensure comprehensive coverage of any earlier relevant literature. There were no studies published between 1946 and 2002, which could be attributed to the fact that it was not until 2002 that South Africa implemented a national routine VAS programme for children aged 12–59 months (Coutsoudis et al., 2019). Given this, it is logical that the first article was published in 2002, paving the way for a steady rise in publications over the subsequent years with intermittent gaps. Following the first publication in 2002, two articles were published in 2007, followed by a four-year gap in the literature until 2011–2017 when three articles were published, and then 2018–2021 when three more articles were published.
Participants
The participants in several studies encompassed parents, caregivers, and both mothers and fathers of children aged 12–59 months receiving VAS (n = 6). One study included a group of participants comprising of health agents, community leaders, and local health and administrative authorities. Similarly, another study involved relevant officials at the regional health office, zonal health office and institution offices, and an additional study highlighted the involvement of people from various levels within the health system. Further studies focused on gathering statistics from children aged 6–59 months (n = 4) and incorporated administrative records from local government sources (n = 1). Notably, many articles featured a combination of populations as described above (n = 4).
Countries
The 10 studies included in the systematic review all focused on countries in Africa. Twelve countries in Africa were mentioned in the articles: Sierra Leone (n = 1), Mali (n = 1), Ethiopia (n = 3), Cote d’ivoire (n = 1), Zimbabwe (n = 2), South Africa (n = 1), Senegal (n = 1), Madagascar (n = 1), Tanzania (n = 1), Uganda (n = 1), Zambia (n = 1), and Burkina Faso (n = 1). All studies but one focused on only one country.
MMAT
The MMAT Qualitative Research Checklist was used to assess the quality of the studies included in the review and to categorize and identify methodological quality through a set of criteria. Supplemental File 2 (S2) provides the MMAT results for all 10 studies. Of the 10 studies included in the review, five adopted qualitative study designs. All five utilized appropriate qualitative approaches to answer the research question and had appropriate data collection methods to investigate aims of the research. Four studies utilized a mixed methods approach. Adequate rationale for using a mixed methods design was addressed by three studies to explore their research questions, whereas one study did not. Three studies adequately addressed the inconsistencies and divergencies in their results and adhered to the quality criteria of each tradition of the methods involved, while one study did not. One study utilized a randomized-controlled trial approach. The participants of the study adhered to the intervention and the study utilized appropriate randomization. Regarding the outcome data and the outcome assessors being blinded to the intervention, it was unclear in this study whether this was considered.
Study objectives of the selected papers
Across all 10 articles, several objectives emerged. The assessment of the factors that favour or limit VAS programs was cited in three studies. Two studies stated that their primary objective was to compare the cost and effectiveness of routine delivery of VAS.
Other study objectives included examining intersections between gender and VAS programs (n = 1), assessing the impact of mobile SMS reminder messages sent to mothers (n = 1), describing a VAS outreach strategy implemented in a remote rural area (n = 1), estimating the effect of Child Health Days (CHD) on public health intervention coverage (n = 1), and investigating factors associated with missed VAS coverage to evaluate the reliability of the intervention (n = 1).
Type of vitamin A supplementation
Across all 10 articles, several types of VAS emerged. Seven studies stated that their type of VAS were Routine (Bauck et al., 2021; Dissieka et al., 2019; Gatobu et al., 2017; Hendricks et al., 2007; Horton et al., 2018; Oliphant et al., 2010). Routine VAS programmes consisted of routine delivery of VAS, appointments at health facilities (Dissieka et al., 2019), CHD, routine visits by health care workers in rural areas (Bauck et al., 2021), routine delivery by community health services (Gatobu et al., 2017). In addition, two studies (Ayoya et al., 2007; Demissie et al., 2003) stated the use of vitamin A capsule programmes, in one article (Ayoya et al., 2007) the program consisted of supplementation of vitamin A capsules during national nutrition weeks, whereas in another article (Demissie et al., 2003), there was distribution and delivery of vitamin A capsules to health institutions in several districts. Lastly, one study (Dube et al., 2014) stated the use of an adapted VAS strategy, in this strategy VAS was implemented in a remote district within the national immunisation days in the district.
Barriers
We identified nine barriers across the nine articles: (1) Capsule stock-outs, (2) Poor healthcare worker attitude and service, (3) Lack of adequate health personnel, (4) Inadequate training of staff, (5) Lack of incentive for participants, (6) Lack of incentive for staff, (7) Lack of budget for transportation, (8) Limited resources (fuel and food supplies) and (9) Inadequate monitoring and evaluation mechanisms. A detailed chart that demonstrates where these barriers were derived from in the articles included is available in Supplemental File 4 (S4). A summary of the barriers is presented in Table 2.
Summary of barriers to VAS.

Capsule stock-outs was identified in three articles (Ayoya et al., 2007; Demissie et al., 2003; Horton, et al., 2018) and was a barrier to implementation of VAS programming in three countries (Mali, Senegal and Ethiopia) due to supply chain disruptions, poor forecasting and inadequate resource allocation, the capsules were out of stock and supply interruptions led to unplanned gaps in capsule distribution and a failure to provide children with the necessary supplementation. One article (Bauck et al., 2021) described Poor healthcare worker attitude and service as a barrier to VAS service delivery, as parents and Public Health Unit (PHU) staff in Sierra Leone emphasized that the attitude of workers, determined whether the parents would bring their children for VAS. Parents and PHU staff noted that disrespectful, indifferent or disengaged healthcare workers discouraged participation in VAS services. Lack of adequate health personnel was identified in one article (Demissie et al., 2003), as a barrier to VAS in Ethiopia, and was reported as one of the major reasons implementing VAS programs was difficult. The scarcity of trained workers resulted in low coverage as staff were struggling to provide services across vast, remote regions. Furthermore, Inadequate staff training emerged as a significant implementation hurdle, as highlighted in one study by Hendricks et al. (2007), causing complications in VAS program execution in South Africa. Insufficient training led to staff forgetting vital vitamin A protocols and neglecting record updates. Lack of incentive for participants was another barrier identified in one article (Bauck et al., 2021) as participants in Sierra Leone voiced their concern for lack of food demonstrations, which impeded on the implementation of VAS programming as it decreased program attendance. Conversely Lack of incentive for staff was another barrier identified in two articles (Horton, et al., 2018; Ouedraogo, et al., 2022), this discouraged workers in Ethiopia and Burkina Faso, as the lack of financial incentive failed to provide encouragement for their efforts. This lack of motivation led to reduced commitment and engagement in VAS campaigns. In addition, Lack of budget for transportation was a barrier identified in one article (Demissie et al., 2003), and described as creating challenges during collection and distribution vitamin A capsules in Ethiopia, especially in rural or remote areas that lacked accessible roads or transportation infrastructure. Limited resources (fuel and food supplies) was another barrier identified in one study (Dube et al., 2014), as staff in Zimbabwe faced challenges regarding leaving sites on time due to mainly fuel and food supplies, which resulted in loss of supplementation. Lastly, Inadequate and evaluation mechanisms (Demissie et al., 2003) was identified in one study in Ethiopia and described as essential program components that were minimal and unavailable, as none of the health institutions submitted reports and workers were left unsupervised.
Facilitators
We identified seven facilitators across the seven articles: (1) Advanced/mobile distribution strategies, (2) Locally organized mini campaigns, (3) Child Health Days, (4) Traditional communication channels, (5) Ensuring awareness amongst implementers and beneficiaries, (6) Message reminder, and (7) Involvement of Community-based Health Workers. A detailed chart that demonstrates where these facilitators were derived from in the articles included is available in Supplemental File 5 (S5). A summary of the facilitators can be found in Table 3.
Summary of facilitators of VAS.
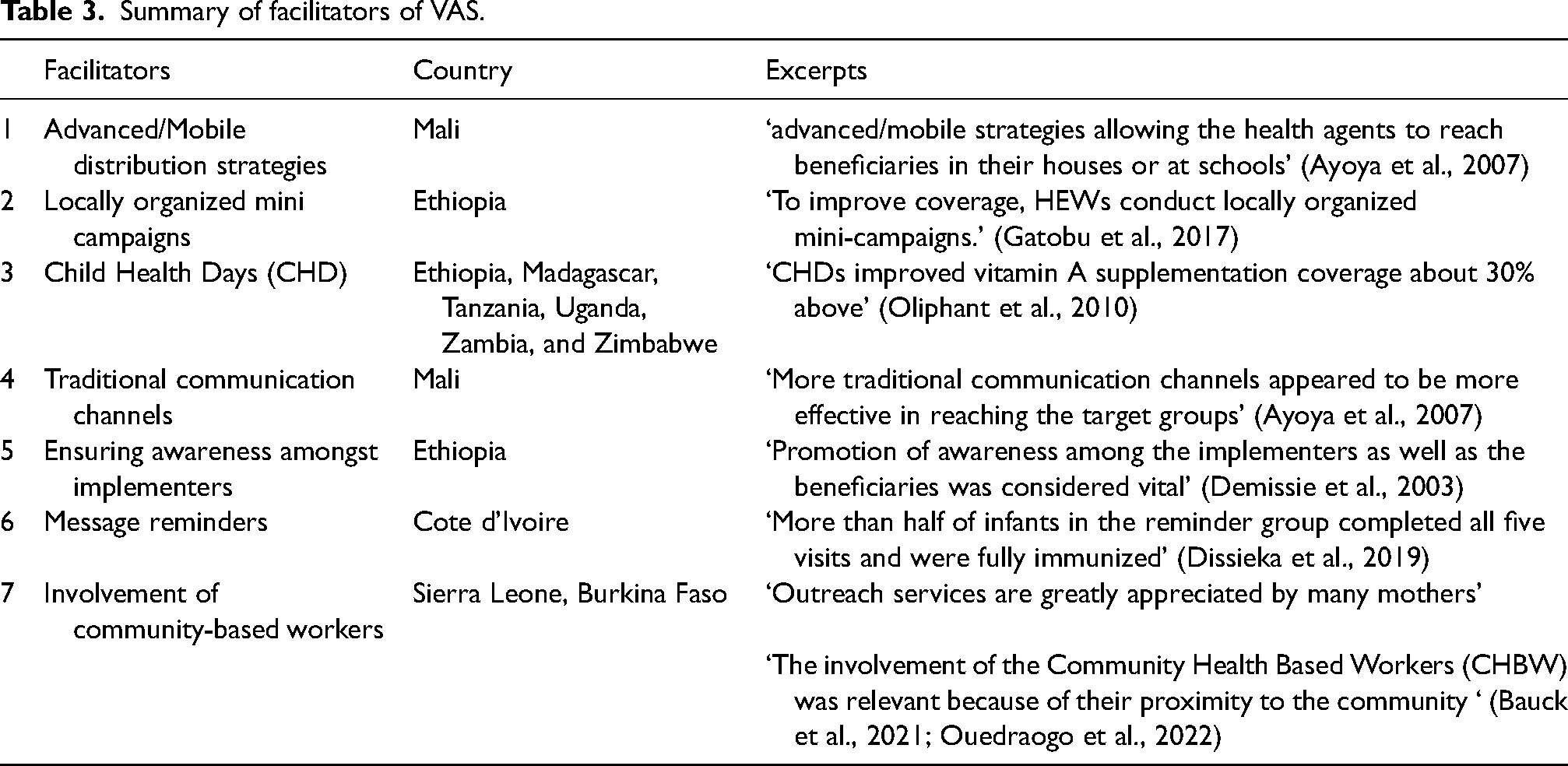
One study (Ayoya et al., 2007) in Mali, described advanced/mobile distribution strategies as a facilitator that allowed health agents to reach beneficiaries in their houses, or at schools in remote areas that did not have access to health facilities, this strategy resulted in an increase in the number of children receiving VAS. Locally organized mini campaigns was another facilitator cited in one article (Gatobu et al., 2017), this campaign in Ethiopia, was described as a hybrid approach in Vitamin A routine areas, it involves either house-to-house service provision or mini-campaigns at a local fixed point, effectively boosting coverage and accessibility. One study (Oliphant et al., 2010) described CHD as a facilitator in six countries (Ethiopia, Madagascar, Tanzania, Uganda, Zambia and Zimbabwe) stating that CHDs, which are designated days focused on delivering comprehensive health services to children, particularly in underserved and remote areas, improved VAS coverage about 50% prior to the previous method that was used in the target populations. One article (Ayoya et al., 2007) stated that more traditional communication channels (town criers, friends and family members) in Mali was a facilitator as it was more effective in reaching target groups than modern methods like radios and television. Ensuring awareness amongst implementers and beneficiaries was another facilitator that was cited in one article (Demissie et al., 2003) as promotion of awareness among implementers and educating them on VAS, was considered vital for the successful implementation of the VAS programme in Ethiopia, In addition, message reminder was a facilitator cited in one article (Dissieka et al., 2019) this facilitator boosted coverage of VAC in Cote d’Ivoire as caregivers of the child received voice message reminders prior to their child's visit. Lastly, two articles cited the facilitator Involvement of Community-based Health Workers (Bauck et al., 2021; Ouedraogo, et al., 2022), as involvement was greatly appreciated by caregivers in Sierra Leone, and the study by Ouedraogo, et al. (2022) stated that Community-based health workers involvement in Burkina Faso ‘removes another burden from health facilities’.
CFIR constructs
A total of 11 constructs out of 70 were found in this review. No facilitators or barriers were noted within the Innovation Characteristics and Outer Setting domain. In the Inner Setting, four studies (40%) identified Available Resources: Materials & Equipment. One study identified Available Resources: Funding, and two studies (20%) identified Incentive Systems as barriers, and one study identified Information Technology as a facilitator. In the Individuals—Characteristics domain, while no facilitators were identified, Innovation Deliverers, Implementation Team Members and Capability were noted as barriers in one study (10%) each. Lastly, in the Implementation Process domain, one study (10%) identified Reflecting and Evaluating-Implementation as a barrier, one study (10%) identified Engaging and two studies (20%) identified Assessing Needs: Innovation Deliverers as facilitators, and Tailoring Strategies emerged as a notable facilitator in three studies (30%), emphasizing the importance of tailored approaches in ensuring effective implementation strategies (Table 4).
Frequency table of cited Consolidated Framework for Implementation Research (CFIR) constructs.

Four constructs were identified within the Inner Setting domain. Available Resources, including Materials & Equipment (20%) and Funding (10%), emerged as a significant barrier across four studies (Ayoya et al., 2007; Demissie et al., 2003; Dube et al., 2014; Horton et al., 2018). These studies highlighted gaps in capsule stockouts and distribution, insufficient budget for necessary transportation, and limited resources such as fuel and food supplies, resulting in a loss of supplementation. Additionally, Incentive Systems were recognized as another barrier in two studies (Bauck et al., 2021; Horton et al., 2018; Ouedraogo et al., 2022), with the lack of food incentives for participants and financial incentives for staff negatively impacting VAS programs. Lastly, Information Technology Infrastructure emerged as a facilitator in one study (Dissieka et al., 2019), where SMS message reminders contributed to an increase in VAS coverage.
In the Individuals Characteristics domain, three key constructs were identified. One study (10%) highlighted Innovation Deliverers as a barrier, emphasizing the critical role of healthcare workers’ attitude and service in VAS delivery in Sierra Leone. Parents stressed the significance of feeling comfortable and acknowledged by the workers (Bauck et al., 2021). Another study (10%) identified Implementation Team Members as a barrier to VAS, citing the lack of adequate health personnel as a major obstacle in implementing the VAS program in Ethiopia (Demissie et al., 2003). Lastly, Capability was recognized by one study (10%) as a barrier, with staff facing inadequate training and struggling to follow protocols and maintain accurate records (Hendricks et al., 2007).
Lastly, four constructs were identified within the Implementation Process domain. Reflecting and Evaluating—Implementation was recognized as a barrier in one study (10%) due to inadequate monitoring and evaluation mechanisms for VAS programs in Ethiopia (Demissie et al., 2003). Engaging emerged as a facilitator in another study (10%), highlighting the importance of promoting awareness among implementers for successful implementation (Demissie et al., 2003). Additionally, Assessing Needs—Innovation Deliverers was identified as a facilitator by two studies (20%), emphasizing the crucial role of Community-based Health Workers in successful VAS program implementation through outreach services and alleviating the workload of health centres (Bauck et al., 2021; Ouedraogo et al., 2022). Lastly, Tailoring Strategies emerged as a facilitator in three studies (30%), involving distribution strategies, locally organized mini-campaigns, CHD and traditional communication channels to enhance VAS program effectiveness (Ayoya et al., 2007; Gatobu et al., 2017; Oliphant et al., 2010).
Discussion
This review identified multiple barriers and facilitators to implementing VAS across 10 articles published between 2003 and 2021. Our analysis was based on 12 countries in Africa, with Ethiopia and Zimbabwe having the most representation in available studies.
VAD remains a significant public health issue in Africa, particularly in low-income countries where access to nutrient-rich foods is limited. VAD is a leading cause of preventable childhood blindness and increases the risk of severe infections and death from common diseases like diarrhoea and measles (demissie et al., 2003). In SSA, VAD disproportionately affects children under five and pregnant women, contributing to weakened immune systems and higher mortality rates (Ayoya et al., 2007). Moreover, VAD is associated with stunted growth and poor developmental outcomes in children, making it a critical area for public health interventions such as VAS (Hendericks et al., 2007). Addressing VAD through targeted supplementation programs has been shown to reduce child mortality by 23% in regions with high deficiency levels (Bauck et al., 2021), highlighting the importance of maintaining adequate coverage in at-risk populations.
Several options exist to boost the implementation of VAS across Africa as derived from the barriers and facilitators identified in this review. First, it is essential to revamp the healthcare structure across various countries, which is related to lack of access to quality healthcare, such as adequate healthcare personnel and trained staff (Demissie et al., 2003; Hendricks et al., 2007). The sub-region is characterized by poor access to quality healthcare, a high doctor-to-patient ratio, a lack of access to infrastructure and many rural communities lacking basic health facilities (Bauck et al., 2021; Demissie et al., 2003; Dube et al., 2014; Hendricks et al., 2007). The infrastructural decadence and lack of a health workforce contribute to poor vitamin A service delivery. Anecdotal evidence shows the huge dependence on volunteers in the implementation of VAS in Nigeria, and often, the conduct of these volunteers affects the uptake of the program.
Second, the eligible articles in this study highlighted the shortage of vitamin A capsules that was underscored in three countries: Mali, Senegal and Ethiopia (Ayoya et al., 2007; Demissie et al., 2003; Horton, et al., 2018). Particularly in Ethiopia, funding emerged as a significant challenge, resulting in various barriers such as incentive systems and transportation issues. While outreaches in VAS campaigns have improved coverage and reached underserved populations, this approach contributes to poor patronage of health facilities and the use of routine health services suffers. Attempt to institutionalize VAS and make mothers to come to the health facilities for the program is to encourage the use of other routine health services. Improving access to essential health services, especially in rural areas, and ensuring an adequate workforce is available for sustainable implementation of critical nutrition and health services is highly recommended (Demissie et al., 2003; Falchetta et al., 2020; Faruk et al., 2020; McLean et al., 2020).
Third, studies across various African countries have identified factors influencing VAS, including poor maternal knowledge and fathers’ disapproval (Adamu and Muhammad, 2016; Kassa et al., 2020; Lartey and Armah, 2019). This underscores the critical need to intensify nutrition education and sensitization efforts regarding the health benefits of vitamin A and VAS. Such efforts are essential for conveying the advantages of VAS to children, parents, communities and governments alike, promoting its acceptance and uptake. Evidence from Ethiopia has shown that nutrition education can potentially increase the uptake of VAS in Africa (Berihun et al., 2023; Kassa et al., 2020). In Kenya, a study has identified increasing access to functional health facilities, expanding outreaches and campaigns, dispelling faith-related misconceptions and complementing nutrition literacy with innovative caregiver reminders as essential strategies to improve the uptake of VAS programs (Oiye et al., 2019).
Fourth, VAS can be further integrated into existing health programs like maternal newborn and child health weeks, measles campaigns and immunization plus days, as widely practiced in Nigeria. Although not explicitly addressed in the articles reviewed due to their broader focus, this approach leverages existing platforms to deliver additional health services to under-five children. In Angola, Chad, Cote d’Ivoire, South Sudan, Tanzania and Togo, VAS has successfully integrated into polio campaigns (Chehab et al., 2016; Haselow et al., 2022). Also, the integration of VAS into CHD increased VAS coverage in Ethiopia, Madagascar, Tanzania, Uganda, Zambia and Zimbabwe (Oliphant et al., 2010). Recently, VAS was also incorporated into the measles campaign in Nigeria. This serves as a facilitator as it may reduce the cost of implementation and leverage resources available in other programs to deliver additional health services to under-five children. This effort will also require strengthening collaboration with other health and non-health sectors in driving the VAS program.
Fifth, compared to the determinants of large-scale nutrition interventions in Africa (Ezezika et al., 2021), which had all CFIR domains represented, determinants for outer setting and Innovation domains were missing for the VAS, specifically, supportive policies and tailoring intervention to local needs. The absence of these facilitators for VAS might signal gaps in VAS implementation strategies that could be leveraged to improve VAS coverage. For example, supportive policies have been shown to facilitate implementation processes and enhance service delivery, and as a barrier, gaps in policy were shown to impact the scalability and sustainability of nutrition interventions (Ezezika et al., 2021).
Finally, the results emphasize the significance of tailored strategies and community involvement in enhancing the effectiveness of VAS programs. Recognizing context-specific barriers and utilizing facilitators like community-based health workers and traditional communication channels to improve program coverage and outcomes is crucial. Furthermore, differences in determinants between VAS and large-scale nutrition interventions highlight the necessity for targeted interventions that address the unique challenges and objectives of each program type. VAS, with its narrower focus on a specific nutritional intervention, required a more focused approach, targeting particular constructs relevant to its specific innovation and target population.
Limitations
This systematic review has several limitations and strengths that should be considered when interpreting the results. One limitation is some regions in Africa may have been underrepresented due to the limited availability of published data from certain countries. Additionally, the review only included academic journal articles, which may have excluded relevant insights from other publication types. The exclusion of non-English articles may also have resulted in a language bias, as valuable research published in other languages could have been overlooked. Moreover, the data collection for this study was conducted in early 2023. Since then, updated research and recent interventions may have been published that are not included in this review. This limits the study's ability to reflect the most current findings on VAS programs. However, this review also has notable strengths. It provides a comprehensive synthesis of barriers and facilitators to VAS programs across multiple African contexts, offering insights that are crucial for scaling an improving VAS intervention. Furthermore, the inclusion of both qualitative and quantitative studies enables a holistic understanding of challenges and opportunities in VAS implementation.
Conclusion
This review uncovered multiple barriers and facilitators organized around the CFIR framework to VAS programs in Africa. The findings of this review hold significant implications for informing the strategic planning and design of both new and existing VAS programs, as well as the development of implementation strategies specifically aimed at overcoming common barriers and capitalizing on established facilitators. The barriers and facilitators identified under the CFIR domains offer valuable insights into adapting VAS interventions to national and local settings. Moreover, they contribute to a nuanced understanding of the implementation and use of VAS in Africa. Nine barriers and seven facilitators were identified, ranging from issues such as capsule stock-outs and inadequate healthcare worker training to innovative distribution strategies and community involvement. These findings underscore the importance of addressing implementation challenges, including capsule stock outs, limited resources and inadequate staff training, while acknowledging the distinctive features and resources inherent in local contexts where VAS programs operate. Effective strategies entail not only addressing barriers like enhancing resource availability and improving healthcare worker training but also leveraging facilitators such as community involvement and innovative distribution channels. By adopting this approach, stakeholders can optimize the efficacy of VAS initiatives, recognizing that successful implementation hinges on overcoming barriers and leveraging existing local assets and opportunities to achieve maximum impact.
Supplemental Material
sj-docx-1-nah-10.1177_02601060241294133 - Supplemental material for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review
Supplemental material, sj-docx-1-nah-10.1177_02601060241294133 for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review by Obidimma Ezezika, Selina Quibrantar, Asua Okolie, Oluwaseun Ariyo and Alanna Marson in Nutrition and Health
Supplemental Material
sj-docx-2-nah-10.1177_02601060241294133 - Supplemental material for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review
Supplemental material, sj-docx-2-nah-10.1177_02601060241294133 for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review by Obidimma Ezezika, Selina Quibrantar, Asua Okolie, Oluwaseun Ariyo and Alanna Marson in Nutrition and Health
Supplemental Material
sj-docx-3-nah-10.1177_02601060241294133 - Supplemental material for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review
Supplemental material, sj-docx-3-nah-10.1177_02601060241294133 for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review by Obidimma Ezezika, Selina Quibrantar, Asua Okolie, Oluwaseun Ariyo and Alanna Marson in Nutrition and Health
Supplemental Material
sj-docx-4-nah-10.1177_02601060241294133 - Supplemental material for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review
Supplemental material, sj-docx-4-nah-10.1177_02601060241294133 for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review by Obidimma Ezezika, Selina Quibrantar, Asua Okolie, Oluwaseun Ariyo and Alanna Marson in Nutrition and Health
Supplemental Material
sj-docx-5-nah-10.1177_02601060241294133 - Supplemental material for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review
Supplemental material, sj-docx-5-nah-10.1177_02601060241294133 for Barriers and facilitators to the implementation of vitamin A supplementation programs in Africa: A systematic review by Obidimma Ezezika, Selina Quibrantar, Asua Okolie, Oluwaseun Ariyo and Alanna Marson in Nutrition and Health
Footnotes
Acknowledgements
The authors would like to thank Omer Choudhary and Sophie Paquet and for their support in piloting the data screening and extraction processes.
Author contributions
Study conception: OE. Study design: OE, SQ and AM. Analysis and interpretation of data: SQ and AO. Draft of the manuscript: OE, SQ and AO. Critical revision of the manuscript for important intellectual content: OE, SQ, AO, OA and AM. All authors read and approved the final manuscript.
Availability of data and materials
All data and related metadata underlying the findings reported are included in the submitted article and its supplementary information files.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding to OE from the Western Faculty Research Development Fund, Faculty of Health Sciences, Western University.
Trial registration
A protocol for this systematic review was developed and uploaded onto the PROSPERO—International prospective register of systematic reviews database (Registration number: CRD42023413057).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
