Abstract
Background:
Relapse and treatment dropout are key challenges in opioid use disorder that need effective intervention strategies. Motivation enhancement therapy may be effective in improving opioid use disorder treatment outcomes.
Objective:
To assess the effectiveness of group motivational enhancement therapy (GMET) in patients with opioid dependence.
Methods:
It is an open-label, two-arm, parallel-group, randomized controlled trial (RCT). Participants include patients with opioid dependence and on opioid maintenance treatment, aged between 18 and 60 years. The intervention consists of six GMET sessions, each lasting about 45 minutes. Assessments will be conducted at four time points—baseline, six weeks, four months, and six months—using the Kessler psychological distress scale (K10), Recovery and Relapse Prevention Inventory Diagnostic (RAPID), WHO Quality of Life-BREF, Harm Reduction Self-Efficacy Questionnaire and Stage of Change Readiness and Treatment Eagerness Scale for drugs (SOCRATES 8D).
Conclusion:
The customized GMET approach tailored to the specific needs of the local population is expected to enhance the effectiveness and feasibility of the intervention and its implementation.
Keywords
MET is an evidence based psychological intervention in the management of substance use disorder. In resource limited settings, GMET may be the answer to this challenge.Key Messages:
Opioids continue to be one of the major contributors to the global illness burden. They account for the majority of the drug overdose deaths. In 2021, 13.2 million individuals were reported to inject drugs, predominantly opioids. 1 Opioids constitute the drugs injected by more than 80% of the people. 2 In India, opioid consumption rates are three times the global average. 3 Also, there are remarkable regional variations in opioid use prevalence. Among people aged 10–75 years in different states, the overall opioid usage rates vary from 0.2% to 25.2%, whereas rates of opioid use disorders are between 0.1% and 6.9%. 4
A combination of physiological, behavioral, and cognitive symptoms because of the use of opioids characterizes opioid dependence. A key feature is the loss of control over its use. 5 Also, those who quit or reduce using opioids not only experience acute withdrawal symptoms but also intense craving for them. 6 The ensuing discomfort and other situational factors often result in opioid use lapses and relapses despite the associated negative consequences. 7 Opioid maintenance treatment is the treatment approach with the most evidence based for managing opioid dependence. 8 However, a major challenge is the limited effectiveness and high relapse rates with pharmacological intervention, especially if provided alone, resulting in relapse rates frequently exceeding 50%.9,10 Thus, it is combined with psychological interventions for enhanced effectiveness and adherence. 9
Motivational interviewing (MI) is an empirically supported psychological intervention that aids individuals in reducing or quitting their drug and alcohol use through the resolution of ambivalence for behavior change. 11 Motivational enhancement therapy (MET) can help a person remain calm and focused in the face of health, psychological, and social stress. When used in combination with necessary medications and traditional therapy methods, MET can be a helpful component of a complete treatment program. It is associated with improved treatment outcomes, including higher treatment retention. 12 A study with 89 participants misusing opioids reported group motivation enhancement therapy (GMET) combined with cognitive behavioral therapy sessions as highly helpful. 13 Another study with individual and group MI conducted on 90 and 79 patients on methadone maintenance treatment found both interventions to be equally effective in reducing average daily drug use. 14
MET is mostly delivered over multiple one-to-one sessions; however, owing to the scarcity of workforce in resource limited setting, GMET will be the answer to this challenge. MET carried out in a group might be the answer to this challenge. However, there are only a few group MET studies on the opioid-using population and none from India. Thus, there is a need for research assessing the feasibility and efficacy of MET delivered to patients with opioid dependence syndrome seeking treatment in the Indian setting. The present study aimed to determine the impact of GMET on treatment adherence, relapse rates, and quality of life in comparison to general counseling among patients with opioid dependence.
Methods
The study used an open-label, two-arm, randomized controlled trial (RCT) design. It will be conducted among patients with opioid dependence receiving daily supervised opioid substitution therapy (OST) from an OST clinic operating at a tertiary care center in north India.
The eligibility criteria for the participants will be as follows: (a) individuals with opioid dependence aged between 18 and 60 years; (b) newly registered in the OST clinic or those who have relapsed and reinitiated maintenance treatment within the past month, and (c) willing to provide informed consent. On the other hand, individuals with serious medical conditions that could have prevented study participation or hindered engagement in therapy and those with comorbid psychiatric illnesses or current substance dependence (excluding tobacco dependence) were excluded from the study.
Each patient’s participation in the study lasted six months. The study will compare the outcomes of the participants who received GMET with those who received general counseling. Participants of both genders will be included, and to reduce gender-sensitive concerns, separate formulations for the male and female groups will be created.
Sample Size
The sample size was estimated using the software G*power 3.1.9. Considering ANOVA repeated measures within-between interactions for two groups and four measurements, effect size (f) = 0.2, α error of 0.05, power = 0.95, and assuming correlation among repeated measures 0.5 and nonsphericity correction 1, the sample size was 56. We kept the sample size for the study at 64 to encompass the 10% attrition.
Description of Intervention for the Active Intervention Group and Control Group.
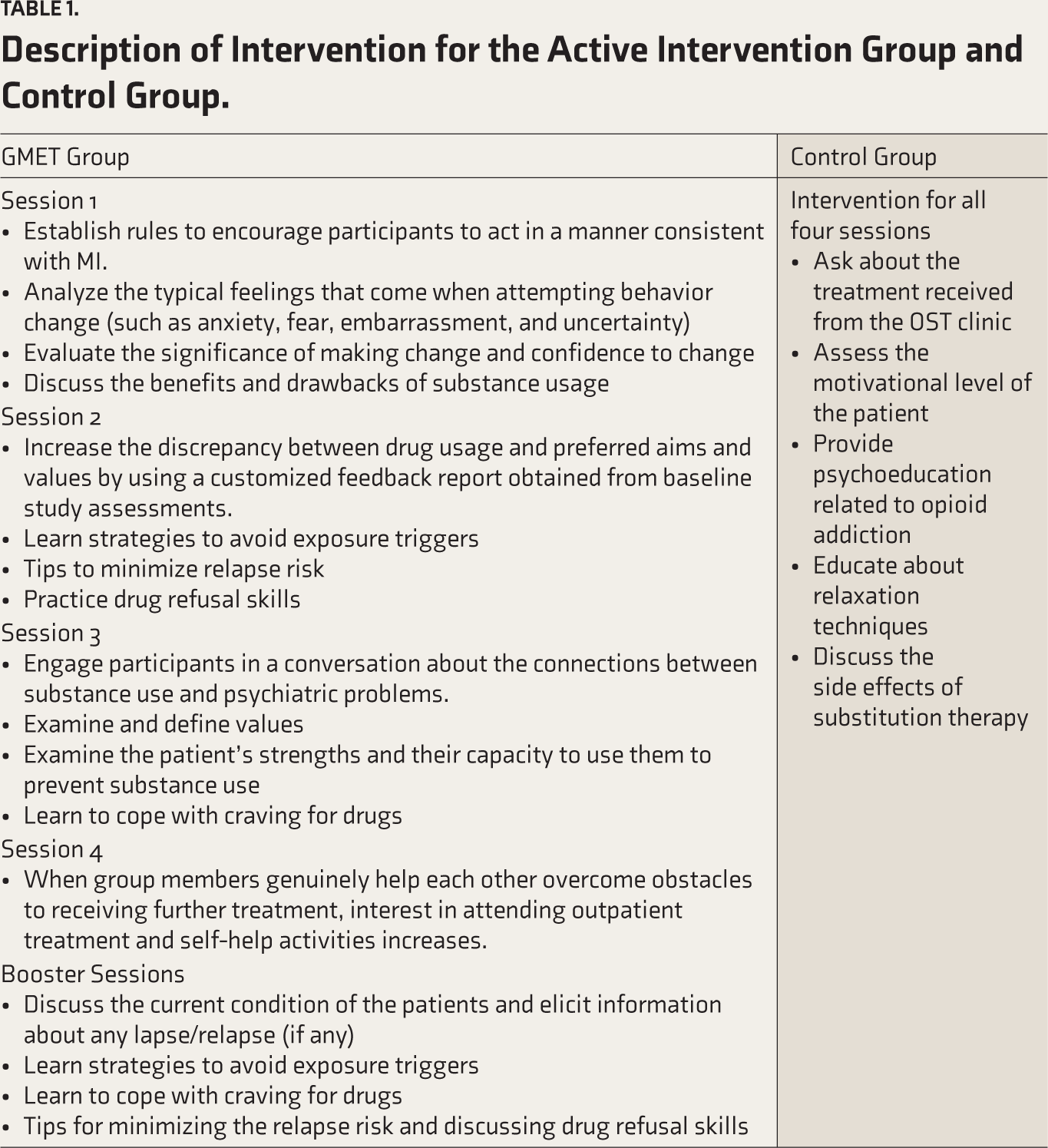
Intervention
Patients will be recruited in two arms: (a) the “control group” received treatment as usual consisting of opioid maintenance treatment and group counseling and (b) the “intervention group” (or GMET group) received opioid maintenance treatment along with GMET.
The GMET intervention consisted of four GMET sessions of about 45 minutes each. After the initial session at baseline (week 0), the subsequent sessions will be scheduled at the end of week 1, week 2, week 3, and week 4. Booster sessions will be conducted at the end of the second and third months. Reassessment of the participants using the various study tools will be done at the end of week 6, four months, and six months after enrolment in the study (Table 2). Participants will be required to attend a minimum of three GMET therapy sessions, or else they will be considered dropouts. Each session began with a summary of previous sessions and ended by summarizing the current session.
Intervention and Assessment Schedule During Group Motivational Enhancement Therapy.
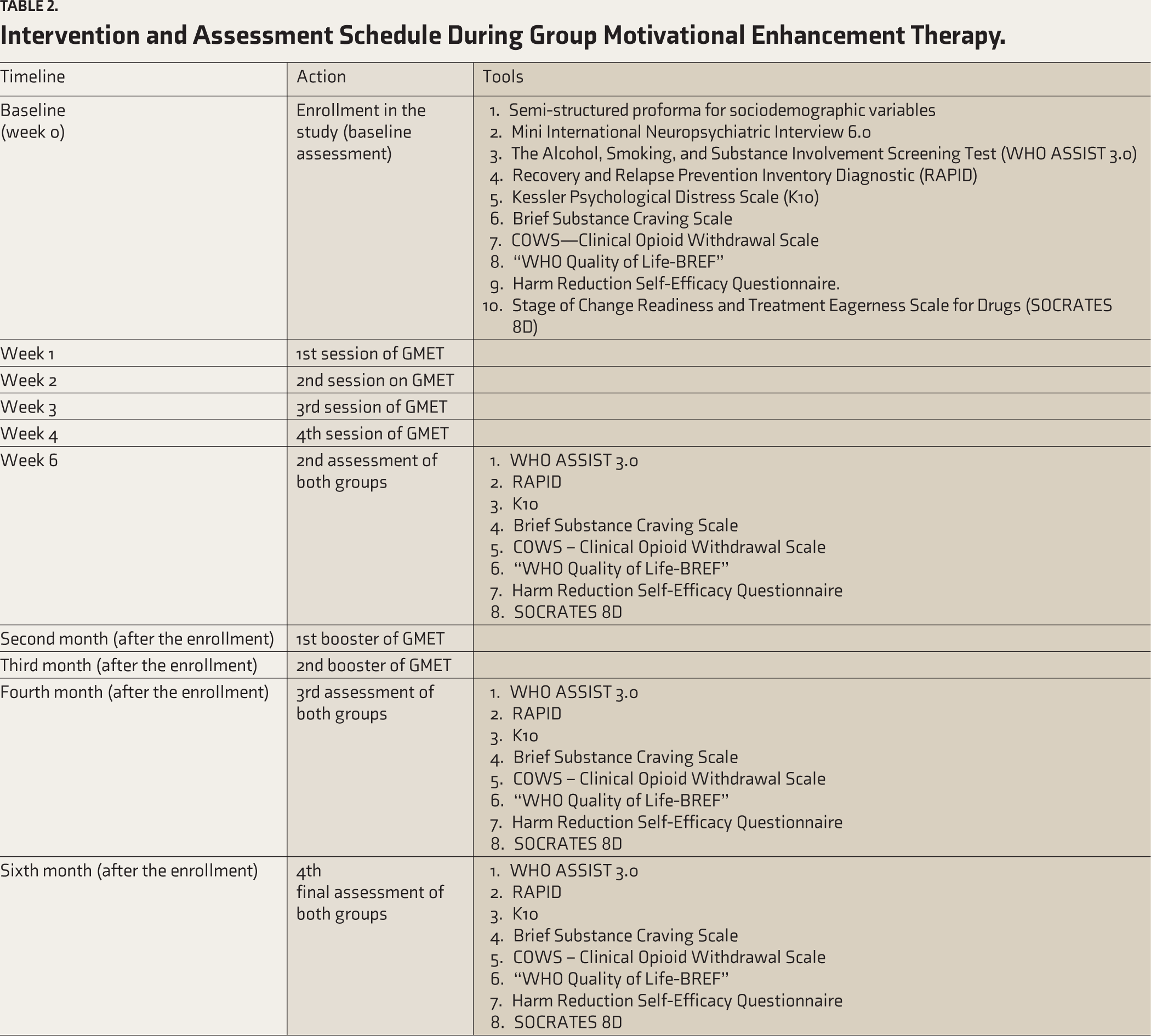
Study Instruments
A semi-structured proforma will be used to collect the sociodemographic (including age, gender, educational attainment, occupation, family’s monthly income, religion, area of living, religion, and type of family) and clinical data (including usage patterns of opioids and other substances, related harms, history of other illnesses, findings of physical and mental status examination, and treatment details). Other tools used for screening and assessment include the following:
The Mini-International Neuropsychiatric Interview (MINI 6.0) 15 : This structured interview schedule screens and diagnoses the major psychiatric disorders listed in the Diagnostic and Statistical Manual of Mental Disorders, fourth edition and International Statistical Classification of Diseases and Related Health Problems, 10th revision. It comprises 16 modules, each corresponding to a different diagnostic category. The schedule will be utilized in the study to identify psychiatric comorbidities.
WHO Alcohol, Smoking, and Substance Involvement Screening Test (WHO ASSIST 3.0) 16 : The Alcohol, Smoking, and Substance Involvement Screening Test (ASSIST) was created by an international team of researchers and clinicians for the World Health Organization (WHO). It is a technical tool that helps identify substance use disorders and health risks associated with substance use in primary health care, general medical care, and other settings.
Clinical Opioid Withdrawal Scale 17 : There are 11 items on the Clinical Opiate Withdrawal Scale (COWS). This tool is used to rate the severity of typical opiate withdrawal symptoms.
Stage of Change Readiness and Treatment Eagerness Scale for Drugs (SOCRATES 8D) 18 : This tool provides information on drug users’ motivation to enter treatment. The instrument consists of 19 items and yields 3 factorially derived scale scores: Recognition (Re), Ambivalence (Am), and Taking Steps (Ts).
Recovery and Relapse Prevention Inventory Diagnostic (RAPID) 19 : This is a valuable tool for assessing one’s recovery progress and potential for relapse. It is based on the “five rules of recovery” and involves a series of questions. A score above 70 indicates a low risk of relapse at present. Scores between 50 and 70 are typical during early recovery. Conversely, a score below 50 suggests a higher risk of relapse.
Kessler Psychological Distress Scale (K10) 20 : This 10-item survey asks about the anxiety and depression symptoms a person has encountered in the last four weeks to produce a general measure of psychological distress. Each item is scored from 1 “None of the time” to 5 “All of the time.” It has a Cronbach a of .909.
Brief Substance Craving Scale 21 : The Brief Substance Craving Scale is a 16-item self-report tool that treats drug cravings over a day. It has a reliability of 0.798. On a 5-point Likert scale, desire frequency and intensity are noted.
WHO Quality of Life-BREF (WHOQOL-BREF) 22 : This 26-item questionnaire assesses an individual’s quality of life. The items are rated on a scale of 1–5, with higher scores indicating a better quality of life. It has a Cronbach a of .948.
Harm Reduction Self-Efficacy Questionnaire 23 : This is a self-report questionnaire to determine how confident drug users are in their ability to adopt coping mechanisms to lessen the biological and psychological effects of injecting drugs in high-risk situations. The items are rated on a Likert scale of 0 (not at all confident) to 10 (very confident). This questionnaire has a Cronbach a of .779.
All tools were translated into Hindi, except WHO ASSIST 3.0 and WHOQOL-BREF (used Hindi version).
Validation of Tools
The questionnaires were translated from English to Hindi by qualified translators. A proficient bilingual performed the back translation. Five subject matter experts were consulted for formal validation of the tools, after which the tools were corrected and sent again for approval. The forward translation was then revised, and its grammar and spelling were reviewed. Subsequently, an advance copy of the Hindi questionnaire was made available, and a final version was developed following the authors’ acceptance.
Expected Outcomes
Primary outcome: GMET will lead to a reduction in improvement in treatment adherence and relapse rates.
Secondary outcomes: GMET will lead to improvement in recovery and relapse scores (assessed using RAPID), harm reduction self-efficacy (assessed using the harm reduction self-efficacy questionnaire), quality of life (assessed using WHOQOL-BREF), and reduction in psychological distress (assessed using K10).
Research Ethics Approval, Protocol Amendments, and Consent
The Institutional Ethical Committee approved this trial. The principal investigator used trial information materials to inform participants about the contents of this research before their participation. The researcher briefly described the possible risks and advantages of study participation before gaining informed permission. The researchers also notified individuals that their participation was entirely voluntary and might be terminated at any moment. It was made clear that when a subject would agree to participate in the trial, informed consent would be acquired before offering any intervention.
Procedure
All the opioid-dependent patients fulfilling the selection criteria and providing written informed consent will be enrolled in the study. Using a computer-generated permuted block randomization procedure, the participants will be randomly allocated to either the “intervention group” or the “control group.” The allocation will be done using sequentially numbered, opaque, sealed envelopes containing group information. A third party not engaged in this study will produce the randomization list and prepared those envelopes. Owing to the nature of the psychological intervention, the investigator will be aware of the type of intervention given to the participants. The Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) template (Table 1) displays the trial timeline and Consolidated Standards of Reporting Trials (CONSORT) diagram (Figure 1) the flow of participants during the trial. The data collected will be only accessible to those who will be authorized by the lead investigator and will be verified by one of the co-authors for accuracy and completeness.
CONSORT Flow Diagram.
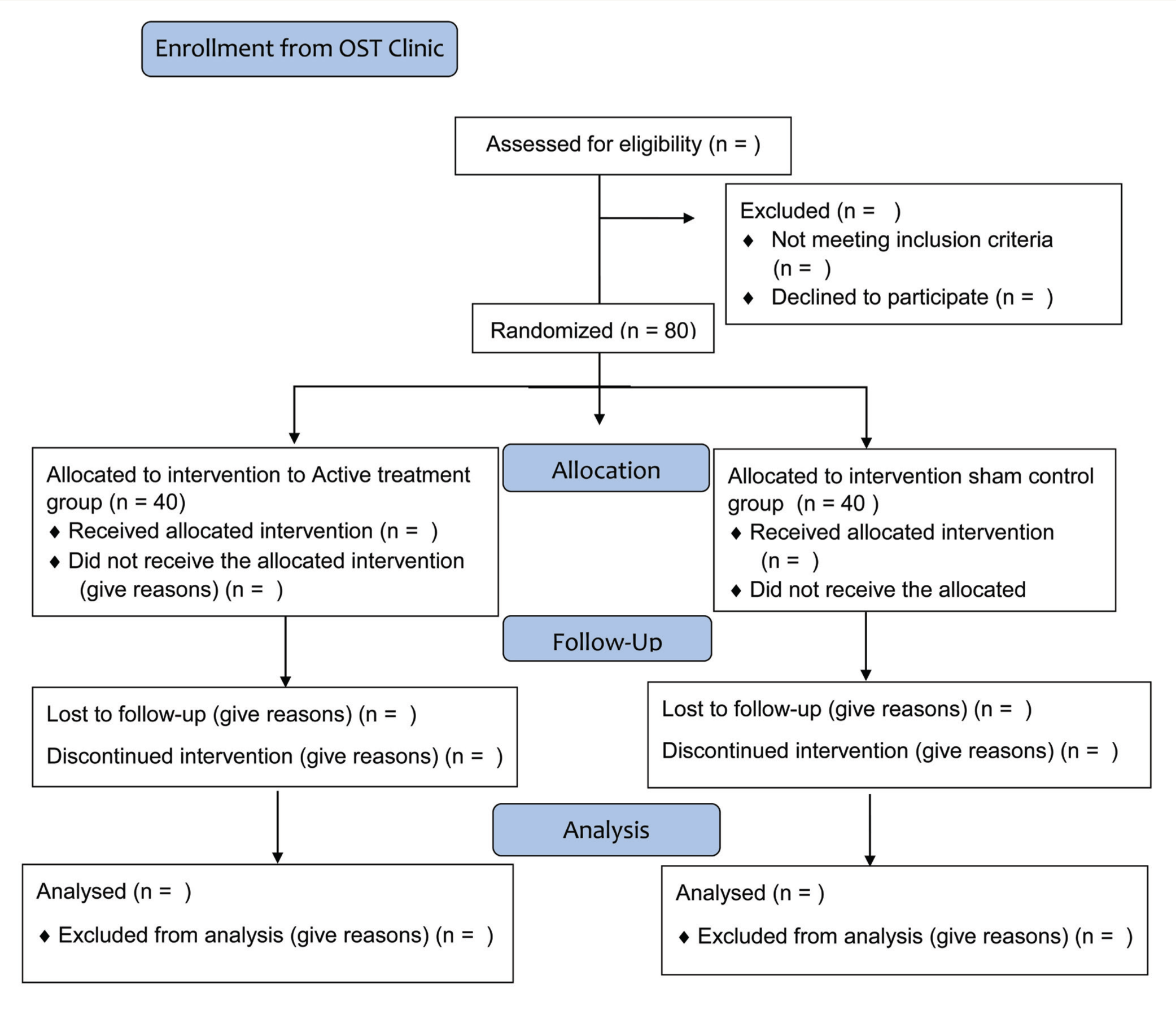
Additionally, regular meetings will be conducted to discuss concerns about trial implementation. The results will be analyzed after data collection. Each participant’s data will be kept for up to five years following the last evaluation.
Confidentiality
The principal investigator securely maintained all study-related data. Each participant’s documentation will be kept separately. The electronic database was stored in a password-protected Excel file, which will be stored together with the survey materials. The electronic database did not contain any information revealing a participant’s identity. Data entry and statistical analysis will be done on a computer that was not linked to the Internet.
Support Services and Follow-up Care
A month after participation will be ended, participants were still able to get in touch with the researchers during the trial period.
Statistical Methods
Univariate statistical tests for clinical variables will be employed using data obtained from all assigned participants. A repeated measures ANOVA will be used to compare the groups, and Pearson’s/Spearman test will be used to analyze the association. A P value of <.05 was used to indicate statistical significance. Data recording will be done on Microsoft Excel, and analysis will be done using the SPSS, version 25. 24
Discussion
Individuals grappling with opioid use disorders face a plethora of challenges that can have detrimental effects on their physical, social, and psychological well-being. Identifying and adapting evidence-based management solutions to the loco-regional context is crucial for enhanced effectiveness. Currently, psychosocial interventions are integral to every comprehensive treatment program for substance use disorders and are recommended by most of the standard treatment guidelines.25,26 Low or diminishing motivation for treatment often results in dropout and relapse. Motivation enhancement therapy aims to target an individual’s motivation to make changes in substance use behavior and adhere to recommended treatment. This, in turn, results in reduced relapses and improves the treatment outcomes.
In developing countries like India, where there is an existing large demand and supply gap for psychologists and trained individuals offering psychological interventions, individual therapy can be given to a limited proportion of patients. Conducting GMET can be a solution to overcome the limitations of resources while still providing effective treatment. However, there is a paucity of evidence regarding the feasibility and efficacy of GMET in developing countries. Thus, to address the treatment needs of patients with opioid dependence, in this study, we attempted to assess the efficacy of GMET in improving treatment adherence, reducing relapse rates, and enhancing the quality of life.
Relapse is a multifactorial phenomenon influenced by various intrapsychic and interpersonal factors. 27 Motivation is a key intrapersonal determinant influencing adherence to the treatment. 19 It can influence the relapse process in two different ways: by driving positive behavior change and by getting rid of maladaptive behavior. These factors are taken into account in the manualized GMET conducted over multiple sessions. As motivation is a dynamic phenomenon and may fluctuate according to psychosocial circumstances, continuous tracking of motivation levels and enhancement measures is important. Thus, we planned booster sessions of GMET in our study.
Conclusion
Evidence in support of GMET will guide clinicians to deliver the therapy effectively in patients with opioid use disorders and other substance use disorders. To the best of the authors’ knowledge, this study will be the first to explore the utility of GMET intervention in opioid use disorder in India. GMET appears to be a feasible and effective approach for the management of opioid use disorder in settings where resources are scarce. This protocol can be customized as per loco-regional circumstances to generate more evidence regarding GMET for opioid dependence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
None used.
Ethical Approval
Ethical approval was taken from the Institutional Ethical Committee at King George’s Medical University No. 1198/Ethics/2022, dated: December 31, 2022, ref. code:111TH ECM IIB Ph.D/P2.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent will be taken from all the participants at the time of data collection.
Trial registration
Clinical Trials Registry - India (CTRI): CTRI/ 2022/12/048618 on December 30, 2022.
