Abstract
Purpose of the Review:
Schizophrenia and bipolar disorder are understood to have neuroinflammatory/neuro-immunological basis in their etiopathogenesis. There are few studies synthesizing the association of schizophrenia and bipolar disorder in type 1 diabetes mellitus (T1DM), a common immunological disorder.
Collection and Analysis of Data:
We performed meta-analyses of studies assessing the prevalence and risk of schizophrenia and related disorders and bipolar disorder in individuals with T1DM. Fifteen studies consisting of a total sample of 9,768,028 (T1DM: 435,553; non-T1DM controls: 9,332,475) were included. Random-effects meta-analyses using the restricted maximum likelihood method for pooling logit transformed prevalence values and the Mantel–Haenszel test for pooling risk ratios were used.
Results:
Pooled (transformed-back-transformed) prevalence for schizophrenia and related psychotic disorders was 0.37% (95%CI: 0.19–0.73), and for bipolar disorder it was 0.39% (95%CI: 0.05–2.99) (together: 0.38% (95%CI: 0.2–0.71)] in T1DM. The prevalence models showed significant heterogeneity but were statistically significant, had low publication bias, and survived sensitivity analysis. The pooled risk ratio for schizophrenia and related disorders together with bipolar disorder was 1.80 (95%CI: 0.64–5.03), and for schizophrenia and related disorders alone it was 1.19 (95%CI: 0.46–3.11), indicating higher rates of these disorders in T1DM. The pooled risk ratios were not statistically significant and did not survive sensitivity analysis. Trial sequential analysis suggested the need for more studies to confirm increased risk.
Conclusion:
With available studies, we could not provide convincing evidence for the hypothesis that the prevalence and risk of schizophrenia and related disorders and bipolar disorder are significantly greater in individuals with T1DM.
Keywords
Schizophrenia and bipolar disorder, both severe mental illnesses, are now being hypothesized as neuroinflammatory or, more broadly, neuro-immunological disorders.1–2 Intriguingly, autoimmune disorders, in general, are known to increase the risk of these disorders. 3 The neuro-immunological damage in schizophrenia and bipolar disorder is associated with an altered innate immunity, 4 which is characteristic of autoimmune disorders like type 1 diabetes mellitus (T1DM). 5 Moreover, insulin deficiency in T1DM, caused by immune/inflammation-mediated damage, 5 is shown to lead to abnormal signaling in insulin-modulated brain neurotransmitter systems, which is in turn associated with schizophrenia and bipolar disorder pathophysiology. 6
While T1DM-associated autoantibodies have not been found to confer the risk of schizophrenia, there has been inconsistency across epidemiological studies in showing the association between T1DM and schizophrenia. 7 Very recently, meta-analytic studies have found that the prevalence of many mental disorders, in particular depression, was greater in patients with T1DM.8–9 However, there are no studies synthesizing the association of schizophrenia and bipolar disorder in T1DM. We aimed to conduct a systematic review and meta-analysis to determine the prevalence rates and risk of schizophrenia and bipolar disorder in T1DM. We hypothesize that the risk of schizophrenia and bipolar disorder is greater in patients with T1DM compared to controls (i.e., risk ratio >1) and that the prevalence rates in T1DM are greater than the global general population rates, which are about 0.3% and 0.5% for schizophrenia and bipolar disorder, respectively, according to a latest estimate. 10
Methods
The present study was conducted as per the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. The Meta-analyses Of Observational Studies in Epidemiology (MOOSE) checklist (Supplementary Table 1) was used to inspect compliance with it. As this was a synthesis of data available in the literature, no patient consent and ethics committee approval were sought.
The Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Studies Reporting Prevalence Data.
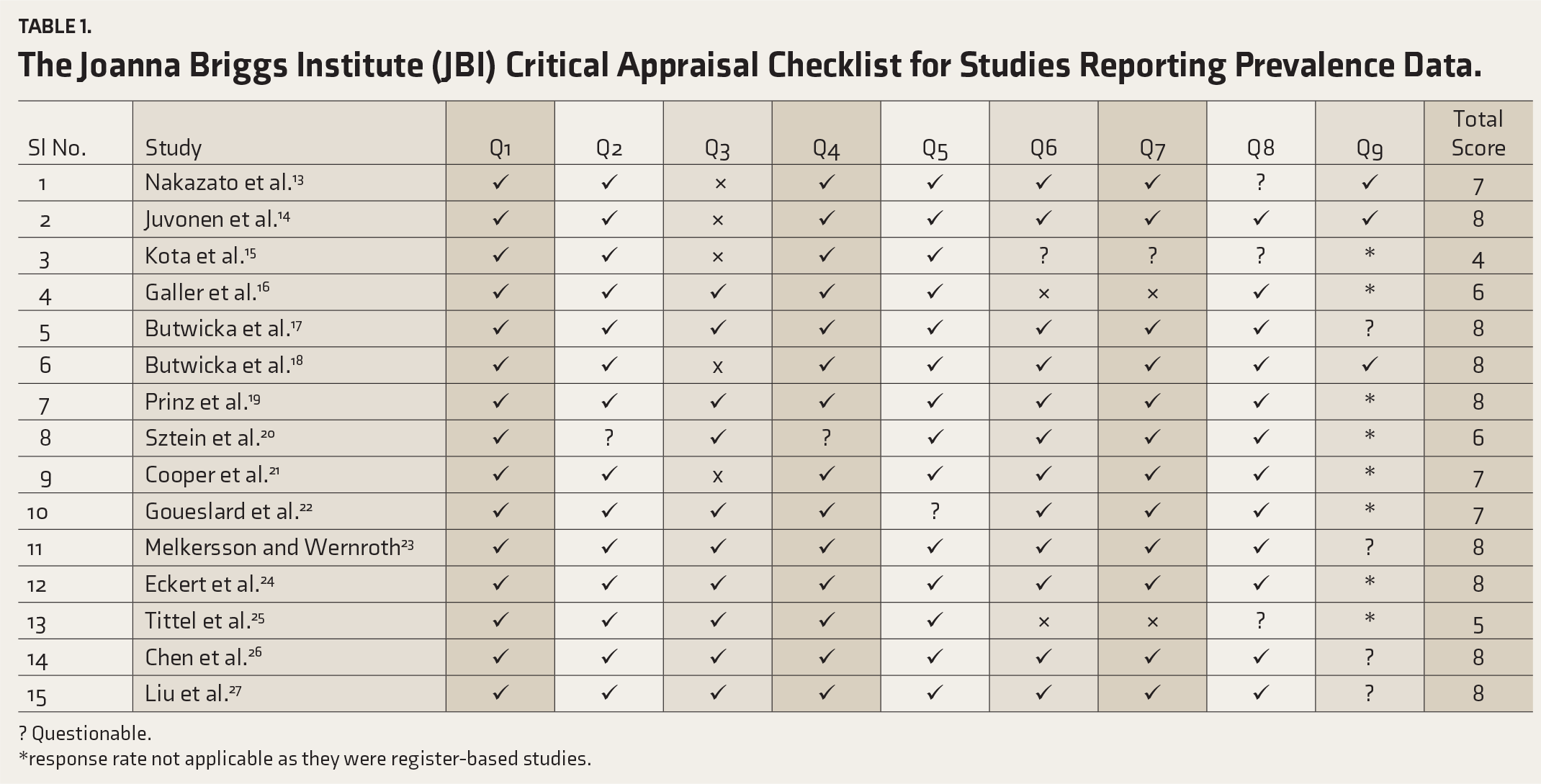
? Questionable.
*response rate not applicable as they were register-based studies.
Eligibility Criteria
The following inclusion criteria for studies were used: a diagnosis of T1DM made in childhood/adolescence; a diagnosis of schizophrenia or related psychotic disorders or bipolar disorder based on standardized diagnostic criteria or standardized diagnostic tools; studies reporting rates of schizophrenia or related psychotic disorders or bipolar disorder in either the group of T1DM patients or both the T1DM group and the control (non-T1DM) group; cohort or cross- sectional or retrospective chart studies; and population of any sex and geographic region. Studies were excluded if the age of onset of T1DM was in adulthood or was indeterminate. Only studies published in English were eligible. Broadly, we considered the exposed group as the one with persons with T1DM and the group with non-T1DM as the unexposed group.
Search Strategy and Study Selection
An initial thorough search on the PubMed and the PROSPERO databases was conducted to ensure no similar meta-analysis had been or was being undertaken.
All types of articles published till 15 February 2024 were searched independently in the PubMed, Scopus, and Google Scholar databases by three authors (ST, CVV, SKT). The keyword search strings that were used for retrieving article information were “Diabetes” and “mellitus” and “type 1” or “types 1” or “type-1” or “juvenile-onset” or “insulin-dependent” and “psychosis” or “psychotic” or “schizophrenia” or “bipolar” and “association” or “prevalence” or “risk” and “children” or “adolescents.” The initial screening was carried out by going through the titles and abstracts. Cross-references were also looked for to complete the search process.
Outcome Measures and Data Extraction
The rates (numbers) of cases diagnosed with schizophrenia or related psychotic disorders or bipolar disorder in the group of T1DM patients and the control (non- T1DM) group, where available, were retrieved along with the total sample of T1DM and healthy controls. Where rates were depicted in graphs and not in terms of values, values were extracted from graphs using an open-source platform (
Quality Assessment
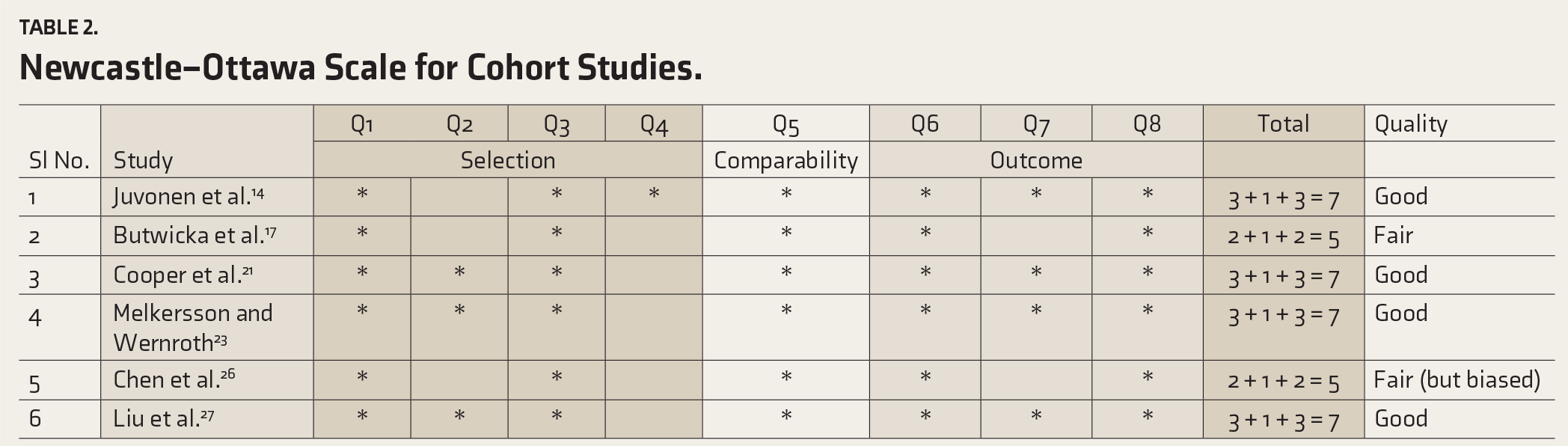
Quality assessment was independently conducted by two investigators (MSS and SAZ). The Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Studies Reporting Prevalence Data 11 and additionally the Newcastle–Ottawa Scale for cohort studies 12 were used for assessing the quality of studies for cohort studies that were included.
Data Analysis
Meta-analyses were conducted using the open-source software
Sensitivity Analysis
Sensitivity analyses were conducted by excluding studies that scored “low” or “biased” on the quality assessment.
Trial Sequential Analyses
To improve the validity of the meta- analysis of risk ratios, we conducted a supplementary “trial sequential analyis” (TSA). An open-source software
Results
Screening and Study Selection
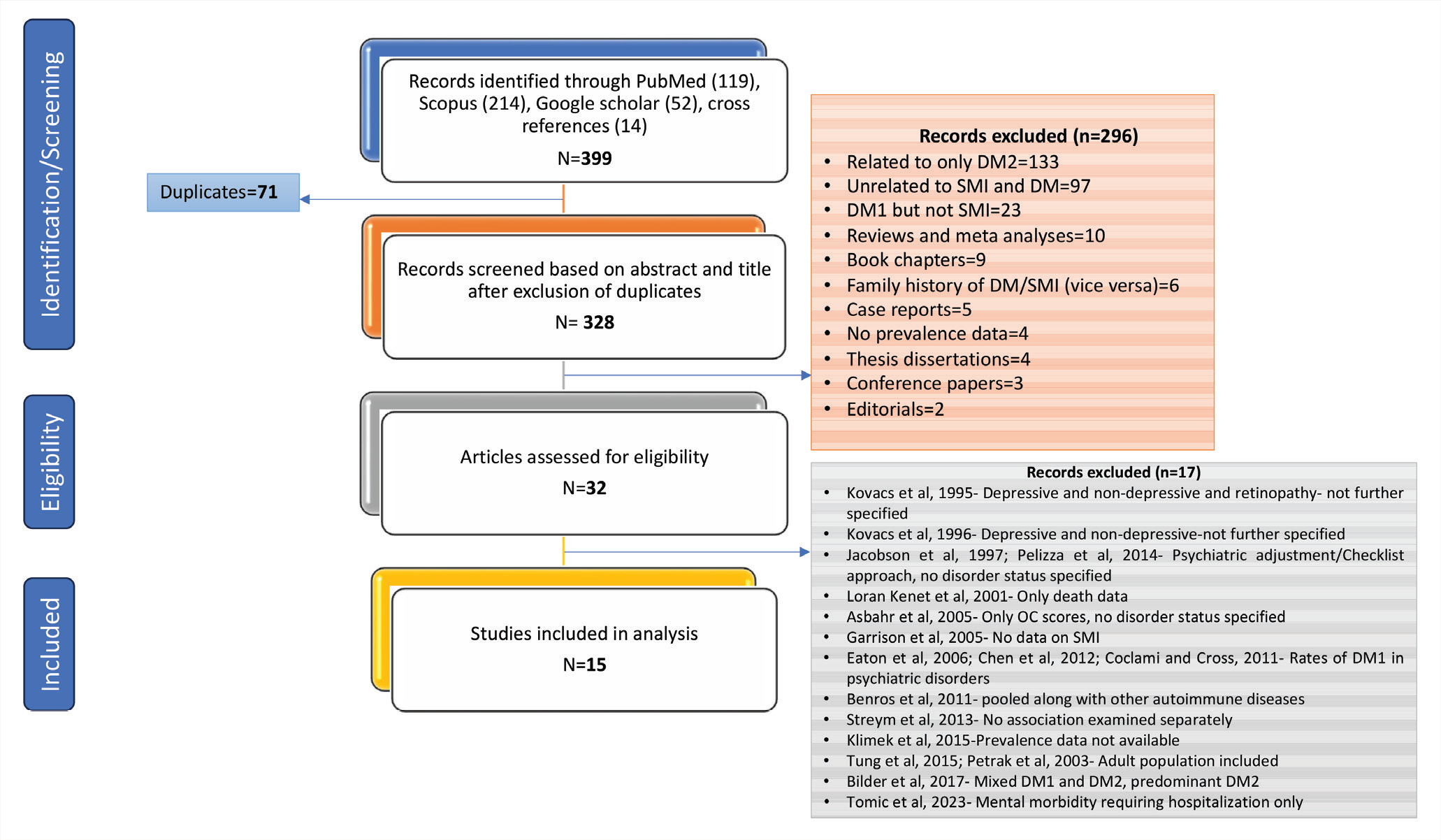
A total of 399 articles were retrieved. After initial screening, 32 articles were reviewed in detail. Finally, for the synthesis and meta-analysis, we included 15 articles. Details of screening and eligibility are shown in the PRISMA flow chart (Figure 1).
PRISMA Flow Chart.
Study Characteristics
The study characteristics of the 15 studies13–27 included in the review are provided in Supplementary Tables 2a and 2b. The list of citations to articles excluded at the second screening stage (eligibility;
While prevalence data was available from all the 15 included studies, risk ratio could be calculated only from 6 studies14,17,21,23,26,27 While 14 of the 15 studies reported prevalence rates for schizophrenia and related disorders, only 3 studies21,26,27 reported rates for bipolar disorder. Correspondingly, while all 6 studies that could be assessed for risk ratios reported data for schizophrenia and related disorders, only 2 of those 6 studies reported data for bipolar disorder.
Quality Assessment
While none of the 6 studies assessed on the Newcastle–Ottawa Scale scored “low,” 4 of the 15 studies evaluated on the JBI Critical Appraisal Checklist scored low. See Tables 1 and 2. However, although none of the studies evaluated on the Newcastle–Ottawa Scale scored low, the Chen et al. study, 26 which was noted to have a biased selection of the non-exposed cohort where even diabetes mellitus type 2 was excluded, was considered “biased.”
Newcastle–Ottawa Scale for Cohort Studies.
Meta-analyses: Prevalence
Random-effects meta-analyses of prevalence values revealed a pooled (transformed-back-transformed) prevalence of 0.37% (95%CI: 0.19–0.73) for schizophrenia and related psychotic disorders and 0.39% (95%CI: 0.05–2.99) for bipolar disorder (together: 0.38% (95%CI: 0.2–0.71) in T1DM patients. All three analyses were statistically significant in terms of effect, implied no publication bias, but showed considerable heterogeneity. The pooled prevalence for bipolar disorder was imprecise (wide confidence interval). See Table 3, Figure 2A, and Supplementary Table 4.
Meta-analyses: Risk ratio
Results of the random-effects meta- analysis of risk ratio data revealed that the pooled risk ratio of schizophrenia and bipolar disorder, together, in T1DM was 1.80 (95%CI: 0.64–5.03;
Sensitivity Analysis
On excluding the four studies that score low on quality assessment according to the JBI Critical Appraisal Checklist for prevalence data, the pooled prevalence rate for schizophrenia and bipolar disorder together was noted to reduce to 0.30% (95%CI: 0.15–0.61). For schizophrenia and related disorders, the rate dropped to 0.33% (95%CI: 0.15–0.74). Model significances for sensitivity analyses compared to the primary analyses remained unaltered. Sensitivity analysis was not conducted for bipolar disorder as the study number was reduced to only two.
A sensitivity analysis for risk ratio meta-analysis showed that the pooled risk ratio was reversed (0.88% (95%CI: 0.49–1.56;
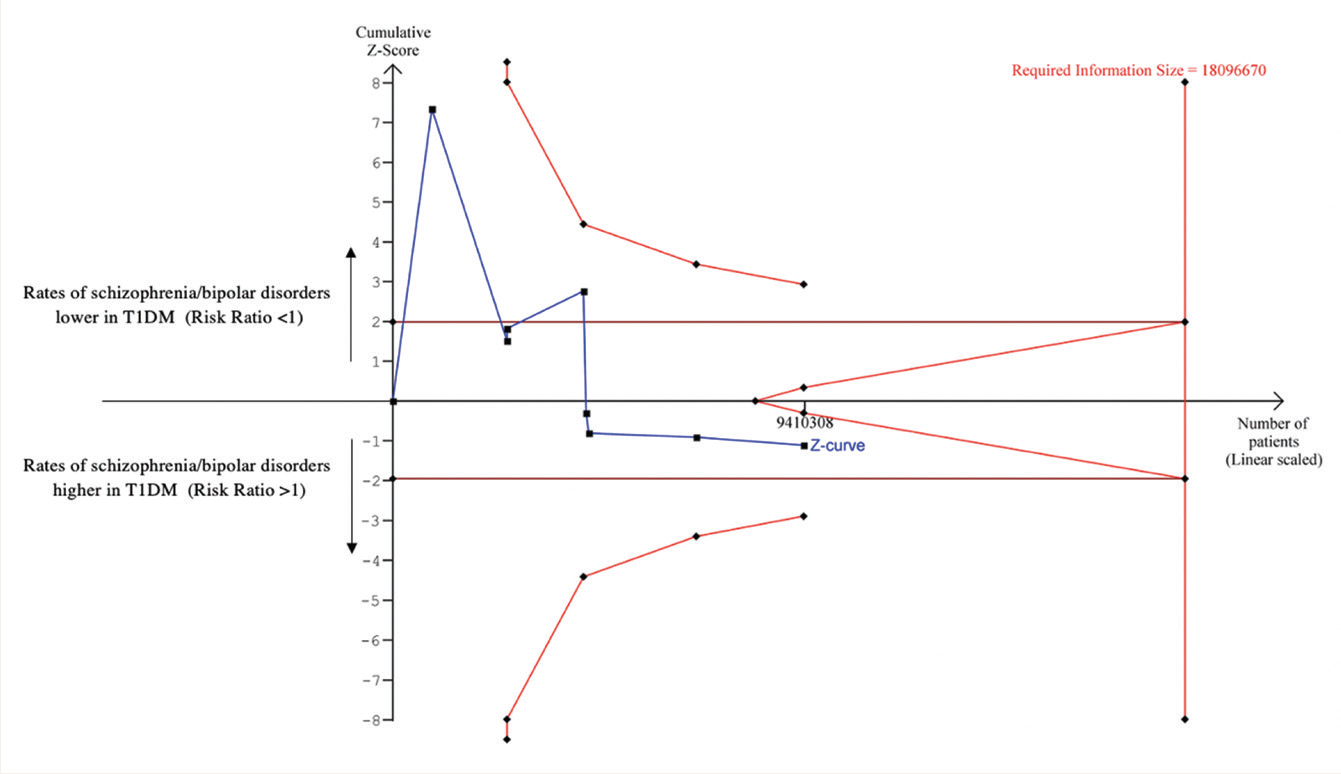
Trial Sequential Analysis
Details of the input values for the TSA are given in Supplementary Table 5. Figure 2 shows the TSA graph. The cumulative Z-curve was seen to end on the side of a higher risk of schizophrenia/bipolar disorder in T1DM and not to cross any of the trial sequential boundaries—conventional, monitoring, and futility—suggesting an inconclusive nature of the findings and a need for more studies to confirm the increased risk.28,29 The diversity-adjusted required information size was estimated as 18,096,670, which was greater than the current information size of 9,410,308.
Results of the Random-effects Meta-analyses of the Prevalence of Schizophrenia and Related Psychotic Disorders and Bipolar Disorder in Type 1 Diabetes Mellitus.
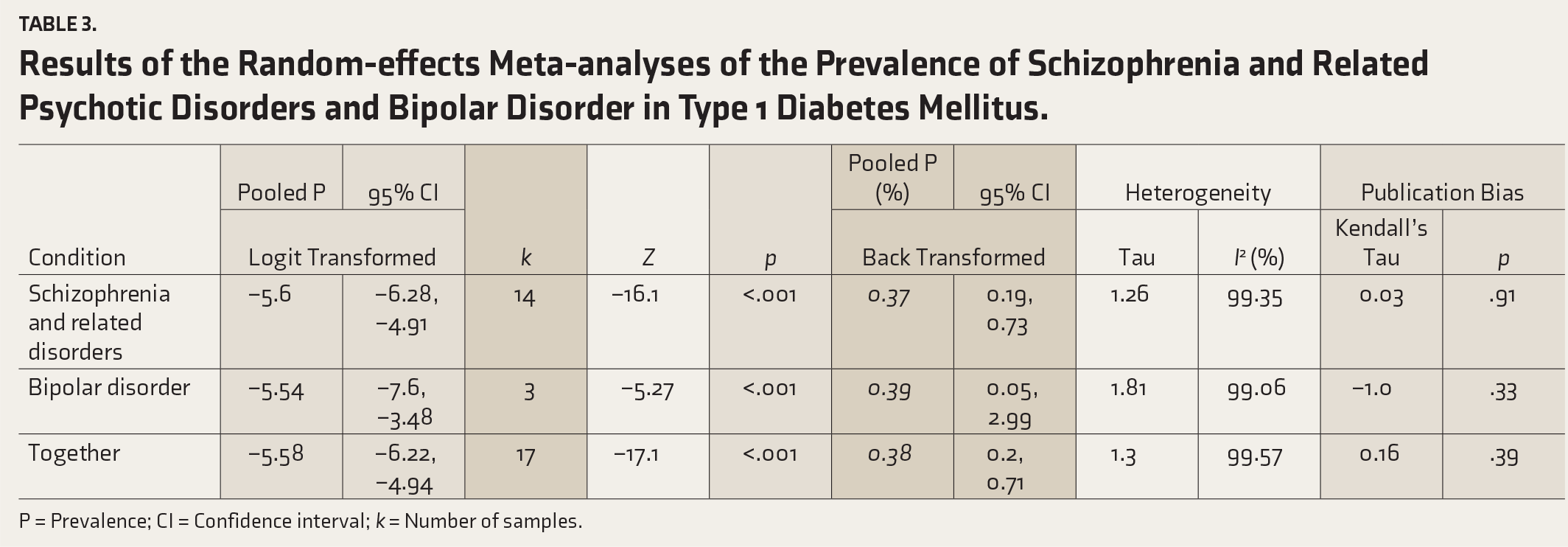
P = Prevalence; CI = Confidence interval;
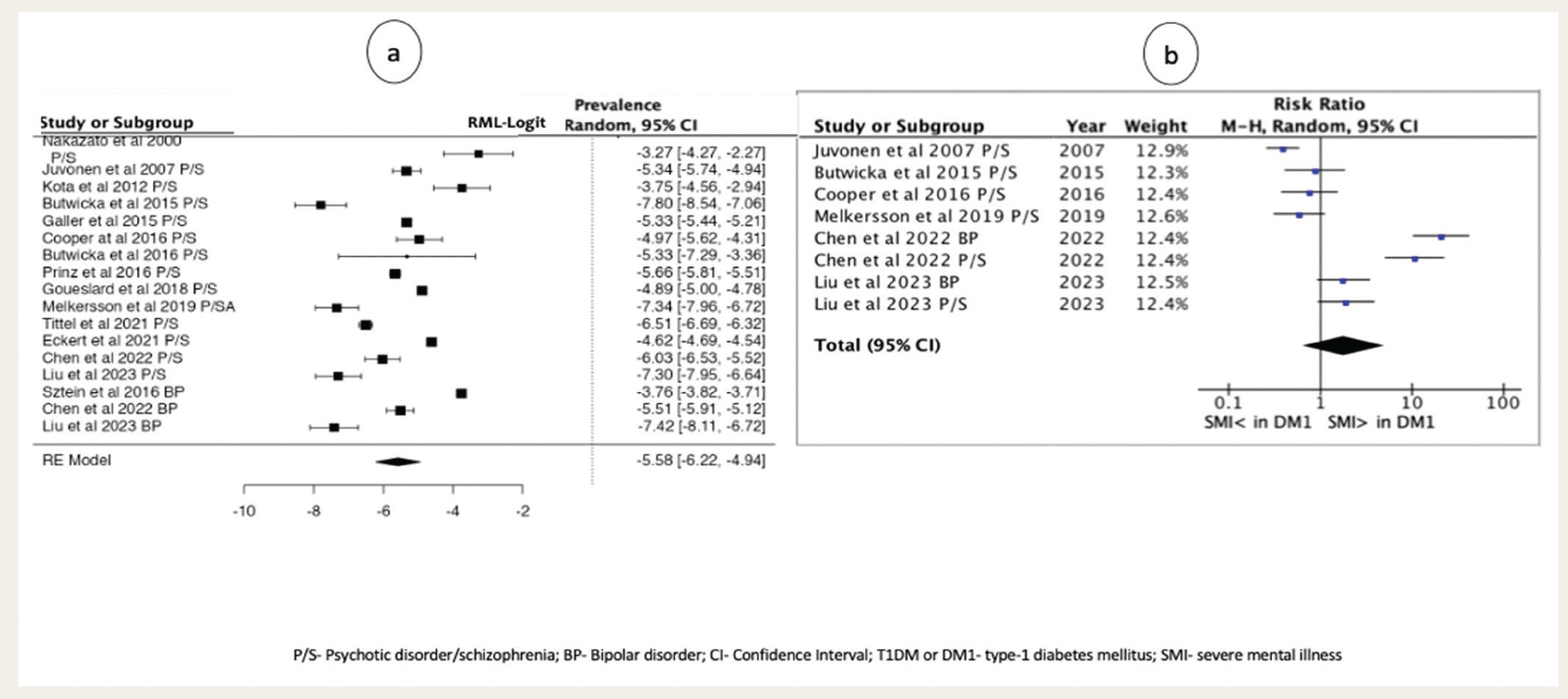
P/S: Psychotic disorder/schizophrenia; BP: Bipolar disorder; CI: Confidence interval; T1DM or DM1: Type-1 diabetes mellitus; SMI: Severe mental illness.
Trial Sequential Analysis Graph.
Discussion
Our results could not support the hypothesis that the pooled prevalence and risk of schizophrenia and related disorders and bipolar disorder are significantly greater in individuals with T1DM. While the pooled prevalence rates were sensitive, they were not significantly greater in T1DM than in the general population. On the other hand, while the risk ratio of schizophrenia and bipolar disorder was higher in T1DM than in non-T1DM, this result could not survive sensitivity analysis. Moreover, this result was limited by a lack of statistical significance for the meta-analysis of risk ratios and, more importantly, high heterogeneity.
We suppose that various factors contributing to methodological and clinical diversity are likely to influence our results. Methodological diversity might have risen due to variations in factors such as age and gender, treatment and glycemic control status of T1DM, etc. Meta-regression controlling for such factors might allow us to understand their influence on the results of meta-analysis. On the other hand, variations in the immune response patterns within both T1DM and schizophrenia/bipolar disorder and their influence on each other might contribute to clinical diversity. 30 TSA, which adjusts for all forms of diversity, suggested that the required information size is roughly 2 times that of the available information size, implying a need for more studies in this regard. Good quality data on the prevalence of schizophrenia and bipolar disorder in T1DM from countries such as India, where the prevalence of T1DM is higher than in many other countries, 31 can help meet this need. Nevertheless, there is a fair amount of data on the association of depression in T1DM from India. 32
Conducting an article search on a more comprehensive list of databases and performing a meta-regression might provide more valuable information.
Conclusion
Our review cum meta-analysis could not provide convincing evidence for the hypothesis that the prevalence and risk of schizophrenia and related disorders and bipolar disorder are greater in individuals with T1DM. We conclude that more studies are needed to ascertain the association.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
None used.
Ethics Approval and Informed consent
As this was a synthesis of data available in the literature, no patient consent and ethics committee approval were sought.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
